Abstract
This research introduces a novel hydrogel polyester fabric treated with mixture of synthetic/natural hydrophilic polymers along with in situ synthesis of nano silver through reduction of silver nitrate. Polyvinyl alcohol (PVA) and tragacanth were used as the most common synthetic and natural hydrophilic polymers, citric acid as a friendly cross-linking agent, and sodium hypophosphite as catalyst. Various characteristics of the treated fabrics including water absorption capacity just before dripping, drying time of the wetted fabric, moisture regain, water retention value, vertical wicking, and antimicrobial properties against a Gram positive (Staphylococcus aureus) and a Gram negative (Escherichia coli) bacteria were investigated. SEM images and EDX patterns were also obtained to characterize the surface morphology and elemental analysis of the finished fabrics. The size of small crystalline region was calculated through XRD data and the chemical structure of the samples was identified with FTIR spectra. The finished fabric showed the higher moisture absorption and time of drying with very good anti-bacterial properties.
Introduction
Hydrophilic gels as hydrogel are cross-linked materials absorbing large quantities of water without dissolving. Softness, smartness, and the capacity to store water are some properties of hydrogels [1–4]. The ability of hydrogels to absorb water is due to the presence of chemical groups such as hydroxyl (–OH), carboxyl (–COOH), amide (–CONH–), primary amide (–CONH2), sulfonic (–SO3H), and other hydrophilic groups that can be found within the polymer backbone or as lateral chains. The favorable property of hydrogels is their ability to swell, when absorbing water. In this case, the unabsorbed glassy phase is separated from rubbery hydrogel region with a moving boundary. Regularly the meshes of the network in the rubbery phase will start expanding, allowing other solvent molecules to penetrate within the hydrogel network [5]. The equilibrium swelling degree and the elastic modulus of hydrogels depend on the cross-linking and charge densities of the polymer network as well as on the cross-linked polymer concentration after the gel preparation [6]. Synthetic and natural hydrogels have been used in tissue engineering, pharmaceutical, and biomedical fields. They have been used in wound dressing, drug delivery, dyeing absorption, sensors, agriculture, sanitary pads as well as implants, injectable polymeric systems, ophthalmic applications, hybrid-type organs due to the high water absorption capacity, and biocompatibility [7–19]. Glampedaki et al. investigated moisture absorption capacity of polyamide 66 fabrics functionalized with chitosan-based hydrogel and polyester textile functionalized through incorporation of pH/thermo responsive N-isopropyl acrylamide-chitosan micro gels. They found higher wettability and moisture absorption on the treated fabrics [20,21]. Lavric et al. assessed interaction between water and stimuli-responsive micro gel (poly-NiPAAm/chitosan) on cotton fabric. They showed upgrading textile material quality by providing highly attractive feature of stimuli-responsive liquid management properties [22].
Polyvinyl alcohol (PVA) is a polymer of great interest because of its many characteristics such as good mechanical properties, good chemical stability, low toxicity, biocompatibility, and the ability to retain water in the structure [23–32]. PVA hydrogels are used in tissue engineering scaffolds, wound dressings, and drug controlled-release systems such as contact lens production, cartilage reconstruction and regeneration, artificial organs, and many others [23–32]. PVA shows higher tensile strength and elongation before breaking having a suitable molecular weight for preparation of hydrogel. The low molecular weight leads to the weak entanglement of PVA chains forming physical cross-linked network and the high molecular weight produces bubbles that cannot be removed completely from the PVA solution [23–32]. The combination of synthetic and natural hydrogel improves physical, biocompatibility, and irritant-sensitivity properties of both hydrogels [33].
Hydrocolloid is set of polysaccharides and proteins with functional effects such as concentration, gelling, foams stability, and emulsions. Tragacanth hydrogel is an anionic polysaccharide composed of two parts of water-swellable and water-soluble known as bassorin and tragacanth, respectively. Tragacanth is used as stabilizer, thickener, emulsifier, moisture retaining agent, binding agent, anti-freezing, and adhesive [34–36].
Nano silver can also be utilized in the textile industry by incorporating into the fiber or produced as a powder. Silver is a good anti-bacterial agent, non-toxic and natural inorganic metal appears as an interesting material to be used in different textile fibers. Smaller particles with a larger surface area are more efficient in the anti-bacterial activities [37]. There are some limitations to use nano silver in aqueous phase such as controlling the shape, size and stability in solution. PVA hydrogel and nano silver retain their individual features and characteristics in the combined system [38].
