Abstract
Purpose:
Active fixation thoracic endografts have been presumed to cause adverse events in patients with aortic syndromes. This study evaluated the outcomes of a barb-fixated thoracic endograft and compared with a non–actively fixated endograft in patients managed for aortic syndromes.
Materials and Methods:
A single center retrospective analysis was conducted using the STROBE guidelines. Patients treated for aortic syndromes with the barb-fixated Zenith TX2 TAA Endovascular Graft (ZTEG) between January 2011 and June 2024 were included. Patients with replaced proximal landing zones and genetic aortic syndromes were excluded. Primary outcomes were migration and migration distance (MD) in patients with at least 12 months of follow-up. A propensity-score matched comparison (1:1) to patients treated with the non-active fixation Zenith TX2 Dissection Endovascular Graft (ZDEG) was performed, after applying the same eligibility criteria.
Results:
Ninety-six patients treated with ZTEG were included. No perioperative retrograde type A aortic dissection (rTAAD) or intramural hematoma occurred. Both endoleak Ia and proximal reintervention rates were 6%, each, at 20 months (IQR, 1–54.5). Endograft migration was assessed in 65 patients [median (IQR) follow-up: 33 months (IQR 20–71)], showing a 2 and 20% migration at 1 year and maximum follow-up, respectively, with a median MD of 2 mm (IQR 0–5) and 5 mm (IQR 2–9) at the same timepoints, respectively. The propensity score-matched comparison with 23 ZDEG patients revealed significantly lower 1-year migration (4% vs 35%; p=0.04) and shorter median MD [4 mm (IQR 0–9.5) vs 10 mm (IQR 6–15.5); p=0.016] at maximum follow-up for the ZTEG group. No difference was observed in endoleak Ia, proximal reintervention, or rTAAD rates at any point.
Conclusion:
The use of barb-mediated fixation ZTEG endograft in patients with aortic syndromes demonstrated reduced migration and MD compared with the non-actively fixated ZDEG, without differences in rTAAD, endoleak Ia, or proximal reintervention during follow-up.
Clinical Impact
Thoracic endovascular aortic repair with a barb-fixated endograft (Zenith TX2 TAA Endovascular Graft; ZTEG) in patients with aortic syndromes demonstrated migration rates of 2% at 1 year and 20% at maximum follow-up, with no retrograde dissection (rTAAD). In a propensity score-matched comparison with a non-active fixation endograft (Zenith TX2 Dissection Endovascular Graft; ZDEG), ZTEG showed significantly lower 1-year migration and shorter migration distance at maximum follow-up. No difference was observed in endoleak Ia, proximal reintervention, or rTAAD at any time point. These findings suggest that ZTEG may reduce distal migration without compromising safety regarding retrograde dissection, contrary to existing concerns.
Introduction
Thoracic endovascular aortic repair (TEVAR) is the treatment of choice in patients managed for either aortic dissection or intramural hematoma (IMH) and penetrating aortic ulcer (PAU) of the descending thoracic aorta.1–3 Coverage of the proximal entry tear and/or adequate proximal sealing, depending on the underlying disease, is the cornerstone of TEVAR’s durability in acute and chronic cases.1–5 In aortic dissections, the use of TEVAR to cover the main entry tear targets the depressurization of the false lumen and promotes true lumen’s expansion. 6 Additional considerations, including target-vessel management (debranching, stenting or coiling/plugging), extend of coverage and staging of the repair must be carefully individualized to improve outcomes.6–8 Consequently, the successful treatment of these patients remains particularly challenging.
The proximal landing zone in aortic dissections has been among the main considerations for successful treatment, regarding both the short-term technical and clinical success, as well as the long-term durability.4,5 Over time, various endografts have been used, with the cumulative experience suggesting that a low oversizing of no more than 10%, the avoidance of bare metal stents at the proximal end of the device and refraining from ballooning of the graft could help to prevent retrograde type A dissections (rTAAD).9,10 Proximal active fixation elements, like hooks and barbs, used as part of some TEVAR endograft designs, to secure proximal seal and prevent distal migration, have likewise been regarded with caution, lacking, though, adequate evidence.11,12 Nevertheless, beyond preventing rTAAD, secure proximal sealing is essential to ensure the durable treatment of the diseased aortic segment, while avoiding endoleaks and caudal migration.4,5
The aim of this study was to present the outcomes of a barb-fixated thoracic endograft in patients treated for aortic syndromes 13 and compare it to a thoracic endograft without active fixation.
Materials and Methods
Study Design
A single-center retrospective analysis was conducted according to the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) guidelines. 14 Patients treated for aortic dissection, IMH or PAU; all described under the term aortic syndromes, 13 and managed with the Zenith TX2 TAA Endovascular Graft (ZTEG; Cook Medical Inc, Bjaeverskov, Denmark), between January 1, 2011, and June 31, 2024, were included. A comparative analysis was performed between these patients and those managed with the Zenith TX2 Dissection Endovascular Graft (ZDEG; Cook Medical Inc, Bjaeverskov, Denmark) for aortic syndromes during the same time-period. To ensure comparable baseline anatomic characteristics, only patients with imaging follow-up of at least 1 year were included and were propensity-score matched based on predefined anatomic variables, including the presence of dissection, proximal landing zone (according to Ishimaru classification), prior ascending aortic repair, proximal aortic diameter, maximum aortic diameter, and graft oversizing ratio.
