Abstract
Introduction
Kidney transplantation is the optimal treatment for end-stage renal disease. 1 Kidney function maintains the homeostatic balance of calcium and phosphate ions necessary for proper bone development and mineralization. 2 Chronic kidney disease (CKD) culminates in irregular bone remodelling as a result of abnormal mineralization. 3 Chronic disease may lead to the development of hyperparathyroidism, adynamic bone disease, uremic osteoporosis, and other derangements of bone metabolism 4 that may persist even among patients who achieve normal graft function posttransplant. 5 Moreover, posttransplant immunosuppressive agents contribute to alterations of normal bone metabolism. Glucocorticoids alter osteoblast and osteoclast activity, promoting bone resorption over bone deposition, 6 while calcineurin inhibitors (CNI) have been associated with high-turnover bone loss in animal models. 7
The unique physiology of kidney transplant recipients places them at higher risk for bone fractures, 8 with an estimated threefold increase in fracture incidence when compared to the general population. 9 The reported incidence of fractures among kidney transplant recipients is widely variable in the literature. A systematic review conducted by Naylor et al. 10 found incidence rates ranging from 3.3 to 99.6 fractures per 1000 person-years and 5-year cumulative incidence ranging from 0.85% to 27%. The pronounced variability in current literature is due, in part, to differences in fracture sites assessed and in the methods of ascertaining fractures. 10 Few of the studies reviewed by Naylor et al. 10 included patients transplanted after the year 2006. Recent studies have assessed the incidence of fractures in more contemporary kidney transplant populations. Iseri et al. 11 reported an incidence rate of 13.5 major fractures (hip, spine, humerus, and forearm) per 1000 patient-years among 3992 Swedish patients between 2005 and 2016. Among 4821 Canadian patients between 1994 and 2009, Naylor et al. 9 found a 5.5% 10-year cumulative incidence of non-vertebral major fractures (composite of hip, forearm, and proximal humerus) and a 3.5% 3-year cumulative incidence of any fracture. Overall, few studies exist that assess the risk factors and incidence of fracture in contemporary Canadian kidney transplant populations.
Identification of general, and transplant-specific, risk factors for fractures is necessary to guide fracture risk prognostication and preventative strategies. Older recipient age,10–13 female sex,10–13 white race, 13 diabetes,10–13 older donor age, 12 increased time on dialysis,10–12 and history of fracture10,13 have been repeatedly demonstrated to be independent risk factors for fractures. Falls during hospitalization prior to transplant 12 and hyperparathyroidism 14 may also increase fracture risk. However, no studies have assessed the risk factors for recurrent fractures specifically among kidney transplant recipients. Since recurrent fractures have been associated with increased mortality in the general population, 15 identification of contributory risk factors is important in the prevention of recurrent fractures and its associated morbidity and mortality. Given the heterogeneity in the reported fracture burden and the lack of data on risk factors for recurrent fractures, the primary objective was to identify the incidence of all fractures and fractures categorized as major osteoporotic fractures. The secondary objective was to assess risk factors for first and recurrent fractures.
Methods
Design
This retrospective cohort study was approved by the institutional Research Ethics Board and informed consent was obtained from all individual participants included in the study.
Setting
The study was conducted at a large transplant centre in an academic, tertiary care hospital in Ontario, Canada.
Population
The population of interest included adults (age ≥ 18 years) transplanted between January 1, 2004, and December 31, 2013. A total of 1773 adults were transplanted within the study period. The mean recipient age at transplant was 47.6 years and 47.5% received transplants from living donors. The population was 61.7% male, predominately of white race (68.3%), mean body mass index (BMI) of 26.7 kg/m2, and 30.0% had experienced a fracture pretransplant.
Sampling
All patients transplanted within the time period were eligible for inclusion into the study cohort regardless of biological sex, race, cause of end stage renal disease, donor type (ie, living or deceased). Participants were excluded from the study if they: received a previous kidney transplant, received a simultaneous non-kidney transplant, or received a graft that was deemed non-functional within 14 days of transplant, necessitating a return to dialysis (primary graft non-function). Data were collected until there was documentation that patients had experienced the outcome under investigation (first or recurrent fracture), developed graft failure, died, or reached the last study follow-up day (December 31, 2014).
Data Collection
Clinically relevant recipient variables assessed were age, sex, race, BMI, history of pretransplant fracture, peak panel reactive antibody (PRA), time on dialysis, history of diabetes, history of smoking, and history of cardiovascular disease. Donor variables collected were age, sex, BMI, living or deceased donation, and history of hypertension (yes/no). Delayed graft function (yes/no), induction type (depleting/non-depleting agent), and CNI regimen at discharge (tacrolimus/cyclosporine) were transplant variables assessed as potential confounders.
