Abstract
Ultrasonic modification was used as a simply-operated and efficient method for improving the hydrophilicity and cytocompatibility of polyglycolic acid (PGA) and poly lactic-co-glycolic acid (PLGA, lactide:glycolic acid (LA:GA) = 10:90) fibers, and maintaining the tensile property at the same time. The fibers were pre-treated ultrasonically by dipping in the mixed solution composed of absolute ethyl alcohol and polyphosphoric acid (PPA) (volume ratio 1:1) at 250 W ultrasonic power for 6 min. Scanning electron microscopy was used to observe the surface morphology of PGA and PLGA fiber before and after modification. Fourier transform infrared spectroscopy was used to investigate the change of fiber chemical composition. X-ray diffraction and differential scanning calorimetry analysis showed that the crystalline degree of modified PGA and PLGA fibers decreased. The results of tensile test indicated that compared to that before modification, the breaking strength of modified PLGA increased, while the breaking strength of PGA fiber remained unchanged. The water contact angle of modified fiber was lower than that of unmodified fiber, showing higher hydrophilicity. The cell proliferation assay indicated that fibroblast cells attached and proliferated better on the modified fiber, which means the modified fibers possess good cytocompatibility. These results suggested that ultrasonic modification is an easy-operated and efficient method and the modified PGA and PLGA fibers could be useful in the biomedical textiles field.
Keywords
Introduction
Polyglycolic acid (PGA) is biodegradable polyester-like macromolecule material. It has many excellent performances, such as favorable fiber-forming ability, biodegradability, nontoxic, and so on. It has been treated as one of the most utility biodegradable polymers [1]. Poly lactic-co-glycolic acid (PLGA) is a copolymer of lactide (LA) and glycolic acid (GA). Besides the similar good performances to PGA, PLGA has different degradation rate and physical properties by changing the molar ratio of LA and GA [2]. PGA and PLGA are widely used as surgical sutures, drug delivery devices and so on [3,4]. In addition, the excellent fiber-forming and spinning property of PGA and PLGA means they can be spun, weaved, knitted, and braided. In short, they can be used as biomedical textile.
Biomedical textile is a new interdiscipline which combines classical material, textile and biomedicine. Up to now, biomedical textile has used in grafting product, external therapy product, hygiene product, and protective equipment [5]. Especially, the application of biomedical textile in tissue engineering field becomes a new research hotspot [6,7]. As excellent biomedical textile materials, PGA and PLGA have been widely used in various tissue engineering scaffolds. However, being lack of specific cellular and molecular interacting sites on their surfaces make them behave poorly in promoting cells adhesion, proliferation, differentiation, and mineralization [8]. Therefore, based on various applications, improving the specific cellular cytocompatibility of PGA and PLGA is very important and necessary, and modification is an efficient method to attain the objective. Sodium hydroxide is used to surface hydrolyze PGA mesh, which enhances vascular smooth muscle cells adhesion by transforming the ester bonds to carboxylic acid and hydroxyl groups, but the breaking strength drops sharply because of strong hydrolysis [9]. Two cell adhesive peptides, Arg-Gly-Asp(RGD) or Tyr-Ile-Gly-Ser-Arg(YIGSR), are used to strengthen the efficacy of PLGA skeletal muscle tissue engineering for guiding cell arrangement to form parallel myotubes. Bone marrow concentrate has been combined on PGA scaffolds can supplement to supply the bone marrow stimulation in rabbit articular cartilage repair [10]. Peptide interfacial biomaterials have been used to alter the surface of PGA and effectively promote endothelial cell adhesion and spreading [11]. However, few tests of mechanical properties have been done in this paper [12]. In conclusion, these biomaterial modifications, including both chemical and physical modifications, had an actively affect on surface protein adsorption and cell interaction [13–15]. However, the mechanical properties, which are very important as tissue engineering scaffolds, are weakened, or not mentioned. If tissue engineering scaffolds are lack in mechanical properties, they cannot provide enough supporting force in organ regenerate process.
