Abstract
The relative rate of degradation of poly(ethylene terephthalate) geotextiles in various monovalent alkali hydroxides was studied in both aqueous and alcoholic systems. In an aqueous system, the reaction of all three alkalis on poly(ethylene terephthalate) geotextiles is restricted to the surface since tenacity loss was insignificant and no surface cracks was noticed, whereas in an alcoholic system, significant loss in tenacity occurred due to the formation of surface crack. Furthermore, the relative rate of reactivity of metal hydroxides in aqueous and alkaline media was temperature dependent. However, in equimolar concentration, at 80℃, the relative rate in an aqueous system is of the following order: LiOH > NaOH > KOH in the ratio of 1.65:1.1:1.0. Above 80℃, the reaction is reversed, as aqueous KOH reacts faster than aqueous NaOH. Two different activation energies were found for aqueous KOH at a threshold temperature of 80℃. Furthermore, in heterogeneous systems using dimethyl terephthalate, aqueous KOH was slightly faster than aqueous NaOH in the temperature range of 70–80℃. In an alcoholic system, KOH is almost 1.5 times faster than NaOH at 20℃ and at 60℃.
Keywords
Introduction
Poly(ethylene terephthalate) (PET) has been widely used in the field of geosynthetics because of its high strength, low creep and ease of manipulation. The major established areas of applications are landfills [1], soil reinforcement [2] and erosion control filter [3]. However, PET is susceptible to hydrolysis in both acidic and alkaline environments. Hydrolysis in alkaline conditions is much more severe than in acidic conditions [4].
PET in geosynthetic applications may encounter many hazardous chemicals depending on the nature of application. For example, in municipal solid waste landfills and in soil reinforcement application, PET encounters acidic or alkaline soil [5]. Furthermore, in alkaline soil, many alkali metal ions, such as Ca+2, Na+1 and K+1, could be present. Recently, the use of PET geosynthetics has been extended to many mining operations, where the fibre is exposed to harsh mining chemicals [6,7]. Although the nature (aqueous or alcoholic) and severity of the chemical is dependent on the types of mining operations, various transition metals (Na+, Ca+2, K+, Mg+2) [8] and very high alkaline concentrations with many cations are found in the mining chemicals [6,9] as well as in lagoon systems [10]. Consequently, degradation of polyethylene geomembranes in aqueous transition metals (FeCl2, MnSO4 and CuCl2) was investigated by van Langenhove [11] and he found that the degradation was accelerated by a factor of 10, or more, depending on the metal. At concentration of up to 2%, the greatest decrease in the oxidative induction time was observed in the presence of Cu, followed by Fe and Mn.
However, for polyester, very few but contradictory reports were published about the reactivity of various metal hydroxides on either apparel- or geotextile-grade PET. Data from Imperial Chemical Industries [12] showed that at 100℃, the hydrolysis reaction of aqueous Ca(OH)2 is 13 times faster than aqueous NaOH on apparel-grade polyester, whereas Kish and Nouri [13] showed that aqueous Ca(OH)2 is only four times faster than the aqueous NaOH. Halse et al. [14,15] found that for geotextile-grade polyester, the aqueous NaOH reacts faster than aqueous Ca(OH)2. However, the relative rate depends on the fabric construction, as it has been reported that after 120 days/25℃/pH = 12, PET-needled, non-woven, heavy weight geotextile fabric lost 30% and 53% strength and PET-heatset, non-woven geotextile fabric lost 20% and 31% strength in aqueous Ca(OH)2 and NaOH, respectively [14,15]. No specific information was provided regarding the types of strength (tensile, tearing and bursting) investigated. Similar contradictory reports were published regarding the effect of high alkalinity on the geosynthetic clay liner materials [6,16]. However, direct comparison of rate of hydrolysis between the apparel and geotextile grade is difficult as most of the research articles that investigated the hydrolysis of geotextile PET failed to report the basic properties of fibres, such as tenacity, fibre type, tex and number of filament.
All the above-mentioned studies have been carried out in the aqueous–alkaline system and compared reactivity of monovalent alkali hydroxide with divalent alkali hydroxide. No studies could be found that investigated the relative effect of various monovalent hydroxides on the polyester geotextiles in aqueous systems. Furthermore, no work could be found on the relative reactivity of alcoholic alkalis on PET geotextiles. Alcohol content in alkaline leachates from landfills [17] and mining operations [18] have been reported. As the application of PET geosynthetic materials expands to various mining and lagoon operations that create different leachates, the effect of various hydroxides in both aqueous and alcoholic systems on PET geosynthetics may affect their long-term performance. The alcoholic environment in lagoons is very challenging to the long-term durability of polyester geosynthetics [19].
In this study, we have investigated the relative reactivity of three monovalent alkali metal hydroxides, namely NaOH, LiOH and KOH, in aqueous system on geotextile-grade PET. However, detailed investigation was carried out on the relative reactivity of NaOH and KOH in both aqueous and alcoholic systems. Furthermore, the relative reactivity of NaOH and KOH in aqueous system was confirmed in the heterogeneous system using dimethyl terephthalate (DMT). The results of this investigation would be useful to understand the long-term durability of PET geotextiles used in various mining and lagoon operations.
Materials
PET materials
Geotextile-grade high-tenacity bright (≈0.1% TiO2) PET, 1100 dtex/200f, flat multifilament yarn was used. The yarn was obtained from HOECHST (Trevira, type 730) and the glass transition temperature (Tg) is 80℃ and the melting temperature is 255℃. For ease of handling during chemical treatment, 1 × 1 weft knitted rib fabric was prepared using a V-bed universal knitting machine with needle gauge of 7 and stitch-cam setting of 12.