Fractures at all skeletal sites were recorded and subsequently grouped as: hip (femoral neck), foot/toes, other lower extremity (femoral shaft and tibia/fibula), pelvic girdle (pelvis, sacrum, and coccyx), rib cage (ribs and sternum), thoracolumbar vertebrae, shoulder girdle (scapula and clavicle), proximal humerus, forearm (radius/ulna), other upper extremity (distal humerus and hand), head/facial bones, and neck (cervical spine). Major osteoporotic fractures (MOFs) were classified as hip, thoracolumbar vertebrae, proximal humerus or forearm fractures for separate analyses. Fractures were confirmed using radiological reports or transcription notes of the medical team. A fracture was reported as acute if the clinical note or radiology report indicated as such. In the case of fractures only reported on a radiological report, a fracture was considered acute if it was a new finding compared to a previous radiologic exam in the same anatomical area performed less than 365 days prior. When a posttransplant fracture was reported to have occurred with no date specified in the clinical note, the date of the note was used as a proxy for the fracture date, and the fracture was classified as likely acute. Both acute and likely acute fractures were included in the analyses. Fractures were included regardless of level of trauma since determination of level of trauma was highly subjective and suboptimal bone health has been found to be associated with increased risk of both low- and high-trauma fractures. 16
Data Analysis
Baseline demographic and clinical variables were described using summary statistics. The distribution of fractures by anatomical site was described by the number of patients who had at least one fracture at each of the skeletal sites, and the number of total fracture cases reported at each skeletal site.
Kaplan–Meier methods were used to assess and graph the cumulative incidence of any first fracture and any MOF over time. Multivariable time-dependent Cox proportional hazards models were used to identify risk factors for first posttransplant fracture. To reduce the potential bias caused by excess graft loss and mortality in the period shortly following transplantation, risk factors for any first fracture were additionally analyzed using the time origin as 3-months posttransplant and 6-months posttransplant. Both of these analyses yielded similar results, thus only the former was selected for presentation.
Multivariable analysis of the effect of risk factors on recurrent fractures was performed using the shared frailty model, allowing the correlation between recurrent fractures occurring within the same patient to be accounted for as a frailty effect. Frailty effect was assigned a gamma distribution while hazard distribution was analyzed using lognormal, exponential, Weibull, and Cox models. Analyses using the exponential and Weibull hazard distributions yielded similar results as the Cox model and were thus excluded from presentation. Analyses were performed using the Stata/MP version 16 (College Station, Texas). Missing values were imputed using multiple imputation method.
Procedure
Basic demographic and baseline clinical information was collected from the Comprehensive Renal Transplant Research Information System (CoReTRIS), a post-kidney transplant research database developed at the study centre. Fracture events were ascertained from a review of the electronic medical records used by the kidney transplant program at the study centre.
Results
After applying the exclusion criteria, 1285 participants were included in the final study cohort (Figure 1). Table 1 outlines the baseline characteristics of participants in the study sample. The mean recipient age was 50.2 years, the majority were of white race (65.2%) and more than half of the population was male (61.0%). Additionally, 27.2% of patients experienced fracture prior to transplant.
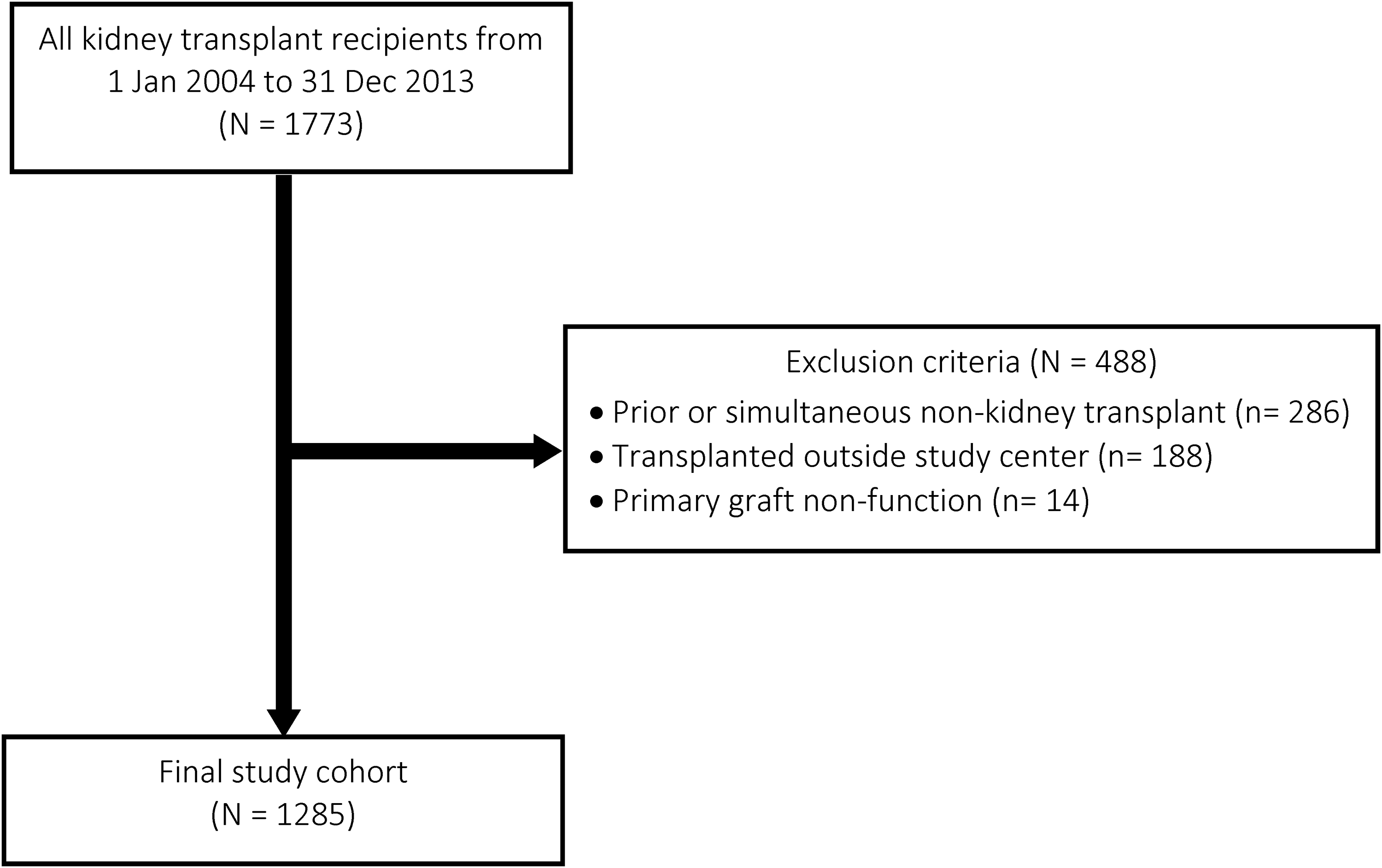
Patient selection for first and recurrent fractures.
