Abstract
Keywords
Introduction
Avascular necrosis, also known as aseptic necrosis, or ischemic bone necrosis is a well-recognized complication among kidney transplant recipients. In 1969, Cruess reports one-third of kidney transplant recipients suffered from this debilitating complication. In recent years, the incidence of avascular necrosis has decreased due to the standardization of the induction and maintenance of immunosuppressive agents, mainly due to the use of calcineurin inhibitors-based regimens, and early steroid withdrawal. 1
The intrinsic pathophysiology of avascular necrosis is characterized by insufficient flow of blood to the bone, inducing necrosis and bone loss, with the femoral head being the most likely bone structure to succumb to ischemic necrosis. In addition to steroids, other risk factors for avascular necrosis include sickle cell anemia, fatty emboli, alcoholism, dyslipidemia, hemostatic disorders, osteopenia, and secondary hyperparathyroidism. 2 Some of the transplant-specific risk factors for avascular necrosis include the development of delayed graft function, a higher degree of human leukocyte antigen mismatches, and pretransplant serum calcium and phosphorus levels. 3
Although few studies have assessed the risks of avascular necrosis in kidney transplant recipients, to the best of our knowledge, there are no studies to date that delineate the kidney transplant outcomes among this population.
Specific Aim
The aim was to compare rejection, death-censored graft failure, and patient mortality in kidney transplant recipients who had avascular necrosis post-transplantation with matched controls.
Design/Methods
Design
Design: This was a single-center retrospective observational comparative evaluation of all adult kidney (including simultaneous pancreas-kidney) transplant recipients transplanted between January 1, 2001, and December 31, 2018. This study was approved by the local institutional review board. The evaluation received a waiver or written informed consent.
Population: The target population of 5570 kidney and simultaneous pancreas-kidney recipients were transplanted between 2001 and 2018. The mean age was 49.5 years; 60.1% were male, and predominantly white, with 18.5% as non-white recipients. In comparison to the United Network for Organ-Sharing (UNOS) kidney and pancreas population during the same period, the population was comparable in terms of male recipients at 60.7% and mean age at the time of transplant of 48.5 years. The proportion of non-white recipients was significantly lower compared to UNOS data at 47.1%.
Sampling: All adult kidney-only transplant recipients or recipients of simultaneous pancreas and kidney transplants at this center were included. The sample included simultaneous pancreas-kidney recipients, as most of the pathophysiological conditions and kidney disease burdens were similar. All data were extracted from the center's transplant database. The study period was selected to start after 2001, as since then the immunosuppressive induction and maintenance protocol were relatively stable and protocolized. Transplant recipients with a post-transplant diagnosis of avascular necrosis were identified and compared with a control selected by incidence density sampling in a 1:3 ratio by post-transplant interval who did not have avascular necrosis. That is, for example, if one transplant recipient had avascular necrosis 24 months post-transplant, then 3 controls were selected who did not have avascular necrosis by 24 months post-transplant and were transplanted within ±1week range of the avascular case and still had functional graft. Multiorgan transplant recipients except for simultaneous pancreas-kidney were excluded. Recipients with a diagnosis of pretransplant avascular necrosis were excluded.
Data Collection
Acute rejection, death-censored graft failure, and patient mortality after avascular necrosis diagnosis were outcomes of interest. Death-censored graft failure was defined as a return to dialysis or retransplant after graft failure. Patients were followed until death, death-censored graft failure, or until the end of the data analysis in April 2022.
All data were collected from an institutional database. Recipient's and donor's baseline characteristics including immunological risk factors were collected, that included: recipient's age at the time of transplant, body mass index, gender, race, cause of end-stage kidney disease, previous transplant, transplant types (living donor or deceased donor), recipients with pretransplant dialysis or not, time on dialysis and post-transplant delayed graft function. Average human leukocyte antigens mismatch, sensitization as assessed by panel-reactive antibody > 10%, induction immunosuppression, calcineurin inhibitor-based immunosuppression, and early steroid withdrawal were collected. Donor baseline characteristics collected were age at donation, kidney donor profile index, gender, race, and body mass index.