The study complied with the Declaration of Helsinki. No approval was required from the local ethics committee according to the current state law (§12 HmbKHG) due to its retrospective nature and unidentifiable data.
Study Cohort
The indication for repair regarding aortic dissection [acute (0–14 days), subacute (15–90 days), or chronic (>90 days)], PAU or IMH (acute or chronic setting) was based on the available guidelines at the time of repair.1–3,15–17 Patients treated with ZTEG with a proximal landing in Ishimaru zone 2 or distally were eligible. Cases with more proximal landing zone (Ishimaru zones 0–1) were excluded to avoid the impact of aortic arch’s curvature and hemodynamics on endografts’ apposition and potential migration during follow-up. 18 Patients, treated fora thoracic aortic diameter ≥55 mm (thoracic or thoracoabdominal aortic aneurysm) were excluded. 19 To detect potential device migration events, patients with an available imaging surveillance of at least 1 year were selected.
Regarding further inclusion criteria, all investigated devices should have landed in a non-dissected healthy native aortic segment. Patients with genetic aortic syndromes, previous proximal open (frozen elephant trunk, hemiarch or total arch replacement) or endovascular repair (TEVAR, fenestrated/branched TEVAR, parallel graft technique), affecting the proximal landing zone, were excluded (Figure 1).
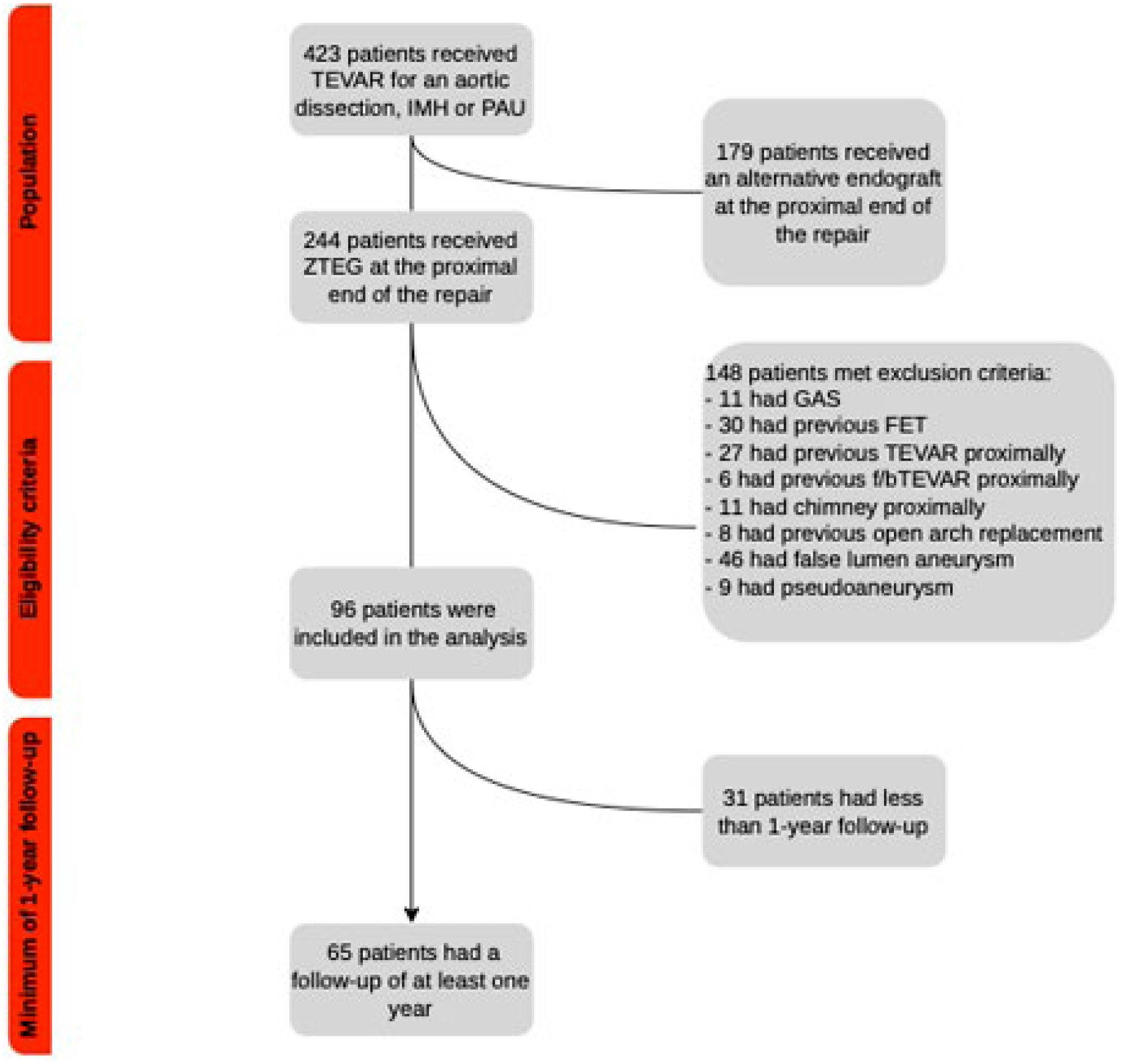
Flowchart of the population included in the study. Reasons for exclusion according to eligibility criteria are provided. TEVAR, thoracic endovascular aortic repair; IMH, intramural hematoma; PAU, penetrating aortic ulcer; GAS, genetic aortic syndrome; FET, frozen elephant trunk; f/bTEVAR, fenestrated/branched thoracic endovascular aortic repair.
