Abstract
A thermal resistance coating of different high temperature resistance polymers, e.g. polysilazane and polysiloxane was applied to a carbon roving by a continuous dip coating method. Improved mechanical and thermal resistance properties of the coated carbon roving were evaluated by thermogravimetric analysis and tensile property testing under thermal stress. Investigation of the mechanical properties of coated carbon roving revealed a significant increase in their breaking strength, which amounted to more than 50% as compared to uncoated roving. The thermal stability as well as the oxidation protection of the coated sample in ambient air at temperatures up to 800℃ was much higher than that of the control sample. Topographical phenomena occurring due to the coating on the microscale were examined by scanning electron microscopy and energy dispersive X-ray analysis. The surface energy and the adhesion property of the coated substrate were determined by measuring the contact angle.
Introduction
Nowadays, carbon fibres possess the most preferable position among all engineering materials because of their technological importance in high-tech applications. They are used extensively in different fields because of their light weight, high specific strength, stiffness, chemical resistance, thermo-shock and fracture toughness, especially under high temperatures. Furthermore, the thermal expansion of carbon fibre is almost zero [1] and it retains its strength at high temperature about 3000℃ in non-oxidation atmospheres [2]. Owing to their minimal thermal expansion, they are a unique material for high-temperature applications such as structural components of space vehicles, defence industries, automotive, aviation, electronic industries, marine, protective textiles and the promising area of textile-reinforced concrete [3,4]. However, carbon materials are stable up to about 2100℃ in the inert atmosphere [5]. Due to their low oxidation resistance, they lose weight and mechanical properties significantly at temperatures of over 400℃ in the air [6,7]. Additionally, Baker et al. [8] and Konkin and Varshavskii [9] reported that carbonized fibre will start oxidizing in the air at about 300℃ followed by graphitized fibre at a temperature of about 400℃. For this reason, carbon fibres have to be protected from extreme heating conditions (especially in the case of fire) or of conditions in which the temperature of carbon reinforcements in use is above the oxidation temperature. Therefore, the thermal- and oxidation-protected carbon fibre is not only a desired property for imparting sufficient stability to heat but also for extending the life span of carbon-based composites and other carbon-reinforced products. Over the long term, much research has been carried out to overcome the oxidation problem, including research on thermal spraying, electroplating, physical and chemical vapour deposition and plasma coating [10–13]. This coating can protect the carbon material from adverse oxidation conditions much like sunscreen protects our skin. Besides, coating can improve the properties of filament yarn and fabric. As a result, the coating is not only important for the stability of carbon fibre against oxidation, but it can also improve the adhesion property between filament and matrix material [14]. It must be mentioned that high temperature resistance coating of carbon fibre is the most challenging issue because of its chemical inertness, very smooth surface and negative thermal expansion coefficient. However, polysiloxane and polysilazane chemistry offer a great potential for providing a thermal barrier property to carbon fibre. These preceramic polymers become cross-linked by thermal, chemical or photochemical treatment. Moreover, both polymers convert into SiCN and SiCO ceramic layers [15–18] at high temperatures which could convey a good oxidation protection property of carbon fibre. Thermal resistant coatings based on polysilazane and polysiloxane polymers not only improve the functional property of carbon fibre but these coatings will also enable new functions for high temperature applications. Additionally, these functional coatings will meet the growing demand of carbon fibre based technical textile.
The main task of this research study is to design a heat resistance coating to achieve both structural and resistance to the thermal degradation of carbon fibre. To fulfil this task, polysilazane and polysiloxane polymers were applied to carbon filament (CF) rovings by continuous dip coating method. Then, the surface topography, thermal and tensile properties of coated carbon roving were investigated through different experiments.
Materials and methods
Materials
List of polymers used for coating of carbon roving.
