Abstract
Purpose:
Peripheral arterial disease (PAD) is a major global health burden often requiring endovascular intervention. Complex lesion morphologies such as calcification or long occlusions limit procedural success. Vessel preparation (VP) techniques, including atherectomy and intravascular lithotripsy, are increasingly adopted, but consensus on their definition, purpose, and clinical role is lacking. The main objective was to define VP in endovascular PAD interventions, determine its primary aims, and identify key barriers and enablers for its adoption in clinical practice.
Materials and Methods:
A modified Delphi process was conducted involving 103 international experts across vascular surgery, interventional radiology, angiology, and cardiology. Two rounds of online surveys and 11 structured interviews were completed, with ≥70% agreement predefined as consensus. Qualitative thematic analysis was used for free-text and interview data.
Results:
VP was defined as “the initial step in an endovascular procedure to facilitate subsequent interventions by modifying lesion characteristics” (93% agreement). Six core aims were established: luminal gain, improved vessel compliance, plaque/calcification reduction, enhanced drug delivery, reduced complications, and improved technical success. Barriers included high device costs and insufficient evidence. High-level agreement supported VP use in femoropopliteal and popliteal segments, with intravascular lithotripsy preferred for calcified lesions. Imaging recommendations and modality-specific VP guidance were also developed.
Conclusion:
This work provides the first international definition and structured aims of VP in PAD. It identifies practical guidance, barriers to adoption, and priorities for future research. Findings will support standardisation in clinical practice, research, and health policy regarding VP technologies in PAD treatment(s).
Clinical Impact
This international consensus provides the first standardised definition, core aims, and practical guidance for vessel preparation in endovascular treatment of peripheral arterial disease (PAD), enabling more consistent clinical practice, research design, and health policy development worldwide.
Introduction
Peripheral arterial disease (PAD) is a growing global health issue, with significant impact on quality of life, amputation risk, and mortality.1–6 Endovascular revascularization has become the preferred strategy in many patients with symptomatic PAD, particularly in the femoropopliteal segment.4,5 However, lesion complexity, heavy calcification, and anatomical challenges frequently compromise the long-term success of standard endovascular tools such as plain balloon angioplasty (PGA) and drug-coated balloons (DCBs). 7 This has led to the growing adoption of vessel preparation (VP) techniques—such as atherectomy, intravascular lithotripsy (IVL), and specialty balloons—intended to optimise lesion compliance, enhance drug uptake, and potentially reduce the need for stenting.
Despite increasing use of VP devices, there is currently no universally accepted definition of what VP entails, and its goals remain inconsistently stated across trials and clinical practice. This is despite international guidance advocating the consideration of VP when intervening in people with PAD. 8 Moreover, evidence for VP efficacy/effectiveness is limited and often conflicting. 7 A recent systematic review and network meta-analysis highlighted that while VP with atherectomy or IVL may reduce bailout stenting, it might not improve key patient outcomes such as freedom from target lesion reintervention, major amputation, or mortality; it also identified significant gaps in VP evidence, in particular for patients with chronic limb-threatening ischaemia (CLTI), long or heavily calcified lesions, and popliteal artery disease. 7
There is a pressing need for expert consensus to define VP in PAD interventions, outline its aims, identify current barriers and enablers to its adoption, and guide future research. This work aimed to address these unmet needs through a structured international modified consensus process involving experts in PAD intervention, to establish a clear and practical position statement on VP in endovascular PAD treatment.
Materials and Methods
Aim and Objectives
Main aim
Define the term VP in endovascular PAD revascularization and the aims/intentions of VP in this clinical context.
Secondary aims
Explore barriers and enablers regarding the use of VP, best practices using currently available VP devices/technologies, and critical areas for future research in this clinical context.
Objectives
Perform a series of global online surveys and qualitative work to:
Define the term VP in PAD revascularization, using a modified Delphi consensus approach;
Define the term aims of VP in PAD revascularization, using a modified Delphi consensus approach;
Explore the barriers and enablers regarding the use of VP in this clinical context per anatomical area;
Explore principles of best clinical practice when using VP based on experts’ opinions;
Explore important areas for future research relating to VP;
Produce a position statement, following review and approval by collaborators, regarding the above objectives.