Technical Details
Preoperative planning was performed, using a computed tomography angiography (CTA) no older than 6 months. Patients were managed under general anesthesia, in a hybrid operating room with a fixed imaging system and fusion guidance (Vessel Navigator, Philips Healthcare, Best, the Netherlands). Bilateral percutaneous femoral access was preferably used and TEVAR was performed in standard fashion. Cardiac output reduction method during the endograft deployment was at the discretion of the operator, using either inferior vena cava balloon occlusion or the Valsalva maneuver and pharmacologic support.
Both endografts (ZTEG and ZDEG) were available in diameters from 22 to 42 mm, including tapered configurations, while sharing an identical delivery system and nearly identical graft design, both lacking a proximal bare-metal stent. However, the ZTEG featured proximal active fixation barbs. According to the Instructions for Use (IFU), ZTEG was indicated for thoracic aortic aneurysms and dissections, whereas ZDEG is intended for aortic dissections- PAU and IMH are not specifically listed as indications for either device. Although ZTEG is no longer available in Europe, its successor, the Zenith Thoracic Alpha (ZTA), indicated for aneurysmal disease, retains proximal fixation barbs, incorporating an additional proximal bare metal stent. The choice between the 2 stents was mainly driven by the surgeons’ preference and availability of appropriate sizes for each case. The ZTEG endograft was the primary TEVAR device stocked in our department and, thus, it was readily available in a wider range of lengths and diameters. Debranching with a left carotid-subclavian bypass or left subclavian artery (LSA) transposition was performed either simultaneously or at a separate stage, when necessary.2,3 Plug occlusion of the LSA was selectively performed when a zone 2 landing was chosen.
Imaging Evaluation
Computed tomography angiography of slice thickness ≤1 mm with arterial phase was available in all cases. Appropriate measurements and assessments were conducted using the preoperative, first postoperative (within 30 days), first year, and latest available follow-up CTAs. 20 When a proximal reintervention was performed during follow-up, no further assessment of the imaging outcomes was performed.
The preoperative CTA was used to assess the underlying pathology and initial aortic diameter in all cases (ZTEG and ZDEG). Regarding the proximal landing zone, the maximum cross-sectional outer to outer wall aortic diameter of the proximal landing zone with 10 mm intervals (0 mm, 10 mm, 20 mm) was assessed in the first postoperative, first year and latest follow-up CTA of the ZTEG group, while the same measurements were performed for the comparative ZDEG group. For the measurement of distal migration and migration distance (MD), the distance between the proximal edge of the endograft and the distal end of the respective supra-aortic vessel ostium [zone 2: left common carotid ostium, zone 3: left subclavian artery (LSA) ostium] was measured in the first year and last-follow-up CTA and was compared to the first postoperative scan in both groups.20,21
Diameter and length measurements were performed using the Aquarius iNtuition Viewer (Version 4.7.2, TeraRecon, Foster City, California) after the application of outer curvature line from the aortic root down to zone 4 by 2 independent experienced investigators (G.A. and P.N.).22,23 For the first 10 cases, the measurements were blindly performed from each investigator and subsequently evaluated by the senior author (T.K.). No difference ≥2 mm was detected in any measurement.
Data Collection
A dedicated local database of pseudonymized data was created. Information on baseline characteristics, anatomy, and details of the applied repair was collected. Data on 30-day major adverse events, as well as imaging-related outcomes, reinterventions, and mortality, were recorded at 30 days and follow-up.20,21
Definitions
Migration was defined as any ≥10 mm endograft caudal movement, based on the reporting standards. 20 Migration distance (MD) was defined as the distance of endograft displacement measured in millimeters (Supplementary Figure 1). Bird-beak was defined as a wedge-shaped gap >5 mm between the first covered stent of the endograft and the inner aortic curvature.21,24 (Supplementary Figure 1). Oversizing was defined as the percentage difference between the proximal endograft diameter and the mean diameter of the targeted proximal sealing zone. Proximal landing zone dilation was calculated as the percentage of difference in the mean value of proximal landing zone diameter (measured at 0, 10, and 20 mm), between the first postoperative and first year and last available follow-up CTA. 25
Outcomes
The primary outcome were migration and MD in the ZTEG cohort at 1 year and at maximum follow-up, defined as the latest available follow-up for each patient. Proximal landing zone dilation and bird-beak presence were evaluated as secondary outcomes. Endoleak Ia, rTAAD, and proximal reinterventions were also assessed.