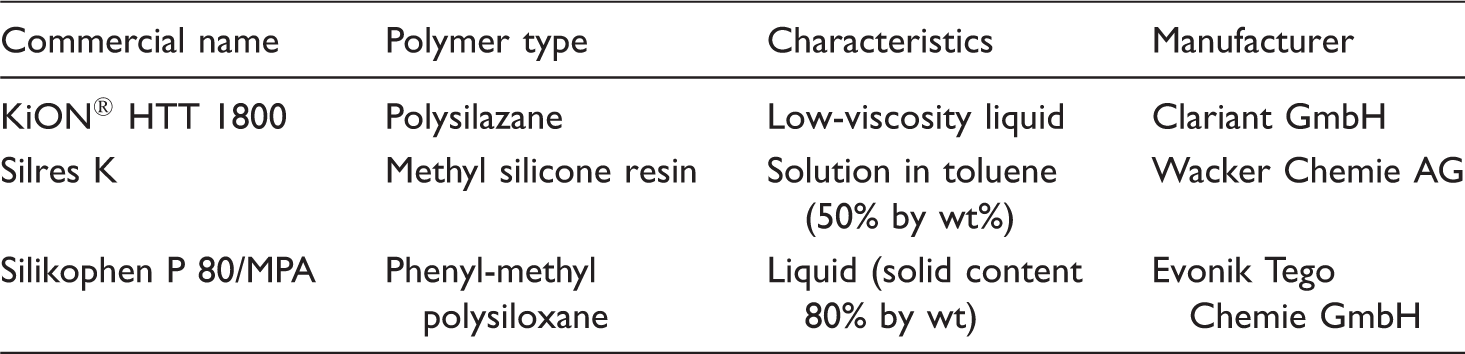
Acetic acid ester (ester) supplied by Biesterfeld, Germany was used as the solvent. It is a clear, colourless and low viscosity organic solvent available in liquid form.
Coating process of carbon roving
Formulation and process parameters for coating.

CF: carbon filament.
According to the formulations, the homogeneous coating solution was produced by appropriate stirring and put into the coating bath. Then, the CF roving was passed through the coating bath at room temperature. The coating fluid was agitated appropriately by glass rod to have uniform coating on the roving. After impregnation, the roving was passed over to the squeezing rollers. No pressure was applied on the carbon fibre during squeezing since the material is very brittle. The process parameters such as speed, tension and curing time were maintained constant for the whole experiment. Only curing temperatures were varied for different coating polymers because the recommended curing temperature of each polymer is not the same. Next, the roving was dried and cured in the drying unit at the required temperature and for the required time.
During curing, polymers were cross-linked to each other and formed a siloxane (SiOSi) film on the top of the carbon fibre surface. The mechanism of polysiloxane resin coating is shown in Figure 1. Finally, coated carbon roving was wound up by a special spool. The amount of coating gained by carbon roving was determined by weighing of each 5 m sample before and after coating (dried sample). To get accurate results, weight gain was calculated in three stages of coating (at initial, middle and final stages).
Mechanism of coating with polysiloxane resin.
Tensile property testing
The stress–strain behaviour of coated carbon roving was tested according to the ISO 3341 standard at room temperature and under thermal stress at 300℃, 400℃, 450℃ and 500℃ to evaluate the coating effect on the mechanical and thermo-mechanical properties of the roving. The testing was performed with a Zwick-100 material testing instrument equipped with special return clamps, a stain measuring photo sensor and an infrared heating system (Figure 2).
Determination of stress–strain behaviour of CF roving under thermal stress (ITM, TU Dresden).
The test program was controlled and monitored by the software Testxpert-II. The breaking force (forces to break the yarn) versus elongation was recorded during testing and five measurements for each sample were taken to get an average value of the results from each sample. The samples were conditioned for a minimum of 24 h in standard climate 20℃ ± 2℃ and 65% ± 2% relative humidity. The sample was heated at 20℃/min for applying thermal stress.
Scanning electron microscopy and energy dispersive X-ray analysis
The surface topographical nature of coated carbon fibres was analysed by scanning electron microscopy (SEM). These experiments were conducted with a Zeiss Digital SEM (DSM 982 GEMINI). Energy-dispersive X-ray (EDX) was used to determine semi-quantitative chemical analysis and mapping. The Zeiss DSM 982 GEMINI instrument was equipped with an EDX micro analyser system.