Preparation of materials for hydrolysis treatment
PET fabric preparation for hydrolysis treatment
(a) Grey fabric. This fabric was used for hydrolysis treatment without any prior preparatory (scouring) treatment. (b) Scoured fabric. The grey knitted fabric was scoured to remove fats, oil and wax, which is a common practice in textile industry. The scouring process was carried out in a rotary wash wheel with 550 ml of scoured solution in the dyeing canister using a rotational speed of 40 ± 2 rev/min. No standard test methods are available for scouring. The scouring process was carried out using the following method, which is a standard method used in textile industry—Na2CO3: 2 g/l; sandosin: 2 g/l; temperature: 60℃ and time: 30 min.
Preparation of DMT disc
DMT is used to understand the hydrolysis reaction in a heterogeneous system that is found in geocomposites (geomembrane+clay). The DMT disc was prepared from a fine powder, produced with a mortar and pestle, using the ‘Apex Hydraulic Jack’, a device used for the preparation of potassium bromide discs for infrared spectra. A total of 0.5 g of DMT powder was weighed out and placed inside the 13 mm evacuable pellet die. The pressure of the machine was kept as 7110 kg and the time was 5 min.
Chemicals
The Na2CO3 (99.5%) and LiOH (99%) were supplied by Vickers while aqueous and alcoholic sodium hydroxides (AnalarR) and potassium hydroxides (AnalarR) were supplied by Aldrich. Sandosin NI (99%) was obtained from Sandoz Chemicals and DMT (99%) was supplied by BDH.
Methods
Hydrolysis treatment
All the hydrolysis experiments were carried out in a rotary dyeing machine (ROTADYER) in a water bath using plastic dyeing tube for hydrolysis temperature of up to 80℃ and in an oil bath using stainless steel dyeing tube for hydrolysis temperature of 90℃ and above. The samples were treated with 100 ml alkaline solution of different concentrations under a range of temperatures for a predetermined period of time. After the completion of each treatment, the alkaline-treated samples were washed with distilled water followed by acid treatment to neutralise the remaining alkali in the sample. The sample was dried for 48 h at room temperature
Analytical techniques
Gravimetric method
The weight loss (%) of the conditioned (65% relative humidity, 20℃) samples was measured using the following formula
Mechanical properties
Mechanical properties of the individual filaments taken from the hydrolysed yarns were determined using Instron-1026 Universal Tester. The cross-head speed was 50 mm/min and the load cell was 500 g with a full scale load of 100 g, according to ASTM D 2256 [20]. The yarn samples were first glued on cardboard squares with an inner dimension of 20 mm. The framed fibres were then clamped in the machine and testing was done. Before testing, all yarns were conditioned in a standard atmosphere of 20 ± 2℃ and 65 ± 2% relative humidity.
Photographs
The photographs of DMT samples were taken at the University of Leeds using standard camera.
Scanning electron microscopy
To obtain the scanning electron microscopy (SEM) microphotograph, a Cambridge Stereoscan S360 was used, operating at an accelerating voltage of 20 kV and photographs were taken by a camera directly attached to the instrument. The fibre samples were coated with thin gold (20 nm) using a Polaron E500 sputter coating unit at a pressure of 13.3 Pa. The samples were kept under this condition for 6 min with air current flow of 18 mA.
Results and discussion
Reactivity of NaOH, KOH and LiOH: Aqueous system
Weight loss (%)
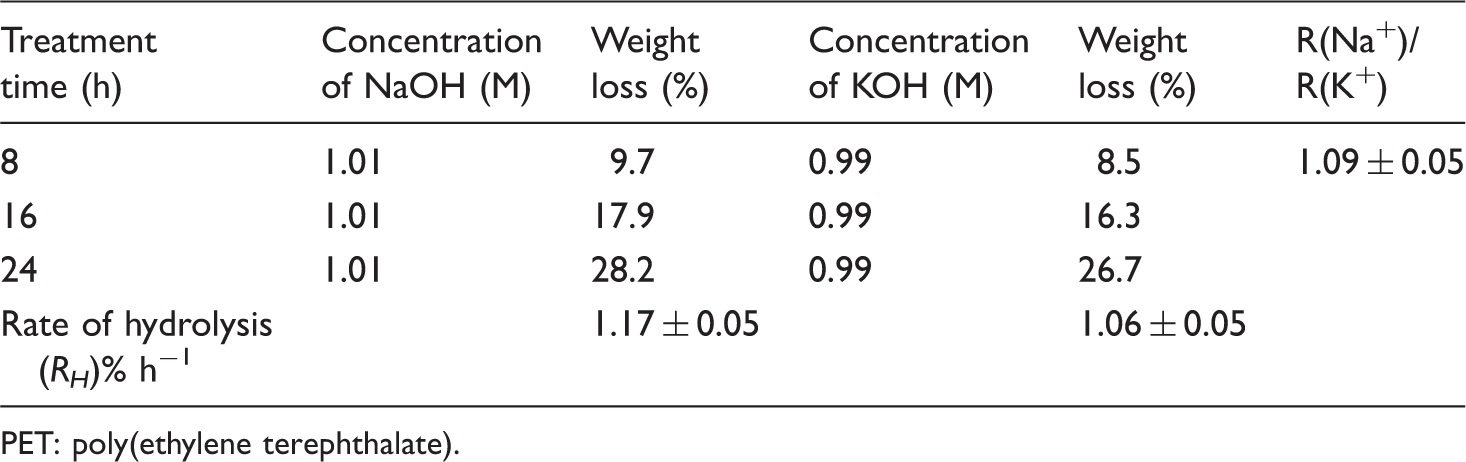
Table 1 shows the weight loss (%) and rate of hydrolysis [(RH)% h−1] alongside standard deviation of scoured PET fabric in aqueous LiOH, NaOH and KOH for different treatment times at 80℃. For all three hydroxides, the weight loss (%) increases with increasing treatment time, with the correlation coefficient (R2) of 0.9999, 0.9872 and 0.9947 for NaOH, KOH and LiOH, respectively, but the rates of hydrolysis are different (Figure 1). The rate of reactivity or rate of weight loss (%) was 2.64% h−1 for LiOH, 1.74% h−1 for NaOH and 0.81% h−1 for KOH. Using equimolar concentration data of LiOH and NaOH from Table 1, the relative reactivity of LiOH and NaOH [R(Li+)/R(Na+)] is 1.52. Table 2 shows the weight loss (%) data and relative reactivity of NaOH and KOH for grey PET fabric treated at 80℃ with equimolar concentration. Under these conditions, aqueous NaOH is about 10% faster reacting than aqueous KOH and the relative reactivity ratio of [R(Na+)]/[R(K+)] is 1.09. Considering the weight loss (%) data (Tables 1 and 2) of these three aqueous hydroxides in equimolar concentration, it can be stated that at 80℃, the reactivity is in the following order: LiOH > NaOH > KOH in the ratio of 1.65:1.1:1.0 at equimolar concentration of 1.00 (M).