Statistical Analysis
Continuous data were presented through means ± standard deviation, if they were normally distributed and through medians with corresponding interquartile ranges (IQR), if they were non-normally distributed. Normality was assessed both visually (boxplots) and by applying the Shapiro-Wilk test. Categorical data were presented as absolute numbers and percentages. Follow-up was assessed through Kaplan-Meier curves. Propensity score matching (1:1) was applied, considering multiple anatomical parameters using the nearest neighborhood model with a caliper value set to 0.02 (“MatchIt” package in R). 26 Six variables were taken into consideration during propensity score matching: the presence of dissection, the proximal landing zone, the presence of a previous ascending aortic repair, the proximal diameter (greater or smaller than 35 mm), the maximum diameter (greater or smaller than 40 mm) and the oversizing ratio (greater or smaller than 10%). Differences between groups were assessed using the exact binomial McNemar test for binary outcomes and the Wilcoxon signed-rank test for continuous outcomes. 27 Missing data were random and not imputed. p value was considered significant when <0.05. Statistical analyses were performed with RStudio (Version 2025.05.1+513; Integrated Development Environment for R, Posit Software, PBC, Boston, Massachusetts).
Results
Patient Characteristics
Ninety-six patients managed with the ZTEG endograft met the eligibility criteria [median age 70 years (IQR 61–76), 32 (31%) females]. The most treated disease was aortic dissection in 63 patients (66%). In two, dissection coexisted with PAU (2%), while 6 patients (6%) presented with both PAU and IMH (Table 1).
Patients’ Baseline Characteristics.

Values in median (Q1-Q3) or n (%).
Abbreviations: COPD, chronic obstructive pulmonary disease; CAD, coronary artery disease; MI, myocardial infarction; TIA, transient ischemic attack; PAD, peripheral arterial disease; CKD, chronic kidney disease; TEVAR, thoracic endovascular aortic repair; f/bEVAR, fenestrated/branched; IMH, intramural hematoma; PAU, penetrating aortic ulcer.