Contact angle and surface energy calculation
Contact angle (CA) measurement is a useful technique to estimate surface energy (SE) and surface tension of liquid droplets at solid interfaces. This technique is extremely useful in detecting wetting, adhesion and absorption property in monolayers [19]. The static CA was measured by DSA 100 Krüss. SE was calculated by the Owens–Wendt method [20]. DSA 100 software was used for analysing the SE and surface tension of liquid droplets at solid interfaces. All measurements were carried out at 20℃. Ten individual measurements were randomly taken from different places on each sample to calculate the average and the SD value.
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was used to study the thermal stability and decomposition of the material. TGA measures mass changes of material as a function of temperature and time and it is controlled by an extremely sensitive electronic microbalance [21,22]. TGA experiments were performed using TA instrument Q500 with a heating rate of 10℃/min. The samples were heated from 25℃ to 940℃ to determine the complete thermal degradation of coated and uncoated carbon fibres. All tests were carried out in nitrogen and air atmosphere using a flow rate of 60 mL/min.
Results and discussion
Investigation of stress–strain behaviour
Tensile properties of coated and uncoated carbon roving.

CF: carbon filament.
It is evident from Figure 3 that all coating polymers enhance tensile strength of carbon fibre approximately 50–75% at room temperature. It was observed that the tensile strength increases by approximately 75% at room temperature for sample R3 compared to CF (as received). This increased strength at room temperature may be attributed to the individual fibres bonding to each other in a polymer matrix and as a result CF roving acts as single fibre and reduces the fibre defects during testing. Besides, coating improves the filament micro friction/adhesion within the roving.
Comparison of tensile strength of different coated CF rovings.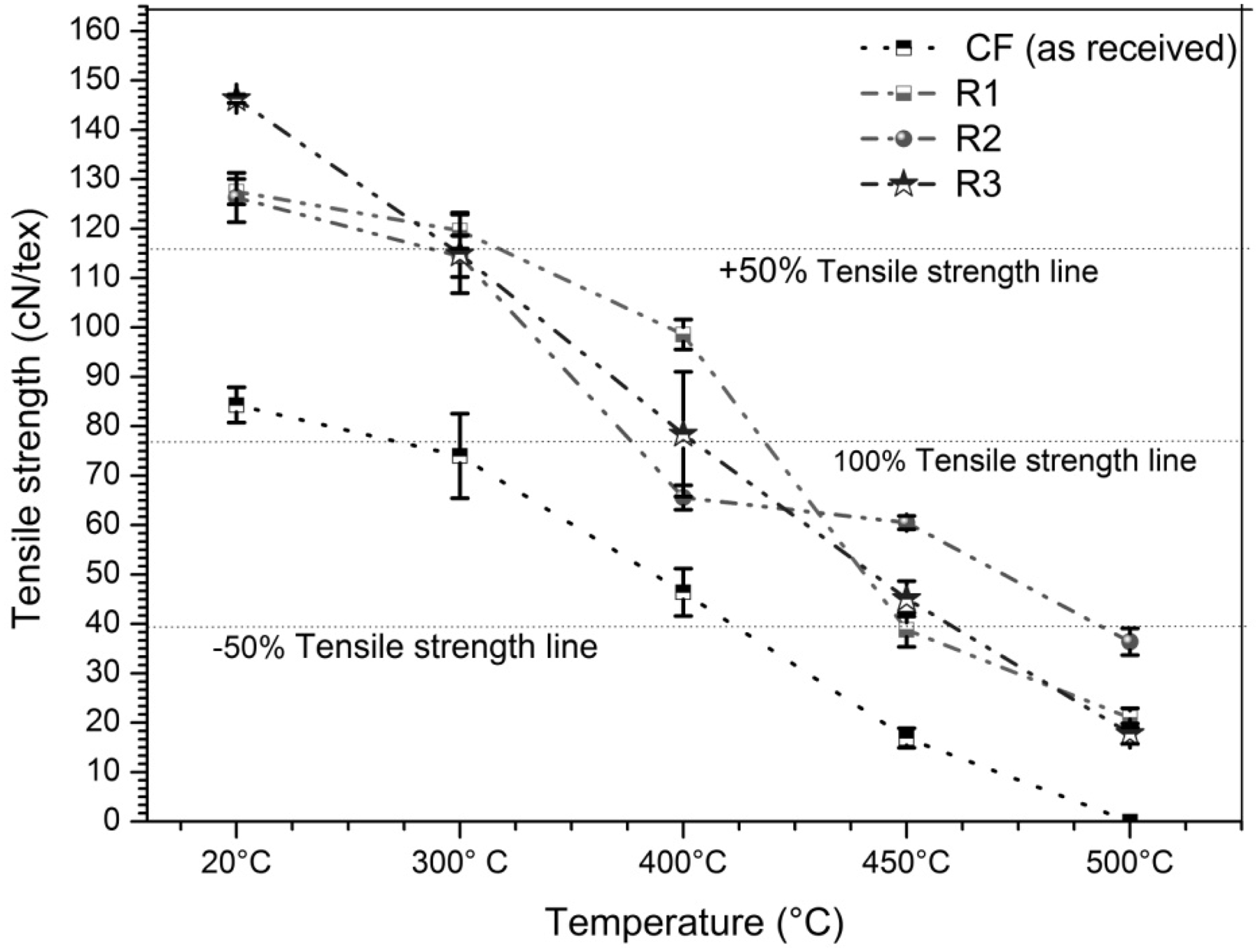
On the other hand, uncoated CF loses its 50% strength at 400℃ and 80% strength at 450℃. Although sample R1 shows the best result at 400℃, it loses its strength drastically (more than 55% compared to original CF) at 450℃. The strength loss tendency of sample R2 is relatively slower than others. Whereas other coated samples lost their strength over 75% compared to uncoated CF at 500℃, sample R2 retains its 46% tensile strength (compared to uncoated CF) at 500℃. From the above discussion, it can be concluded that methyl silicone resin-coated carbon fibres show better stability at high temperatures than polysilazane and phenyl-methylpolysiloxane-coated carbon fibres. These results may be attributed to the fact that phenyl-methylpolysiloxane and methyl silicone resins are cross-linked to each other during heating at high temperatures and form a three dimensional (−Si−O−Si−) matrix which is more stable and imparts more oxidation barriers than a polysilazane resin (−Si−N−Si−) matrix.