In this study, we aim to improve the properties of PGA and PLGA (LA:GA = 10:90) fibers, so that they can be applied to braid nerve regeneration scaffolds in our next study. Whether the tensile performance of fibers maintain well after modification is very important, because it has a considerable influence on mechanical properties of nerve scaffold. Meanwhile, as a biomedical material, the surface hydrophilicity influences the adhesion of nerve cells and delivery of neurotrophin. For achieving our goals, a modification method is needed not only to enhance the hydrophilicity and cytocompatibility of PGA and PLGA fibers, but also to maintain the mechanical performance. Ultrasonic is a considerable potential method for material surface modification and has been used on many kinds of fibers: it has been used to improve the wettability of wool fibers [16], and has enhanced the hydrophilicity of ultrahigh molecular weight polyethylene (UHMWPE) fibers with the help of chromic acid solution [17]. However, ultrasonic modification method has not been applied on PGA and PLGA fiber. Furthermore, PGA and PLGA are degradable material. Water or strong oxidizing medium, which has been used in other ultrasonic modification literatures, cannot be used on PGA and PLGA because this will cause a big loss of mechanical performance. In this study, we attempted for the first time using ultrasonic to modify PGA and PLGA fibers, with the help of mixed absolute ethyl alcohol and polyphosphoric acid (PPA) solution as appropriate medium. Furthermore, the morphology, chemical component, structure, mechanical property, hydrophilicity and cytocompatibility before and after modification are compared and discussed.
Materials and methods
Materials
PGA and PLGA (molar ratio LA:GA = 10:90) fibers were provided by Shanghai Tianqing Biomaterial Co., Ltd, China. PGA fiber is 2.95 dtex and PLGA fiber is 3.66 dtex. Both fibers are taken from multifilaments. All chemicals were analytically pure and used without further purification. Human fibroblast cells were supplied by Shanghai Jurui Biomaterials Co., Ltd, China.
Ultrasonic modification
Absolute ethyl alcohol, as diluent and dispersant, and PPA were mixed according to 1:1 volume ratio. Because the relative density of alcohol (0.79 g/cm3) is smaller than PPA (2.1 g/cm3), the mixed solution is divided into two layers. So the mixture was pre-treated with 250 W ultrasonic power and a frequency of 40 Hz for 30 min for mixing uniformly. PGA and PLGA fibers, in the form of loose bundles, were dipped in the pre-treated mixture without tension, and treated with ultrasonic machine (JAC-2010P, Shanghai Junyi, China) with 250 W ultrasonic power and a frequency of 40 Hz for 6 min. Treated fibers were taken out and cleaned with deionized water for three times and freeze-dried in vacuum freeze-drying dryer (FD-1 A-50, Shanghai Bilang, China) for 3 h.
Characterizations
The surface morphology of fibers before and after ultrasonic modification were examined by scanning electron microscopy (SEM; Hitachi, S-4800, Japan). To check the chemical structure of PGA and PLGA fibers, fibers were detected using a Fourier transform infrared spectroscopy (FTIR) spectrometer (NEXUS 670, Thermo Nicolet Corporation, US) in the range of 400–4000 cm−1 with a resolution of 1 cm−1. For analyzing the structure change of fibers before and after modification, X-ray diffraction (XRD; D/max-2550 PC, Rigaku, JP) was used to get the crystallinity. A differential scanning calorimetry (DSC, Pyris-1, PerkinElmer) was used to investigate the thermal property change of fibers. All the measurements were carried out under nitrogen atmosphere.
Tensile performance in degradation
Modified and unmodified PGA and PLGA fibers were immersed in phosphate-buffered saline (PBS, pH 7.4) and placed in the incubators (HH.CP-T, Shanghai Sanxin, China). The breaking strength of fibers was tested with single fiber strength tester (LLY06E, Laizhou electron instrument Co., Ltd, China) at day 1, day 3, day 5, and day 7 (
Water contact angle measurements
For testing the varying hydrophilicity of PGA and PLGA fibers after modification, a contact angle goniometer (JC2000A, Shanghai Zhongchen, China) was used by the sessile drop technique under ambient laboratory conditions and gauge length was 5 cm.
Cell proliferation assay
According to GB/T 16886.12-2000 [18], for comparing the cytocompatibility of unmodified and modified PGA and PLGA fibers, each sample was weighed 20 mg, put into plastic injector, and pressed into round shapes with the diameter 8.5 mm. The sheets were sterilized by immersing in 75% (w/v) alcohol for 1 h three times, washed three times with PBS (pH 7.4), and immersed in 10% (w/v) fetal bovine serum (FBS) and Dulbecco’s modified eagle’s medium (DMEM) for 24 h. Then, sheets were placed individually into a six-well culture plate, seeded with human fibroblast cells at a density of 2.0 × 104 cells/well and the culture medium was replenished every other day.
