Abstract
Introduction
Solid organ transplantation is a life-saving treatment for individuals with end-stage diseases of the heart, lung, liver, kidney, and pancreas. With better medical management and immunosuppression regimens, transplant recipients are living longer and there is a growing need to improve their long-term outcomes. 1 Physical rehabilitation, which includes structured exercise programs such as treadmill walking, cycling, and lifting weights is beneficial for transplant candidates and recipients as it improves exercise capacity, physical function, and quality of life.2,3 The authors of systematic reviews and meta-analyses have been unable to draw strong conclusions from the data due to small sample sizes and lack of consistency in outcomes reported in clinical trials of exercise training.3–6 For example, in a systematic review of 35 randomized controlled trials (RCTs) examining the effectiveness of exercise training, the authors identified that a wide range of 26 outcome domains and 126 different outcome measurement instruments were used among these trials, most of them related to body functions and structures (ie, aerobic capacity, muscle strength, and body composition). 7 The synthesis of findings from RCTs of physical rehabilitation in transplantation using meta-analysis would strengthen the evidence for physical rehabilitation. However, the heterogeneity of outcomes limits the ability to conduct meta-analysis, as has been noted by our group3,7 and others.5,6
To advance the evidence in this field, it is essential that common outcomes are used in future clinical trials, which could improve the ability to compare results across studies and also to conduct meta-analyses.8,9 Hence, the development of a core outcome set (COS), defined as the minimum set of outcomes that have been agreed upon to be measured and reported in all clinical trials of a specific clinical population, is considered a key initiative. 9 We believe that establishing COS for clinical trials involving the transplant population can help to improve research methodology, to reduce risk of bias due to selective reporting (ie, only those outcomes with significant differences being reported in the literature), and to address issues of inconsistency in outcomes reported across trials in the area of physical rehabilitation and transplantation.
Study Objective
The purpose of this study was to explore the perspectives of patients and professionals (researchers and clinicians) to identify the key outcomes domains that are important and relevant for physical rehabilitation of transplant recipients in 3 different phases: pretransplant, early posttransplant (within 1-year posttransplant), and late posttransplant (more than 1-year posttransplant). As a secondary objective, we aimed to differentiate the most important outcome domains according to different stakeholder subgroups: patients and professionals.
Methods
Design
We used a Delphi method to identify which outcome domains were most important for adults undergoing transplantation. 9 The study was approved by the University Health Sciences Research Ethics Board (protocol number 33783), and all participants provided informed consent prior to participation. This study was registered in the Core Outcome Measures in Effectiveness Trials (COMET) initiative website: http://www.comet-initiative.org/studies/details/977.
Setting
The electronic survey was distributed by email from a university between May and July 2017. Participants completed the survey in the community, at their home, or workplace.
Population
The population from which we drew participants for the Delphi survey included transplant recipients and professionals (clinicians and researchers) in the field of transplantation across all organ groups. The participants were restricted to residents of Canada. In 2017, there were 2352 solid organ transplants done in Canada of which 36% were in female recipients, and the majority of transplant recipients (52%) were between the ages of 45 and 64 years. 10 Among transplant professionals in Canada, there are 58 members of the Canadian Society of Transplantation Allied Health and Nursing group (83% female), which represents the types of professionals working in transplant rehabilitation programs (personal communication, Yvonne Squires, Project Manager, and Canadian Society of Transplantation, September 9, 2022).
Sampling
Purposeful sampling was used to recruit participants for the Delphi survey. Invitation letters with a consent statement and a short questionnaire related to eligibility to participate in the study were emailed to potential participants through the Canadian Donation and Transplantation Research Program (CDTRP), which includes researchers (N = 151), and a patient family donor group (N = 21); and the Canadian Network for Rehabilitation and Exercise for Solid Organ Transplant Optimal Recovery (CAN-RESTORE) rehabilitation program directory to target clinicians in Canadian rehabilitation programs (20 programs across 5 Canadian provinces). A snowball sampling recruitment strategy, in which participants were asked to voluntarily provide emails from potentially eligible colleagues and peers, was also used to increase the number of participants.