Figure 4 illustrates the stress–strain curves of uncoated and different polymer-coated carbon rovings at 20℃, 300℃ and 400℃. It is clearly observed that all coated samples show similar stress–strain behaviour at 20℃, 300℃ and 400℃. It can clearly be seen that coated samples (R1, R2 and R3) show a linear stress–strain curve at different temperatures than uncoated roving. In contrast to original carbon roving, stress–strain curves of phenyl-methylpolysiloxane and methyl silicone resin-coated carbon rovings (samples R2 and R3) are more linear. At 500℃, stress–strain values of samples R2 and R3 were 36.4 cN/tex, 17.8 cN/tex and 0.37%, 0.20%, respectively, whereas uncoated carbon fibres were fully oxidized.
Stress–strain curves of uncoated and different coated carbon rovings (a) CF(as received); (b) R1(HTT 1800); (c) R2(Silires K); (d) R3 (Silikophen P80/MPA).
Surface topography
The topographical changes of coated and uncoated carbon roving have been investigated using SEM which shows the changes of the fibre surface characteristics of coated and uncoated samples. Figure 5(a) shows the SEM images of CFs (as received) which present a smooth and homogeneous surface. The surfaces look smooth, the only feature being longitudinal ridges which extend over the whole length of the fibre.
SEM images of uncoated and coated carbon roving (a) CF (as received); (b) Coated CF with R1; (c) Coated CF with R2; (d) Coated CF with R3.
It might be observed that the sizing materials aggregate on the surface which is applied to CF roving during production to protect the fibre from damage during handling and processing. The surface of sample R1 (Figure 5(b)) shows regular, smooth coating layers on the surface under SEM photographs. It may also be observed that some coating polymers and sizing materials aggregate on the surface. This aggregation may have occurred due to the re-deposition of sizing material (polyurethane) of supplied carbon roving. The samples R2 and R3 (coated with phenyl-methylpolysiloxane and methyl silicon resin, respectively) show very regular and smooth surfaces (Figure 5(c) and (d)). The deposited coating layers seem to be thin and the filaments are strongly attached to each other.
EDX analysis
EDX results of coated and CF (as received) roving.

