Abstract
Anatase titania (TiO2) nanoparticles were prepared from natural minerals (rutile sand) using acid extraction by sol–gel method. The obtained X-ray diffraction results show that the particle possesses crystallite size of 12 nm. The colloidal silica and TiO2 nanoparticle-embedded colloidal silica solutions were prepared using sol–gel method followed by sonication process. The particle size was measured for the prepared colloidal TiO2/SiO2 sol. The prepared solution was coated on the surface of the cotton fabric through pad-dry-cure method. Elemental analysis confirms the presence of TiO2/SiO2 nanocomposite along with cellulose on the surface of the fabric. The washing durability of the coated fabrics after fifth and 10th washes indicates that the nanoparticles strongly adhered to the fabric surface. The ultraviolet resistance, burning performance, and antibacterial activity against Gram-positive
Introduction
Nanotechnology is extensively used in textile industries because it confers unique properties on fabrics such as ultraviolet (UV) protection, antibacterial activity, fire resistance, and high durability [1]. The large surface-to-volume ratio of the nanoparticles is effectively interacted with fabric surface due to the Van der Waals forces involved in both [2]. The inorganic materials like metals and metal oxide-coated fabrics have created a lot of awareness appropriate to improve their functional properties [3]. Owing to their cost consideration, a small amount of metal nanoparticles, compared with the usage of metal oxide nanoparticles, are used in textile industry. The metal oxide nanoparticles, like Al2O3, SiO2, MgO, ZnO, TiO2, etc., are extensively studied for better physico-chemical and functional properties for textile applications [4–8]. Especially, the anatase TiO2 nanoparticles are extensively used to improve multifunctional properties of cotton and wool fabrics, namely UV protection, fire resistance, and antibacterial activity [9].
TiO2 is the one of the functional materials and is essentially used in different areas like electronics, biomedical, optical, dermatological, and photocatalytic due to their excellent physico-chemical properties [8]. The colloidal silica sol, chitosan, and acrylic binders with TiO2 nanoparticles are used to incorporate functional properties on fabrics [10–12]. The monodispersed colloidal silica particle is one of the attractive materials not only because it acts as a binder for coating nanoparticles on fabrics but also because it improves their softness [13]. The matrix of polyethylene glycol (PEG) is added into the sol, which makes the surface protective and biocompatible, and also enhances the physical and antistatic properties of the fabrics [14,15]. There are few reports on silica (SiO2) binders with TiO2 nanoparticles coated on cotton fabrics. Thus, it is essential to study the function of TiO2/SiO2 composites on the surface of cotton.
In this study, the anatase TiO2 nanoparticles and TiO2/SiO2 composites are prepared using sol–gel method. PEG is added with the prepared composites to improve the biocompatibility and stability of the prepared solution. The prepared TiO2 composites are coated on cotton fabrics by pad-dry-cure method. This study focuses mainly on the analysis of functional properties of TiO2/SiO2-coated fabrics, like UV protection, flame retardation, and antibacterial activity against
Experimental
Materials
Mercerized and bleached cotton fabric (100%, mass 138.84 g m−2, 116 ends per inch × 84 picks per inch) was pretreated as substrate for coating. Tetraethyl orthosilicate (TEOS; Merck; 99%), PEG (PEG 400; Merck; 99.5%), ethanol (Merck; 99.9%), and de-ionized water were used for the preparation of silica sol and nano-TiO2 from rutile sand (Kanyakumari, Tamil Nadu, India) and sulfuric acid (H2SO4; Merck; 99%). For both samples, ammonia (Merck; 25%) was used as the reducing agent.
Synthesis and characterization of titania nanoparticles
In this study, TiO2 nanoparticle was synthesized using 5 g natural mineral (rutile sand) using acid extraction via sol–gel method [8]. Raw rutile sand was fired in a muffle furnace at 873 K and digested with 500 ml sulfuric acid at 353 K for 1 h. The obtained titanium sulfate was taken as a precursor and then added with a reducing agent (25 ml ammonia) and maintained at a pH 9. The mixture was stirred (400 r/min) at room temperature for 3 h in order to form titanium hydroxide solution. The precipitate was centrifuged at room temperature at 10,500 r/min, and then, the collected powder was rinsed thrice with double-distilled water to remove the ionic content (NaSO4). Finally, the obtained rinsed powder was washed with ethanol for a few minutes to remove the moisture content. The obtained powder was dried at 673 K under muffle furnace for 3 h to produce the white TiO2 nanopowder.