Ethical Approval and Regulatory Considerations
This project did not involve patients, or anyone receiving medical/invasive treatment as part of this process. All participants were qualified health care professionals, recruited internationally (Tables 1 and 2, Supplemental Appendix 1). Written informed consent was sought from all participants taking part in all relevant activities, either remote (online) or face-to-face. No identifiable data were recorded or shared at any point. All participants agreed to the analysis and recording of their views (written consent). The preparation of the surveys and the conduct of the Delphi consensus were carried out between November 2024 and March 2025. The East Midlands Research Ethics Committee provided confirmation that this project does not require ethical approval from a National Health Service (NHS) Committee as it does not involve patients or use of patients’ data/information. No payments were made to those taking part in the project. The University of Leicester (United Kingdom) ethical approval committee approved this work in December 2024 (Reference: 2195).
Summary of Key Characteristics of the 103 Participants.
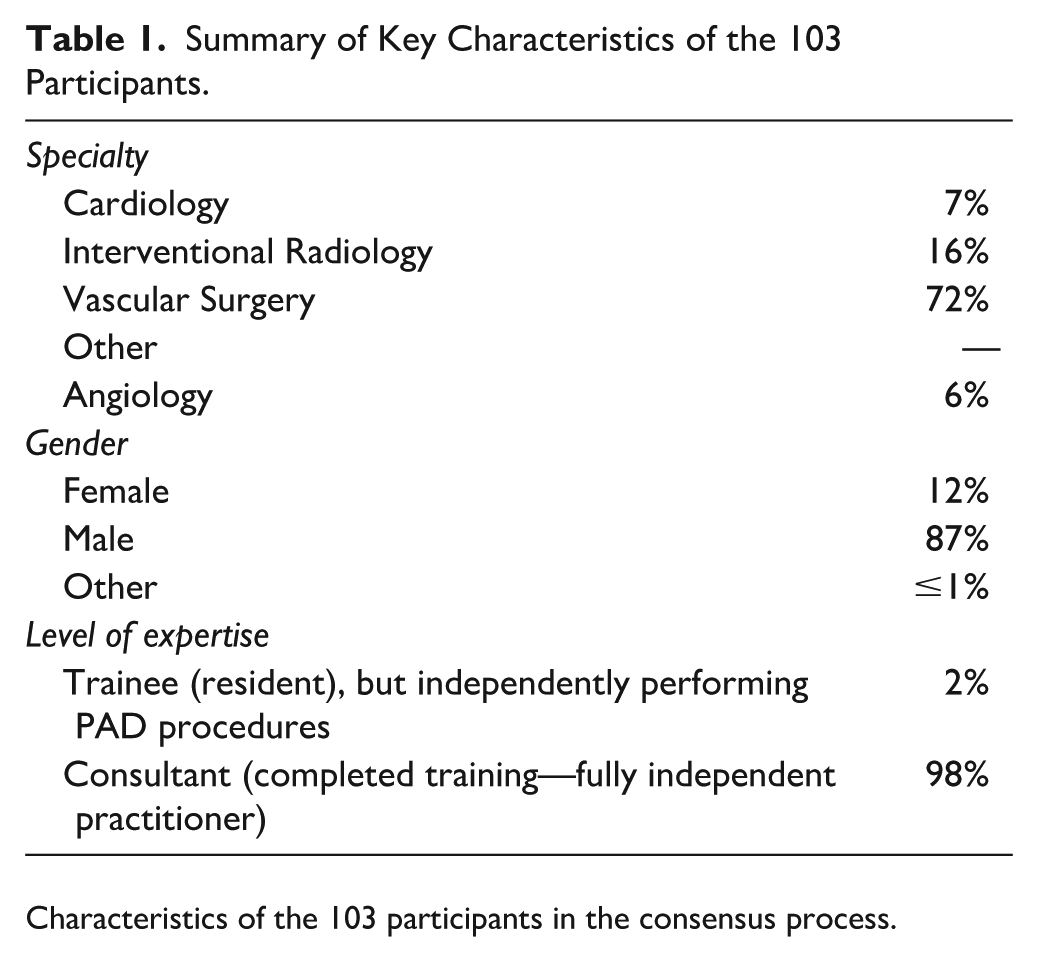
Characteristics of the 103 participants in the consensus process.