Baseline characteristics of sample with donor information.
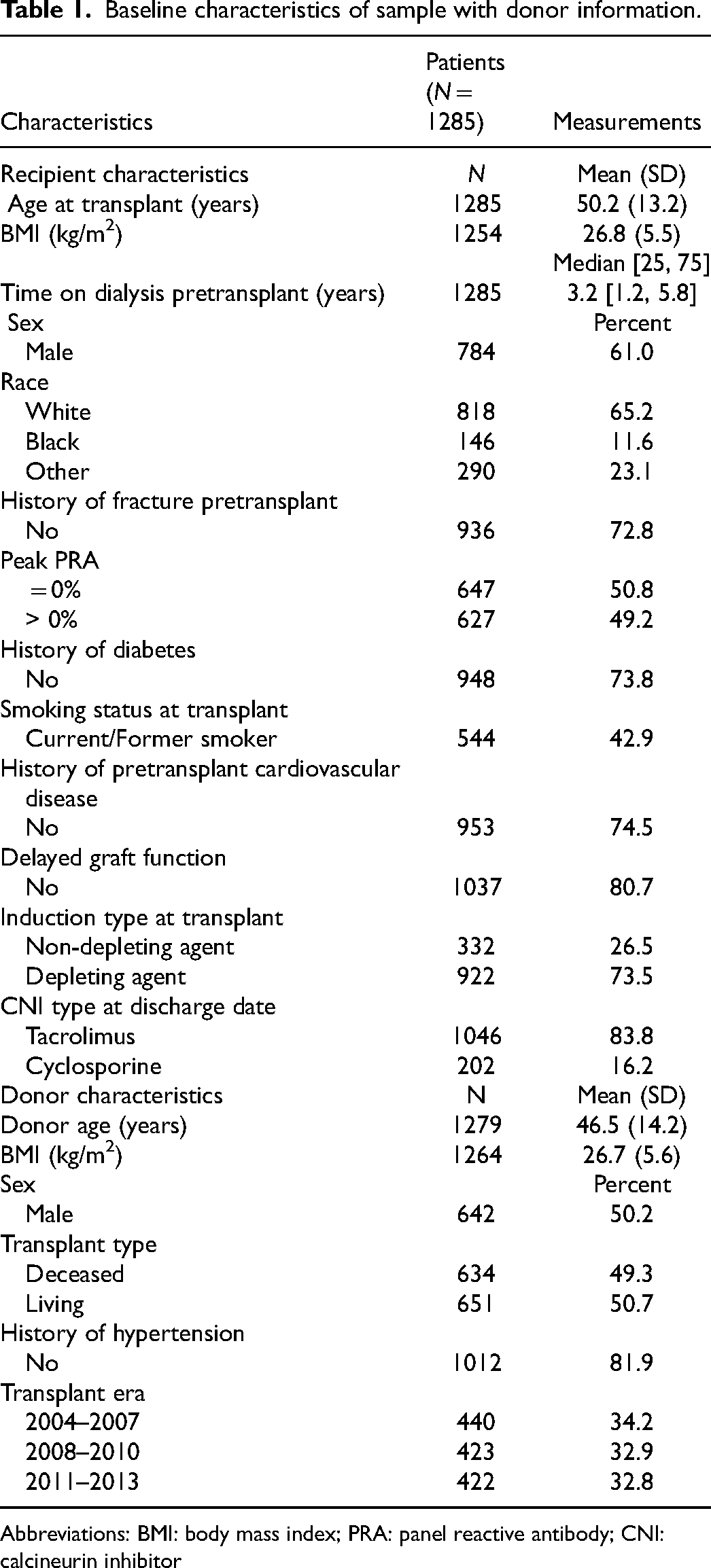
Abbreviations: BMI: body mass index; PRA: panel reactive antibody; CNI: calcineurin inhibitor
Incidence of First Fracture and Fractures Categorized as major Fractures
For the outcome of any first fracture, there were 174 events observed in 1285 patients, with a median follow-up time of 4.00 years (IQR: 2.13–6.31). There were 85 first MOFs observed in 1285 patients, with a median follow-up time of 4.33 years (IQR: 2.38–6.60). The 10-year cumulative incidence observed for any first fracture and any first MOF was 27.1% (95% CI: 22.5, 32.4) and 17.8% (95% CI: 13.4, 23.5), respectively (Figure 2). The distribution of all posttransplant fractures by anatomical site is shown in Figure 3. Fractures of the forearm (radius/ulna), feet/toes, and other lower extremity bones were the most frequent while neck fractures (cervical spine) were the least frequent. Over the course of follow-up, the incidence rate of any first fractures was 3.08 per 100 person-years (95% CI: 2.65, 3.57), while the incidence rate of all MOFs was 1.42 per 100 person-years (95% CI: 1.15, 1.76) (Supplemental Table 1).
