Abstract
Background
This study assesses the efficacy of Heli-FX EndoAnchors (EA) in the prevention and treatment of proximal failures after endovascular aortic aneurysm repair (EVAR).
Methods
Outcomes of 92 consecutive patients treated with EAs, between June 2010 and March 2018, were retrospectively analyzed. The EAs were deployed during primary EVAR (primary arm [PA]) or revisions for failing proximal fixation (revision arm [RA]). The primary endpoint was freedom from type 1a endoleak (T1aEL) and/or stent-graft migration; secondary end points were proximal reintervention-free survival and overall survival.
Results
The median follow-up among the 92 patients was 4.1 years (interquartile range=2.0-6.4). The PA consisted of 73 patients, of which 85% had hostile infrarenal neck morphology. In the RA, 19 patients received EAs, combined with an infrarenal extension cuff in 90% of cases. Treatment success, defined as absence of T1aEL on the first (<60 days) post-operative imaging studies, was 97% in the PA and 74% in the RA, respectively. Five-year freedom from T1aEL and/or stent-graft migration was 87.3% in the PA and 58.8% in the RA. Overall survival at 5 years was 65.1% and 51.4% in the PA and RA, respectively, and freedom from proximal reintervention was 89.3% in the PA and 79.7% in the RA.
Conclusions
In this single-center retrospective series, EndoAnchors used during primary EVAR in hostile neck anatomy demonstrated high mid-term freedom from T1aEL and/or stent-graft migration. The use of EAs to treat proximal failure of aortic endografts showed less benefit. In these complex cases, fenestrated EVAR or open repair may be more appropriate.
Clinical Impact
This study demonstrates that the use of EndoAnchors during primary endovascular aortic aneurysm repair (EVAR) is associated with high mid-term freedom from type 1a endoleaks and stent-graft migration. These findings suggest that EndoAnchor placement may improve EVAR durability, particularly in patients with hostile neck anatomy, and reduce the need for secondary interventions in this high-risk group. In contrast, the more limited benefit observed in revision settings highlights the need to carefully consider alternative treatment strategies when treating proximal failure after EVAR.
Introduction
Endovascular aortic aneurysm repair (EVAR) is considered a good alternative to open surgical repair (OSR) for the treatment of abdominal aortic aneurysms. Its main advantage is lower perioperative and 30-day mortality, whereas a meta-analysis has shown long-term mortality comparable to OSR. 1 The EVAR, however, has a higher long-term reintervention rate.1,2 Approximately 12% of EVAR reinterventions are performed to repair proximal failure, such as type 1a endoleak (T1aEL) and/or stent-graft migration.3,4 Therefore, achieving a durable proximal seal and fixation is important in improving long-term outcomes.
In 2009, the Heli-FX EndoAnchor system (originally developed by Aptus Endosystems, Sunnyvale, California; now part of Medtronic Vascular, Santa Rosa, California) was introduced as a new method for enhancing proximal endograft fixation. The EndoAnchor (EA) system is designed to fixate the proximal stent-graft fabric to the arterial wall by a helical screw that penetrates the stent-graft fabric and secures itself in the aortic wall. The system is considered to augment radial fixation and sealing, thereby preventing or treating T1aEL and/or stent-graft migration.
Most of the currently available evidence regarding the efficacy of EAs originates from the Aneurysm Treatment using the Heli-FX EndoAnchor System Global Registry (ANCHOR) registry. Results from this large, multicenter, global registry mainly describe relatively short-term outcomes.5–9 The current, single-center study aims to evaluate the mid-term efficacy of EndoAnchors for the prophylaxis and/or treatment of T1aEL and/or stent-graft migration in a consecutive series of patients.
Materials and Methods
Study Design and Population
All consecutively treated patients who underwent a procedure in which EAs were placed between June 2010 and March 2018 in the St. Antonius Hospital were retrospectively analyzed. Patients were eligible for inclusion when EAs were deployed in primary EVAR or secondary procedures to treat failing proximal fixation after EVAR. Inclusion criteria were elective procedures, infrarenal aortic aneurysms, EAs in the infrarenal neck, and complete data regarding the primary end point.
Patients were stratified according to the methodology of the ANCHOR registry 7 and divided into 2 study arms: those treated at the time of an initial EVAR procedure (primary arm [PA]) and those undergoing a revision procedure to treat failing proximal sealing after EVAR (revision arm [RA]). Patients were further subdivided by indication for EA use. The PA consisted of primary cases performed prophylactically to prevent T1aEL or stent-graft migration and a subgroup to treat procedural T1aEL. Uses of EAs in the RA were revision procedures for T1aEL, for stent-graft migration, or for a combination of T1aEL and stent-graft migration. The index procedure was defined as the first intervention during which EAs were deployed. Accordingly, the index procedure referred to the initial EVAR in the PA and referred to the revision procedure in the RA. Data were retrospectively collected from medical records, including imaging follow-up. The study was conducted in line with the Declaration of Helsinki and adhered to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines. 10 The Ethics Review Board approved the protocol, and given the use of anonymized and retrospective data, the requirement for individual informed consent was waived.
End Points and Definitions
Imaging follow-up was performed according to the standard of care in our hospital. Computed tomography angiography (CTA) was routinely performed within 4 to 6 weeks after the index procedure. Subsequent surveillance was done using duplex ultrasonography (DUS) or CTA at 12 months and annually thereafter. Treatment success was defined as the absence of a T1aEL at the first post-procedural CTA performed within 60 days after the index procedure.