Demographics of Participants.
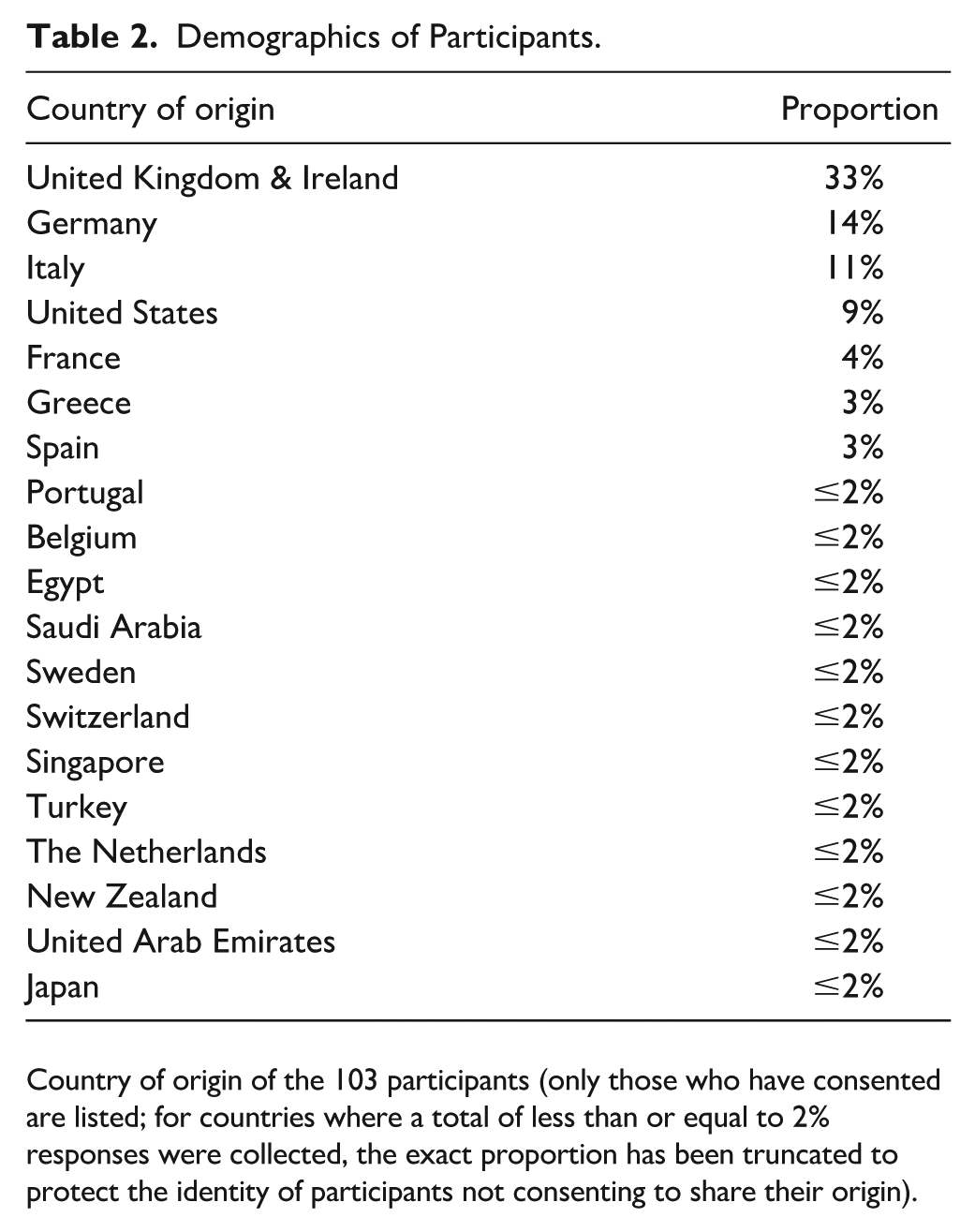
Country of origin of the 103 participants (only those who have consented are listed; for countries where a total of less than or equal to 2% responses were collected, the exact proportion has been truncated to protect the identity of participants not consenting to share their origin).