Regarding aortic dissections, 50 (79%; 50/63) were classified as acute/subacute and 13 (21%; 13/63) as chronic. Among those treated in the acute/subacute phase, 27 (54%; 27/50) presented with aortic rupture or malperfusion. The remaining 23 (46%; 23/50) patients were treated based on the presence of high-risk features; in all cases at least 2 high-risk features were present. Intramural hematoma was mostly treated urgently (83%; 10/12), while PAUs were mostly treated in the chronic phase (81%; 17/21; Table 2). Proximally, the implanted endograft landed in zone 2 in 62 cases (65%), in zone 3 in 15 cases (16%) and in zone 4 in 19 cases (20%). Proximal landing zone median diameter was 33 mm (IQR, 30–35) and median oversizing was 14% (IQR, 11%–18%).
Anatomical Characteristics and Indication for Repair.
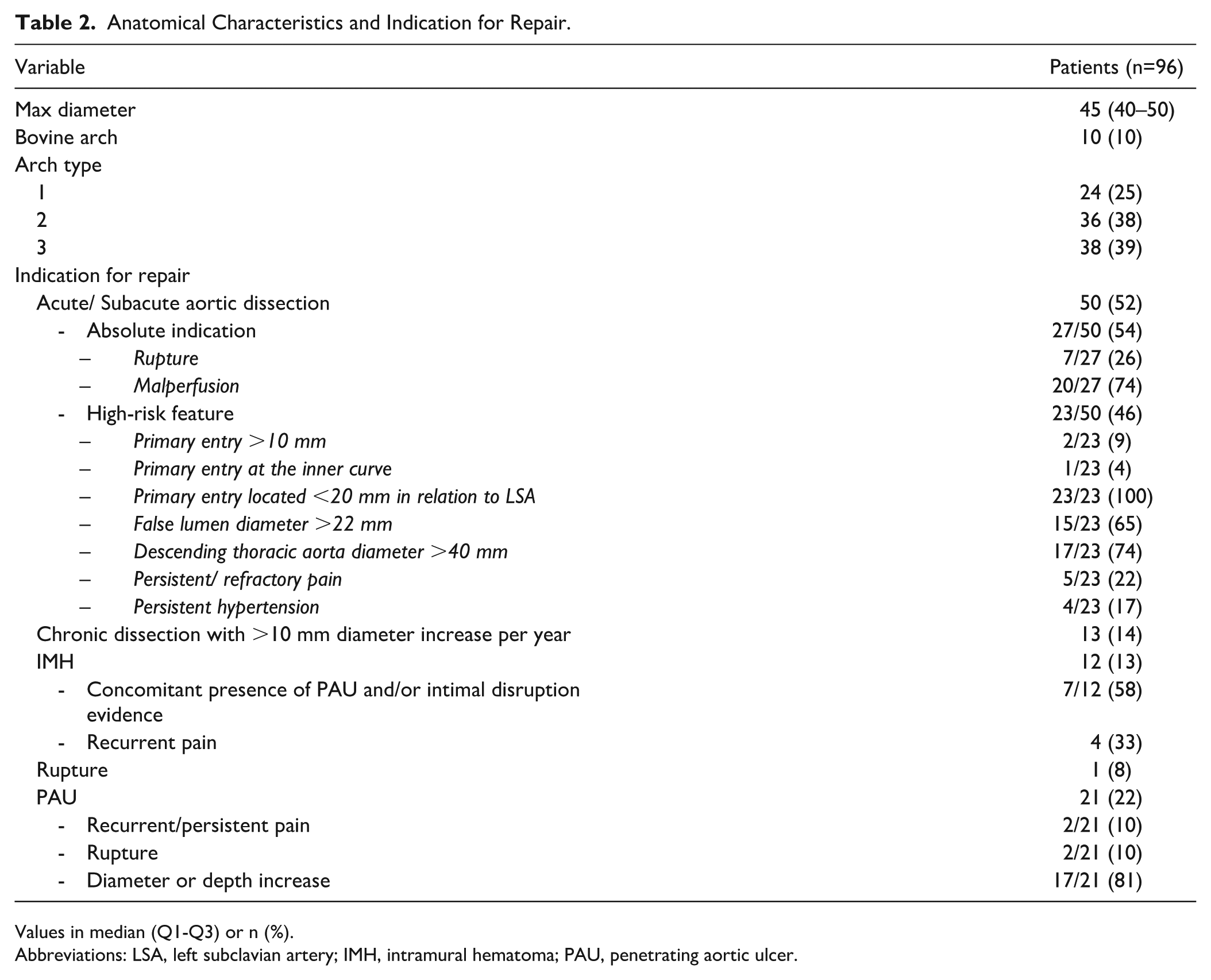
Values in median (Q1-Q3) or n (%).
Abbreviations: LSA, left subclavian artery; IMH, intramural hematoma; PAU, penetrating aortic ulcer.
Thirty-two patients (33%) had an LSA debranching (94%; 30/32 LCCA-LSA bypass and 6%; 2/32 LSA transposition); 9 (28%; 9/32) with simultaneous TEVAR, one (3%; 1/32) during the first post-TEVAR day due to spinal cord ischemia, and the rest (96%; 22/32) before TEVAR. Among the patients receiving an LSA debranching, 23 (24%) had an adjunctive LSA plug occlusion.
Early Outcomes
No rTAAD was detected intraoperatively (0%) or in the pre-discharge CTA. The completion angiography revealed a type Ia endoleak in 7 cases (7%), with only 4 (4%) detected in the 30-day CTA, and 2 persisting at follow-up (2%; one led to a proximal reintervention). Eight cases of bird-beak were detected in the 30-day CTA. Early proximal reintervention was performed in 1 case: a planned frozen elephant trunk due to IMH in zone 0, proximal to the treated type B dissection, already existent in the preoperative CTA. Major adverse events are presented in Supplementary Table 1.
Follow-up Outcomes
The median follow-up for the ZTEG cohort was 20 months (IQR, 1–54.5). Survival at 84 months was 86.6% (SE 7.4%; Figure 2). Proximal reinterventions were performed in 6 patients (6%), and only one was planned (Table 3). No patient presented with rTAAD. Endoleak Ia was present in 6 patients (6%) during follow-up; 4 of them were treated with proximal extension (Table 4).