EDX: energy dispersive X-ray; CF: carbon filament.
It has been observed that the uncoated CF contained nearly 100% carbon atoms. In contrast, the coated carbon fibres have oxygen (O) and silicone (Si) element along with carbon (C) and nitrogen (N) elements. These oxygen (O) and silicone (Si) elements prove the presence of silicone- and oxygen-based polymeric material on the surface of carbon fibres. It is shown that samples R2 and R3 contain relatively much oxygen (O) and silicone (Si) elements compared to sample R1 (Table 4).
CA and SE results
CA and SE results of coated sample.

CA: contact angle; SE: surface energy.
It should be mentioned that the CA values against diiodomethane for sample R3 was found to be zero, the drop of diiodomethane on the surface of coated carbon roving disappeared immediately. The highest SE (50.94 mN/m) was found for the sample R3 and the SE values (33.75 and 23.76 mN/m, respectively) were obtained for samples R1 and R2. From the above discussion, it can be concluded that the surface of phenyl-methyl polysiloxane coated CF roving being higher in SE than the surface of polysilazane coated CF rovings. This higher SE tends to increase the adhesion property with a matrix as well as reduces energy by absorbing contaminants from the environment.
Thermogravimetric analysis
TGA in air atmosphere
Figure 6 illustrates that the uncoated carbon fibres started to oxidize at 280℃ very slowly and strong oxidation begins above 500℃. On the other hand, oxidation starts at about 500℃ and the strong oxidation zone begins at above 600℃ for coated carbon. Figure 6 shows that the initial stage of the mass change rate from 25℃ to 280℃ is very slow for all coated and uncoated carbon fibres. At this stage, a mass reduction of 1.63% of original CF (as received) occurred due to the reduction of sizing material and the evaporation of volatile oligomer of carbon fibre.
Selected TGA graph of initial 25% mass change.
On the other hand, coated CF slightly loses its mass at this stage due to the evaporation of solvent and water of coating. In the second stage (from temperature 300℃ to 600℃), slow oxidation occurred. At this stage, original carbon fibres oxidized slowly. As a result, they lost their mass and strength. For coated carbon fibres, mass reduction occurred due to the volatilization of oligomeric coating material and the oxidation of organic groups (methyl, ethyl, phenyl, propyl and vinyl) which are attached to silicon. Finally, we found a rapid decomposition stage (above 600℃) which could be referred as phase 3. At this stage, very quick oxidation occurred. As a result, all coated and uncoated carbon fibres lost their mass very rapidly.
Mass change at high temperature
Mass percentage at different temperatures.
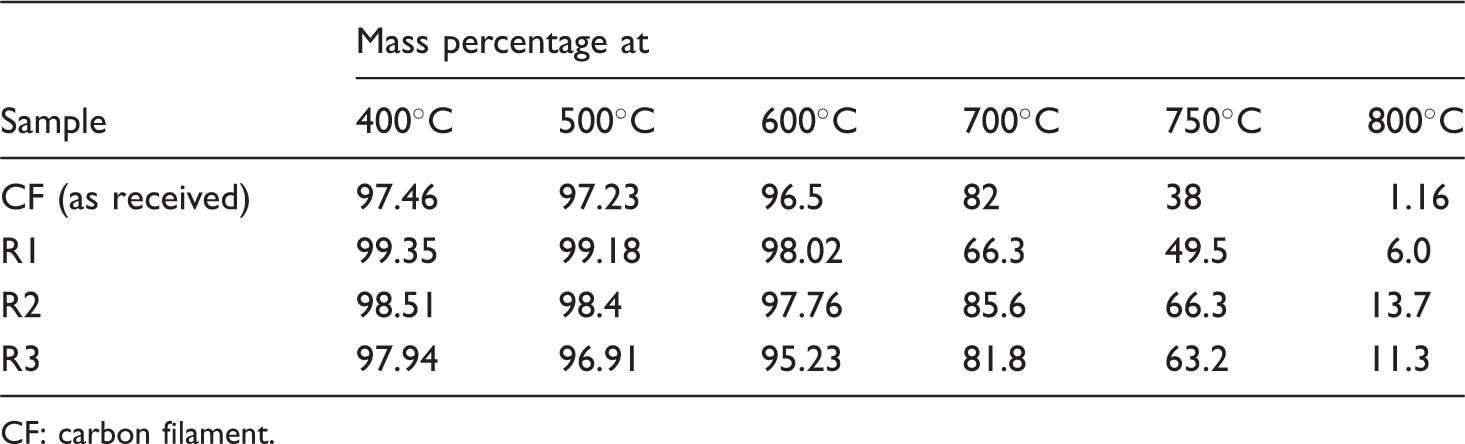
CF: carbon filament.