Cell proliferation was assessed with cell counting kit-8 (CCK-8, Dojindo Melecular Technologies, Inc, Kumamoto, Japan). After culturing for different times, the sheets containing cells were rinsed with PBS for three times, immersed in dilution CCK-8 reagent (volume ratio 1:10), and then put in the 5% CO2 incubator at 37℃. After 2 h, 100 µL medium with CCK-8 solution was aliquoted into a 96-well culture plate for optical absorption measurement at 450 nm wavelength on an enzyme-linked immunosorbent assay instrument (M2E, Melecular Device, USA). The same volume of culture medium and CCK-8 reagent without cells was also incubated as the background. Optical density values were calculated from triplicates of each group. Cell morphology on the fibers was observed by SEM.
Statistical analysis
Statistical analysis was performed using Origin (Origin lab, USA). Data results were averaged and expressed as means ± standard deviation (SD). Statistical differences were determined by one way ANOVA. The statistical significance was assessed by
Results and discussion
Characterization of the ultrasonic modified fiber
Figure 1 shows the SEM micrographs of PGA and PLGA fibers before and after ultrasonic modification. The long groove on the left side was formed during the course of fiber spinning and is independent of this paper. Compared with unmodified PGA fiber, modified fiber had more protrusion on its surface, even appeared some microgroove and exfoliation. The surface of modified PLGA fiber is similar to PGA fiber with more obvious protrusion and microgroove, which means ultrasonic modification had erosive effect on the surface of PGA and PLGA fiber.
SEM micrographs of surfaces of (a) PGA fiber, (b) ultrasonic modified PGA fiber, (c) PLGA fiber, and (d) ultrasonic modified PLGA fiber. PGA: polyglycolic acid; PLGA: poly lactic-co-glycolic acid; SEM: scanning electron microscopy.
As shown in Figure 2, the FTIR spectra of four kinds of samples are very similar. However, compared with flat curve of unmodified PGA at 3414.9 cm−1 region, a diffuse absorption band appeared at the same region on modified PGA because of hydroxyl dimer. It may be due to partial hydrolysis of ester bonds which caused terminal hydroxyl and carboxyl increased and formed intermolecular hydrogen bond. The polar groups, hydroxyl and carboxyl, have affinity with water molecules and cells, so the change is