The obtained TiO2 nanopowder was characterized using X-ray diffraction (XRD; X'Pert PRO, PANalytical, Almelo, the Netherlands) spectrometer with CuKα as a radiation source (
Colloidal silica and hybrid composite sol
The colloidal silica sol was obtained by adding TEOS (10 ml) into ethanol (180 ml) under vigorous stirring. PEG, ammonia, and water (molar ratio 2:1:1) were drop-wise added as a stabilizer and a reducing agent, respectively, under continuous stirring (400 r/min) for 90 min at room temperature [17]. TiO2 nanoparticles (1 g) were added to the silica sol under stirring (400 r/min) for 15 min. Then, the obtained hybrid TiO2/SiO2 composite solution was kept under ultrasonic irradiation with low frequency at 30 kHz for 30 min. The flow chart for the preparation of SiO2 and TiO2/SiO2 composites is shown in Figure 1.
Preparation of SiO2 and TiO2/SiO2 nanocomposites.
Nanoparticle coating and characterization of cotton fabrics
The 15 × 15 cm cotton fabric samples were rinsed with 1 wt% of NaOH solution along with de-ionized water twice at room temperature. The cleaned fabrics were dried in an oven at 303 K for 5 min. In addition, the fabric substrate was immersed four times in a homogeneous solution containing silica sol and TiO2/SiO2 hybrid solution, using pad-dry-cure method [17] with a suitable pressure (4 kg cm−2) and speed (5 r/min). Finally, the coated fabric was cured in an oven at 333 K for 3 min under dry heat to remove impurities. To evaluate the nanoparticle ascent in the textile fibers, we subjected the coated fabrics to comprehensive analysis. The proposed securing mechanism of TiO2/SiO2 composite systems on the surface of cotton fabrics is shown in Figure 2. Hereafter, the un-coated fabrics, fabrics coated with silica [17], and fabrics coated with TiO2/SiO2 hybrid composite nanosol are termed as UC, SC, and TSC fabrics, respectively.
Schematic representation of chemical reaction during coating on fabrics.
UC and TSC fabric sample properties were further explored through comprehensive characterization techniques. The surface morphology of the UC and TSC fabrics was investigated using SEM with EDS. The structural analysis of fabric substrate was carried out using XRD studies. The functional group of the fabric samples was examined through Fourier transform infrared spectroscopy (FTIR) combined with attenuated total reflectance (ATR) sampler (Spectrum 100; PerkinElmer, USA) with the range from 4000 to 400 cm−1.
The wash durability (fastness) of UC, SC, and TSC fabrics was carried out as per the IS: 687 – 1979 standard. The physical properties of UC, SC, and TSC fabric samples were conditioned at standard atmospheric temperature (293 ± 3 K). The thickness of the all fabric samples was measured employing fabric thickness tester as per the ASTM D 5729-97 guidelines. The tensile strength of the fabrics was measured by strip method using tensile testing machine (E091; Eureka, India) in accordance with ASTM D5035-95 standard. Similarly, the tear strength of the fabrics was obtained using falling pendulum-type (Elmendorf) apparatus (Premier, India) in accordance with ASTM D1424:2007 standard. The crease recovery tester (EC-41; Eureka) was used to analyze the crease recovery angle by AATCC 66-1998 standard with a scale range from 20° to 80° and an applied creasing load of 300 g for 2 min. The testing was carried out using warp and weft yarns of the fabric samples. The air permeability values of the fabrics were obtained according to IS: 11056-1984 and DIN 53887 standards with the flow rate of passing the air perpendicularly at 100 Pa using an air permeability tester (M021A; Premier). The whiteness index of the fabrics was measured using a spectrometer (CM-3600d; Minolta, Singapore) with D65 light source.