In the present work, PVA as a desirable hydrogel was combined with tragacanth as a novel natural polymer with silver nitrate to prepare PVA-tragacanth/nano silver hydrogel for modification of polyester fabric surface to produce hydrophilic fabric with anti-bacterial properties as a disposable medical and sanitary textile.
Materials and methods
Materials
The raw polyester fabric was used with warp density of 39 yarns/cm, weft density of 18 yarns/cm, and weight of 210 g/m2 (Hijab Textile Co., Shahrekord, Iran). PVA ( Chemical structure of tragacanth.
Methods
Preparation of samples
Preparation of various samples.

W1 and W2 are samples weight before and after treatment, respectively.
Anti-bacterial test
Samples with similar dimension were placed in the tubes autoclaved at 120℃ for 15 min and then 100 mL bacteria suspension (Escherichia coli and Staphylococcus aureus) containing 106 bacteria were added. The tube was closed and placed at 37℃ for 16–20 h. Ten milliliters of sterile normal saline were then added and bacteria was separated from fabric and entered to normal saline. Dilutions of 1:10 of each tube were prepared (100 µL sample and 900 µL normal saline). Eight thousand one hundred microliters of each dilution were cultured on Muller Hinton agar and kept at 37℃ for 18 h. After incubation the number of colonies on each sample was counted and then multiplied in dilution until number of bacteria in mL were obtained (equation (1)).

Blank and test are number of bacterial colonies from untreated and treated samples, respectively.
Drying time of wetted fabric
Samples were impregnated in the distillated water for 24 h and then removed and left on the beaker surface for few seconds and the fabric weight was measured 1 min intervals to obtain a constant weight.
Moisture regain
The fabric samples were placed in room conditions for 24 h and weighed and then placed in an oven at 105℃ for 1 h and reweighed quickly. The moisture regain of each sample was calculated according to equation (2).

Water absorption capacity of the fabric just before dripping
The fabric samples were placed on the beaker and the water droplets were dropped on the fabric surface by a pipette till the water dripped from the sample to the beaker. The amount of water absorption was measured by pipette scale.
Water retention value
The cotton fabric samples were immersed in distilled water for 24 h and then centrifuged at 7800 r/min for 2 min. The fabric weight before drying (Ww) and the dried fabric weight (Wd) at 105℃ were then measured and the water retention values (WRVs) were calculated using equation (3) [39].

Vertical wicking
The water vertical wicking through the fabric was examined using 3 × 20 cm2 fabric sample as 1 cm height was dipped into the distilled water for 2 h and then the wetted height of the samples were recorded.
Results and discussion
Chemical mechanism
The hydrolysis of polyester and formation of PVA–tragacanth hydrogel along with in situ synthesis of nano silver particles are shown in Figure 2. The hydroxyl groups were created on the hydrolyzed polyester fabric (reaction 1). CA as a cross-linking agent in the presence of SHP and heating converted to the cyclic anhydride intermediate to be able to react with functional groups of polyester and PVA–taragacanth (reaction 2) [40]. This provides more hydroxyl groups on the polyester fabric surface due to loading of PVA and tragacanth on the fabric surface (reaction 3). Also, H ions can be replaced with Ag ions synthesis Ag nanoparticles cross-linking between chains of various presented polymers including PVA, taragacanth, and hydrolyzed polyester forming a three dimensional network and stabilizing the PVA–targacanth on the fabric surface (reaction 4).