beneficial to the hydrophilicity of PGA fiber. In unmodified PLGA spectra, a wide absorption band appeared at 3385.5 cm−1 region, which is due to the intermolecular hydrogen bond of νOH. However, after ultrasonic modification, this absorption peak disappeared, and a new peak, which represent intermolecular hydrogen bond of νOH dimer, appeared at 3491.1 cm−1 region instead. This result implies the decrease of intermolecular hydrogen bond and the increase of intermolecular distance after modification. Considering the absolute superiority of GA component, ester bonds of PLGA fiber may also partly hydrolyze after modification. The results may be due to the sound cavitation of ultrasonic modification. On one hand sound cavitation can induce hydroxyl free radical in the mixed solution, on the other hand the effect of sound cavitation promotes the hydroxyl free radical in solution to react with fiber surface. It is beneficial for the hydrolysis of ester bonds and the increase of polar groups on fiber surface.
FTIR spectra of PGA and PLGA fiber before and after ultrasonic modification. FTIR: Fourier transform infrared spectroscopy; PGA: polyglycolic acid; PLGA: poly lactic-co-glycolic acid.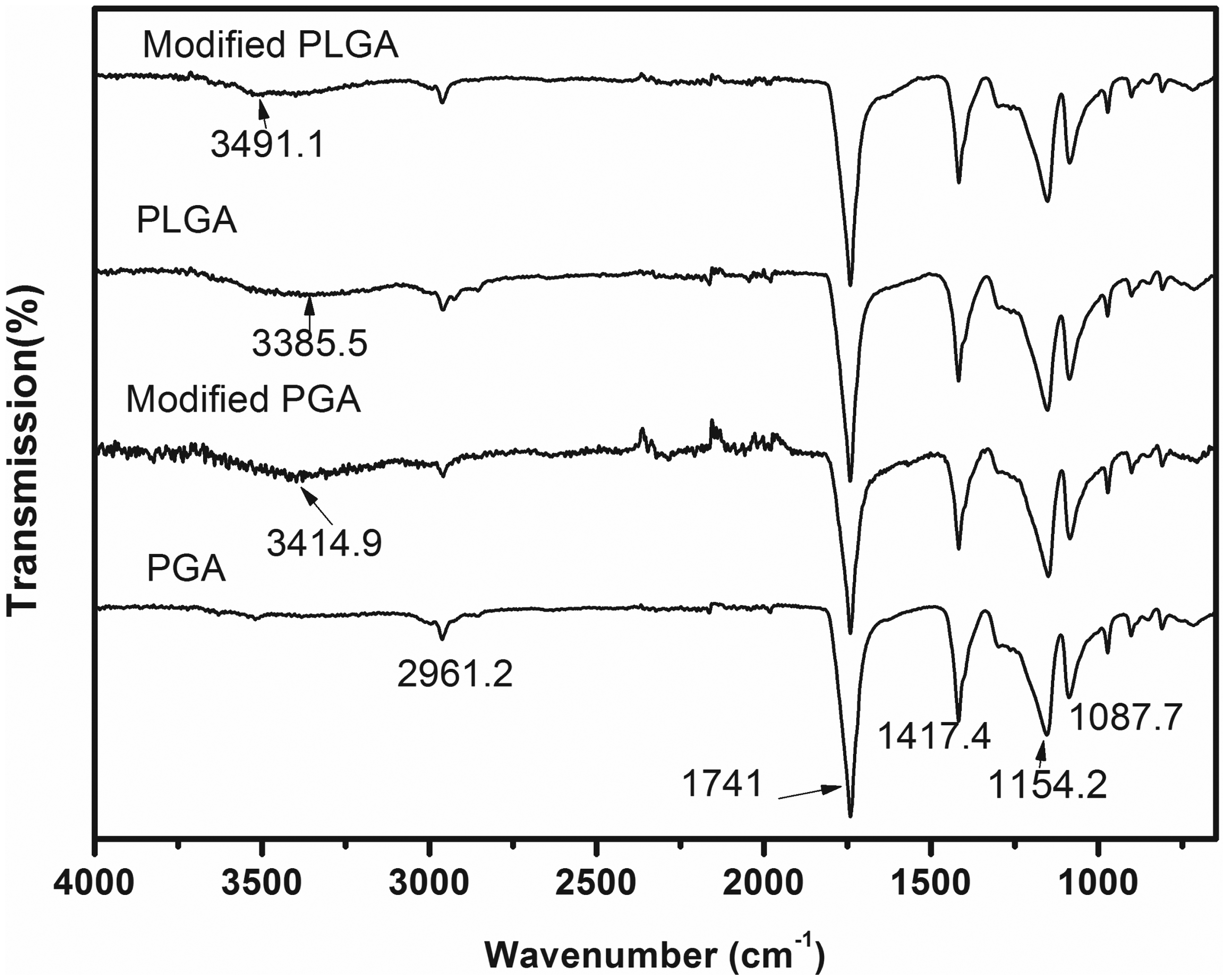
The XRD patterns of PGA and PLGA fibers before and after ultrasonic modification have been shown in Figure 3. The unmodified PGA fiber showed two peaks at 2θ of around 22.32° and 28.64°, which indicates that the PGA fiber had crystalline region. Compared with unmodified PGA, the peaks position (22.34°, 28.78°) of modified PGA had almost no change. However, the peak value of modified PGA fiber dropped obviously. The similar situation appeared in XRD patterns of unmodified and modified PLGA fiber. Furthermore, the drop in PLGA peak value is larger than PGA. It implies that after ultrasonic modification, the crystallinity of PGA and PLGA fibers, especially PLGA fibers drops sharply. It is confirmed with the crystallinity results: modified PGA fiber has a lower crystallinity (52.08%) than unmodified one (57.25%), and the crystallinity of PLAG dropped from 45.05% to 25.52%. It means that ultrasonic modification could decrease the crystallinity and increase amorphous state of PGA and PLGA fiber, especially PLGA fiber. The FTIR spectra of PLGA also confirm the increase of amorphous state. This change is beneficial to water molecules entering into fibers.