Patients were included if they were adults (≥ 18 years old) and had received an organ transplant. Clinicians were included if they were healthcare professionals working in transplant rehabilitation programs. Researchers were included if they were scientists with experience conducting research on exercise or rehabilitation. Individuals who did not have email or internet access; or were unable to understand and complete the questionnaire in English were excluded from the study.
Data Collection
Description of Questionnaire
The questionnaire used in the first round of the Delphi process included a section on participant characteristics (eg, transplant characteristics for patients; employment and transplant-related experience for professionals), followed by a list of outcome domains to be rated for each of 3 phases: pretransplant; early posttransplant (less than 1-year posttransplant); and late posttransplant (1-year or more posttransplant). The outcome domains that were considered for the Round 1 questionnaire were selected by 2 investigators, based on the most frequently reported outcomes used in exercise training studies in transplantation from a previous systematic review. 7 The International Classification of Functioning, Disability and Health (ICF) framework, developed by the World Health Organization, was used to classify the selected outcome domains into 3 categories: body functions and structures; activities and participation; and other for outcomes that did not fit in the ICF classification.7,11 The ICF is frequently used in rehabilitation and is an important tool to classify human function and to assist with the goals of exercise training and physical rehabilitation programs. 11 As the outcome domains initially selected tended to be mainly related to body functions and structures, additional outcome domains focusing on other areas such as quality of life, activity, and participation were selected based on further literature review and included in the questionnaire. A total of 27 outcome domains were finally included in the Round 1 questionnaire.
Ratings were made using a 9-point Likert scale (1-3: not important; 4-6: important but not critical; and 7-9: very important–critical), with an option of “don‘t know”. 10 An example or a brief explanation of each outcome domain was provided to ensure clarity. For example, exercise capacity was described as “walking capacity, aerobic capacity” and quality of life was described as “perceived well-being in different aspects of the life: physical, mental, social”. The questionnaire for patients had the same content as that for professionals but was modified slightly to incorporate more lay language. The questionnaire was pilot tested for clarity of language with 1 patient, 1 clinician, and 1 researcher and slight modifications to wording were made prior to distribution to the participants.
Data Analysis
Data collected through QualtricsTM were exported into Microsoft ExcelTM for analysis and reported in frequencies or percentages after each round of the questionnaire. The data were presented for the percentage of patients and professionals that ranked the outcome as very important–critical (ratings of 7 to 9) or not important (ratings of 1 to 3). These categories were chosen as they were used to make decisions on which outcomes moved to the next round of the questionnaire. The weighted average of responses from both groups was calculated where the weights were proportional to the number of respondents in each group [ie, percentage of patients ˟ 14 + percentage of professionals × 24)/38].
Chi-square analysis was done to compare the percentage of patients versus professionals scoring an outcome as very important–critical (ratings of 7 to 9), using Statistical Package for the Social Sciences (SPSS; version 22. IBM Corp.). P value of <.05 was considered statistically significant.
Procedure
The questionnaires were distributed to the participants and responses were collected through Qualtrics™—Online Survey Software Tool. Two rounds of Delphi questionnaires were performed. Participants had 2 weeks to complete each round; 2 reminders were provided for each round to increase response rates. The 2 rounds of questionnaires were completed between May and July 2017.
In Round 1, participants were asked to rate the importance of each outcome domain (the same list for all 3 phases of transplant). Participants were also requested to suggest any additional outcome domains they considered important and were not in the initial list. Based on the results of Round 1, outcome domains were selected by 2 investigators to move to Round 2 if they received a score of 7 to 9 (very important–critical) by 70% or more participants and a score of 1 to 3 (not important) by less than 15% participants. 8 To account for the difference in the number of participants who were patients and professionals, the weighted average score for each outcome domain was also calculated.
In Round 2, participants were provided with the summarized results with anonymous responses. Participants were provided with a list of outcome domains for each phase of transplant, which was developed based on participants’ ratings in the first round, using the predetermined criteria. Also, additional outcomes were added to the lists based on the participants’ suggestions during the first round. Participants were again asked to score the outcome domains in Round 2 using the 9-point Likert scale.