Alkali hydrolysis of polyester and synthesis of PVA–tragacanth/Ag hydrogel on the hydrolyzed PET.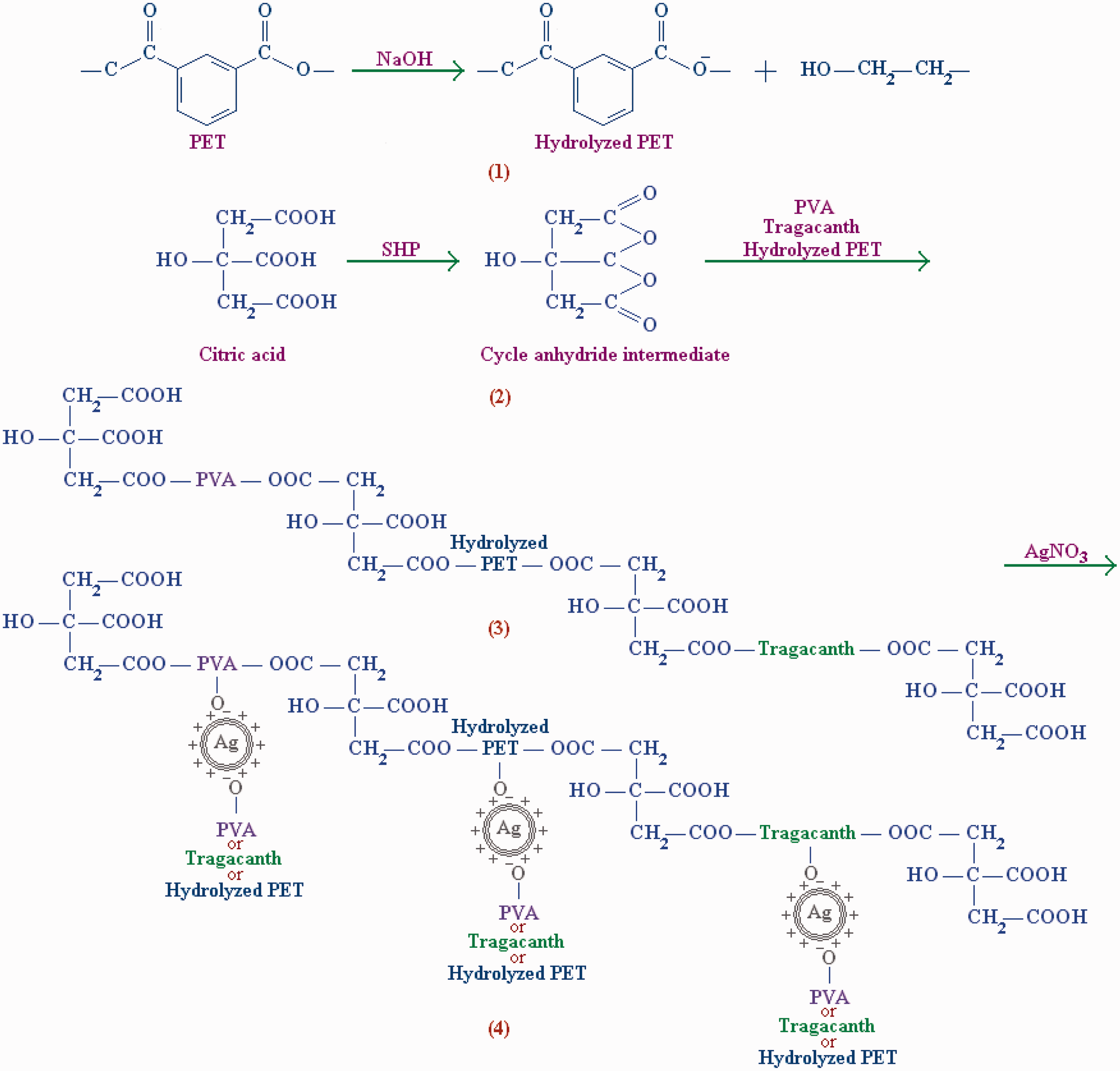
X-ray diffraction
XRD defines the crystalline nature and determines the crystal size of particles. The bulk sample was put on the warm up device, preset the scan speed and 2θ angles from 0° to 100°, the sample was then exposed to the electrons by applying 40 kV and 30 mA.
The XRD patterns of samples treated with nano silver in Figure 3 confirmed the presence of silver crystals on the treated samples. All the prominent peaks on 2θ scale are about 37.47, 44.30, 64.95, 77.27, 82.27 and 37.85, 44.20, 64.26, 77.40, 82.38 for samples 3 and 5 exhibiting (1, 1, 1), (2, 0, 0), (2, 2, 0), (3, 1, 1), and (2, 2, 2) plates. The highest intensity peak around 2θ = 44 is related to the untreated polyester fabric [41]. The main peak was considered at 2θ = 37.47 for sample 3 and 2θ = 38.60 for sample 5. Also the small peaks showed the low amount of nano silver particles on the treated fabric surface. XRD patterns and SEM images confirmed the presence of nano silver on the surface of treated samples and energy-dispersive X-ray spectroscopy (EDX) analysis showed the low amount of nano-Ag particles loaded on the treated fabric surface. The particle size was measured by Scherrer equation, equations (4) and (5).