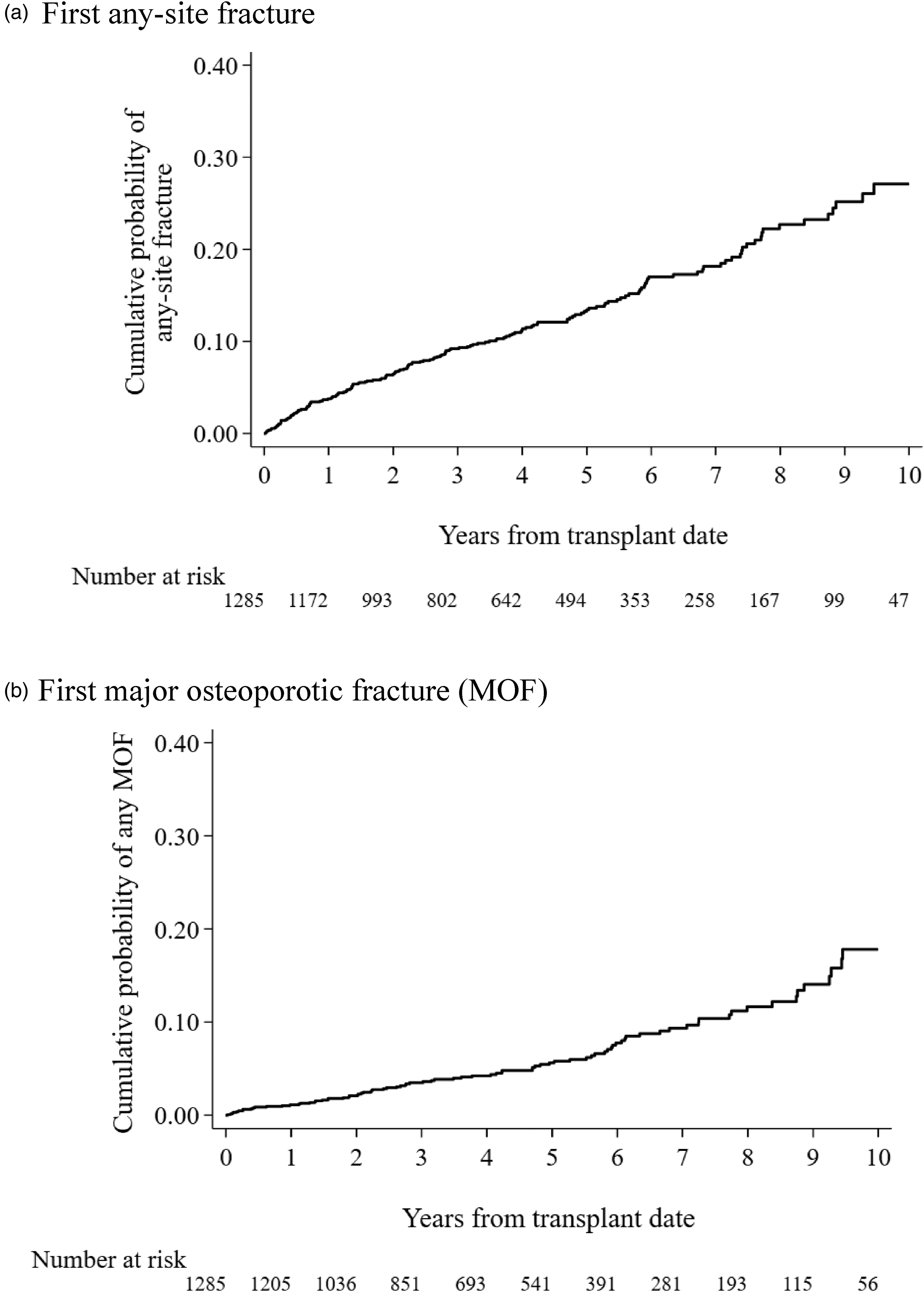
Kaplan–Meier curves of overall fracture incidence (a) overall incidence of any fracture (b) overall incidence of major osteoporotic fractures.