The flame-retardant property of UC, SC, and TSC fabric samples was analyzed using a flame tester (Auto flame I, Premier) at an angle of 45° in accordance with ASTM D1230-97 standard. The UV blocking properties of the fabric samples were tested by UV transmission spectra (Lambda 35; PerkinElmer, USA) with the wavelength ranging from 280 to 400 nm. The percentage of blocking of UV was calculated in accordance with ASTM D6603 standard [18]. Further, the UV protection factor (UPF) value is calculated using the following equation
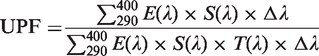
The antimicrobial activities of UC, SC, and TSC fabrics were tested by Kirby–Bauer method (1966). Bacterial strains such as Gram-positive
The quantitative antimicrobial activity was analyzed according to the AATCC test 100-207 (AATCC 2007) in all fabrics. Freshly prepared nutrient broth was transferred to sterile test tube (10 ml each), and then, the fresh cultures of
Results and discussion
Characterization of nanoparticles
Figure 3(a) shows the powder XRD pattern of the prepared sample obtained after calcination at 673 K. The observed XRD pattern of sample is in fine agreement with the anatase structure of TiO2 nanoparticles (JCPDS file no. 21-1272). The peaks observed at (101), (004), (200), (105), (204), and (215) planes correspond to the arrangement of pure anatase structure of TiO2 nanoparticles. The obtained crystallite size of the sample is 12 nm. The SSA of TiO2 nanoparticles is found to be 35 m2g−1 which is confirmed through BET plot as shown in Figure 3(b).
Characterization of TiO2 nanoparticles: (a) XRD pattern, (b) BET surface area, (c) particle size distribution of TiO2 nanoparticles, and (d) TiO2/SiO2 hybrid nanosol.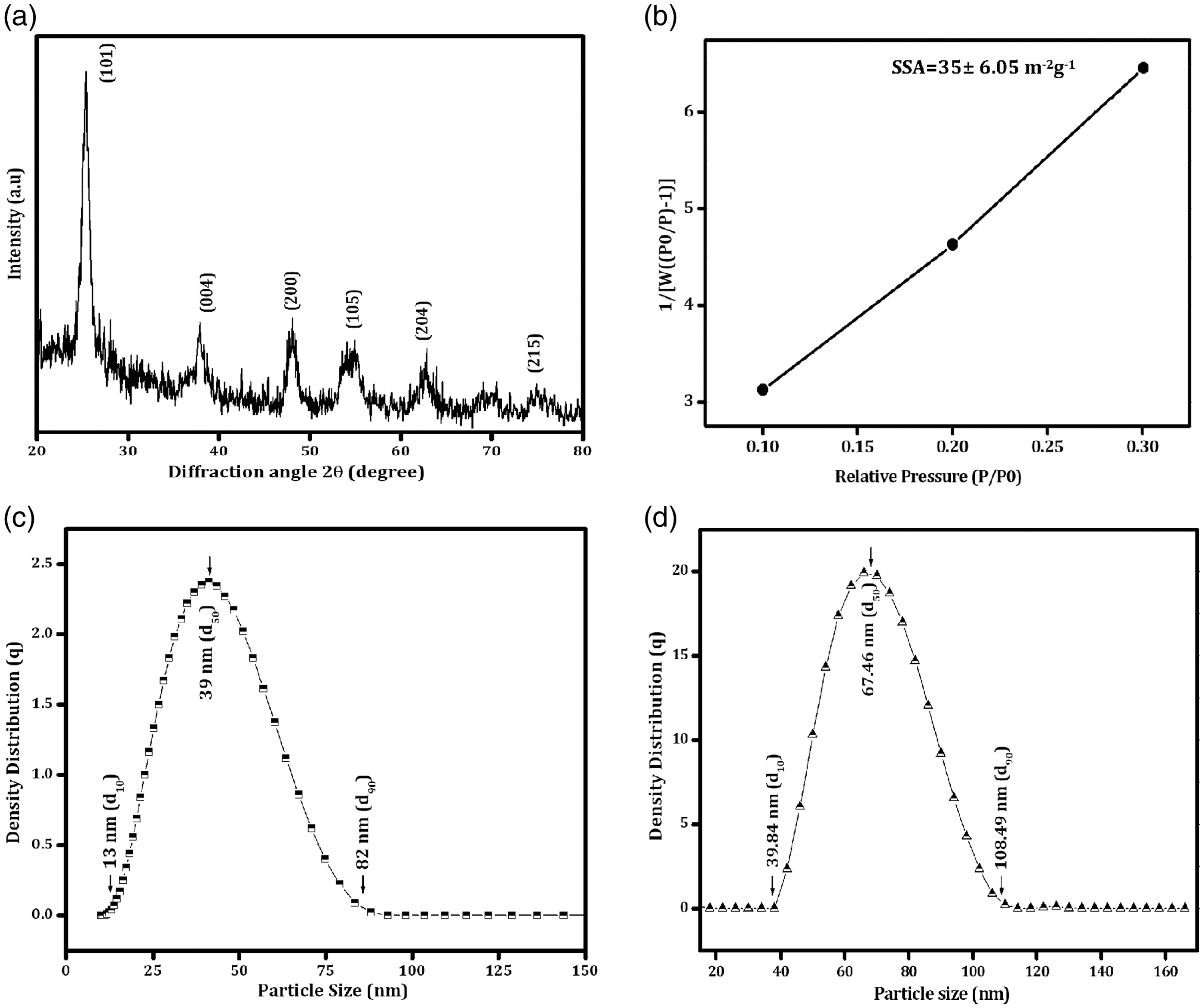