XRD patterns of various samples (a) 1, (b) 3, and (c) 5.


In this equation, λ = 1.54° is the wavelength of X-ray of Cu radiation, β = FWHM is full width at half maximum of the peak based on radian, Wi is sample peak, W0 is 0.15868, and θ is the diffraction angle. Higher FWHM showed the smaller crystals and lower FWHM defined the bigger ones [42]. The crystal size of silver in samples 3 and 5 were 17.09 and 14.99 nm, respectively.
Fourier transformed infrared spectroscopy
FTIR spectroscopy is a technique based on the determination of the interaction between an IR radiation and a sample. It measures absorbed frequencies by the sample and also the intensities of the absorptions. FTIR patterns of untreated and treated polyester fabrics are presented in Figure 4. The peaks from 500 to 1800 cm−1 indicate the original signals, such as stretching vibration band of C=O and aromatic structure confirming ester linkages [43]. The peak at 852 cm−1 and 1429 cm−1 showed the stretching and bending modes of CH2 group, respectively [44]. The peaks at 1690–1760 cm−1 are assigned to C=O stretching [45]. This peak is strong and concern to aldehyde, ketone, carboxylic acid, and ether groups. The peak at 1340–1470 cm−1 is assigned to C–H bending concerns to alkanes group [38]. The peaks at 1050–1300 cm−1 are assigned to C–O stretching concern to alcohol, ether, and carboxylic acid groups [45]. The peaks at 1200–1350 cm−1 are assigned to O–H bending that is sharper for the treated fabric with PVA–tragacanth/Ag than untreated one showing more hydroxyl groups on the surface of PVA–tragacanth/Ag hydrogel treated sample. The peaks at 610 cm−1 and 850 cm−1 are assigned to O–H and C–H vibrations in PVA [46]. The FTIR spectrum of PVA-tragacanth/Ag nanocomposite indicated the linkage between O–H and C–H vibrations due to bonding interaction between O–H and silver nanoparticles. This type of linkage has great influence on the improvement of durability.
FTIR spectra of various samples 1 (red graph) and 5 (black graph).
SEM and EDX
The morphology of the treated and untreated samples is illustrated in Figure 5. The morphology of hydrolyzed polyester fabric is presented in Figure 5(a). PVA–tragacanth hydrogel, PVA–tragacanth/nano-Ag hydrogel, PVA–tragacanth hydrogel with CA cross-linking agent and PVA–tragacanth/nano-Ag hydrogel with CA cross-linking agent coated on hydrolyzed polyester surface are shown in Figure 5(b), (c), (d), and (e, f and g), respectively. Some cracks observed on the fiber surface due to the hydrolysis of polyester in alkali conditions. The formation of polymeric layer and adhesion between the fibers can be observed on the treated samples. Nano silver particles were also seen in Figure 5(c, e–g) as spherical white points. The higher resolution Figure 5 (f and g) indicates the cracks filled with PVA–tragacanth/nano-Ag hydrogel. Also the agglomerated nanoparticles determined particle size of 42.06 nm.
SEM image of various samples (a) 1 (1.00 kx), (b) 2 (1.00 kx), (c) 3 (1.00 kx), (d) 4 (1.00 kx), (e, f, g) 5 (1.00 kx), (10.00 kx), (30.00 kx), respectively, and (h) EDX of sample 5.
Figure 5(h) shows EDX spectrum of the treated sample coated with PVA–tragacanth/Ag along with CA cross-linking agent. The presence of Au element in this pattern is due to the gold layer covered the treated sample to prepare for SEM and EDX analysis. The presence of nano silver on the PET fabric surface was proven by EDX patterns. The peak intensity shows the low amount of nano-Ag particles on the treated fabric surface.
Drying time of wetted fabric
Characteristics of different treated fabrics.

Moisture regain
The finished samples with PVA–tragacanth hydrogel showed higher moisture regain than raw sample (Table 2). The created hydroxyl groups on the polyester surface help to absorb water molecules that then release slowly. The treated sample with CA and hydrogel indicated the highest moisture regain due to the presence of remaining hydroxyl and carboxyl groups of CA within the complex structure of the formed 3D hydrogel.