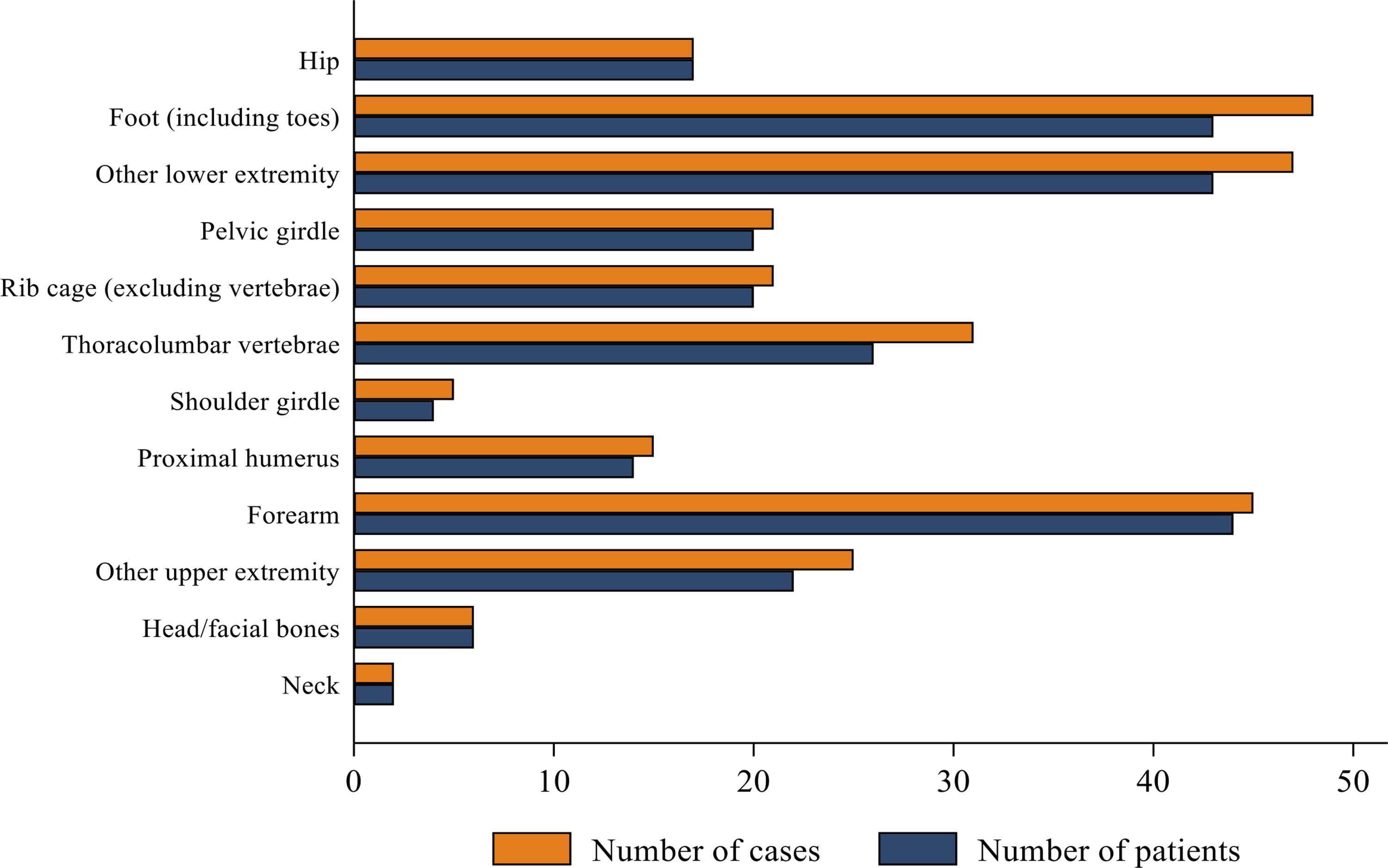
Distribution of posttransplant fracture sites for any fracture.
Risk Factors for First Fractures
Using the transplant date as the time origin, the multivariable Cox proportional hazards model demonstrated female recipient sex (HR = 1.64 [95% CI: 1.20, 2.26]), history of any fracture prior to transplant (HR = 1.54 [95% CI: 1.12, 2.11]), and recipient history of diabetes (HR = 1.85 [95% CI: 1.29, 2.65]) to be independent risk factors for any first fracture ( Table 2 ). Both increased recipient BMI (HR = 0.97 per 1 kg/m2 [95% CI: 0.94, 1.00)]) and donor BMI (HR = 0.97 per 1 kg/m2 [95% CI: 0.94, 1.00)]) were found to have a statistically significant protective effect against any first fracture.
Multi-variable Cox model for effect of risk factors on any first posttransplant fracture.