The follow-up period was defined as the period between the index procedure and the last available CTA or DUS imaging follow-up. End points were defined according to the ANCHOR registry and were similar for both the PA and the RA. 7 Study endpoints were evaluated using radiology reports in combination with assessment performed by the attending vascular surgeon. The primary endpoint was T1aEL and/or stent-graft migration-free survival. T1aEL was determined on CTA or DUS but always confirmed on CTA. Stent-graft migration was assessed on CTA follow-up due to the inability to detect stent-graft migration on DUS. Stent-graft migration was present when the proximal part of the endograft had migrated >10 mm relative to the chosen anatomical landmarks or when the migration resulted in clinically adverse events. 11 Secondary end points were proximal revision-free survival and overall survival. The endpoint for proximal revision was defined as any reintervention performed for a T1aEL or proximal stent-graft migration, including endovascular procedures (eg, proximal extension cuff or fenestrated endovascular aortic repair [FEVAR]) and/or open surgical procedures (eg, graft explanation or aortic banding).
Pre-operative Anatomical Measurements
Aortic neck characteristics were measured in all patients in the PA on pre-operative CTAs. Measurements were performed using 3mensio Vascular (Pie Medical Imaging, Maastricht, the Netherlands) following a standardized protocol. 12 Measurements were performed by 1 researcher and verified by a second researcher when deemed necessary (eg, in borderline or unclear measurements). The following anatomical infrarenal neck characteristics were assessed: neck length, infra-/supra-renal angulation, presence of circumferential neck thrombus and/or calcification, conical neck configuration (gradual neck dilatation of >2 mm per 10 mm of neck length), and maximum aneurysm diameter. A hostile neck was considered when it met at least one of the following criteria: neck length <10 mm, neck diameter >28 mm, infrarenal angulation >60°, conical neck configuration, or >2 mm average circumferential thrombus or calcification. 13
Statistical Analyses