Reactivity of NaOH, KOH and LiOH at 80℃, scoured PET fabric. Reactivity of aqueous NaOH, KOH and LiOH at 80℃, scoured (S2) PET fabric. PET: poly(ethylene terephthalate). Reactivity of aqueous NaOH and KOH at 80℃, grey PET fabric. PET: poly(ethylene terephthalate).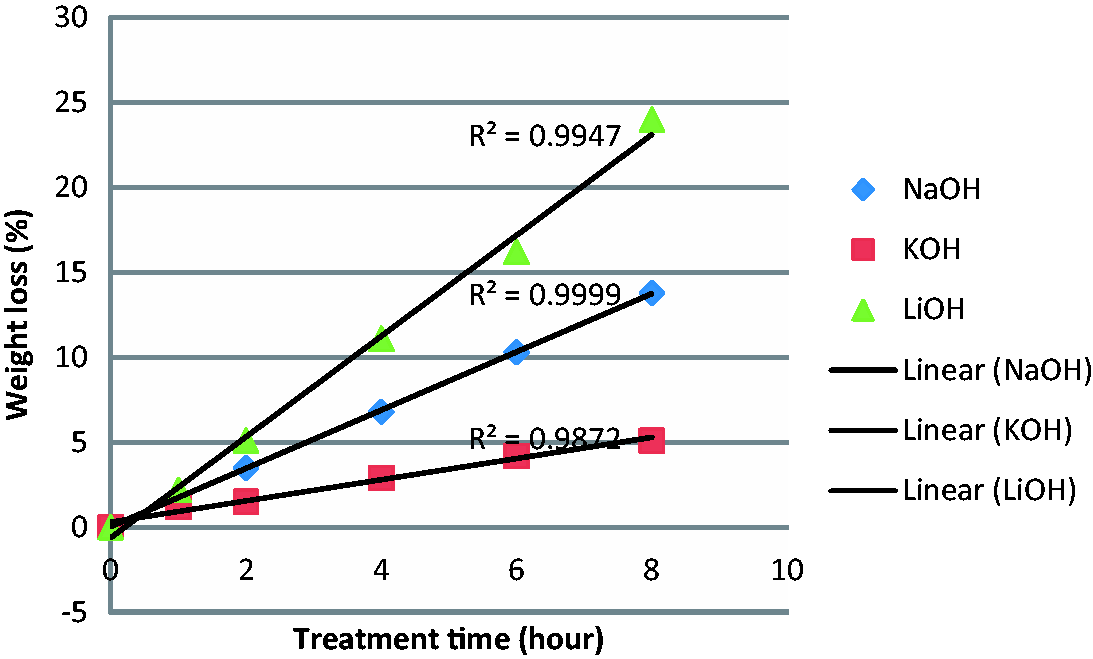

Mechanical properties of aqueous NaOH, KOH and LiOH hydrolysed PET
Mechanical properties of NaOH-, KOH- and LiOH-treated fibres at 80℃.

Scanning electron microscopy
The surface of original PET (Figure 2) is smooth with no visible cracks. However, the treated samples for aqueous NaOH (Figure 3), KOH (Figure 4) and LiOH (Figure 5) show surface pitting in the longitudinal direction. No cracks can be seen in the axial direction. Usually, if there are no other factors involved, aqueous alkaline hydrolysis is a surface reaction with consecutive layers of the PET surface being removed by OH− ions, for which no tenacity loss should occur. However, tenacity loss is often reported in the literature [18]. The longitudinal surface pittings found in the present study are similar to those found on the PET geotextiles [14] and apparel polyesters [18,21]. These longitudinal pits on the ‘apparel-grade polyesters’ are generated due to the presence of titanium dioxide and other particulate impurities [18,21]. The PET used in this study is bright type and contains about 0.1% titanium dioxide, which might be responsible for such longitudinal surface pitting.