Results
Recruitment and Response Rate
A total of 76 individuals (56 professionals and 20 patients) were invited by email to participate in the Delphi study. Forty individuals (25 professionals and 15 patients) were eligible and consented to participate in the study. Thirty-eight participants completed Round 1 of the questionnaires (24 professionals; 14 patients) and 36 participants completed Round 2 (23 professionals; 13 patients) of questionnaires. There was a 90% to 95% retention rate over the 2 rounds and no incomplete responses.
Description of Participants
The patient group consisted of 14 individuals (8 women and 6 men) with a mean age of 51 years, (SD = 9) and included different transplant types (
Characteristics of Patients on the Delphi Panel.

Allowed for multiple responses.
Characteristics of Professionals on the Delphi Panel.

Allowed for multiple responses.
Selection of Outcome Domains From Delphi Survey
All outcomes with their ratings from patients and professionals for Rounds 1 and 2 are presented in
Results of the Delphi Survey for the Pretransplant Phase Showing the Percentage of Participants Scoring “Very Important – Critical” and Scoring “not Important” by Outcome Domain in Rounds 1 and 2.
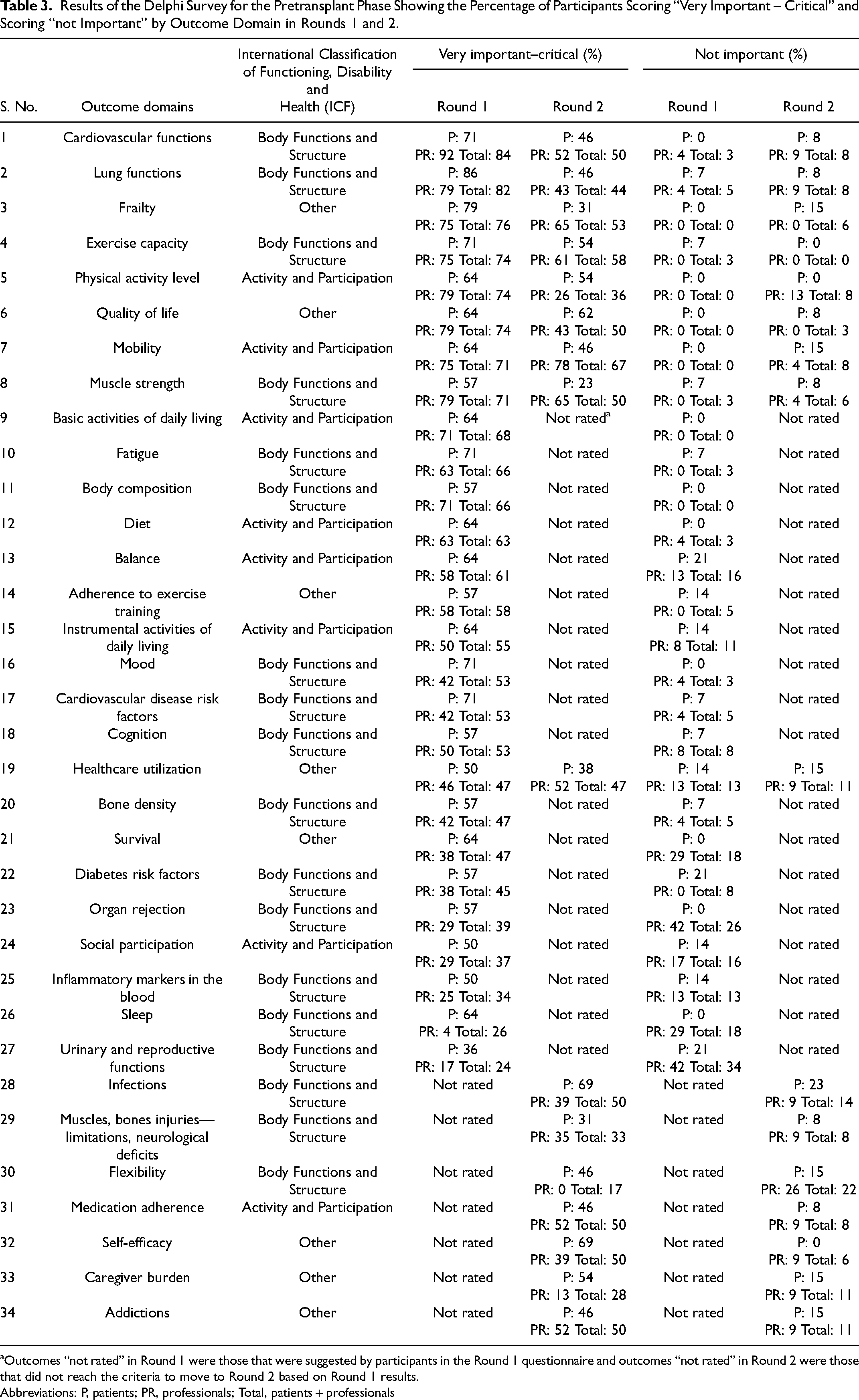
Outcomes “not rated” in Round 1 were those that were suggested by participants in the Round 1 questionnaire and outcomes “not rated” in Round 2 were those that did not reach the criteria to move to Round 2 based on Round 1 results.