Process to Achieve Consensus and Finalise Recommendations
An online and face-to-face modified Delphi process 9 was performed adhering to recommendations by COMET9–11; we opted for the use of a modified version of the Delphi process, whilst still adhering to COMET/CONSORT/CROSS guidance,9–11 to ensure that all participants had sufficient experience using the devices/technologies included in this work, as per the participant inclusion criteria listed below:
Inclusion Criteria
Health care professional with expertise in endovascular treatment of PAD with a minimum of 3 years independent experience as first operators in PAD interventions.
Experience in using VP technologies for at least 1 year prior to the survey being disseminated.
Exclusion Criteria
Clinician without significant involvement in PAD treatment or VP.
Immediate conflict of interest that may immediately bias their participation (employed by a relevant company producing VP technologies, holding shares in any relevant company).
The below sequential steps were followed in the modified Delphi approach, to address all objective and produce the required position statement:
Literature review to inform the subsequent survey questions and interview discussions; published separately. 7
Structured online survey to explore key areas of interest.
Core team online meeting to explore results of the survey.
Interviews to resolve unclear areas and finalise the second online survey, until data saturation was reached.
Second structured online survey.
Core team online meeting (second round).
Finalisation of recommendations and statements.
External review of recommendations and statements (E.A.S., A.D., L.P.).
Final review of report by all collaborators..
This process provided the necessary qualitative and quantitative data which were then used to reach agreement based on the modified Delphi approach and complementary qualitative analysis of both free-text (surveys) entries and views expressed during interviews (until saturation).
Recruitment
Identification
All participants were identified prospectively based on the above inclusion criteria, via the Research Collaborative for Peripheral Arterial Disease (RCPAD; rcpad.org) membership list. A total of 202 RCPAD members fulfilling the inclusion criteria were identified and notified to take part via email invitations in January 2025 (electronic invitations). Details regarding the RCPAD group are available on rcpad.org. This is an international cross-specialty group of vascular specialists.
Invitation
Personalised invitations via email were sent using a generic email invitation via the RCPAD main office, outlining the project.
Follow-up
Reminder emails to enhance response rates, ensuring broad and representative participation were sent weekly in January and February 2025 via a central RCPAD administrator on behalf of the lead authors (A.S., K.S.).
Literature Review (First Step)
A systematic literature review and subsequent network meta-analysis was completed by our group prior to this work and was the basis of the survey questions, for both rounds 1 and 2. 7
Online Structured Surveys (Second Step)
An online structured survey was designed by the lead authors (A.S., K.S.) and RCPAD committee, based on the above review and considerations regarding pre-/intra-/post-operative use of VP in this clinical context. The survey used both closed questioning and free-text entries; the survey was finalised and disseminated in January 2025, as above.
Participants were asked to rank the importance of each query explored in the survey (identified in the first step) using a Likert scale, as recommended by the Grading of Recommendations, Assessment, Development and Evaluation working group and/or based on binary (Yes/No) questions, expressing their preferences. Participants ranked items and differences in rankings between stakeholders were explored to finalise potential topics of discussion during the subsequent interviews and second round survey and eventually finalise the recommendations. Supplemental Appendix 2 lists all questions in the online surveys (online components of the work undertaken). Those interviewed were selected randomly from across 103 participants, ensuring at least 50% of the sample were female operators (6 of the 11) and spread across Europe, 5 the United States, 3 and Australasia. 3
Definitions
All definitions regarding anatomy and clinical considerations were as per the latest global guidelines on CLTI. 4
Analysis
The online survey results were analysed in tabular format and key areas of interest were then discussed 2 online meetings by the core investigators (RCPAD group): preoperative, intraoperative, and postoperative considerations, surrounding clinical and technical decision-making. Following that, a total of 11 interviews were completed with 5 male and 6 female survey participants to resolve areas of uncertainty and finalise the format and content of the second online survey.