Original PET. NaOH-treated PET; weight loss, 28.2%. KOH-treated PET; weight loss, 26.7%. LiOH-treated PET; weight loss, 24.0%.



This study showed that at 80℃, the relative rate of reactivity is LiOH > NaOH > KOH. This relative rate for geotextile-grade PET is in agreement at 70℃ (NaOH> LiOH> KOH) on ‘apparel-grade polyester’ but the relative rate is inconsistent at 95℃, which was LiOH > NaOH> KOH [22]. The similar contradictory relative rate was obtained for monovalent (NaOH) and divalent [Ca(OH)2] hydroxides [12–15] on both geotextile and apparel-grade PET. ICI (1978) data shows that aqueous Ca(OH)2 reacts 13 times faster than the aqueous NaOH at 100℃, whereas at 25℃, NaOH reacts faster than Ca(OH)2 [14,15]. Kish and Nouri [13] reported that the relative rate of hydrolysis for Ca(OH)2 is always higher than that for NaOH but the reactivity rate ratio of Ca(OH)2/NaOH is increased with increasing treatment temperature. It seems that several factors contribute to this contradictory rate of hydrolysis including glass transition temperature (Tg), apparel versus industrial-grade PET, percentage concentration versus molar concentration of alkali and material–liquor ratio in the treatment bath.
The linear rate of relationship between weight loss (%) and treatment time, the direct proportional relationship between weight loss (%) and breaking load loss (%) and insignificant loss in breaking tenacity suggest that the hydrolysis reaction for all three aqueous hydroxides takes place on the fibre surface. In this reaction (Figure 6), the hydroxyl ions attack the electron-deficient carbonyl carbons of the polyester to form an intermediate anion. Chain scission follows and results in the production of hydroxyl and carboxylate end-groups, which is not further attacked by hydroxyl groups.
Reaction mechanism of alkaline hydrolysis [13].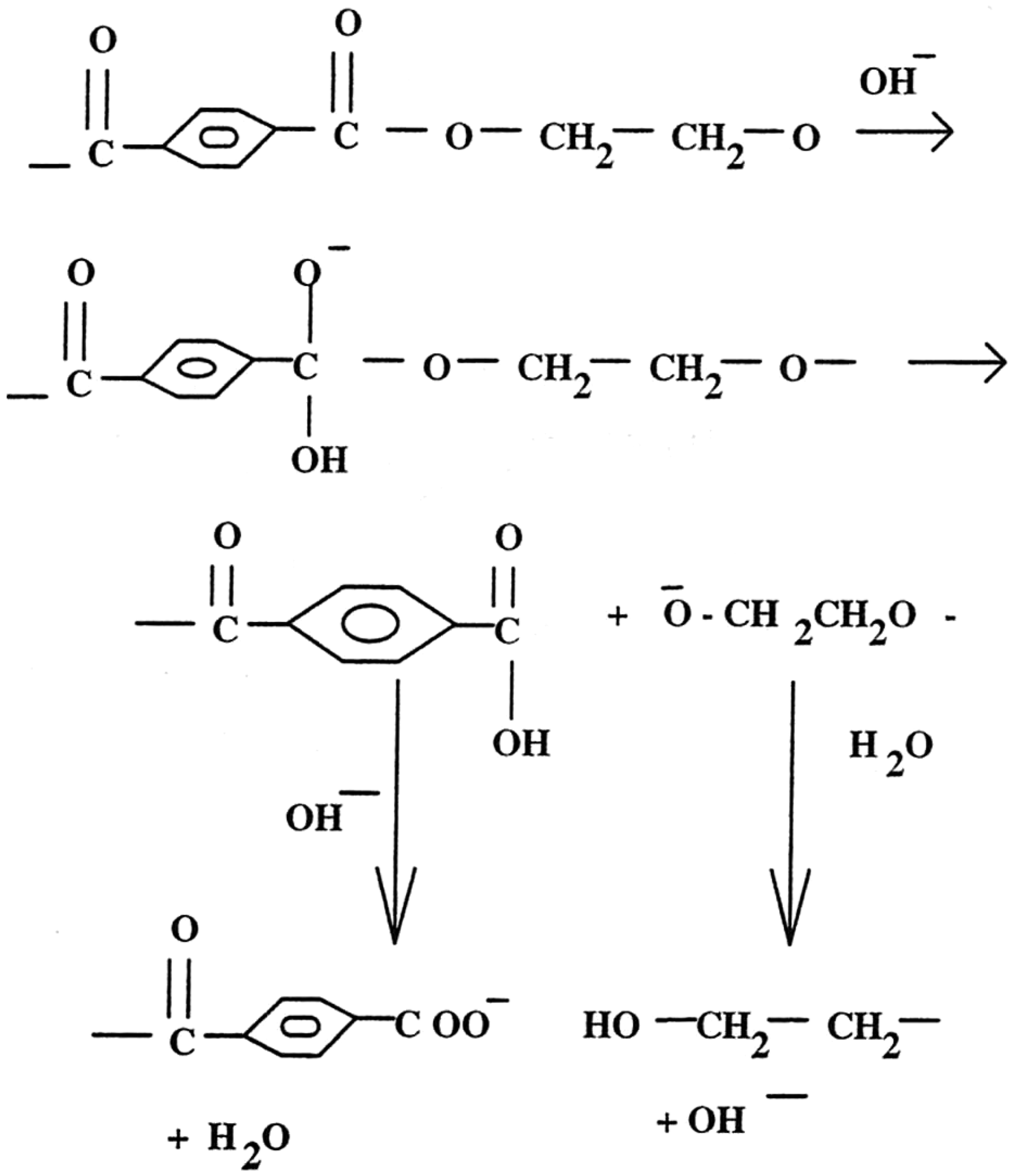
Effect of temperature on relative reactivity of NaOH and KOH: Aqueous system
Weight loss (%)

Effect of temperature on relative reactivity in terms of weight loss (%) of aqueous NaOH and KOH.