The particle size distribution of TiO2 nanoparticles and colloidal TiO2/SiO2 hybrid nanosol is shown in Figure 3(c) and (d). The particle size distribution of TiO2 nanoparticles is in the range from 13 to 82 nm (Figure 3(c)), while the same is from 40 to 108 nm (Figure 3(d)) in case of TiO2/SiO2 hybrid nanosol. However, the average particle size distribution of TiO2 and TiO2/SiO2 hybrid nanosol is, respectively, 39 (d50) and 67 (d50) nm.
Figure 4(a) and (b) shows the morphology of the prepared sample examined using SEM and TEM studies. The observed result indicates no interfacial gaps between the individual particles. The interconnected globular-shaped nanoparticles with an average particle size of ∼110 nm with a slight agglomeration are revealed from SEM image (Figure 4(a, i)). The EDS spectra confirm the purity of TiO2 nanoparticles as 100% (Figure 4(a, ii)). The elemental composition of the titanium and oxygen is 52.98 wt% and 47.02 wt%, respectively.
Morphological analysis: (a) surface morphology of TiO2 nanoparticles ((i) SEM image; (ii) EDS pattern), (b) TEM image ((i) TiO2 nanoparticles; (ii) Colloidal SiO2 nanoparticles; and (iii) Nano TiO2/SiO2 hybrid particles) and (c) SAED pattern ((i) TiO2 nanoparticles; (ii) Colloidal SiO2 nanoparticles; and (iii) Nano TiO2/SiO2 hybrid particles).
The primary nanostructured particles are evidently revealed from TEM image (Figure 4(b, i)). The particle size is relatively uniform with spherical morphology with an average diameter from 10 to 20 nm. The obtained concentric ring in SAED pattern (Figure 4(c), i)) shows the diffraction pattern that confirms the crystalline nature of the prepared TiO2 nanoparticles. Figure 4(b, ii and iii) shows the primary particle size (25 nm) of colloidal SiO2 nanoparticles and TiO2/SiO2 hybrid nanosol obtained from TEM studies. Figure 4(b, ii) reveals the spherical morphology of SiO2 nanoparticles with chain formations. TiO2 nanoparticles are adhered to SiO2 nanoparticles surface as shown in Figure 4(b, iii). The amorphous and polycrystalline nature of SiO2 nanoparticles and TiO2/SiO2 nanosol is evident from Figure 4(c, ii and iii, respectively).