Comprehensive field notes were taken by the team during the interviews and during online meetings, which were also recorded and transcribed verbatim. Thematic analysis was used to organise the views expressed in the interviews and the free-text replies of the survey into themes. 12 All participants were invited to leave their email address to be contacted for comments they did not wish to share openly (2 participants shared further views with the lead author). No identifiable data (eg, date of birth or address) were collected; email addresses were stored in a secure hard disk drive for future contact (with the participants’ consent).
The shortlist of statements was reviewed by the core study group (K.S., A.S.) to agree on the wording of the final set of standards, as recommended by the COMET Initiative and was also shared with all stakeholders prior to publication. These were then shared with all stakeholders completing the survey and taking part in interviews, for final feedback and subsequent consensus to be reached.
To reduce bias, a predetermined agreement threshold was used: Standards ranked of critical importance6–9 by >60% or of little importance1–3 by <15% by participants were deemed to have reached the threshold for consensus for inclusion. Any items/recommendations that were ranked of critical importance by <10% of stakeholders or of little importance by >60% of the group were excluded from the final list of agreement points, following the online surveys and subsequent interviews. In terms of agreement regarding the subsequent phrasing of the recommendations, unanimous agreement was stated when all participants agreed regarding a statement, high-level agreement was reached when ≥70% agreed and moderate agreement when 50% to 70% had agreed; of note, all statements described in this wok reached at least 70% or higher agreement.
Results
Participant Characteristics
A total of 103 individuals took part (Table 1) of the 202 invited (51% response rate) in the 2 online surveys, followed by 11 interviews. All participants filled in all components of the surveys. All participants had at least a minimum of 3 years of experience using VP devices in routine clinical care (range 3–22 years; median: 7 years); the vast majority (98%) were independent practitioners (“consultants”) for at least 5 years. Participants reported that their host centres perform a mean of 560 procedures (standard deviation [SD]: 82) for PAD per year and a mean of 120 procedures are led by themselves (SD: 33). Procedures were discussed in a multidisciplinary team meeting in 76% of centres prior to proceeding with the intervention. Most participants (76%) were vascular surgeons, with a range of specialties having taken part, including radiology, angiology, and cardiology (Table 1). The majority (76%) work in teaching centres, across all major continents (Europe, Americas, Asia); see Table 2 for further details.
Definition and Aims of VP
Following 2 rounds of online surveys and qualitative interviews, there was almost unanimous agreement that VP in endovascular PAD treatment(s) should be defined as: “The initial step in an endovascular procedure to facilitate subsequent interventions by modifying lesion characteristics”—93% agreement.
The following aims of VP in endovascular PAD treatment(s) were identified via thematic and framework analysis of the free-text replies and interviews’ transcripts; there was again 93% agreement in the second round of online survey regarding these 6 aims:
1. Luminal Gain and Vessel Expansion
Increase vessel lumen diameter before using subsequent treatment.
2. Modification of Vessel Compliance
• Change vessel wall characteristics to improve flexibility • Reduce recoil of the arterial wall
Enhance pulsatility
3. Plaque and Calcification Burden Reduction
• Remove/reduce atherosclerotic, fibrotic, and thrombotic material within the lesion • Fracture or modify calcification • Reduce plaque burden to facilitate optimal stent expansion
Lower the incidence of distal embolisation
4. Drug Delivery and Retention
Enhance drug uptake by modifying vessel wall properties
5. Reduce Procedural Risks and Complications
Minimise dissections, elastic recoil, and vessel rupture
Reduce need for bailout stents
Reduce loss of collateral circulation
6. Technical Success and Procedural Efficiency
Enhance procedural safety with lower balloon pressures necessary and optimised dilation/stent-expansion.
The above are summarised in Figure 1.