Reactivity of aqueous KOH for different concentration and treatment temperatures, scoured fabric.

The relative reactivity using different concentration of aqueous NaOH and KOH had been studied at 100℃, where it was claimed that the relative reactivity of aqueous NaOH and KOH is concentration dependent [12]. This relative rate is shown in Figure 7 from the ICI equations (equations (2) and (3)) for aqueous NaOH and KOH. According to ICI equations (equations (2) and (3)), up to 3% concentration, aqueous KOH is more reactive than aqueous NaOH and above this level, NaOH is more reactive than KOH
Relation between weight loss (%) and concentration of alkali at 100℃ (from equations (2) and (3)).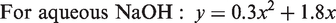


The difficulty with the above two ICI equations (equations (2) and (3)) is that the alkali concentration (x) is expressed as a percentage (weight/volume). For comparing the two different alkalis, one needs equimolar concentrations of the alkalis, where equal numbers of hydroxyl ions would be present. We have modified the ICI equations (2) and (3) using molar concentrations as shown in equations (4) and (5) for NaOH and KOH, respectively


Figure 8 shows the relationships between rate of hydrolysis and molar concentrations of aqueous NaOH and KOH obtained from revised ICI equations (equations (4) and (5)). From these revised equations, it can be seen that at 100℃, KOH is more reactive than NaOH by a factor of 1.2–1.7, depending on the molar concentration. This result is somewhat in agreement with the result of the present study. The comparison of aqueous NaOH and KOH at 100℃ (Table 4) showed that KOH is 5–10% more reactive than NaOH in 1.0 (M) concentration.
Relation between weight loss (%) and concentration of alkali at 100℃ (from equations (4) and (5)).
The ICI data conclusively show that KOH is more reactive than NaOH at 100℃. The data obtained in this study are suggesting a similar reactivity for aqueous NaOH and KOH. However, at 20℃, 60℃ and 80℃, this study showed NaOH to be more reactive than KOH, which is also supported by activation energy, though not by a large factor. On balance, it does appear that at low temperatures, NaOH is more reactive while at higher temperatures, KOH is more reactive. Such a conclusion could be valid because the activation energy for the KOH was higher than that for NaOH between the temperature ranges of 80–100℃.
Reactivity of aqueous NaOH and KOH: Heterogeneous system
Further investigation of relative reactivity of aqueous NaOH and KOH was carried under the heterogeneous conditions using DMT discs.
Weight loss (%)
Reactivity of aqueous NaOH and KOH on DMT disc at different temperature and concentration of ∼1 M.

DMT: dimethyl terephthalate.
The reactivity of the alkali metal hydroxides on DMT powder was higher than that on DMT disc. For example, after 1 h of treatment in 85 m1 of 0.987 (M) NaOH, all DMT powder (0.504 g) was completely dissolved at 80℃ (data not shown here), whereas for the same temperature and treatment time in 1.002 (M) aqueous NaOH, it was found that only 13.3% of the DMT disc had dissolved. At 70℃, after 1 h treatment in 0.987 (M) aqueous NaOH, the weight loss for DMT powder was 88.0% and the weight loss for the DMT disc was only 6.0%. No significant changes in weight were observed for DMT powder or disc in water for 1 h at 80℃. The powder, having a higher surface area, was able to react much more rapidly. The weight losses of both DMT powder and disc were higher than that of PET under the same conditions. When treatment was carried out in 0.987 (M) aqueous NaOH, PET-scoured fabric lost only 2.1% of weight after 1 h at 80℃, which is much lower than the value obtained for a DMT disc for the same treatment conditions (∼14% h−1 in 1.002 M aqueous NaOH).