Data analysis was performed using RStudio (Posit Software, Boston, Massachusetts). Continuous variables are presented as mean (standard deviation) when normally distributed and as median and interquartile range (IQR) when not normally distributed. Categorical variables are presented as counts and proportions. The Kaplan-Meier survival analyses were performed for the primary and secondary end points.
Results
Baseline Characteristics
Between June 1010 and March 2018, 107 patients underwent EA deployment in combination with EVAR; of these, 15 were excluded due to missing data (n = 3), EA placement in the iliac landing zone (n=5), ruptured aneurysm (n=1), or juxtarenal aneurysm anatomy with EAs were used in combination with chimney stent grafts (n=6). The remaining 92 patients were included in the analysis. The median follow-up time was 4.1 years (IQR=2.0-6.4 years). The EAs were used in 73 patients (79%) in the PA group. Of these, 63 EAs (86%) were placed prophylactically, and 10 EAs (14%) were deployed because of a procedural T1aEL. The RA consisted of 19 patients (21%): 14 (74%) were to repair a T1aEL, 2 (11%) for stent-graft migration, and 3 (16%) because of a combination of these 2. Fenestrated repair was not performed in these patients due to challenging visceral or renal arteries and logistic reasons (no time to wait for custom-made fenestrated cuff). Baseline characteristics at the index procedure are summarized in Table 1.
Baseline Patient Characteristics.
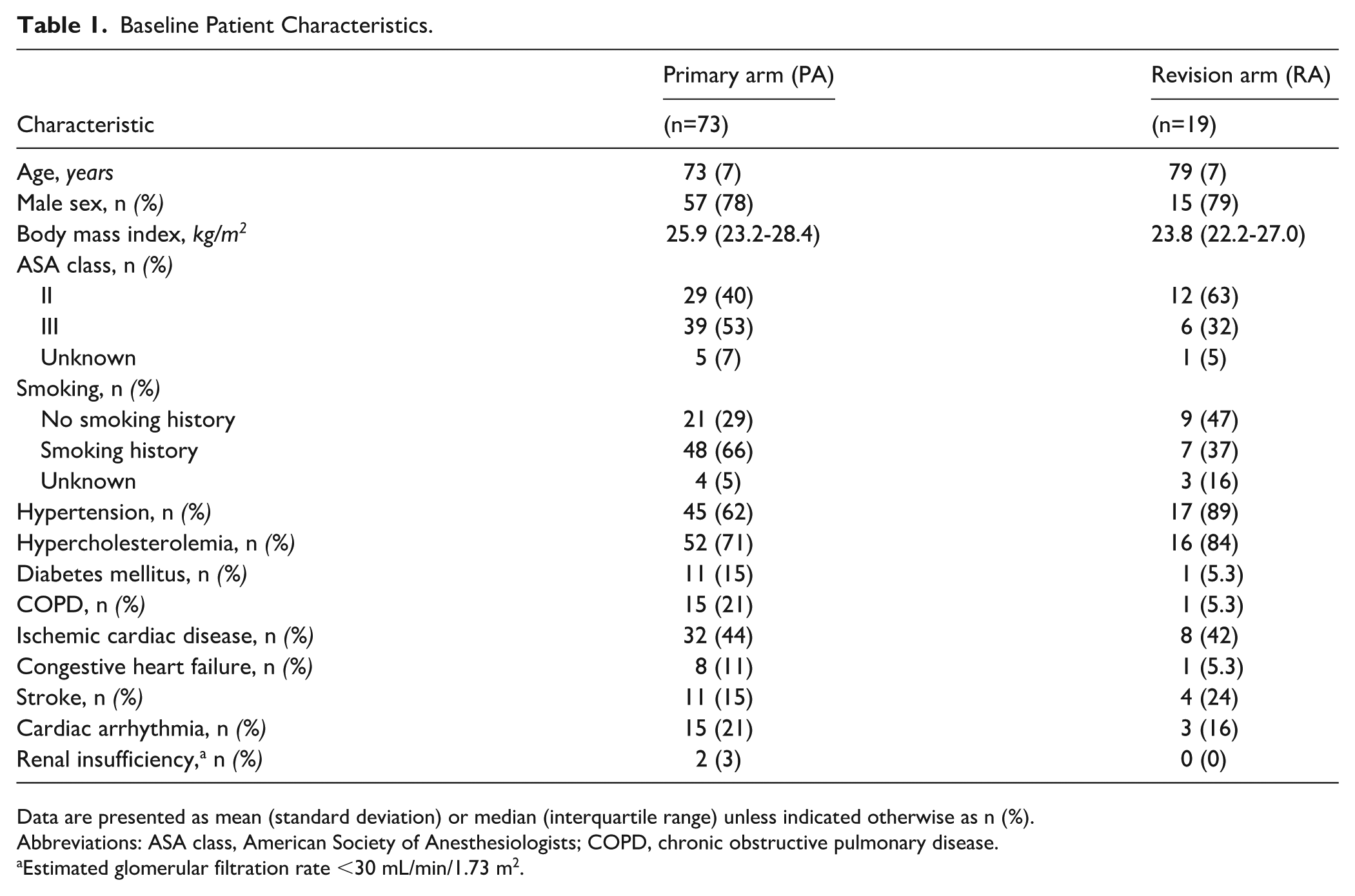
Data are presented as mean (standard deviation) or median (interquartile range) unless indicated otherwise as n (%).
Abbreviations: ASA class, American Society of Anesthesiologists; COPD, chronic obstructive pulmonary disease.
Estimated glomerular filtration rate <30 mL/min/1.73 m2.
Device Modality
Table 2 presents device selection at the time of the primary EVAR procedure in both the PA and RA. The majority of procedures (54%) were performed using a Medtronic Endurant (Medtronic Vascular, Santa Rosa, California). A combination of an AFX endograft (Endologix, Irvine, California) and a Valiant proximal cuff (Medtronic Vascular) was used for the primary repair in 4 cases. This configuration was selected in patients with a wide infrarenal neck diameter combined with a narrow aortic bifurcation. In these procedures, the EAs did not penetrate the fabric of the AFX graft but attached the Valiant cuff to the aortic wall.
Device Modality.
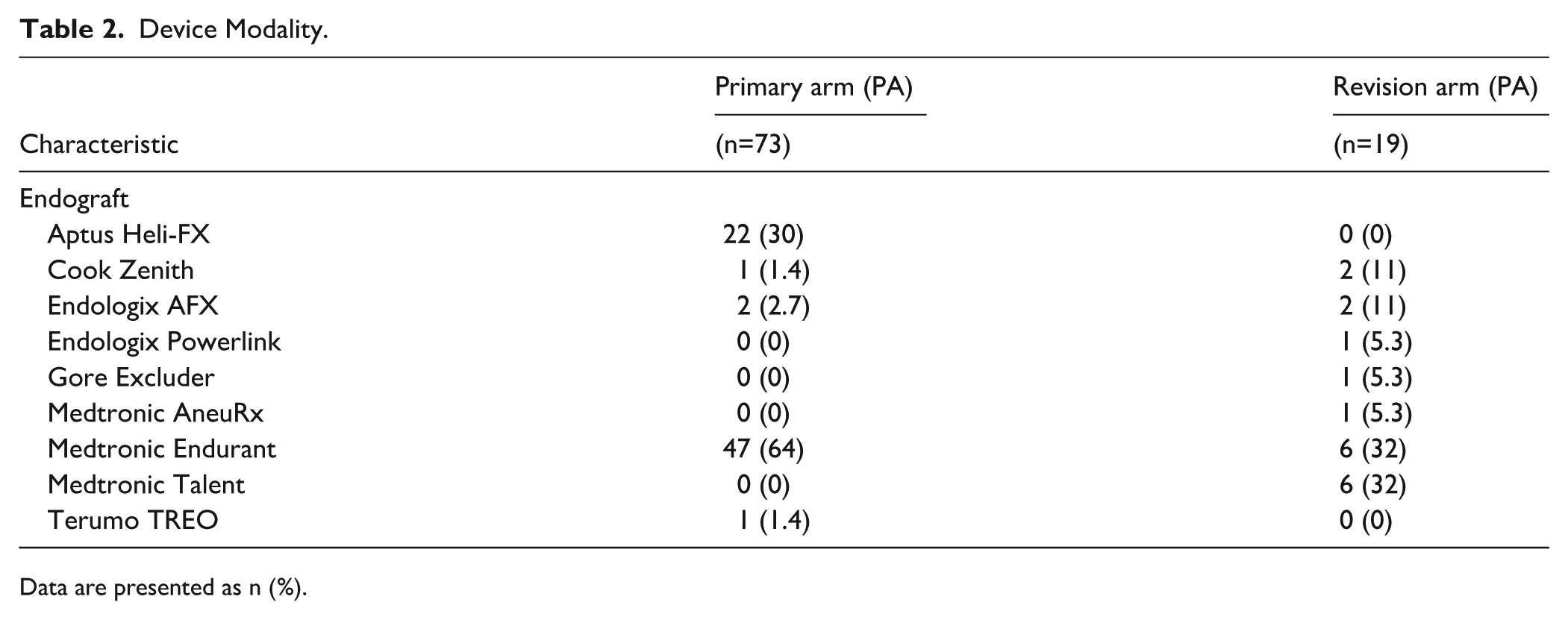
Data are presented as n (%).