Abbreviations: P, patients; PR, professionals; Total, patients + professionals
Results of the Delphi Survey for the Early Posttransplant Phase Showing the Percentage of Participants Scoring “Very Important – Critical” and Scoring “not Important” by Outcome Domain in Rounds 1 and 2.
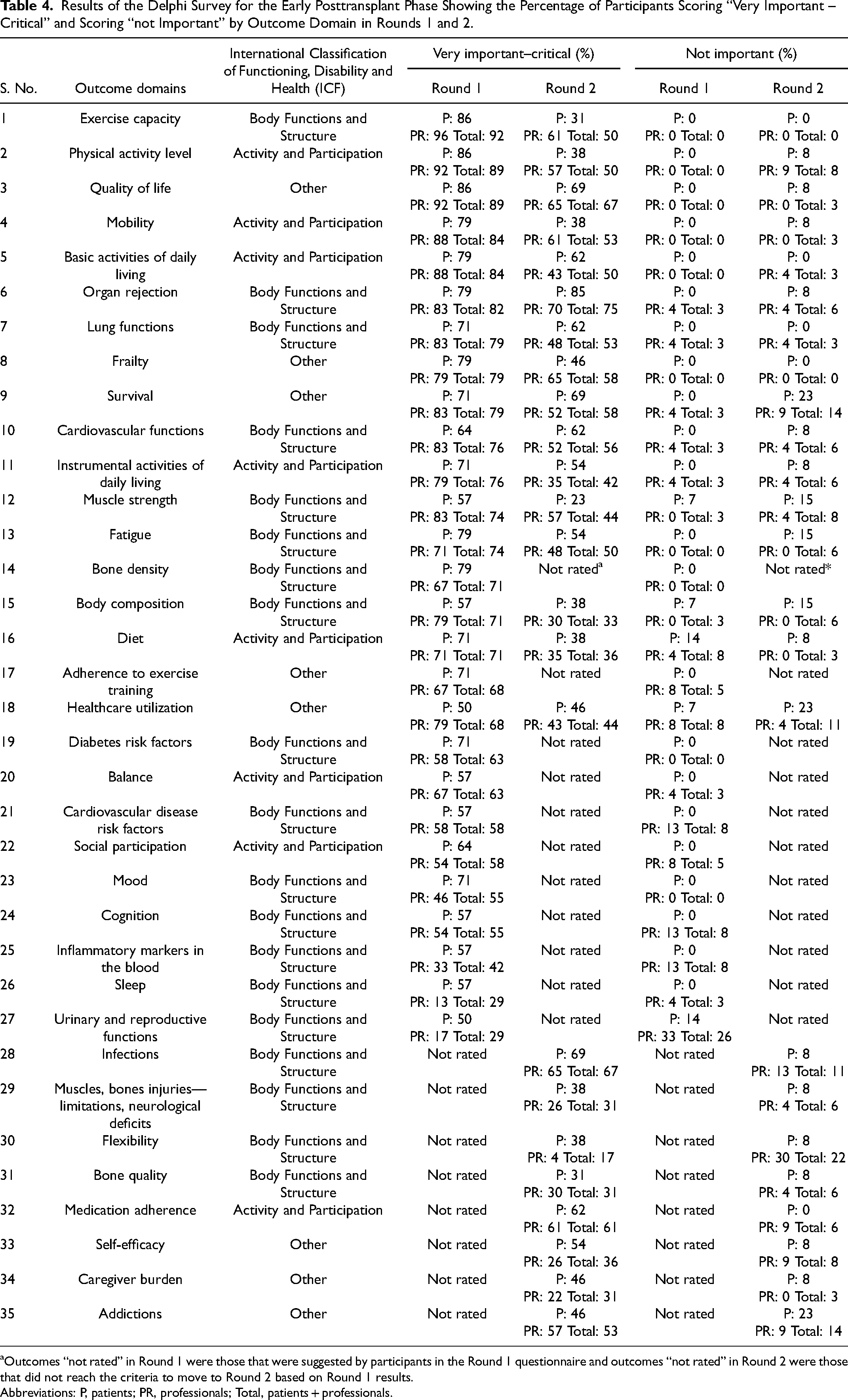
Outcomes “not rated” in Round 1 were those that were suggested by participants in the Round 1 questionnaire and outcomes “not rated” in Round 2 were those that did not reach the criteria to move to Round 2 based on Round 1 results.