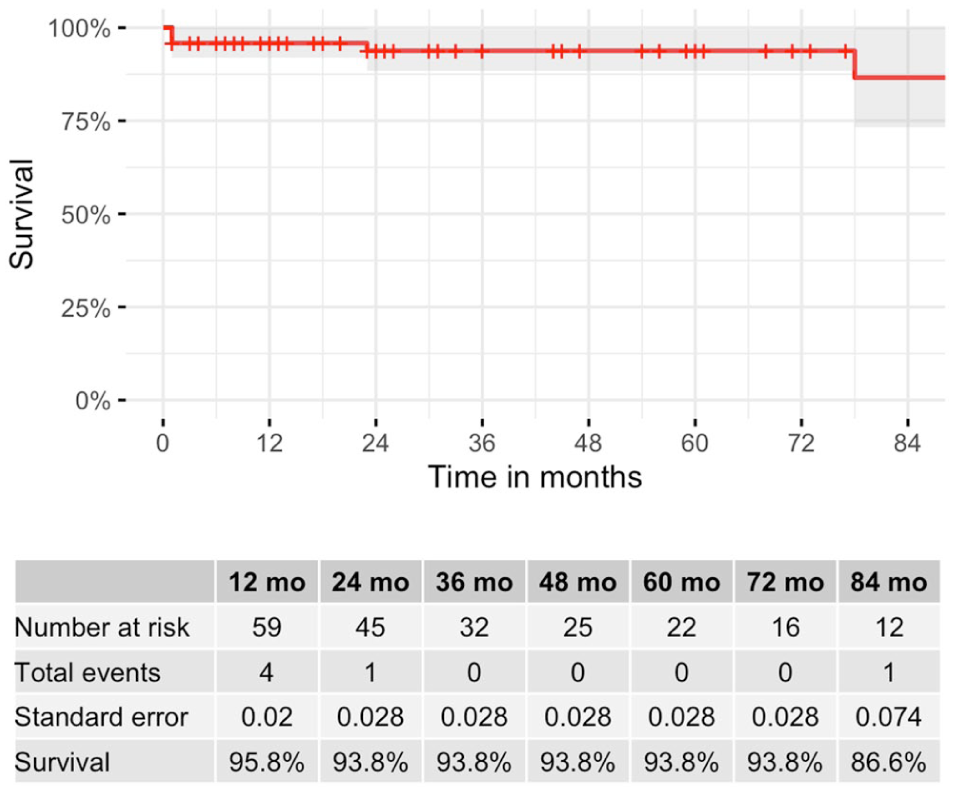
Kaplan-Meier curve for survival.
Proximal Reinterventions.

Abbreviations: FET, frozen elephant trunk; fTEVAR, fenestrated thoracic endovascular aortic repair; TEVAR, thoracic endovascular aortic repair; bTEVAR, branched thoracic endovascular aortic repair; IMH, intramural hematoma.
Endoleak Ia During Follow-up.
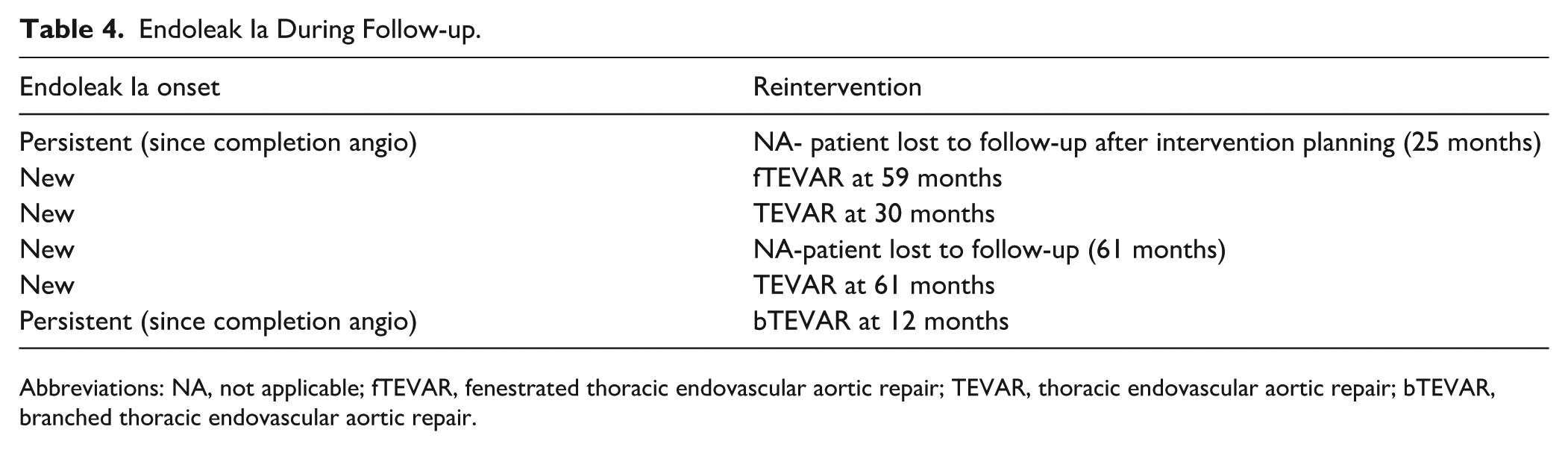
Abbreviations: NA, not applicable; fTEVAR, fenestrated thoracic endovascular aortic repair; TEVAR, thoracic endovascular aortic repair; bTEVAR, branched thoracic endovascular aortic repair.
Regarding patients with adequate follow-up of at least 1 year (n=65), one ZTEG endograft migrated at 1 year (2%) and 13 (20%) at the maximum available follow-up [median follow-up: 33 months (IQR, 20–71)]. Median MD was 2 mm (IQR, 0–5) at 1 year and 5 mm (IQR, 2–9) at maximum follow-up. Bird-beak was present in 13 (20%) cases at 1 year and in 22 (34%) at maximum follow-up. Median proximal landing zone diameter dilation was 5% (IQR, 0–11) at 1 year and 10% (IQR, 4–16) at maximum follow-up.