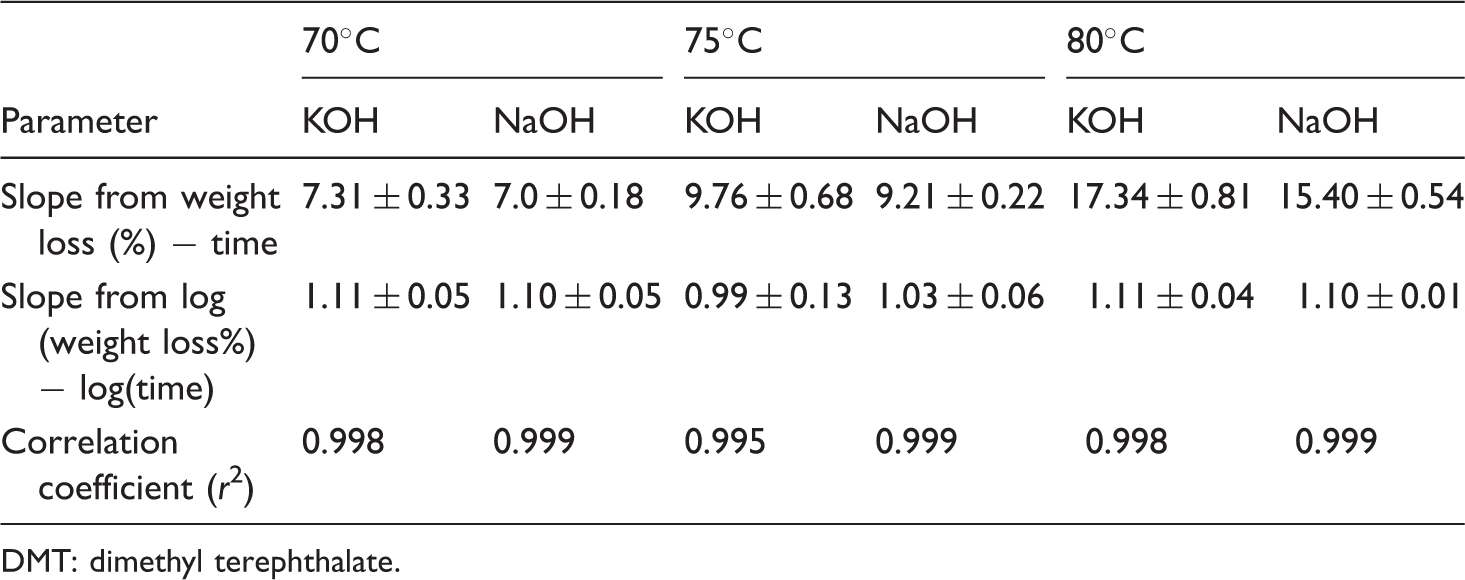
The slopes from weight loss(%)−time and log(weight loss%)−log(time) and correlation coefficients of aqueous NaOH- and KOH-treated DMT are shown in Table 7, which is calculated from Figure 9. The slope for an aqueous KOH treatment is always higher than that of the slope of the equivalent aqueous NaOH treatment for all three temperatures. As can be seen from Table 7, the ratio of slopes for KOH to NaOH at 70℃ is 1.04, at 75℃ this value is 1.06 but at 80℃, the ratio of slopes is increased to 1.13. Therefore, the reaction with a disc of DMT, both alkalis react with similar rates at lower temperatures, with the KOH being perhaps about 10–15% more reactive at 80℃.
Scanning electron microscopy of alcoholic NaOH-treated fibre at 20℃ for 16 h. Slope and correlation coefficient for DMT disc hydrolysed with ∼1 M aqueous NaOH and KOH at different temperatures. DMT: dimethyl terephthalate.
Appearance of DMT disc
The DMT disc did not undergo uniform attack by the aqueous alkalis, as can be seen from the surface of the disc. The original disc was circular and smooth surfaced but the discs treated at 80℃ for 4 h with aqueous NaOH and KOH became rough and irregular in shape (loss of circularity), with many surface cracks. The loss of circularity of the DMT disc is a distinctive characteristic of hydrolysis in a heterogeneous system as no such materials ‘fall-off’ was observed during PET hydrolysis in aqueous systems. The surface cracks were quite visible to the naked eye.
Reactivity of NaOH and KOH on PET geotextiles: Alcoholic alkaline system
Weight loss (%)
Reactivity of alcoholic NaOH and KOH on PET grey fabric at different temperature.
PET: poly(ethylene terephthalate).
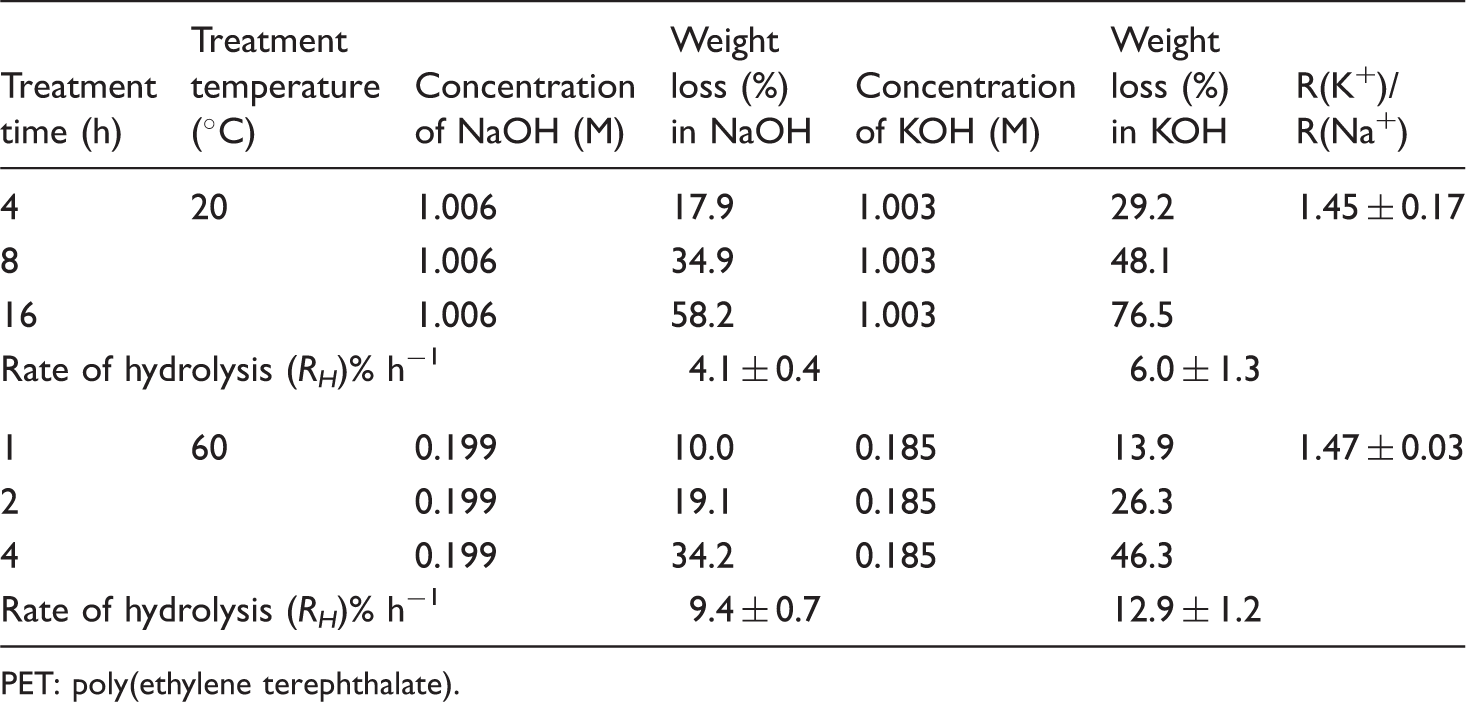
The rates of weight loss in both alcoholic NaOH and KOH were much higher than in the respective aqueous solutions of these two alkalis. At higher temperature, the rate of hydrolysis in alcoholic KOH was so severe that at 80℃, after 1 h treatment, all the PET grey knitted fabric samples had dissolved in 1.003 (M) alcoholic KOH, whereas under identical conditions of treatment, after 1 h in 1.16 (M) aqueous KOH, the same fabric lost only 1.4% weight (Table 5). At 