Abbreviations: P, patients; PR, professionals; Total, patients + professionals.
Results of the Delphi Survey for the Late Posttransplant Phase Showing the Percentage of Participants Scoring “Very Important – Critical” and Scoring “not Important” by Outcome Domain in Rounds 1 and 2.
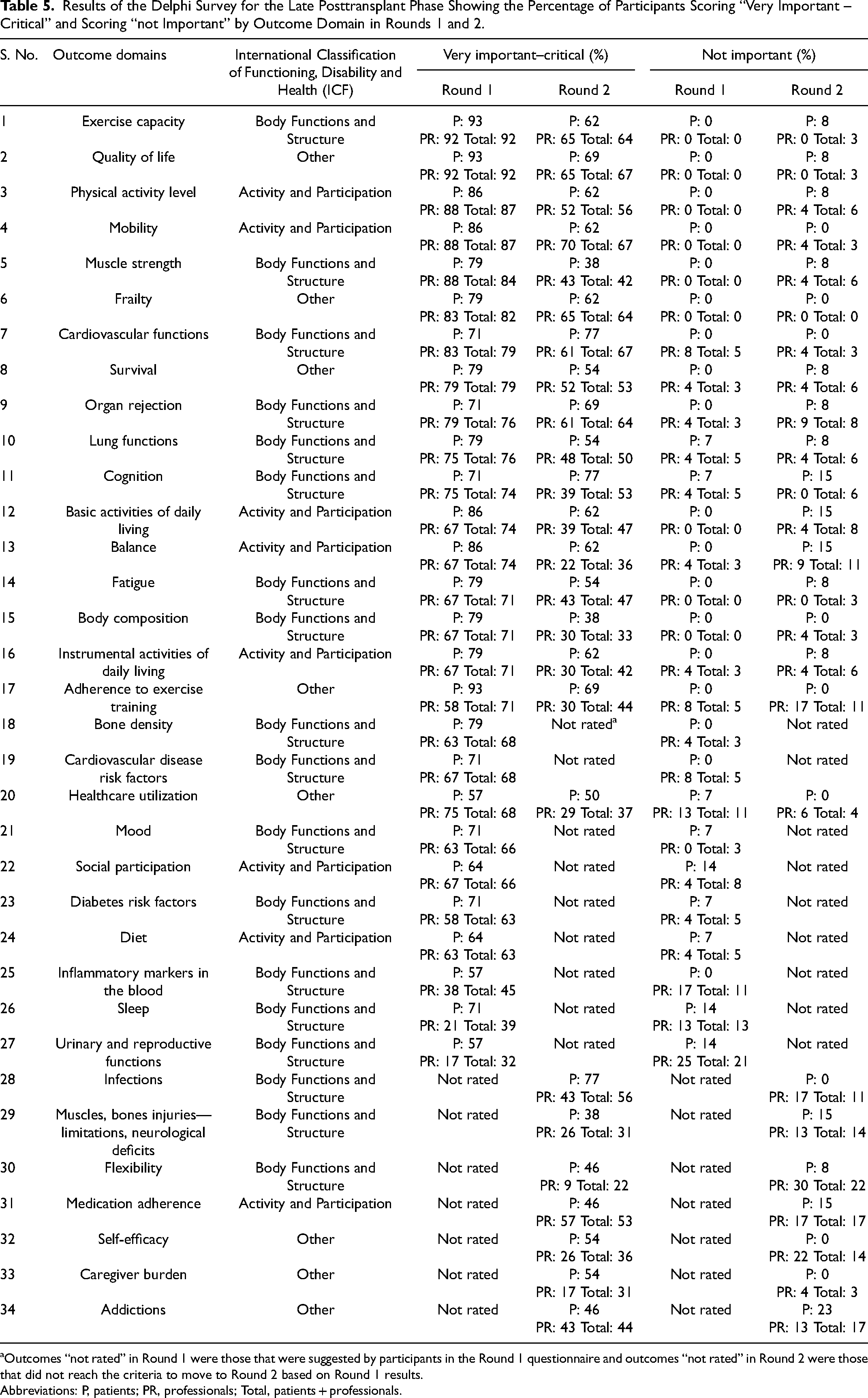
Outcomes “not rated” in Round 1 were those that were suggested by participants in the Round 1 questionnaire and outcomes “not rated” in Round 2 were those that did not reach the criteria to move to Round 2 based on Round 1 results.