Water absorption capacity of the fabric just before dripping
The hydrogel treated samples indicated more ability to absorb water drops on the fabric surface before passing through or dripping compared to raw sample (Table 2). The created layer of PVA–tragacanth hydrogel on the fiber surfaces block the fiber pores preventing the water to pass through. Also, the sample coated with CA cross-linking agent absorbs the most water drop comparing to other samples. The presence of CA cross-linking agent increased the hydrogel stability and sited more hydroxyl groups on the fabric surface.
Water retention value
More water retention reported on the finished fabrics than the raw sample (Table 2). In raw polyester fabric, water remains on the surface preventing passing through the fabric leading to quick release through centrifuge. However, the water molecules penetrated into the finished fabrics producing hydrogen bonding between PVA–tragacanth hydrogel and polyester chains led to difficult removing from the fabric. Also the water retention in the treated fabric with hydrogel and CA was more than others due to the presence of CA. CA increases the hydrogel stability and produces more hydrophilic sites on the fabric surface.
Vertical wicking
The vertical wicking of the finished fabrics was shorter than the raw one (Figure 6) proving slow water molecules transfer in the treated samples. The mechanism of water transfer in textile is similar to the wicking of a liquid in capillaries. The capillary action is determined by diameter and surface energy as the smaller diameter or higher surface energy leads to more vertical capillary. The spaces between the fibers in fabric structure effectively form capillaries. Thus, the narrower spaces between the fibers lead to the greater ability of the water to wick into textile [47]. The polyester fabric treated with hydrogel has the fibers loaded with PVA–tragacanth gel produces spaces with the wider diameter on the fiber surfaces decreases the vertical capillary. Also the wetted hydrogel swells produced wider free spaces for water absorption leads to shorter vertical wicking. As long as presence of free spaces, the water will not rise through the fabric.
Vertical wicking of various samples (a) 1, (b) 2, (c) 3, (d) 4, and (e) 5. (The water vertical wicking is illustrated based on cm.)
Anti-bacterial efficiency
The anti-bacterial activities of the raw and treated samples against E. coli and S. aureus are shown in Figure 7. The number of E. coli colony forming units is 246, 0, and 8 for samples 1 (raw), 3, and 5. This indicates very low colony forming for the treated samples with silver nitrate. The number of S. aureus colony forming units is 523, 54, and 0 for samples 1 (raw), 3, and 5, respectively. Thus, the anti-bacterial activities of the treated samples are 100% and 96% against E. coli for samples 3 and 5, respectively. Also the anti-bacterial activities of samples 3 and 5 were 89% and 100% against S. aureus, respectively. These results indicate excellent anti-bacterial activities against both E. coli and S. aureus for the treated samples with silver nitrate. These results are in accord with other findings showing good anti-bacterial properties on polyester or other fabrics treated with nano silver [48–51].
Anti-bacterial activity against E. coli and S. aureus for various sample (a,d) 1, (b,e) 3, and (c,f) 5, respectively.
Conclusion
In this work, silver nanoparticles were synthesized on polyester fabric during application of hydrogel for first time as anti-bacterial agent however in the other reported works just hydrogel applied on polyester fabric based on the other polymers. Therefore, a novel natural based hydrogel was applied on polyester fabric along with synthesis of nano silver in one simple step as a time and cost effective method. The PVA–tragacanth hydrogel treated polyester fabric indicates higher water absorption due to the presence of hydroxyl groups on the fabric surface. Introducing CA on the finished fabric produces more hydrophilic groups on the surface leads to enhanced water uptake. The presence of PVA, tragacanth, and hydroxyl groups was confirmed with FTIR spectra and SEM images. XRD patterns and SEM images also proved the presence of nano silver on the surface of treated samples and EDX patterns showed the low amount of nano-Ag particles on the treated fabric surface. The crystal size of silver was measured by XRD results and SEM images determined the agglomerated nanoparticles. Further, no significant color change occurred on the treated fabric samples.
Also, in situ synthesis of nano silver on the polyester fabric creates excellent anti-bacterial property with lower water absorption due to the blocking of some hydrophilic groups through silver ions binding. This treatment is an appropriate finishing for enhanced water absorption on the polyester fabric. The treated polyester fabric can also be used in medical applications for absorbing wound liquids or in sport-wear for absorbing sweat preventing from wetting.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