Pre-endovascular aortic aneurysm repair neck characteristics in the primary arm
Table 3 summarizes pre-EVAR neck characteristics from 67 patients in the PA for whom the pre-operative CT scan could be analyzed. Of these, the median neck length was 16.0 mm (IQR=10.0-31.0 mm), with a median neck diameter of 26.1 mm (IQR=23.9-28.2 mm) and a median infrarenal angulation of 44° (IQR=31°-65°). At least 1 hostile neck characteristic was present in 57 patients (85%). Due to the standard use of EAs in combination with the Aptus endograft, some of the prophylactic patients had non-hostile neck parameters.
Neck Characteristics Before Endovascular Aneurysm Repair.
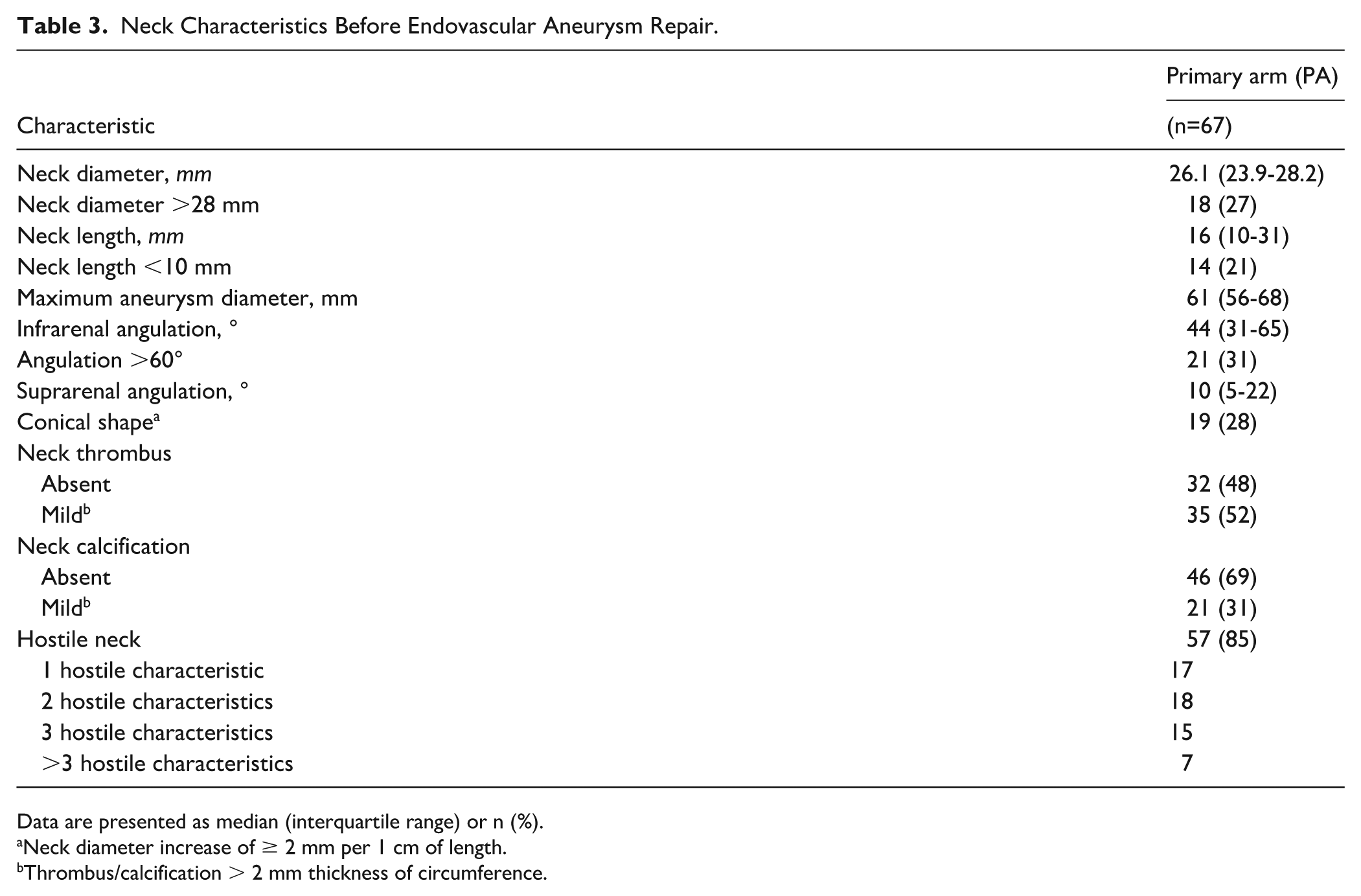
Data are presented as median (interquartile range) or n (%).