Data Analyses
For baseline data, chi-squared tests and Fisher's exact tests to compare differences in frequencies were used. Continuous data were expressed as the mean plus standard deviation for all normally distributed variables. A median plus interquartile range where data showed skewness or non-normality was used. All continuous data comparisons were conducted using a parametric student t-test or the nonparametric Wilcoxon sign rank test. Patient and graft survival using the last date of data collection were used. Rejections using the last date of collected complications were used. Univariate and multivariate survival analysis were conducted using the cox proportional hazards model with log-rank tests, life tables, and Kaplan-Meier survival curves. Multicollinearity was assessed with the variance inflation factor. All variance inflation factor values >10 were regarded as model-disrupting and removed from the multivariate model. Alternatively, a competing risk survival analysis using the methods of Fine and Gray with either mortality or graft failure as the competing event in addition to standard censoring were conducted.
For missing data use of case-wise analysis or multiple imputations for continuous data were used. All P-values ≤.05 are considered statistically significant. All analyses were conducted using STATA version 17 (College Station, TX).
Results
Incidence and Risk Factors for Avascular Necrosis
A total of 88 recipients were identified with the diagnosis of avascular necrosis with an incidence of 1.5%. The mean interval from transplant to avascular necrosis diagnosis was 2.9 (2.75) years. The majority had avascular necrosis of the hip bones except in 3 cases, where 2 had avascular necrosis of the foot bone (talus) and 1 had avascular necrosis of the shoulder bone. Looking for various risk factors for avascular necrosis during the study period (data not shown), including all recipient's and donor's baseline characteristics along with the immunological risk factors, the calcineurin-based immunosuppression was associated with a decreased risk for avascular necrosis (hazard ratio [HR]: 0.46; 95% confidence interval [CI]: 0.22-0.92, P = .03) in the univariate analysis, but this was not true after adjustment of multiple variables in the multivariate analysis (HR: 0.53; 95% CI: 0.25-1.09, P = .09). Neither induction (depleting vs nondepleting induction immunosuppression) nor early steroid withdrawal was associated with the risk for avascular necrosis. Of these recipients with avascular necrosis, 34 underwent hip replacement surgery.