XRD patterns of PGA and PLGA fiber before and after ultrasonic modification. PGA: polyglycolic acid; PLGA: poly lactic-co-glycolic acid; XRD: X-ray diffraction.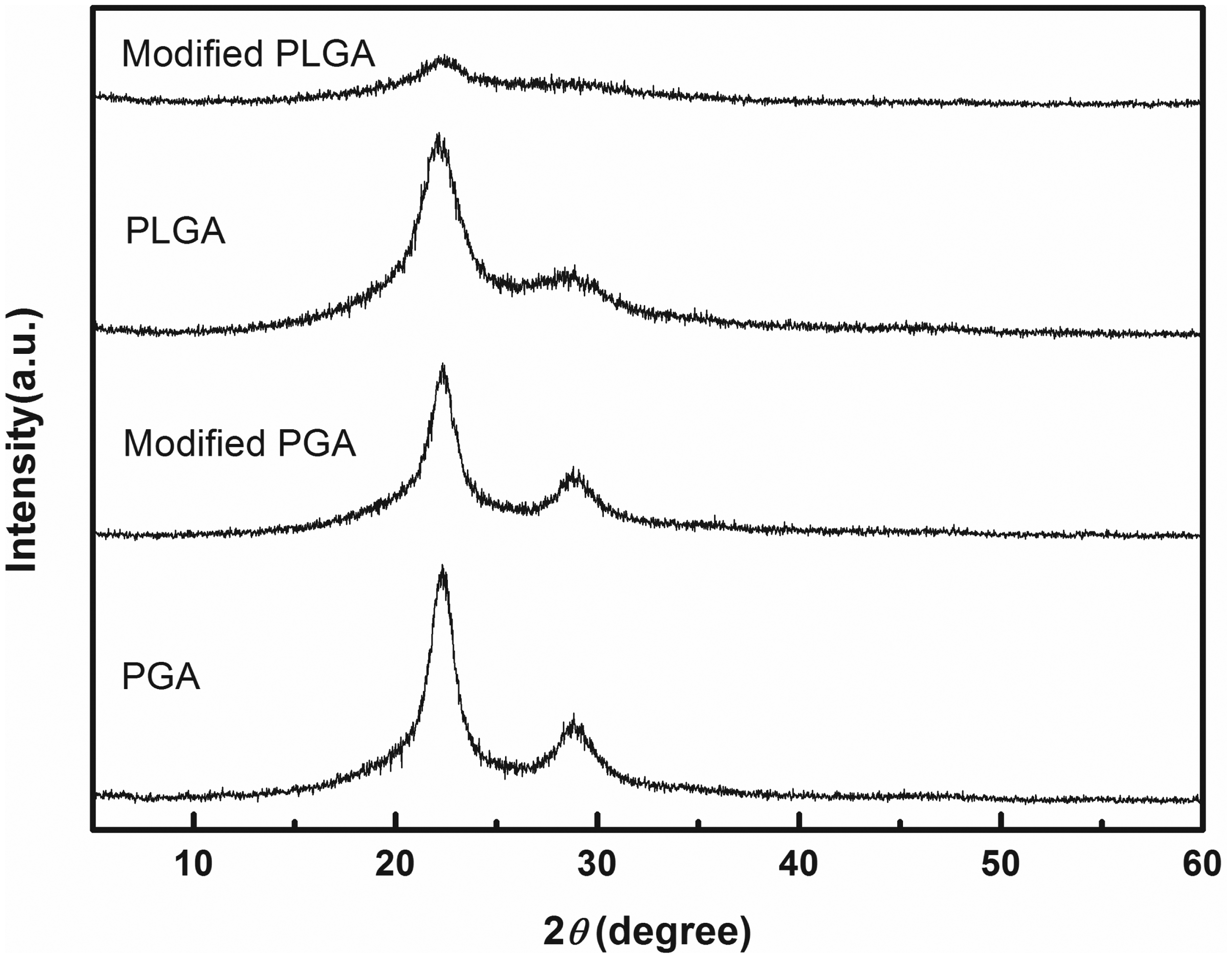
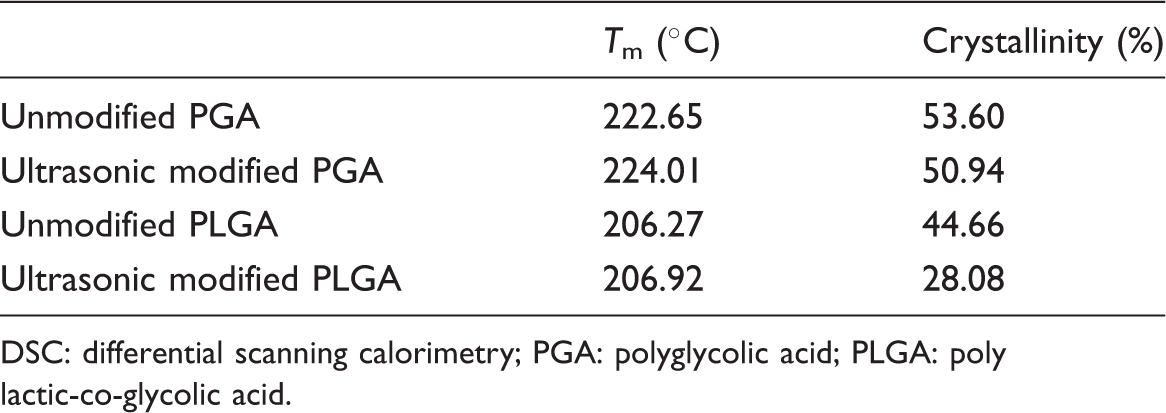
DSC: differential scanning calorimetry; PGA: polyglycolic acid; PLGA: poly lactic-co-glycolic acid.