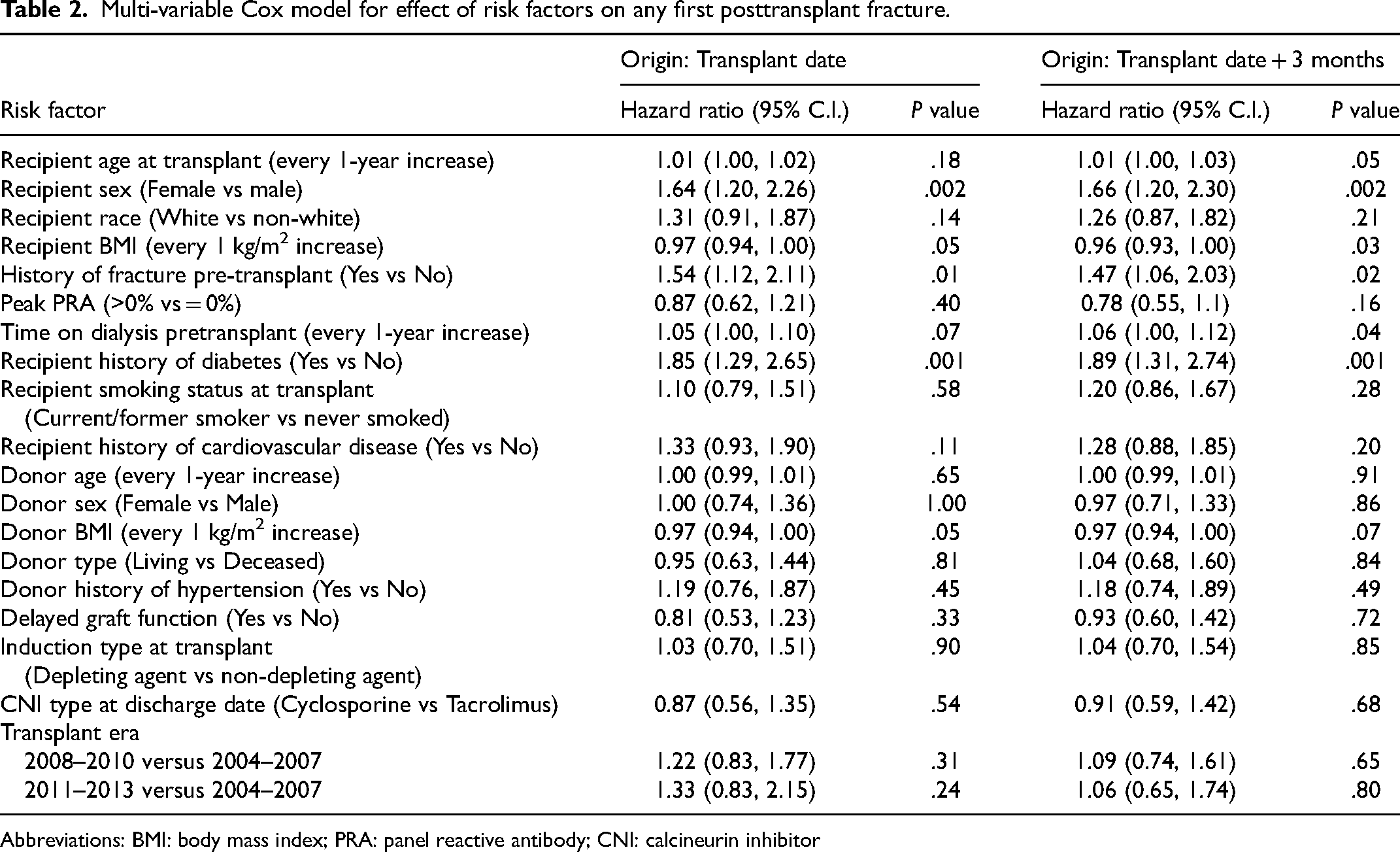
Abbreviations: BMI: body mass index; PRA: panel reactive antibody; CNI: calcineurin inhibitor
The fracture risk factors persisted with similar magnitudes of effect when the time origin of analysis was at 3-months posttransplant. In addition, 1-year increases in recipient age at transplant (HR = 1.01 [95% CI: 1.00, 1.03]) and 1-year increases in time on pretransplant dialysis (HR = 1.06 [95% CI: 1.00, 1.12]) were found to increase risk of first fracture among patients who were alive with a functional graft up to 3-months posttransplant. While greater recipient BMI (HR = 0.96 [95% CI: 0.93, 1.00)]) continued to reduce the risk of fracture at the time origin of 3-months posttransplant, donor BMI did not demonstrate this effect (Table 2).
Risk Factors for Recurrent Fractures
Application of the shared frailty model using a lognormal hazard distribution demonstrated female recipient sex (Coefficient = −0.56 [95% CI: −0.97, −0.14]) and recipient history of diabetes (Coefficient = −0.65 [95% CI: −1.13, −0.18]) to be associated with increased risk of recurrent fracture ( Table 3 ). Patients transplanted between 2011 and 2013 had higher risk of recurrent fracture compared to those transplanted between 2004 and 2007 (Coefficient = −0.56 [95% CI: −1.09, −0.03]). There was a reduction in risk of recurrent fracture per 1 kg/m2 increase of recipient BMI (Coefficient = 0.04 [95% CI: 0.01, 0.08). Using the Cox model for the hazard distribution, recipient female sex (HR = 1.74 [95% CI: 1.21, 2.51]), history of diabetes (HR = 1.76 [95% CI: 1.17, 2.66]) and receiving a transplant from 2011 to 2013 versus 2004 to 2007 (HR = 1.81 [95% CI: 1.07, 3.08]) were associated with an increased risk of recurrent fracture (Table 3).
Multi-variable shared frailty model for effect of risk factors on recurrent fractures.