Comparison of Demographic, Immunologic, and Donor Characteristics With the Control Group
After the identification of the avascular necrosis group, controls were selected using incidence density sampling based on the post-transplant interval for the diagnosis of avascular necrosis. Although an attempt was made to select 3 controls for every case, it was not possible in all cases. For the 88 avascular necrosis group, 257 controls instead of 264 were identified. For each avascular necrosis case, at least 1control was identified. The comparison of the various recipient, immunologic, and donor factors between the 2 groups are outlined in
Demographic, Immunologic, and Donor Data Comparing the 2 Groups.
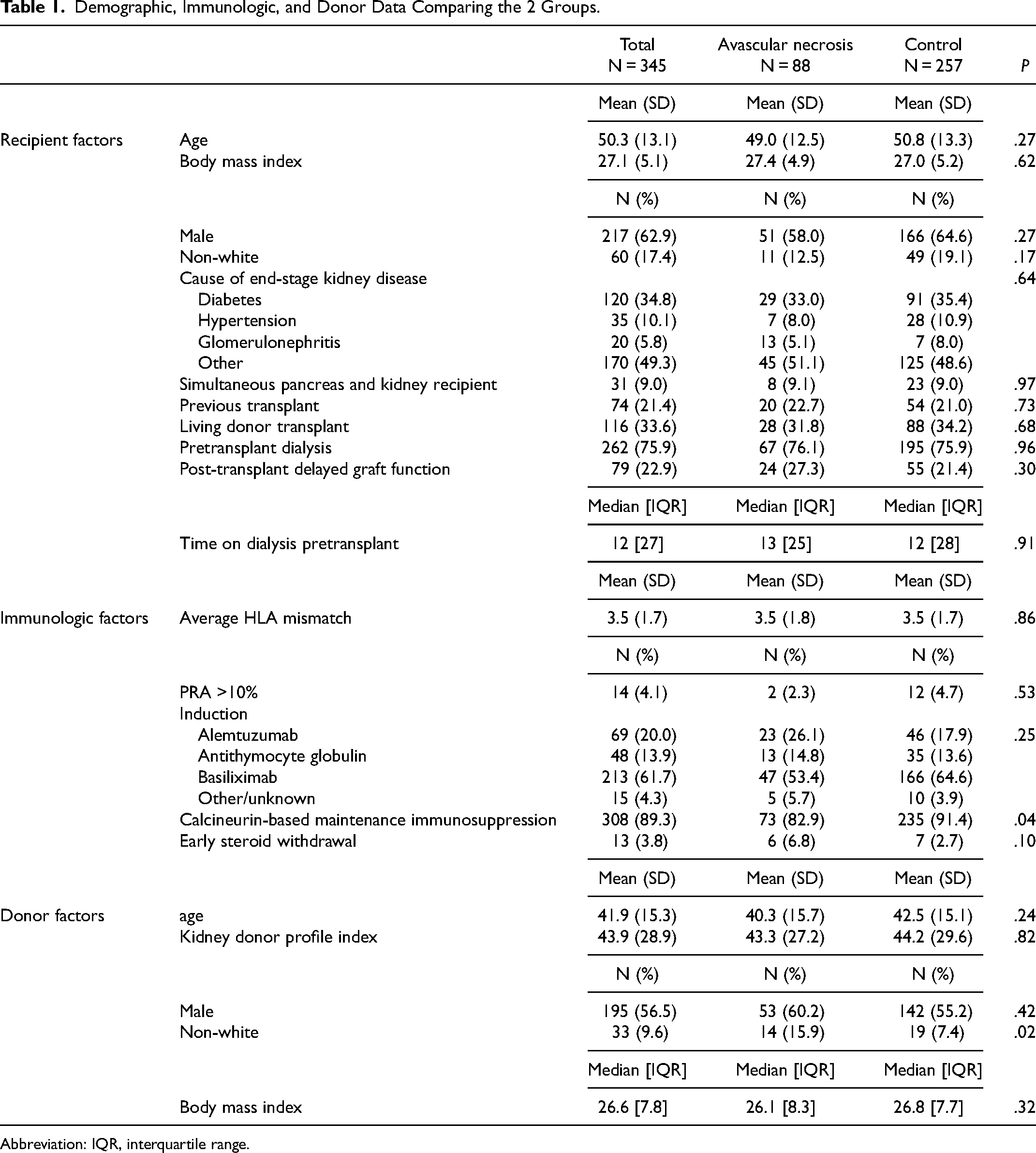
Abbreviation: IQR, interquartile range.
Acute Rejection
Among the entire cohort, 18 had acute rejection, 1 (1%) was in the avascular necrosis group and 17 (7%) (P = .05) in the control group. In the univariable analysis, avascular necrosis was not associated with rejection (HR: 0.17, 95% CI: 0.02-1.27, P = .08). As with the univariable analysis, the multivariable analysis showed no association between avascular necrosis and risk for acute rejection (HR: 0.17, 95% CI: 0.02-1.27, P = .08). The only variable significantly associated with an increased risk of acute rejection was a higher degree of human leukocyte antigen mismatch (HR: 1.44, 95% CI: 1.03-1.99, P = .03) (data not shown).
Death-Censored Graft Failure
Among the entire cohort, 82 had death-censored graft failure, 25 (28%) were in the avascular necrosis group and 57 (22%) (P = .24) were in the control group. The mean interval for death-censored graft failure was significantly shorter in the avascular necrosis group compared to the control group 3.35 (3.93) versus 5.90 (4.37) years (P = .009). In a univariable analysis of the risk factors of death-censored graft failure, avascular necrosis was not found to be associated with death-censored graft failure (HR: 1.37, 95% CI: 0.85-2.20, P = .20). In multivariable analysis after adjustment of multiple variables, avascular necrosis was not associated with death-censored graft failure (HR: 1.48, 95% CI: 0.90-2.42, P = .12). Older recipient age was protective for death-censored graft failure in multivariable analyses (HR: 0.96, 95% CI: 0.94-0.98, P = <.001), and older donor age was associated with increased risk (HR: 1.03, 95% CI: 1.01-1.04, P = .002).