Structural, morphological, and elemental analysis of coated and un-coated fabrics
The observed UC and TSC cotton fabric surfaces from XRD studies are shown in Figure 5(i). The observed diffraction peaks at 2 Characterization of fabrics: (i) XRD pattern and (ii) FTIR-ATR analysis of (a) UC and (b) TSC fabrics.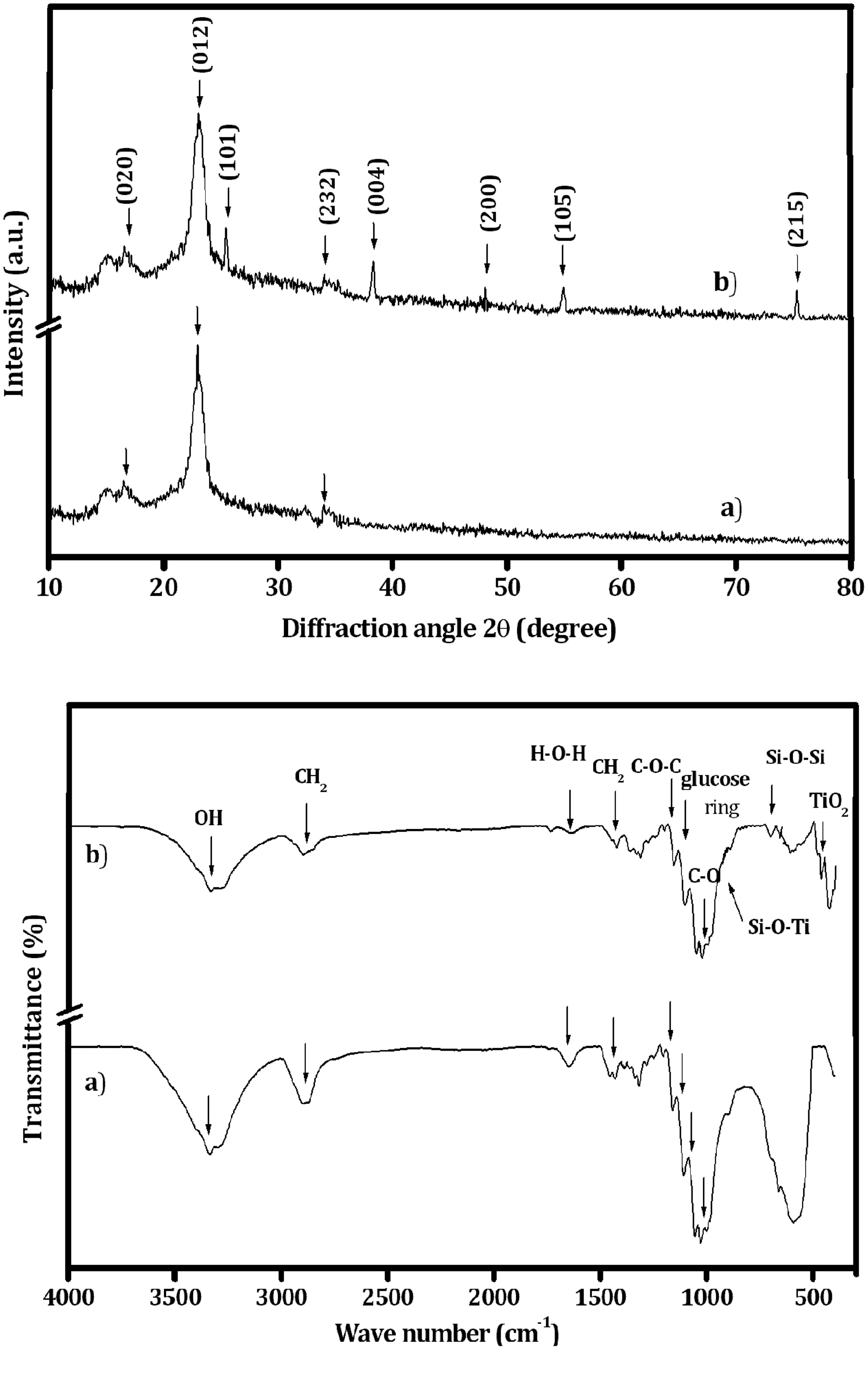
Figure 5(ii) shows the FTIR–ATR transmission spectra of UC and TSC cotton fabrics. The characteristic peaks observed at 3342 and 2883 cm−1 correspond to the stretching modes of O–H and –C–H, respectively. The bending mode of water molecules (H–O–H) is observed at 1642 cm−1 for all cotton fabrics as shown in Figure 5(ii, a). The asymmetric stretching of glucose ring is observed at 1107 cm−1, whereas the band observed at 1026 and 1166 cm−1 corresponds to the C–O bending mode of cellulose [17]. All the discussed peaks are shown for both UC and TSC fabrics. After the stretching and bending peaks of the band appeared at 795 cm−1 are related to Si–O–Si particles [19]. However, Figure 5(ii, b) shows the existence of TiO2 nanoparticles deposited on the surface of cotton fabrics that are clearly revealed at 524 cm−1. Additionally, the composite TiO2/SiO2 nanoparticles-coated fabric is observed at the peak at 948 cm−1, assigned to the Si–O–Ti vibration band [20] which confirms the variation of cotton fabric using TiO2 nanoparticles in the presence of silica network.