Propensity Score-Matched Cohort Comparison
Twenty-three patients, who met the eligibility criteria and were managed with a ZDEG endograft during the same time period, were matched to 23 patients from the ZTEG cohort. Median follow-up was 45 months (IQR, 18–65) for the ZDEG and 33 months (IQR, 18–59.5) for the ZTEG group (p=0.85). A Love plot depicting standardized mean differences between groups before and after matching, for the variables used, is included (Supplementary Figure 2).
At 1 year, 8/23 (35%) ZDEG endografts migrated, while 1/23 (4%) ZTEG endografts migrated (p=0.04). At maximum follow-up, 13/23 (57%) ZDEG and 6/23 (26%) ZTEG migrated (p=0.11). At 1 year, the median MD for both groups was similar [ZDEG 4 mm (IQR, 2–10) vs ZTEG 4 mm (IQR, 0–4.5); p=0.11]. At maximum follow-up, the median MD for the ZDEG was significantly longer, at 10 mm (IQR, 6–15.5), compared with 4 mm (IQR, 0–9.5) for the ZTEG group (p=0.016).
The proximal landing zone dilation was similar between groups, both at 1 year [ZDEG: 6% (IQR, 1–9) vs ZTEG 5% (0–9), p=0.52] and maximum follow-up [ZDEG: 10% (IQR, 6–16) vs ZTEG 7% (3–12), p=0.34]. Bird-beak was also similar between groups at 1 year [ZDEG: 4/23 (17%) vs ZTEG 7/23 (30%), p=0.45] and maximum follow-up [ZDEG: 9/23 (39%) vs ZTEG 12/23 (52%), p=0.6].
An rTAAD appeared in a ZDEG patient, which required a proximal ascending aorta replacement 1 month after the implantation. None of the ZTEG patients presented such a complication. Endoleak Ia occurrence did not differ between groups: no endoleak Ia (0/23;0%) occurred in the ZDEG and one (1/23; 4%) was recorded in the ZTEG group (p=1) at 1 year while at maximum follow-up, there was 1 endoleak in the ZDEG (1/23; 4%) and 2 endoleaks in the ZTEG group (2/23; 8%; p=1; Supplementary Table 2). Proximal reinterventions were performed in 3 ZDEG (3/23; 13%) and 2 ZTEG cases (2/23; 9%; p=1; Supplementary Table 3).
Discussion
The single-arm analysis of the ZTEG endograft in patients with aortic syndromes demonstrated a migration rate of 2% at 1 year and 20% during the maximum follow-up; a suboptimal rate, especially considering the absence of aneurysmatic disease in most patients and the presence of barbs on the ZTEG device. 28 However, as compared to ZDEG, a device without active fixation, the ZTEG migration rate was significantly lower at 1 year and the MD was shorter at maximum follow-up. Despite the migration in the ZTEG cohort, the endoleak type Ia rate remained acceptable; approximately 5% during follow-up, along with an early proximal reintervention rate at 1%, which increased to 6% during follow-up. These results were consistent with previously published evidence on ZTEG. 28 Notably, no cases of rTAAD were reported in the ZTEG cohort, suggesting that barbs may not play a causative role in iatrogenic rTAAD.
Current guidelines suggest TEVAR in patients with complicated acute aortic syndromes.1–3 Similarly, TEVAR is the treatment of choice in anatomically suitable patients with chronic aortic syndromes.1–3 Aggressive oversizing and proximal bare-metal stents have been proposed as risk factors for rTAAD, along with other anatomic (eg, aortic arch/ascending aorta dilation, proximal tear within the arch) and intraoperative features (eg, excessive bird-beak), based on scarce evidence.1–3,9,10 Otherwise, no further guidance exists regarding the optimal endograft design for aortic syndromes. Recently, CE approval within the European Union has established the ZDEG endograft as a recommended option for aortic dissections, while the ZTEG has been gradually withdrawn from the market.