Patient Mortality
Among the entire cohort, 132 patients died: 39 (44%) were from the avascular necrosis group and 93 (36%) (P = .18) from the control group. The mean interval for patient death was not statistically different in the avascular necrosis group compared to the control 8.6 (4.39) versus 9.46 (4.62) years (P = .13). In univariate analysis, avascular necrosis was not associated with the risk for patient death (HR: 1.33, 95% CI: 0.91-1.94, P = .14) (
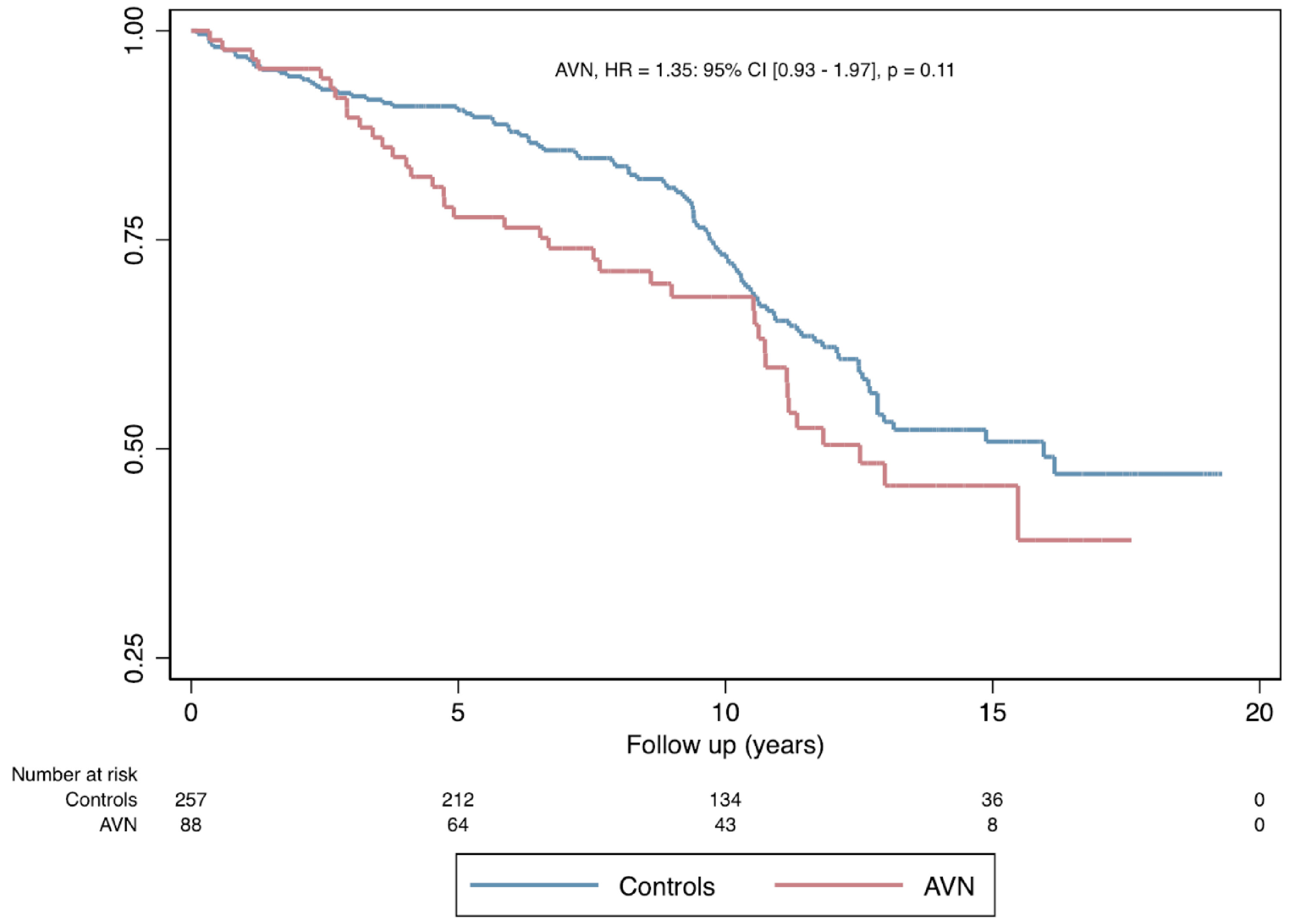
Patient survival probability after AVN. No significant difference in the rate of patient survival between the avascular necrosis group and the control (P = .11). Abbreviation: AVN, avascular necrosis.