Further, the presence of nanoparticles coated on the fabric surface is confirmed through the observed SEM images with EDS spectra (Figure 6(a) and (b)). The TiO2 (39 nm) and TiO2/SiO2 (67 nm) nanocomposites with known average particle size are used for coating. The coating density of TiO2/SiO2 is reduced considerably on the fabrics which is evident from the obtained SEM images (Figure 6(b)). In addition, the presence of cellulose (carbon), TiO2, and SiO2 is shown from the EDS spectra that are given along with the SEM images. The nanoparticles are adhered to the fabric surface after subsequent washes; the same is revealed through the obtained SEM images of the fabrics after fifth and 10th washes. The SEM images of before washing of TSC fabric along with the EDS spectra are shown in Figure 6(b). The images show the durability of the nanoparticles coated on the cotton fabrics that are strongly adhered to the surface even after fifth and 10th washes. The wash durability (fastness) of the coating on the surface of fabrics is further confirmed through EDS measurements (Figure 6(c) and (d)) carried out after fifth and 10th washes. The observed results support the observation made through SEM studies (Figure 6). The percentage of nanoparticles decreases significantly in coating, which is high in the fifth wash than in the 10th wash; that is, the increase in number of washes leads to a decrease in coating removal from the surface of cotton. Thus, the presence of coating after subsequent washes is confirmed.
Morphological analysis: (i) SEM image and (ii) EDS pattern of (a) UC, (b) TSC fabrics before washing, (c) TSC fabrics after fifth washing, and (d) TSC fabrics after 10th washing.
Physical properties of un-coated and coated fabrics
Physical property of UC, SC, and TSC fabrics.

UC: un-coated fabrics; SC: fabrics coated with silica; TSC: fabrics coated with TiO2/SiO2 hybrid composite nanosol.
The longitudinal and transverse weaves are analyzed using tensile and tear strengths for UC and TSC fabrics. The breaking load of interstices (warp and weft yarn) values for tensile and tear strengths of TSC fabric are increased when compared to SC and UC fabrics before washing. The observation is in line with our earlier studies [21] that the overall tensile and tear strengths of the coated cotton fabric specimens are higher than those of the UC fabric. The coated nanoparticles entrap between the fiber structures of the yarn and hence, they restrict the moment of the fiber during elongation. In addition, the coated nanoparticles can also share the load which in turn enhances the tensile strength.
The observed whiteness index value of UC, SC, and TSC fabrics is shown in Table 1. The TSC fabric shows 113% of increased whiteness index value due to the high photocatalytic of TiO2/SiO2 nanoparticles [23] when compared to SC (107%) and UC fabrics. The whiteness index of TSC fabric after fifth and 10th washes is decreased 76% and 69%, respectively, while the SC fabric reveals 77% and 74%, respectively. The removal of TiO2/SiO2 nanoparticles on the surface of the fabric leads to the decrement in the index value after fifth and 10th washes.
The crease recovery angle of the TSC fabric is observed to be higher than that of the SC and UC fabrics. The above studies reveal that the nanoparticles are easily adhering to the cellulous fiber structures [5]. The crease recovery angle of TSC fabrics is increased to 123℃ when compared to UC fabric, which may be due to the nature of nanoparticles. After fifth and 10th washes, this is gradually reduced due to the removal coating on the fabric surface.