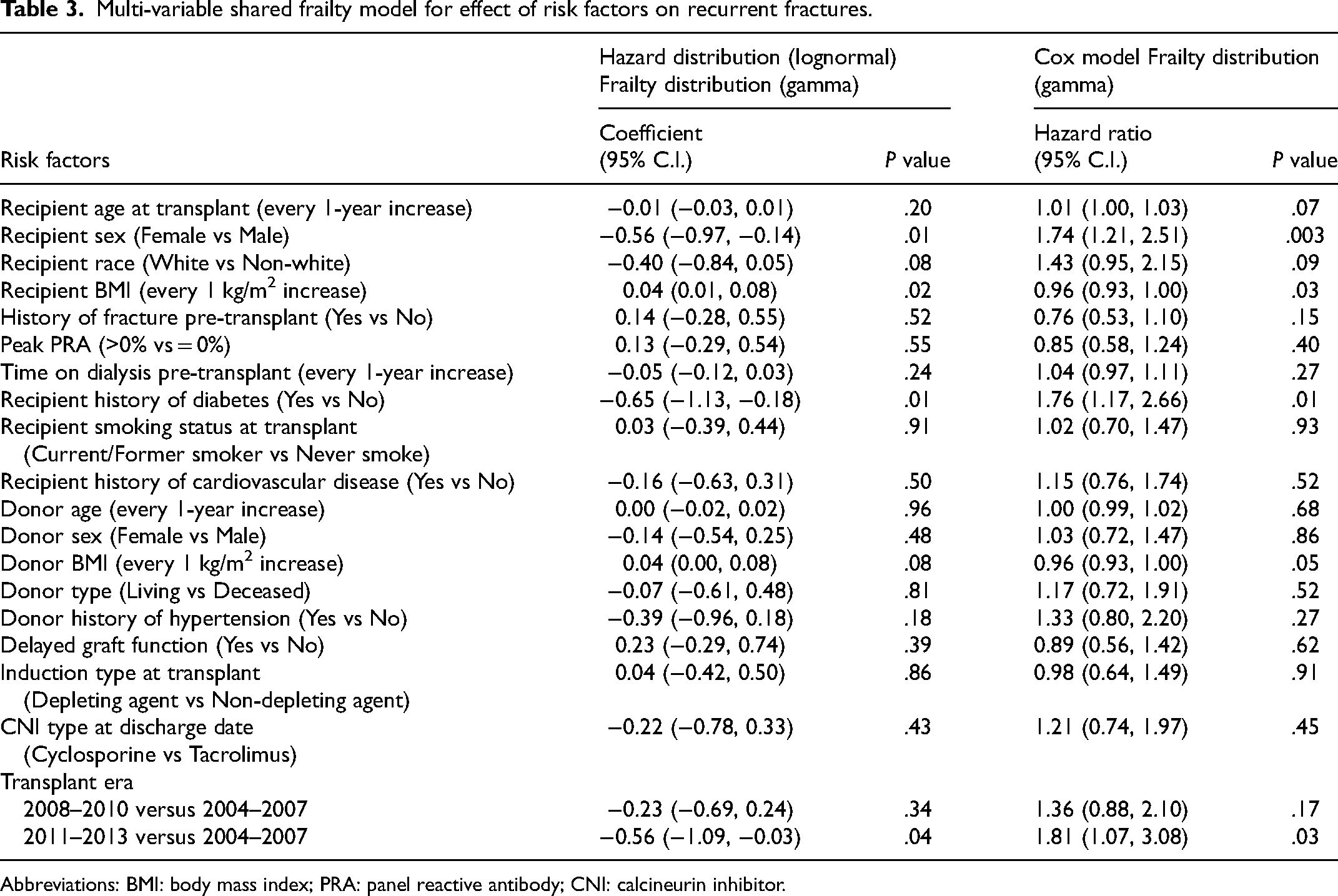
Abbreviations: BMI: body mass index; PRA: panel reactive antibody; CNI: calcineurin inhibitor.
Similar results were observed with exponential and Weibull hazard distributions. On all 4 hazard distribution models analyzed (lognormal, exponential, Weibull, and Cox), every 1 kg/m2 increase in recipient BMI was associated with a statistically significant protective effect against recurrent fracture. In all models used, the likelihood ratio tests for the significance of the frailty effect were statistically significant. As such, the frailty effect could not be ignored. The additional models are shown in Supplemental Table 2.
Discussion
Assessment of the burden of fractures in a contemporary kidney transplant population is important to inform clinical management. This study found a 10-year cumulative incidence of any first fracture and any MOF of 27.1% and 17.8%, respectively, which exceeds what has previously been reported in Canadian transplant populations. A Manitoba population reported 10-year cumulative incidence of MOFs to be 6.3% (95% CI: 3.4, 9.2) 17 and an Ontario population reported a 3-year incidence of 3.5% (95% CI: 3.0, 4.1%) for all fractures and a 10-year incidence of 5.5% (95% CI: 4.6, 6.5) for MOFs. 9 This variation may be attributable to several differences in methodology. Firstly, the studies relied on administrative data, such as billing claims from medical databases, to ascertain fracture events, whereas fracture events in this study were ascertained from individual patient medical records. Reliance on administrative data to ascertain fracture events limits the inclusion of fracture diagnoses that occurred outside the province of study, contributing to the underestimation of fracture incidence. Furthermore, Naylor et al. 9 excluded vertebral fractures from their analysis. Thoracolumbar vertebral fractures included in our cohort accounted for 28% of MOFs. Similarly, other authors have reported vertebral fractures accounting for 9%–33% of MOFs among populations of kidney transplant recipients.11,13–15,18 Hence, exclusion of vertebral fractures underestimates the burden of MOFs.