Abbreviations: P, patients; PR, professionals; Total, patients + professionals.
After Round 2 of the Delphi, there were no outcomes domains that met the criteria for agreement in the pretransplant or the late posttransplant phases (Tables 3 to 5). Only organ rejection was considered very important and met the criteria (>70% in agreement) by 85% of patients and 70% of professionals in the early posttransplant phase.
Comparison of Patient and Professional Subgroup Ratings
The ratings of patients versus the professionals for each of the outcome domains, across all transplant phases and for both rounds were compared using chi-square analysis. While comparing the percentage of patients with professionals that rated outcome domains as “very important–critical”, results from the chi-square test revealed that there were no significant differences in the responses between groups for any of the outcome domains, in any of the phases.
Discussion
The development of consensus-based recommendations for outcome domains has been greatly encouraged for clinical research. This is important to have homogeneity among outcome measures in clinical trials to minimize bias, increase consistency among RCTs and to draw stronger conclusions from systematic reviews and meta-analyses.9,12 Outcome domains related to body structures and functions, activity, and participation were selected in all 3 phases of transplant, which provides a breadth of outcomes to be used in future trials. We found that most outcomes were related to body structures and functions, which was expected since exercise has a larger impact on these types of outcomes. In the second round, no outcome domains met the consensus criteria in any of the 3 transplant phases except for the organ rejection, and only in the early transplant phase. We consider this study as the first step that can inform the development of a COS in this field. Further engagement of the transplant community is needed to come to a consensus on a COS in transplant rehabilitation.
In our study, a fairly large number of outcome domains were considered very important–critical in Round 1 to be used in clinical trials of physical rehabilitation. Therefore, consensus on a core set of outcome domains, which commonly ranges from 2 to 7 outcomes,13–15 was not reached. Although there is no established minimum or maximum number of outcomes that should be included in a COS, it is easier to apply the COS and to increase its uptake if a shorter list of outcomes is recommended. 8
The fact that we have found a larger number of core outcome domains in Round 1, could be because physical rehabilitation is still an understudied area compared to other clinical populations (such as cardiac or pulmonary rehabilitation), so patients and professionals might think that examining many outcome domains is important to build the evidence. Some outcome domains that reached to an agreement may also be more relevant for certain organ groups. For example, lung function reached a consensus in the early posttransplant phase but may be most relevant for lung transplant recipients. As completed by other studies13,16 to refine the most important outcome domains, an open conversation or discussion of the participants to discuss the domains and build consensus for a shorter list of core outcomes should be considered as a next step from this study. Furthermore, domains that are relevant to 1 specific organ group may also be considered as additional outcomes but not as part of the core set.
The 8 outcome domains from the pretransplant phase: cardiovascular functions, frailty, mobility, lung functions, quality of life, exercise capacity, muscle strength, and physical activity level were perceived very important and met our criteria for the consensus in all 3 phases of transplant. Having common outcome domains across the phases of transplant can be helpful to compare the effects of exercise from pre- to posttransplant, and also for longitudinal studies. These common domains also appear to be relevant to all transplant groups, regardless of organ type, which would allow for comparisons for the effects of exercise among the different organ groups.