The effect of ultrasonic modification is mainly due to its cavitation on solid–liquid interface. A very large amount of little bubbles and holes appears in solution by cavitation, sharply change and oscillate. The larger the solution viscosity is, the weaker the cavitation is; the proper addition can decrease the solution viscosity of PPA and increase the cavitation. Cavitation promotes the uniform reaction between fiber and PPA, which produces more polar groups, etches fiber surface, and rearranges fiber microstructure to a certain extent.
Tensile performance in degradation
Tensile performance of fibers is very important when fibers are braiding into nerve scaffold and transplanting in vivo. Figure 4(a) and (b), respectively, shows the breaking strength of PGA and PLGA fibers in different degradation periods. Before degradation, the breaking strength of ultrasonic modified PGA was higher than unmodified one, and the change was statistically significant ( Tensile performance in degradation before and after ultrasonic modification of (a) breaking strength of PGA group, (b) breaking strength of PLGA group, (c) elongation of PGA group, and (d) elongation of PLGA group. Data were mean ± SD, 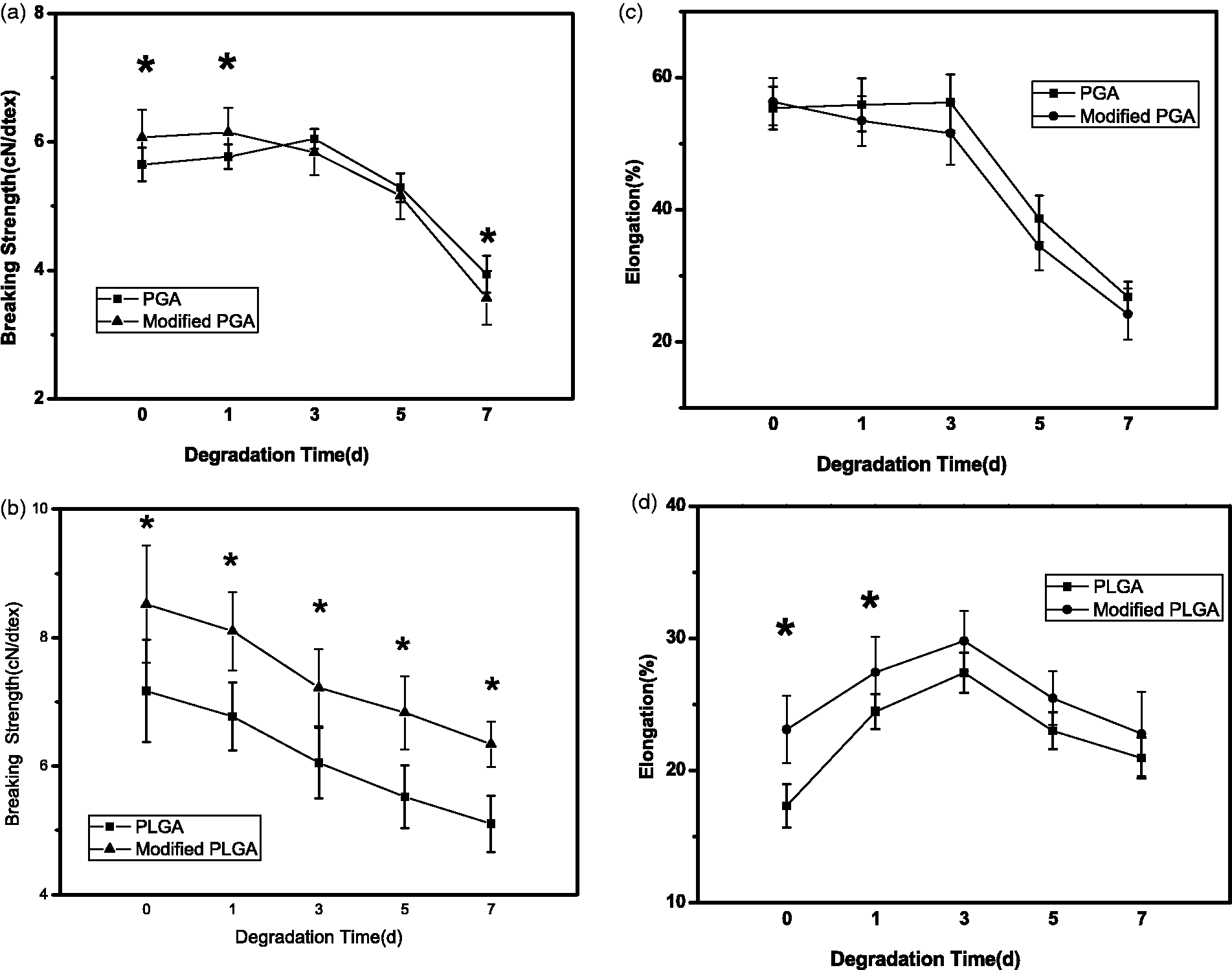
The change of breaking strength of PLGA group in different degradation periods is shown in Figure 4(b). Before degradation, modified PLGA had higher breaking strength than unmodified one (
Figure 4(c) shows the elongation of PGA fiber in different degradation periods. Before degradation, elongation of modified PGA was only a little larger than unmodified one. In the first three days, elongation of modified PGA fiber decreased slowly while elongation of unmodified PGA fiber almost had no change. The reason is as same as the discussion in breaking strength because elongation is related with microstructure of fiber, too. After three days, elongation of both fibers decreased