Multivariable Cox proportional hazards model demonstrated female recipient sex, history of fracture prior to transplant, and recipient history of diabetes to be independent risk factors for any first fracture posttransplant, which was consistent with previous studies.10–13,18 Unique to this study was the assessment of risk factors for recurrent fractures in the patient sample. Recipient female sex and history of diabetes were found to persist as risk factors for recurrent fractures, with similar magnitudes of effect to those observed for any first posttransplant fracture. Pretransplant history of fracture did not persist as a risk factor for recurrent fractures. The analyses also found increasing BMI to be associated with a decreased risk of both first and recurrent fracture. Although some studies have reported no association between BMI and risk of first fracture,15,19 a protective effect of BMI on fracture risk has been previously demonstrated.13,20 The effects of recipient sex, BMI, and history of diabetes on the risk of recurrent fracture were found to be reproducible regardless of whether the hazard distribution was modelled using a lognormal, exponential or Weibull model. Furthermore, patients transplanted in the most recent era, 2011 to 2013, had higher risk of recurrent fracture compared to those transplanted between 2004 and 2007. This association was observed in the lognormal, Weibull and Cox hazard distributions, and was likely due to the expansion of eligibility criteria for recipients. Patients began to receive transplants at older ages and with a greater number of comorbidities, which may explain the increased risk of recurrent fracture in the more recent era. 21
This study presents several contributions to the literature. To our knowledge, this is the first study rigorously testing the effect of general and transplant-specific risk factors on recurrent fractures in a cohort that more closely reflects current paradigms of transplant care. The ascertainment of fracture diagnoses from individual patient medical records captured fracture events that occurred or were treated outside patients’ immediate geographical area, information that might otherwise not be available in the administrative databases used in other studies. There are notable limitations. In 25% of posttransplant fracture cases used in the analyses, we were unable to determine with reasonable certainty whether the fracture occurred on the date reported or within the preceding year. While this may have affected the estimation of fracture incidence, it is not expected that this had a considerable effect on the risk factors identified, as this was a randomly distributed effect. Missing data precluded the inclusion of some fracture risk factors that have been identified in the general population (eg parental history of hip fracture, glucocorticoid use, history of rheumatoid arthritis, bone mineral density). Naylor et al. 17 found no difference in the distribution of these variables when comparing patients who experienced posttransplant MOF and patients who did not, suggesting that these risk factors may not contribute to MOF risk in these patients to the same extent as in the general population. 17 For instance, patients who experienced MOFs had the same bone mineral density T-scores as those who did not. 17 Lastly, this study did not report on the use of anti-resorptive therapy or calcium and vitamin D supplementation on the individual patient level. As of 2010, the protocol was to maintain patients on 1000 IU vitamin D daily, with a target level of greater or equal to 75 nmol/L, and 1000 mg of calcium daily, either from diet or supplements. Prior to 2010 there was no formal, protocolized approach.
The findings suggested that some risk factors for first fracture may not inform risk of subsequent fractures. Early identification of patients at risk for recurrent fractures can guide frequency and type of monitoring as well as optimization of pharmacological, lifestyle, and counselling interventions geared towards reducing fall and fracture risk, and associated morbidity and mortality. For example, increased physical activity and resistance exercises have been found to have protective effects against bone fractures 22 and improve overall bone health in kidney transplant populations. 23 Beyond assessing risk factors for fractures, the effect of first and recurrent fracture events on patient outcomes was not analyzed in this study and is an avenue for future research. This will provide insight into the consequential outcomes associated with fractures and their subsequent management.
Conclusion
Today's kidney transplant recipients remain at increased risk of fracture compared to the general population. Identification of risk factors for first and recurrent fractures among current-era recipients arms clinical teams with the information necessary to guide an individualized, multi-faceted approach to fracture risk reduction and overall bone health maintenance.
Supplemental Material
sj-docx-1-pit-10.1177_15269248221145034 - Supplemental material for Risk Factors for First and Recurrent Fractures among Kidney Transplant Recipients
Supplemental material, sj-docx-1-pit-10.1177_15269248221145034 for Risk Factors for First and Recurrent Fractures among Kidney Transplant Recipients by Norman Atagu, Stefani Mihilli, Huong Thao Nguyen, Alicia Wu, Olusegun Famure, Yanhong Li and S. Joseph Kim in Progress in Transplantation
Supplemental Material
sj-docx-2-pit-10.1177_15269248221145034 - Supplemental material for Risk Factors for First and Recurrent Fractures among Kidney Transplant Recipients
Supplemental material, sj-docx-2-pit-10.1177_15269248221145034 for Risk Factors for First and Recurrent Fractures among Kidney Transplant Recipients by Norman Atagu, Stefani Mihilli, Huong Thao Nguyen, Alicia Wu, Olusegun Famure, Yanhong Li and S. Joseph Kim in Progress in Transplantation
Footnotes
Authors' Note
Norman Atagu, Ajmera Transplant Centre, Toronto General Hospital, University Health Network, Toronto, Ontario, Canada.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