60℃, a similar massive weight loss was observed, where the rate of hydrolysis was 12.9 ± 1.2 h−1 (Table 8) in a 1.003 (M) alcoholic KOH compared with only 1.066 ± 0.05 h−1 (Table 2) for 1.00 (M) aqueous KOH. The degradation in methanol alone was very slow and after 3 h of treatment at 60℃, only 0.96% weight loss was observed, presumably due to finish extraction.
Comparative results between aqueous and methanolic alkaline solution.

Mechanical properties of the alcoholic alkaline hydrolysed PET
Breaking load and tenacity data for PET treated with alcoholic NaOH and KOH at 20℃, 1.00 (M) solutions.
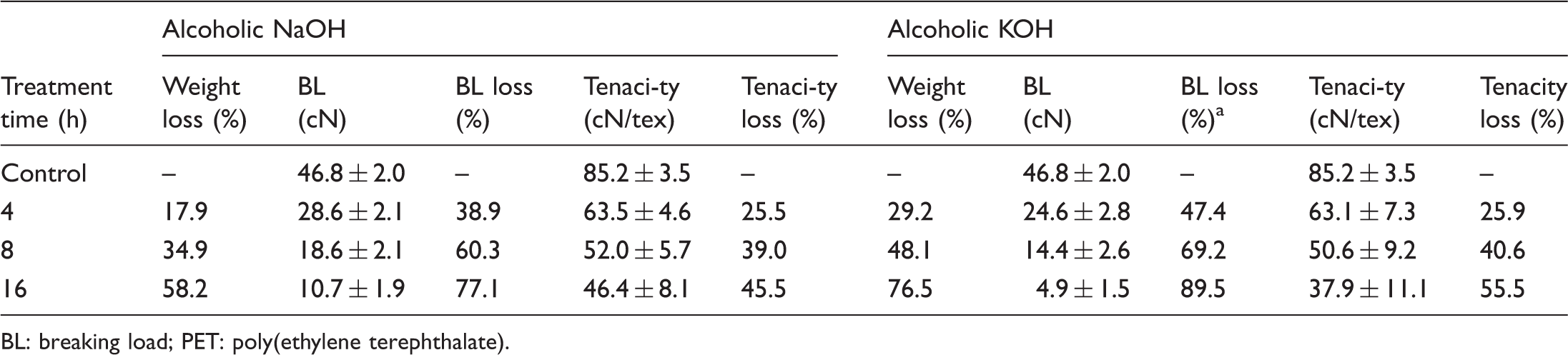
BL: breaking load; PET: poly(ethylene terephthalate).
SEM of alcoholic alkali-treated PET
Scanning electron micrographs of alcoholic NaOH-treated fibres are shown in Figures 9 and 10. Severe cracks are formed in these two micrographs, which was not seen in the aqueous-treated samples. The cracks are all over the surface in blocks and in axial direction. The deeper and wider cracks can be seen in the sample that lost more weight (Figure 9; weight loss, 58.2%) than the sample where weight loss was less (Figure 10; weight loss, 34.2%). It seems that in alcoholic system, the attack of the base (CH3O−) occurs through the amorphous regions, penetrates through these regions and removes the fibre materials from the surface. This explains the higher weight loss and mechanical loss (%) in alcoholic alkaline systems. Furthermore, acidity (H−) function of base CH3O− (14.57) is much higher than the base OH− (12.66) and therefore, it is easier for base CH3O− to penetrate deeper inside the fibre through the amorphous regions, which resulted in larger weight and mechanical properties loss [29].
Scanning electron microscopy of alcoholic NaOH-treated fibre at 60℃ for 4 h.
PET is a semi-crystalline fibre that consists of both crystalline and amorphous regions as well as extended non-crystalline molecules (mesa-amorphous) and tie molecules (Figure 11) [30]. The two main regions (crystalline and amorphous) are linked to each other in the fibre axis direction. During alcoholic treatment, the base (CH3O−) attacks the amorphous regions and produce surface cracks in the axial direction.