Risk factors for patient's death.
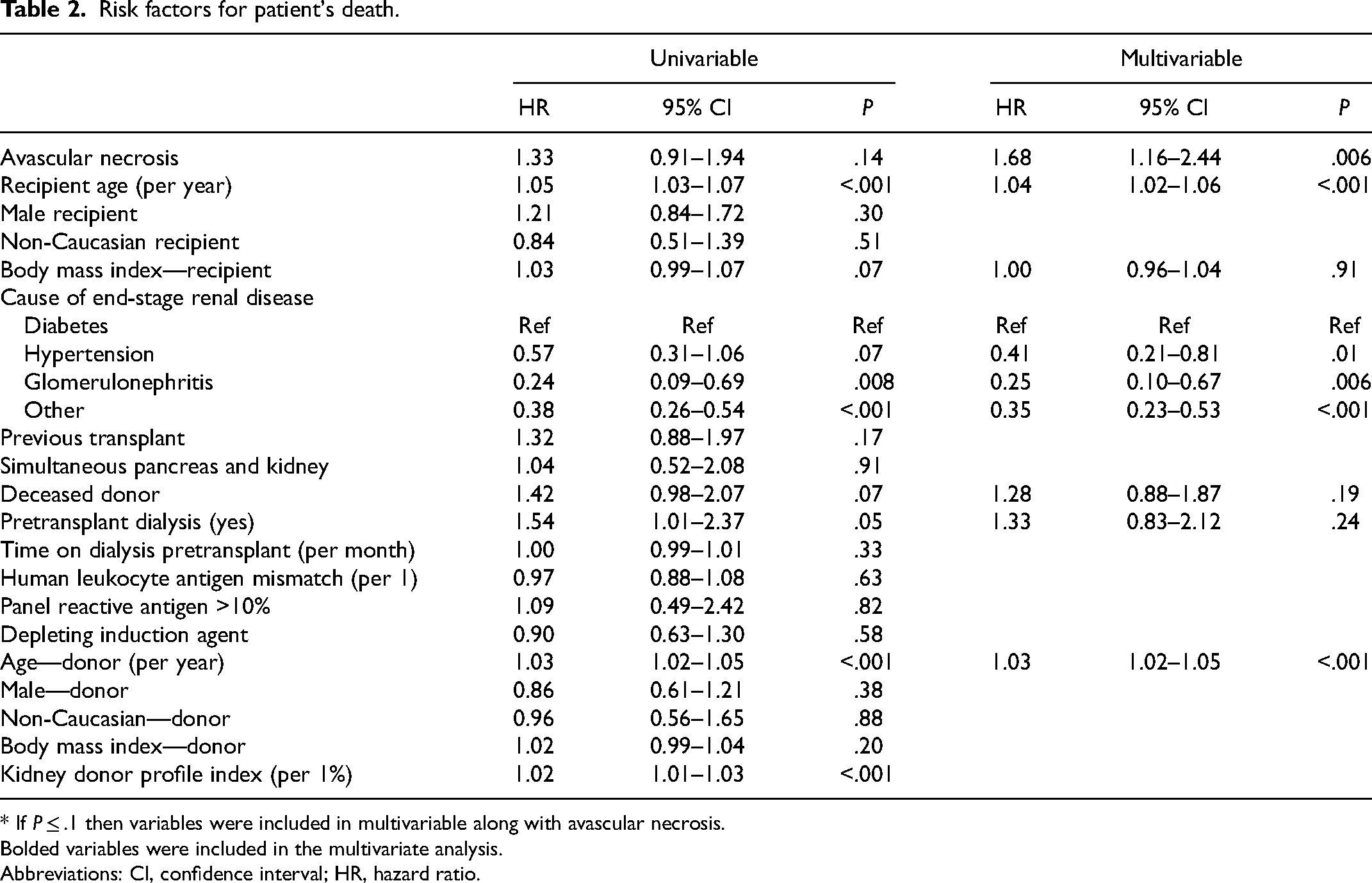
* If P ≤ .1 then variables were included in multivariable along with avascular necrosis.