Neck diameter increase of ≥ 2 mm per 1 cm of length.
Thrombus/calcification > 2 mm thickness of circumference.
Index Procedure
Details of the index procedure are summarized in Table 4. Across all procedures, a total of 553 EAs were placed. A median number of 6 EAs (IQR=5.0-6.0 EAs) per procedure were placed in the PA and 5 EAs (IQR 4.0-6.5 EAs) in the RA. In the PA, 7 procedures (10%) were completed with the use of an additional infrarenal sealing cuff. In the RA, a proximal infrarenal sealing cuff was deployed in 17 patients (90%). No chimney or fenestrated cuffs were performed.
Index Procedure.
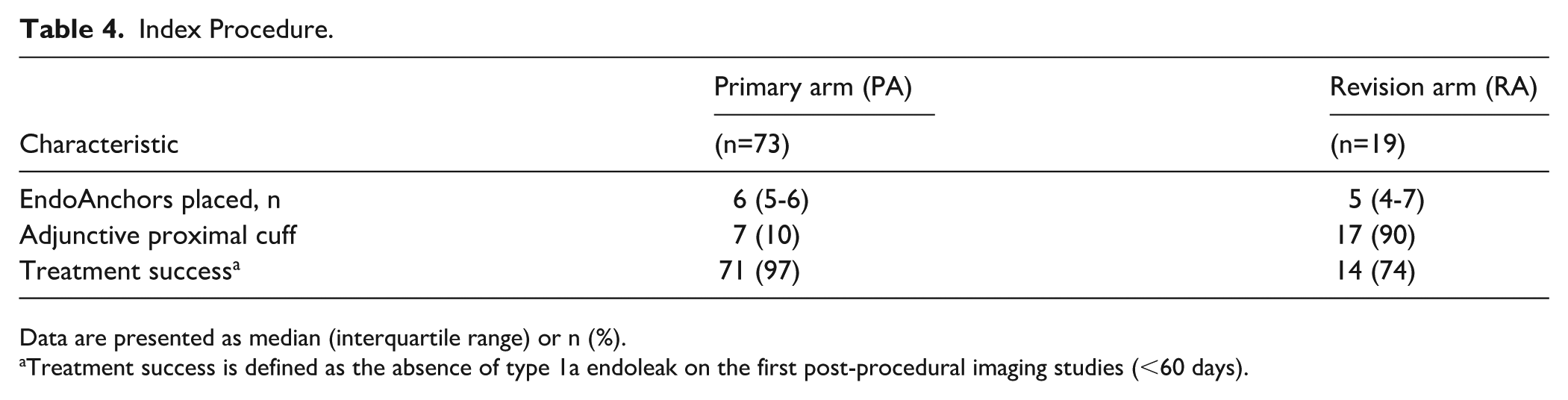
Data are presented as median (interquartile range) or n (%).
Treatment success is defined as the absence of type 1a endoleak on the first post-procedural imaging studies (<60 days).
In the PA, we observed 11 cases (15%) in which findings at procedural completion angiography suggested a low-flow T1aEL. The low-flow pattern of the endoleak, frequently combined with the technical inability to place a proximal extension cuff, rendered the endoleak acceptable. Hostile neck characteristics (with a median of 3) were present in all 11 patients. At the first post-procedure CTA, 10 of 11 endoleaks had resolved. The 1 remaining endoleak was accepted and had fully resolved by the 6-month follow-up. Overall, treatment success, defined as absence of a T1aEL at the first post-procedural imaging study, was present in 97% of cases in the PA.
The treatment success rate in the RA was 74%. Among the 5 patients in the RA with a persistent T1aEL on the first CTA, 1 endoleak was revised by performing embolization. One patient who required OSR because of a lack of endovascular treatment options was deemed unfit and, therefore, did not undergo treatment. Furthermore, the remaining 3 patients were not treated due to a combination of poor clinical condition and because the observed endoleak did not cause a significant expansion of the native aneurysm sac.
Kaplan-Meier Analysis
Figure 1 presents the Kaplan-Meier analysis of freedom from T1aEL and/or stent-graft migration. At 5 years, the T1aEL-/stent-graft migration-free survival rate was 87.3% in the PA and 58.8% in the RA. Notably, all patients (100%) in the PA who experienced a proximal failure event during follow-up had at least 1 hostile neck characteristic. Overall survival at 5 years after the index procedure was 65.1% in the PA and 51.4% in the RA (Figure 2).