No significant difference was demonstrated among the propensity-score matched patients in terms of newly induced rTAAD and/or IMH. No such event was present in the ZTEG, while one event was present in the ZDEG group. The presence of an oversizing of 21% in this patient may have contributed to this outcome.9,10,29 Moreover, no events of early or late rTAAD were present in the single-arm analysis of the ZTEG cohort. This finding contradicts with previous suggestions that patients managed with active fixation TEVAR, through either barbs or proximal bare metal stents are at increased risk for aortic wall injury.11,12 However, no specific evidence on the impact of barbs or hooks on the incidence of rTAAD in patients managed with aortic syndromes exists. Doberne et al 12 concluded that bare-stents or barbs are associated with stent-graft induced aortic wall injury (SAWI). However, their data does not support their conclusion, as the incidence of proximal SAWI of barb-fixated devices (2/45; 4%) was similar to devices without barbs (13/370; 4%). 12 In contrast, prior studies suggest that the absence of hooks and/or barbs on the proximal end of abdominal aortic endografts increases the risk for migration. 30
Between the propensity score–matched groups, significantly more ZDEG patients experienced a migration at 1 year, and more patients demonstrated a significantly higher MD at maximum follow-up. The lack of active fixation in ZDEG may have affected this outcome and led to higher rates of imaging adverse events in this cohort; especially when compared to previous data reporting on a migration incidence of 10% during a 5-year follow-up.30,31 Migration has previously been associated with bird-beak formation, endoleak Ia and proximal reintervention, jeopardizing the efficacy and durability of TEVAR. According to the current analysis, the presence of barbs seemed to decrease the migration rate compared with non-active fixation.24,32 Bird-beak has been related to an increased risk for endograft infolding or collapse. 24 Despite the relatively high rate of bird-beaks in both groups, no major device related complication was detected at least during the initial 3 postoperative years. 24 The median oversizing exceeded 10% in the current cohort and may have affected bird-beak rates, as previously shown. 33
Although proximal landing zone dilation was greater in the ZDEG cohort, this difference did not reach statistical significance. Previous data on proximal sealing zone dilation after TEVAR for aortic dissection demonstrated an estimated proximal aortic landing zone dilation rate of almost 20%.25,34 Proximal landing zone dilation has been, previously, associated with endograft migration and type Ia endoleak.25,35 The presence of barbs and microscopic injury to the intimal layer may trigger an inflammatory response, promoting stronger fixation and improved sealing, thereby limiting migration phenomena. 34 This microscopic wall injury could have facilitated fibrous tissue deposition and prevented further aortic wall dilation in the ZTEG group, limiting sealing zone degeneration. Future adequately powered anatomical studies may clarify this effect.
Despite a significant difference between the 2 groups in terms of endograft migration and MD, no statistically significant difference was observed regarding type Ia endoleaks or proximal reinterventions. 35 Migration, however, has been previously shown to have a substantial impact on these outcomes, as increased graft movement can compromise proximal sealing and predispose to endoleak formation and subsequent need for proximal reintervention. 35 In the present study, both type Ia endoleak and proximal reintervention rates were relatively low during follow-up. The nature of the underlying diseases, with absent large aneurysm sac below the landing zone, potentially explains the fact that migrations did not lead to reinterventions; a sealing even in more distal aortic segments was still achieved. However, optimization of the technique and devices seems mandatory, especially under the lack of long-term evidence.
Limitations
The retrospective design, underpowered sample size, and patient selection bias, introduced by surgeon’s preference in endograft selection, are the most important limitations. Inclusion of aortic syndromes in both acute and chronic setting may have had a significant impact on heterogeneity. Although propensity score matching has been used to address selection bias, the small size inherently inflated potential type I and type II statistical errors, while limiting the application to a wider range of baseline parameters. Anatomic characteristics were chosen for matching due to the greater importance of their nature based on the type of outcomes assessed. Loss to follow-up may have affected the statistical power to detect differences in clinical outcomes. Eligibility criteria may have, also, affected selection and impacted generalizability of the results. The absence of core lab for imaging assessment may have influenced measurement preciseness. The use of aorta’s greater curvature for distance measurements has been preferred due to better performance and higher precision in the aortic arch (zones 2–3 and proximal zone 4). However, the absolute conformity with the reporting standards and direct comparisons to previous evidence may have been disrupted. Under this fact, a potential overestimation of events cannot be excluded. 20 In addition, the lack of a standardized definition of bird-beak in reporting standards led to the use of the most applied definition in the available literature and which may have biased the results. 24
Conclusions
Barb-mediated active fixation in ZTEG may provide a benefit in terms of device migration in patients with aortic syndromes, either acute, subacute, or chronic. Compared with the non-active fixation, ZDEG revealed no difference in terms of rTAAD, endoleak Ia, and proximal reintervention, while decreasing the migration rate in half and MD during follow-up.
Supplemental Material
sj-docx-1-jet-10.1177_15266028261428265 – Supplemental material for A Retrospective Analysis of Barb-mediated Active Fixation Impact on Thoracic Endovascular Aortic Repair Outcomes
Supplemental material, sj-docx-1-jet-10.1177_15266028261428265 for A Retrospective Analysis of Barb-mediated Active Fixation Impact on Thoracic Endovascular Aortic Repair Outcomes by George Apostolidis, Petroula Nana, Giuseppe Panuccio, Alessandro Grandi, Jose I. Torrealba and Tilo Kölbel in Journal of Endovascular Therapy
Footnotes
Acknowledgements
None.
Ethical Considerations
This study complied with the Declaration of Helsinki and no approval by the ethics committee was required due to study design.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tilo Kölbel is a consultant for Cook Medical and Getinge, and proctor for and has intellectual property with Cook Medical, receiving royalties, speaking fees, and research, travel, and educational grants. Petroula Nana is a consultant for Getinge receiving research and educational grants. All authors declare no support from any organization for the submitted work; no financial relationships with any organizations that might have an interest in the submitted work; no other relationships or activities that could appear to have influenced the submitted work.
Data Availability Statement
Data can be provided by the corresponding author upon request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