Bolded variables were included in the multivariate analysis.
Abbreviations: CI, confidence interval; HR, hazard ratio.
Discussion
In this cohort of 88 kidney transplant recipients with post-transplant avascular necrosis over the study period of 18 years, avascular necrosis was not associated with increased or decreased risk for some of the kidney transplant-specific outcomes including acute rejection and death-censored graft failure. Compared to the selected controls based on the incidence density sampling, these recipients with avascular necrosis had a higher risk of mortality. This increased risk of mortality among the avascular necrosis group may be related to the presence of various other comorbid conditions and may not be directly related to avascular necrosis.
The current literature surrounding avascular necrosis does not focus on kidney transplant-specific risk factors and outcomes associated with avascular necrosis. Studies have focused on avascular necrosis occurrence after fractures and treatment of the development of dysplasia of the hip. Avascular necrosis usually involves the end part of a long bone (epiphysis), such as the femoral and humeral heads and the femoral condyles, but small bones can also be affected. Similar to this study, in clinical practice, avascular necrosis was most commonly encountered in the hip joints. 4
No medical treatment has proven effective in preventing or arresting the disease process of avascular necrosis. Conservative measures include limited weight bearing with crutches and pain medications. Treatment with a bisphosphonate, vasoactive prostaglandin, statin, extracorporeal shockwave therapy, and hyperbaric oxygen therapy was shown to be beneficial and was a reasonable initial course of action if the involved segment was smaller.5–7 Advanced avascular necrosis, would eventually require joint replacement surgery. Recently, in one report among 101 kidney transplant recipients, 23% had joint replacement surgery attributed to avascular necrosis. 8
Similar to the previous studies, the overall incidence of avascular necrosis has decreased in the modern era and in this cohort, <2% had this debilitating condition. 1 Although not statistically significant, similar to the previous reports, calcineurin inhibitor-based immunosuppressive agents were protective against avascular necrosis. 1 In this study, early steroid withdrawal was not associated with decreased risk of avascular necrosis. Additionally, in the general population, patients with diabetes may be at higher risk for the development of avascular necrosis, however, in this cohort, there was no association between the causes of end-stage kidney disease as diabetes and avascular necrosis. 9
This study has the expected limitations of a single-center observational study, reflecting the specific population and clinical approach. The data were limited by the small sample size. As the focus of the study was on transplant-specific outcomes, details of other outcomes including physical and functional morbidity were not assessed. As there are multiple studies addressing some of the risk factors for avascular necrosis, details of some known risk factors (eg, calcium/phosphorus metabolism; use of statin, bisphosphonate, etc) were not evaluated. Due to the study being a retrospective observational study, the exact cause of death among recipients who have passed away could not be reported. This evaluation provided more granular data than what is available in registries. Another potential advantage of this single-center evaluation is that it reflects a more homogeneous clinical approach and medical management, in contrast to registry data involving multiple centers.
Conclusion
The incidence of avascular necrosis has decreased in the modern era, but it is still an important complication that is associated with an increased risk of mortality.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Virginia Lee Cook Foundation.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statements
The data that support the findings of this study are available from the corresponding author upon reasonable request.