Functional properties of un-coated and coated fabrics
Figure 7(a) shows the wavelength (280–400 nm) opposed to transmission percentage of UC and TSC fabrics. The transmittance percentage for UV-A and UV-B is almost constant in the UC fabric and its transmittance values are equivalent to 76%. The blocking for both UV-A and UV-B radiations cannot be used for the UC fabric, which indicates the higher transmittance of UV radiation. Conversely, the reduction in transmittance is observed in TSC fabric, which is due to the blocking of UV-B and UV-A radiations. In coated fabrics, the percentage of blocking UV-B radiation is higher than that of UV-A. It is significant to note that the blocking rate of UV-A radiation in coated fabrics remains constant in the entire wavelength range from 315 to 400 nm whereas that of UV-B (280–315 nm) decreases gradually with an increase in the wavelength. In case of TSC fabric, the blocking rate of UV-B radiation is higher than that of the SC fabric [15]. In addition, the blocking rate of UV radiation for TSC fabric after fifth and 10th washes is slightly reduced than that of the unwashed cotton fabrics, which specify the presence of nanoparticle coating after fifth and 10th washes. The blocking rate of UV radiation for the coated fabric indicates that the subsequent washes lead to removal of only a small amount of coating components on the surface of fabrics.
(a) UV protection of coated and un-coated fabrics, (b) Flammability test (i) Post burn of UC and TSC fabric, (ii) During and after burning of TiO2/SiO2-coated fabrics followed by inclined (450) flame testing and (iii) Average value of flammability of UC and TSC fabrics.
UPF value for the UC, SC, and TSC of the fabric samples.

UC: un-coated fabrics; SC: fabrics coated with silica; TSC: fabrics coated with TiO2/SiO2 hybrid composite nanosol; UPF: UV protection factor.
Figure 7(b) shows the average results of five each specimens used to find out flammability of UC, SC, and TSC fabrics. The TSC fabric shows an excellent effect of burning performance than the UC fabric before the washing fastness. However, the decreased burning performance of the fabric is confirmed because of increased washing cycles. From these results, it is clear that the TiO2/SiO2 nanoparticles increase adhesion on cotton fabrics, thereby enhancing the wash durability of the fabric surface. During the flammability test, the UC fabric is completely burned when compared to the TSC fabric. However, the residual cotton is revealed in the TSC fabric (Figure 7(b, ii)). Thus, it confirms that the hybrid metal oxide nanoparticles coated on the cotton fabrics enhanced the functional properties of the fabrics. The exposure of the UC fabric to the flame before wash is easily extended to 6.3 s as reported earlier [17]. The flame retardancy of the TSC fabric is 21.4 s (Figure 7(b, ii)), which is better than that of the SC (11.3 s) [17] and UC fabrics (Figure 7(b, i)). It is clear from the above study that the TSC fabric enhances the fire resistance, thereby increasing the total burning time when compared with that of SC and UC fabrics. The TSC fabric retains its flame retardancy behavior about 70% even after 10 washes, while SC and UC fabrics retain their behavior 60% and 30%, respectively, only (Table 1 and Figure 7(b, iii)).
UC, SC, and TSC fabrics are evaluated for its antibacterial activity against Antibacterial activities of (i) Quantitative antibacterial properties of UC, SC, and TSC fabrics. UC: un-coated fabrics; SC: fabrics coated with silica; TSC: fabrics coated with TiO2/SiO2 hybrid composite nanosol.

The antibacterial effect on the unwashed fabrics is also studied. After fifth and 10th washes, the antibacterial activity rate of the nanoparticles-coated fabrics is decreased due to the removal of nanoparticles from the fabric surface. However, it is evident from the SEM image that some of the nanoparticles are grafted on the cotton surface during fifth and 10th washes. The antibacterial rate of the coated fabrics (TiO2/SiO2 and SiO2) with
Conclusion
In summary, TiO2 nanoparticles and TiO2/SiO2 hybrid nanosol are prepared using sol–gel method and then coated on the cotton fabrics using pad-dry-cure method. The nanoparticles deposited on the fabric surface show higher stability before and after washing, which is again confirmed through SEM–EDS analysis. The observed physical properties and fire resistance of the TSC fabric are better than those of the SC fabric. An excellent UV-protection ability is observed for the TSC fabric. In addition, an excellent antibacterial activity is examined in the TSC fabric against both
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Defence Research and Development Organisation (DRDO), New Delhi (ERIPR/ER/0905103/M/01/1279).