sharply because of ester bond hydrolysis and fiber structure broken. Figure 4(d) shows the elongation of PLGA fiber in different degradation periods. The elongation of modified PLGA fiber was always higher than unmodified one in the whole course, and they increased at the first and similarly decreased after three days. Combined with the result of breaking strength, it can infer that swelling makes the distance between molecular chains farther and makes them slipping more easily. Certainly, molecular interactions of PLGA are originally weaker than PGA because PLGA has less polar groups. On the whole, the elongation change of both modified PGA and PLGA fiber is in reasonable range.
The tensile decrease rate after 7 days degradation of unmodified and ultrasonic modified PGA and PLGA fiber.

PGA: polyglycolic acid; PLGA: poly lactic-co-glycolic acid.
Hydrophilicity of the ultrasonic modified PGA and PLGA fibers
The water contact angles of unmodified and ultrasonic modified PGA and PLGA fiber surface; data were mean ± SD,

PGA: polyglycolic acid; PLGA: poly lactic-co-glycolic acid.
Cytocompatibility of ultrasonic modified PGA and PLGA fiber
The first week of the nerve regenerating is the main period of fibroblast cell migration and proliferation, and concerned with the next regenerating stage. Fibroblast cells spread on the fibers and formed cell membrane among fibers seven days after seeding (Figure 5). Compared with unmodified PGA and PLGA fibers (Figure.5(a) and (c)), fibroblasts cells spread more widely on ultrasonic modified fibers (Figure.5(b) and (d)). Cells were found to integrate well in the fiber aggregates. It seems fibroblasts grow better on modified fibers.
SEM images of fibroblasts growing on (a) PGA fiber, (b) ultrasonic modified PGA fiber, (c) PLGA fiber, and (d) ultrasonic modified PGA fiber. PGA: polyglycolic acid; PLGA: poly lactic-co-glycolic acid; SEM: scanning electron microscopy.