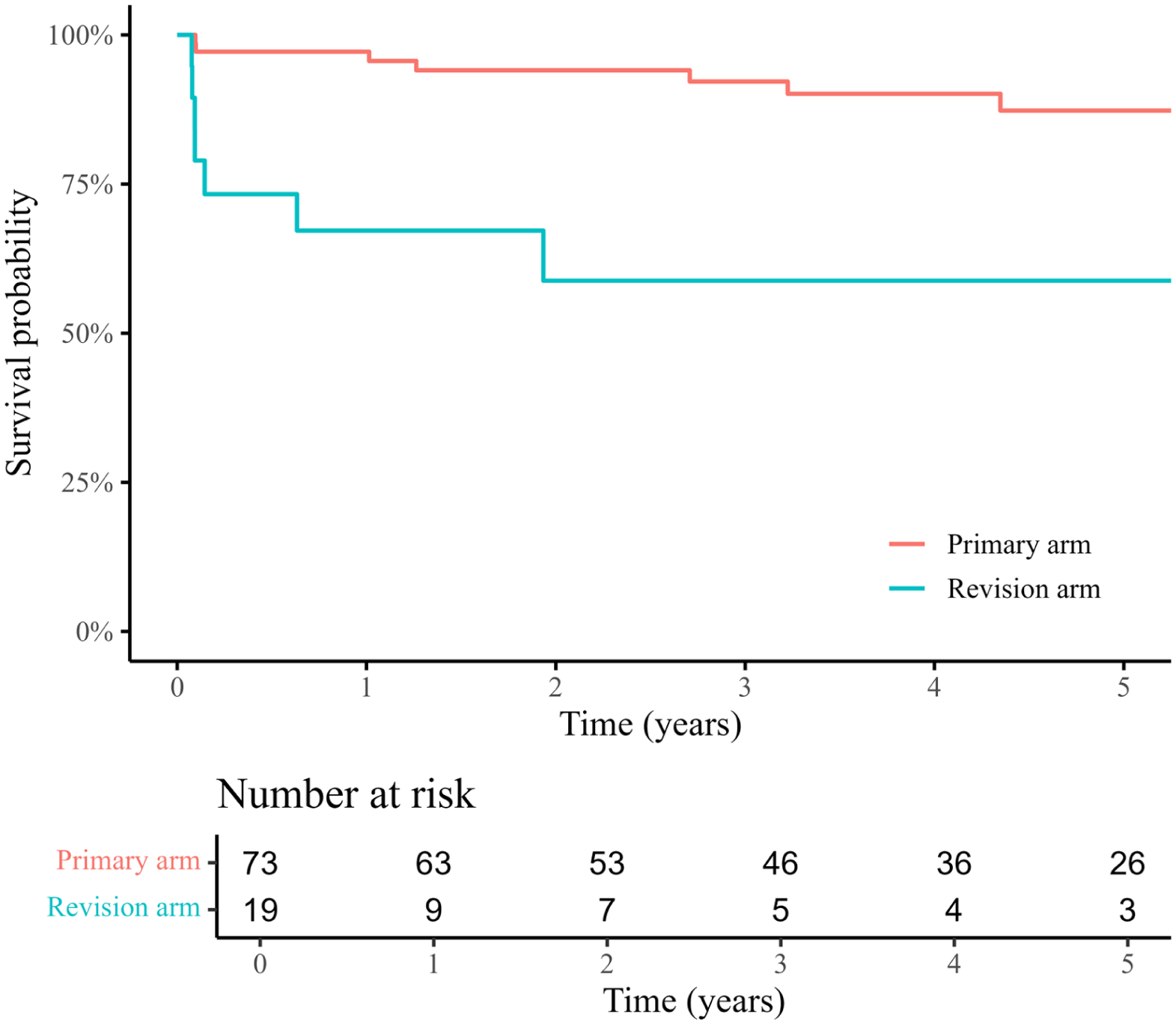
Kaplan-Meier estimation of freedom from type 1a endoleak and/or stent-graft migration after EndoAnchor placement.
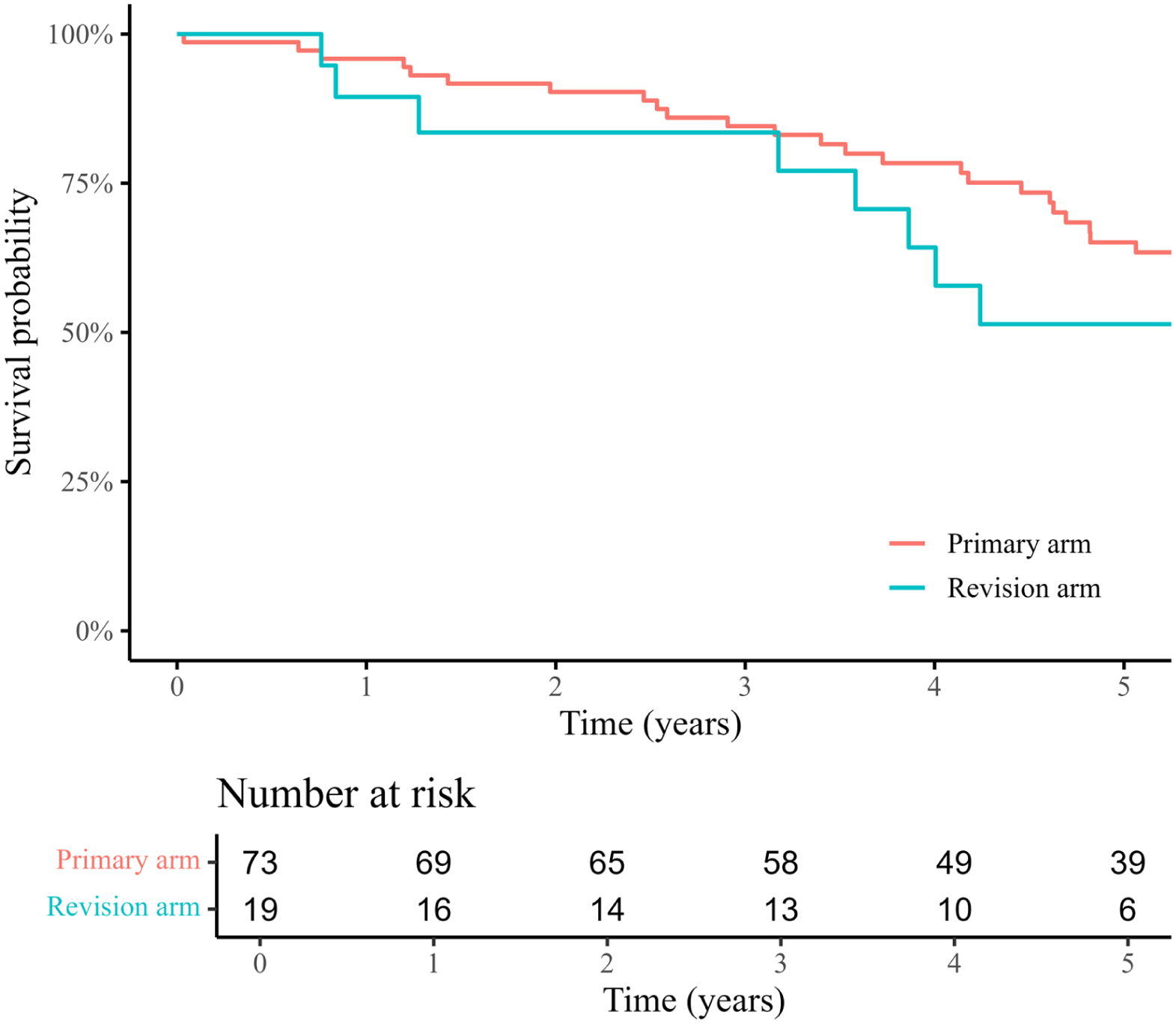
Kaplan-Meier estimation of overall survival after EndoAnchor placement.
A total of 57 reinterventions were performed among 29 patients in both groups. Fourteen reinterventions for T1aEL were performed: 10 in the PA and 4 in the RA. The freedom