On the other hand, polysilazane-coated CF (sample R1) shows better oxidation resistance up to 600℃. It has been observed that samples R2 and R3 show similar mass loss tendencies under high temperatures of over 600℃. The best oxidation resistance in high temperature was found for the sample R2 which had an amount of 85.6%, 66.3% and 13.7% residual mass at the temperatures 700℃, 750℃ and 800℃, respectively. From the above TGA results, it can be concluded that degradation is relatively slower for coated CF than uncoated CF. The possible explanation for improved oxidation resistance of coated CF would be the conversion of coating polymers (polysilazane or polysiloxane) into silicone carbide or silicone nitride ceramic (SiCN or SiCO) structures at high temperatures which may slow down the oxidation rate of carbon.
Relationship between mass loss and tensile strength
Tensile tests on the samples were performed at room temperature, 300℃, 400℃, 450℃ and 500℃. TGA was conducted from 20℃ to 940℃ in ambient air. Figure 7 clearly reveals that uncoated carbon roving lost its strength by 50% and 80% at 400℃ and 450℃ compared to the strength at room temperature; where mass losses of 2.54% and 2.65% were observed.
Relationship between tensile strength and mass loss of coated and uncoated CF roving at ambient conditions.
On the contrary, coated sample R2 lost its strength of 22% and 28% at 400℃ and 450℃, respectively, compared to uncoated carbon roving. On the other hand, coated sample R1 shows the best result at up to 400℃; where the strength was found to be 20% more than the strength of uncoated carbon roving at room temperature and mass loss was only 0.64%. Although sample R1 shows a very slow mass loss tendency up to 600℃, the strength loss tendency is very rapid over 400℃. Sample R3 shows maximum tensile strength at room temperature; however, by increasing the temperature, the strength loss tendency was linear. A similar approach for sample R3 was found at high temperatures in ambient conditions. The strength loss for coated carbon fibre may be attributed to the formation of micro cracks in coating layers under thermal stress which permits the oxygen to attract the fibre. As a result, it loses its barrier effect. Figure 8 shows the micro crack formation of coated carbon fibre after heating at 500℃ under the stress during tensile strength testing.
SEM images of coated CF (R2) after heating at 500℃.
Conclusion
The main objective of this study was to tailor a new thermal resistance and oxidation protective coating system for carbon fibre which will meet the security demand (especially in the case of fire) as well as improve the end use property of carbon fibre reinforcements. The tensile strength of all coated carbon roving was enhanced at room temperature. Tensile properties under thermal stress proved that polysilazane, methyl silicone resin and polysiloxane coating improved the thermal and oxidation resistance properties of carbon fibre substantially. The results of CA and SE show that polysiloxane resin provides a higher SE than methyl silicone resin and polysilazane coated surface. This improved SE can provide good adhesion properties within a matrix system. On the other hand, low SE of methyl silicone resin and polysilazane coated CF refers to the repelling tendency that sufficiently prevents the adhesive from wetting. TGA results prove that an improved thermal and oxidation stability of carbon fibres is achieved by coating them with methyl silicone resin and polysiloxane resin.
Finally, this study can conclude that coatings with polysiloxane and methyl silicone resin on CF roving form relatively good thermal and oxidation resistance surface other than polysilazane. This polysiloxane and methyl silicone resin would be a promising polymer for continuous coating of CF roving.
Footnotes
Acknowledgements
The authors thank the Wacker Chemie AG, Evonik Tego Chemie GmbH and Clariant GmbH, Germany for providing coating materials.
Funding
This work was funded by the German Research Foundation within the project SFB 528, TP D6.