Figure 6 shows the initial attachment and viability of fibroblasts during the culture period. Overall, the amount of cells on modified PGA fiber was always more than the unmodified PGA during the culture course (Figure 6(a)). There is significant difference between unmodified and modified PGA on day 1 ( Fibroblasts proliferation of (a) unmodified and modified PGA fibers and (b) unmodified and modified PLGA fibers. Data were mean ± SD, 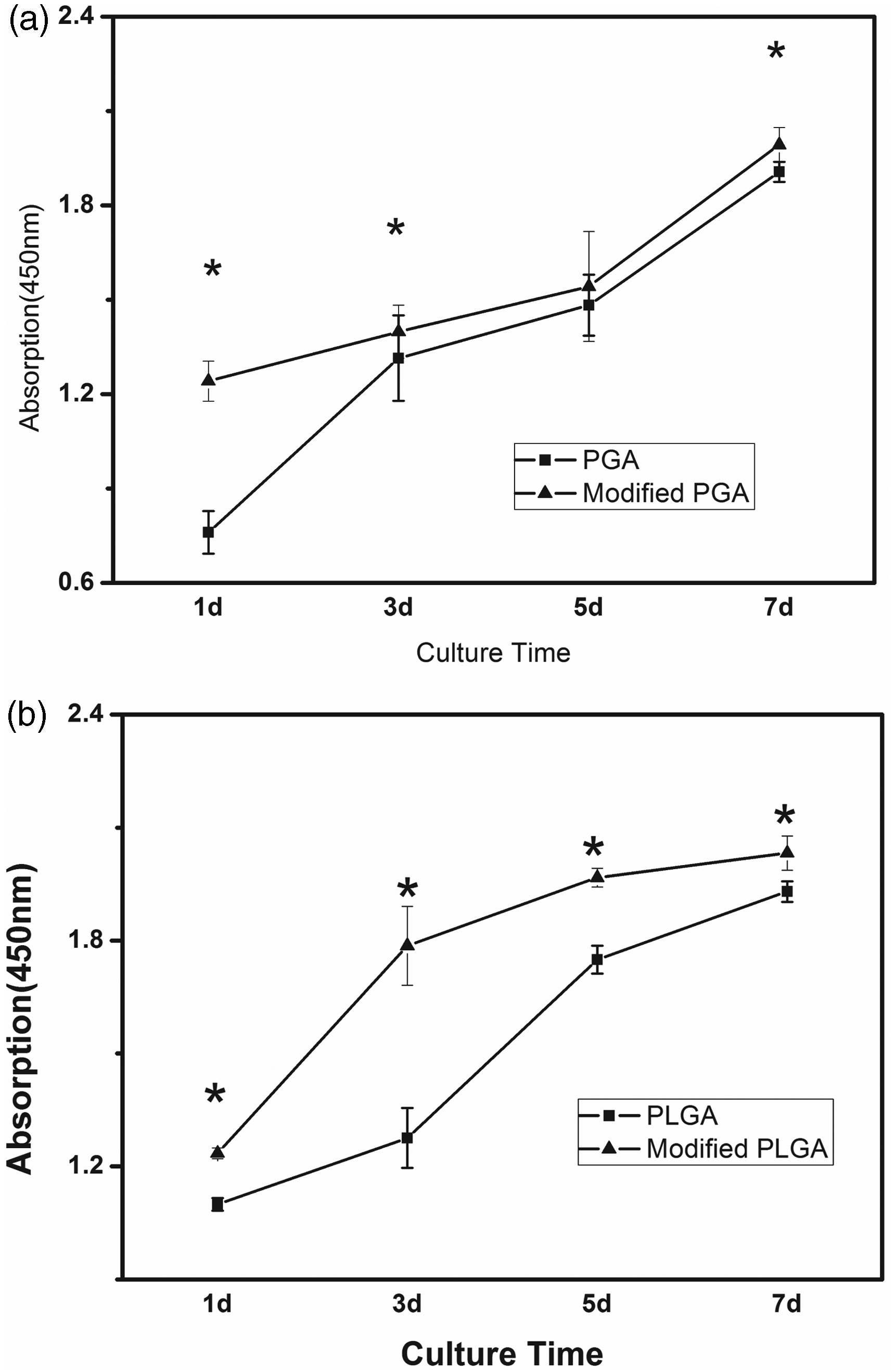
Conclusions
In this paper, ultrasonic modification method is used to enhance the hydrophilicity and cytocompatibility of PGA and PLGA (LG:GA = 10:90) fibers, while maintaining the breaking strength. The fibers were pre-treated ultrasonically by dipping in the mixed solution composed of absolute ethyl alcohol and PPA (volume ratio 1:1) at 250 W ultrasonic power for 6 min for modification. After ultrasonic modification the surface of PGA and PLGA fibers became rougher with obvious protrusion and microgroove, and the surface molecule hydrolyzed slightly and produced more polar groups. Through the investigation of DSC and X-ray, the decrease of crystallinity has been observed, which indicates the increase of amorphous state. The breaking strength in degradation of modified PGA and PLGA fibers is well maintained, and hydrophilicity is improved. Furthermore, fibroblasts grow better on modified fibers, the cells adhesion on the PGA and PLGA fibers is effectively enhanced. These results suggest that ultrasonic modification is an easy-operated and efficient method for PGA and PLGA fibers to achieve requirements of comprehensive performance, so they can be applied to braid nerve regeneration scaffolds in our next study.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shanghai Science and Technical Committee (project number 10411953300); State Key Laboratory for Modification of Chemical fibers and Polymer Materials (project number LZ0902); and Minhang District Industry-study-research Cooperation (project number 2012MH202).