Fibrillar structural model for PET fibres [30].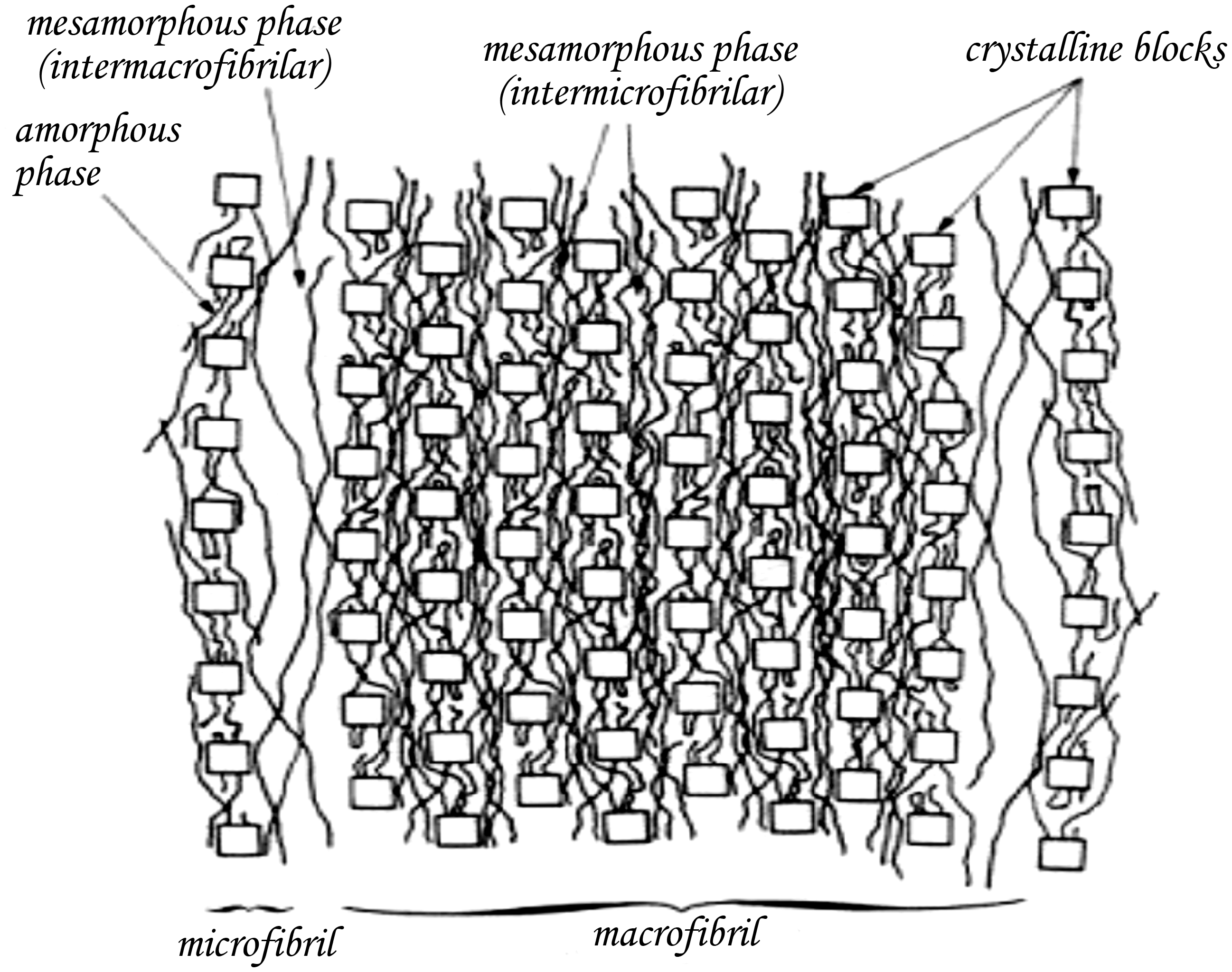
Summary and conclusions
At 80℃, the reactivity of various aqueous alkaline hydroxides on PET is of the following order: LiOH>NaOH>KOH. However, the rate of attack of different aqueous alkaline hydroxides on PET seems to have a complicated dependence on temperature. The present study showed that in aqueous system and at 20℃, 60℃ and 80℃, NaOH is more reactive than KOH, although not by a large factor. At 100℃, KOH is more reactive than NaOH. Such a conclusion in aqueous systems is supported by two different activation energies for aqueous KOH that are below and above 80℃. In heterogeneous system, aqueous KOH was slightly more reactive than aqueous NaOH in the temperature range of 70–80℃. To conclude, at low temperatures, NaOH is more reactive, while at higher temperatures, KOH is more reactive.
In alcoholic system, methanolic KOH reacts about 1.5 times than methanolic NaOH at 20℃ and at 60℃. Overall, the methanolic alkaline solutions attack PET much more rapidly than aqueous alkaline solutions. The larger loss in properties in alcoholic systems was due the formation of surface cracks.
This study is the first of its kind that investigated the impact of alcoholic alkalis on the degradation rate of PET. Degradation of other alcoholic alkalis that can be found in mining operations and lagoon systems should be investigated to understand the long-term durability of PET.
Footnotes
Acknowledgements
The geotextile-grade PET material was supplied by Hoechst (Germany). Ms H Dyson of the University of Leeds, UK, and Dr Ravinder Shidu of University of Manitoba, Canada, prepared the SEM specimens. Their assistance is greatly appreciated.
Funding
Financial support for this study was obtained from Commonwealth Scholarship Fund in the United Kingdom and Clothworkers’ Library in the University of Leeds, UK.