Exercise capacity and quality of life have been previously studied in the transplant population and included in exercise training studies in all organ groups. 3,5,6 However, mobility and frailty have not been extensively studied as outcomes of exercise training. Although mobility reached consensus in the late posttransplant phase in Round 1, this outcome domain may be more relevant in older recipients, who may be frail and have ongoing mobility restrictions for years following transplant. Frailty has been found to be an important predictor for pre- and posttransplant clinical outcomes (eg, mortality and hospital length of stay).17,18 Nevertheless, frailty has only recently been included as an outcome of exercise training. 19 With a greater interest in mobility limitations and physical frailty in transplant patients, we expect that patients and professionals would see these outcomes as very important–critical.
We found that a greater number of outcome domains reached consensus in the early and late posttransplant phase of Round 1; and included unique domains such as survival, organ rejection, fatigue, activities of daily living, and body composition. Although these outcome domains were perceived as important during the early and late stage posttransplant phases, it is important to note that the potential relationship of some of these outcome domains to exercise training is not well established. Studies specifically examining the effects of exercise training on survival and organ rejection are lacking for transplant recipients as they require long-term follow-up, which is not often feasible to conduct. Based on our study participants, it appears that these are relevant outcomes to patients and professionals that warrant consideration for future exercise trials.
Interestingly, consensus on importance of outcome domains was similar among patients and professionals for all 3 phases of transplant. We included both patients and professionals as participants in this study because we believed that professionals might be considering improvements in outcomes as a result of the exercise, and patients might be considering their outcomes as a result of the transplant. The contribution of patients to a consensus panel is needed for research to ensure that primary focus of rehabilitation and outcome assessment should be patient-centered. 20
In Round 2, only organ rejection met the criteria for agreement in the early posttransplant phase. This outcome appears to be of importance for both patients (N = 80%) and professionals (N = 75%). No other outcome domains met the consensus criteria in any of the 3 transplant phases. The relationship between exercise capacity or training and organ rejection is complicated and has not been well studied so it is unclear why this outcome was selected as important by both stakeholder groups.
Further in-person discussion between patients and professionals may help to elucidate the challenges and might suggest better ways to come to consensus on outcome domains for development of COS for transplant recipients. During the face-to-face meeting, we recommend that a smaller group of individuals (approximately 10 people) should be invited. This was based on the recommendation that a group size of 5 to 11 people is found to be both valid and effective for consensus discussions. 21 The panel should include representation from researchers, clinical experts, and patient partners and it should be a mix of Canadian and international panellists.
Limitations
This study had several limitations. We had to stop the Delphi process at Round 2, as our participants did not reach a consensus on any outcome domains to move forward to a third round. This may indicate the need for a face-to-face (in-person or virtual) meeting in order for participants to ask questions to ensure clarity of understanding of the definitions of outcome domains, and how they relate to exercise training. Our participants were limited to those with internet access and English-speaking Canadians. Future work in this area should consider an international panel of professionals and patients to ensure broader relevance and uptake of the COS. 8 We recommend that future studies may also consider including caregivers in addition to patients as stakeholders. Another potential limitation could be the fact that we had a predefined list of outcome domains in Round 1. Some Delphi studies start with an open Round 1 (with no preexisting questions). To address this limitation, participants were asked an open-ended question in Round 1 to suggest any additional outcome domains that they considered important.
Conclusions
Eight outcome domains were identified as critical across the 3 different phases of adult transplantation, however due to a large number of outcomes domains being identified as important in Round 1 and only 1 outcome domain being identified as critical in Round 2, we were unable to reach a consensus on the domains that could be included in a COS for physical rehabilitation. Our results provide a preliminary foundation for further research leading to the development of COS that can be used in clinical trials of physical rehabilitation in transplantation. As a next step, we suggest face-to-face meeting and expanding to a panel of international experts for developing and establishing consensus on COS.
Footnotes
Acknowledgments
The authors would like to acknowledge Sabrina Figueiredo, PhD for her assistance in the study design.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