from proximal revisions at 5 years was 89.3% in the PA and 79.7% in the RA (Figure 3). Other indications for reinterventions were type 2 endoleak (n=11), type 1b endoleak (n=10), graft limb occlusion (n=7), access vessel injury (n=2), lower extremity revascularization (n=11), and endograft infection (n=2).
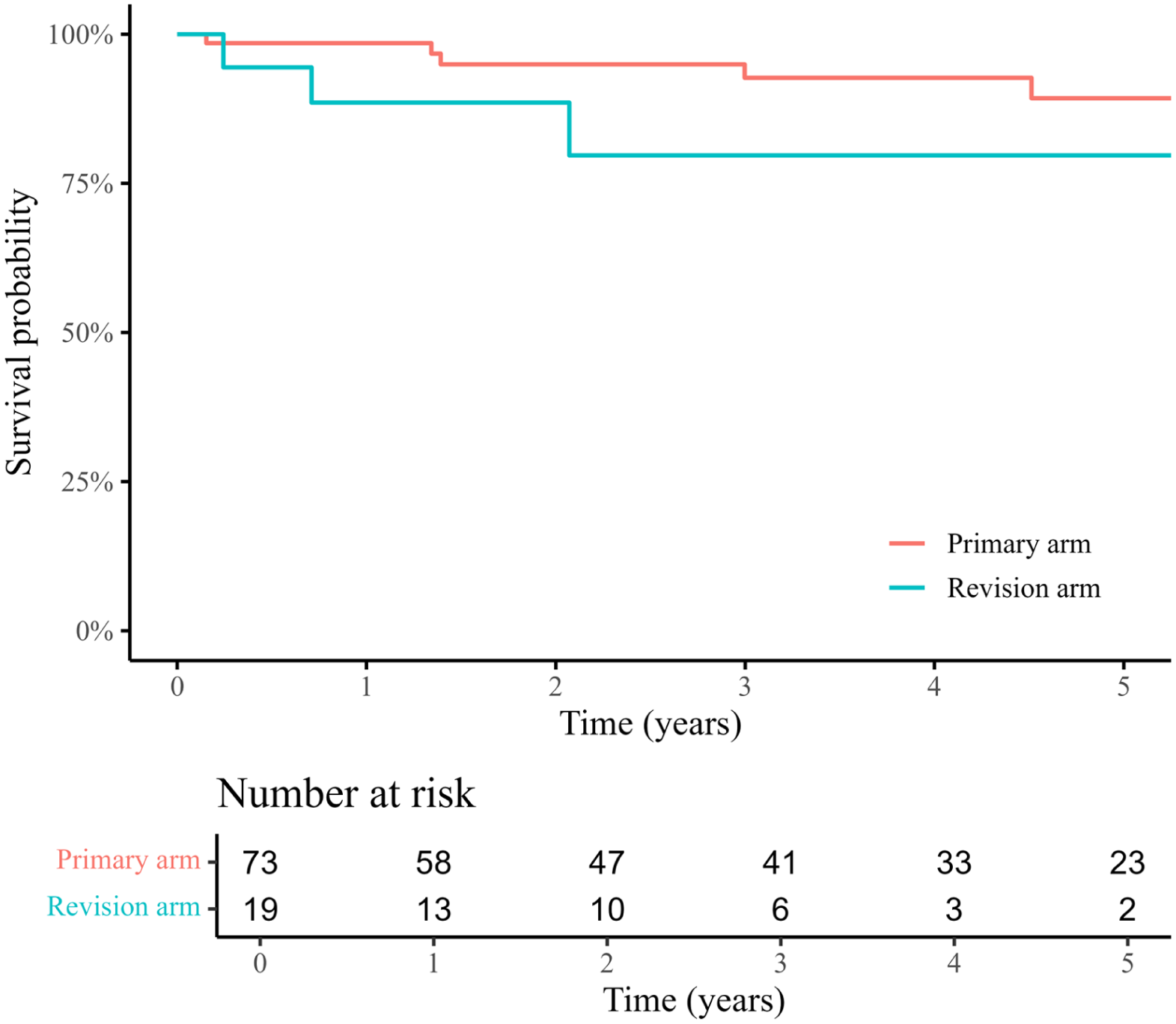
Kaplan-Meier estimation of freedom from proximal reintervention after EndoAnchor placement.
Discussion
The present, single-center study addresses the mid-term outcomes of EA use in a consecutive series of patients. The EA placement in the PA had a high treatment success rate and a high 5-year proximal event-free survival rate. In the RA, the treatment success rate was lower. Furthermore, the patients in the RA demonstrated a 5-year T1aEL-/stent-graft migration-free survival rate of 58.8%.
This study shows a relatively high incidence of persistent T1aELs on completion angiography in the PA (15%). The rate of procedural endoleaks is comparable to the rate reported in the ANCHOR registry. De Vries et al 5 and Jordan et al 9 reported procedural T1aELs in 28% and 17.3% of patients with EA deployment in the PA of the ANCHOR registry. Many of the observed endoleaks appeared as low-flow and occurred in cases without a technically feasible option for additional proximal extension. Most resolved spontaneously by the first post-operative CTA, underscoring that completion angiography may overestimate clinically relevant T1aEL. These results align with findings by Bastos Gonçalves et al, 14 who reported spontaneous resolution of completion T1aELs in 93% of cases. Accordingly, post-procedural CTA seems a more reliable determinant of clinically meaningful T1aEL and thus was used as the basis for our definition of treatment success.
Most of the EAs in our cohort were used for the prophylaxis of proximal apposition failure in hostile necks. In the PA, 85% of patients had hostile neck anatomy, which is a well-established risk factor for T1aEL or stent-graft migration.15–18 The ANCHOR registry reported a T1aEL- and/or stent-graft migration-free survival rate between 94% and 97% at a maximum of a 2-year follow-up.5–8 Results vary depending on the follow-up duration and the proportion of hostile necks included. Mid-term data on the use of EAs remains scarce; current literature reports a mid-term T1aEL-free survival rate of 84% to 93% for primary EVAR with EAs.19,20 In addition, Arko et al 21 described a 5-year reintervention-free survival of 77% for primary EVAR with EAs in short aneurysm necks. The rate of T1aEL- and/or stent-graft migration-free survival that is observed in patients undergoing EA deployment is broadly consistent with previously published data regarding EVAR procedures in aneurysms with a hostile neck where EAs were not deployed. For example, Aburahma et al 22 reported T1aEL- and/or stent-graft migration-free survival of 89% at mid-term follow-up. The comparatively higher incidence of proximal events in our cohort may be explained by the severity of neck morphology, because a large proportion of patients in our cohort presented with 3 or more hostile neck characteristics.
Although the lack of direct comparative data limits the ability to draw definitive conclusions on the additional effectiveness of EAs, 2 matched cohort analyses suggest potential long-term benefits. Muhs et al 6 and Reyes Valdivia et al 23 both showed a significantly higher rate of sac regression among patients treated with EAs. A higher rate of sac regression could indicate a beneficial long-term effect of EAs, because sac regression is an established protective factor for long-term mortality and aneurysm rupture. 24
In the RA, most proximal failures occurred within the first year (Figure 1) and can be divided in persistent and new failures. Treatment success was not achieved in 26% of patients; these findings are consistent with the ANCHOR registry, in which 18% to 25% showed persistent T1aEL at 1 year.5,7 Notably, almost all patients in the current RA (90%) underwent at least 1 adjunctive measure in addition to the use of EAs. During 17 of 19 procedures, an infrarenal proximal sealing cuff was deployed simultaneously with the EAs. Deployment of a proximal cuff in revision procedures is routinely performed because isolated EAs strengthen the proximal seal but cannot extend the sealing zone. A good alternative for EAs with a proximal cuff is the use of FEVAR. In a systematic review by Perini et al, 25 the clinical success rate of the use of FEVAR to treat T1aEL was 94%. In the current series of patients, no fenestrated cuffs were deployed due to several reasons. In some patients, the diameter of the renal arteries was too small for bridging stent grafts. Moreover, in some patients with large aneurysms, the waiting time for a custom-made fenestrated endograft was judged too long. Nowadays, in these patients, a physician-modified endograft could be considered. EndoAnchor deployment can often be performed under local anesthesia, thus offering a viable alternative for high-risk patients with an indication for a proximal revision procedure.
This study has limitations. Its retrospective, single-center design limits generalizability, and the small RA sample reduces statistical precision of endpoint estimates. In addition, the lack of a control group without EA use prevents direct comparative assessment of efficacy and limits the ability to draw definitive conclusions.
Conclusion
In this single-center retrospective series, EndoAnchors used during primary EVAR in hostile neck anatomy demonstrated high mid-term freedom from T1aEL and/or stent-graft migration. The use of EAs to treat proximal failure of aortic endografts showed less benefit. In these complex cases, fenestrated EVAR or open repair may be more appropriate.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