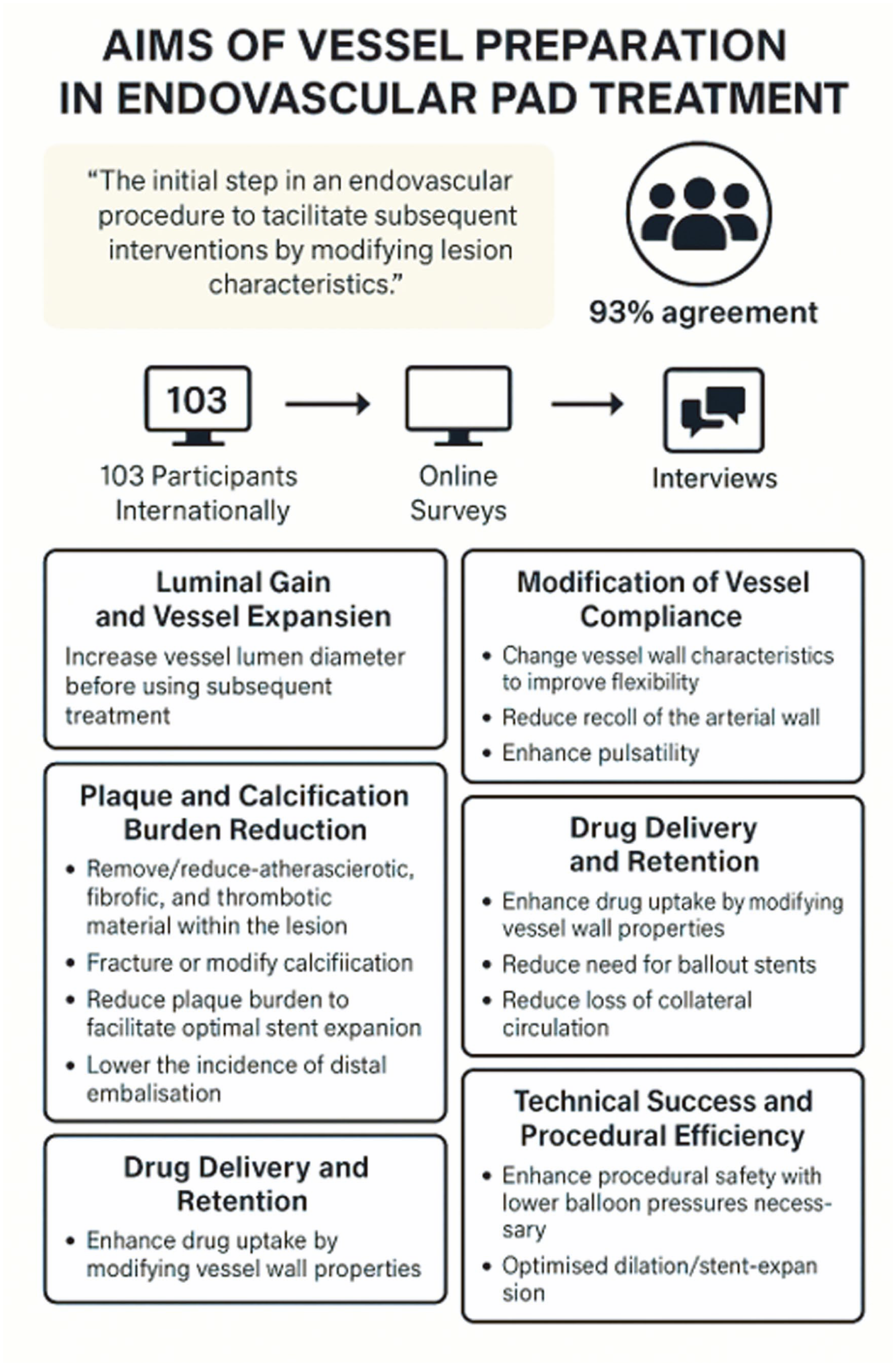
Summary of key findings relating to use of vessel preparation (VP) in peripheral arterial disease (PAD) including definition and aims of VP.
Types of VP Used by Participants in Routine Care and Anatomical Considerations
The vast majority of participants (98%) use PBA most commonly in their routine clinical care; however, 93% used additional modes of VP to PBA in the last 1 year, with IVL being the most widely used modality (52%), followed by high-pressure angioplasty (23%), rotational atherectomy (20%), and directional atherectomy (10%). The majority of participants routinely use VP in the superficial femoral artery (94%), with high-level agreement (73%) across both surveys and also in the qualitative interviews. This was followed by routine VP use in the popliteal artery at 86% (see Supplemental Appendix 3). Of note, there was high-level agreement (73%) that VP should be used in the superficial femoral artery across both surveys and also in the qualitative interviews. Finally, 93% of participants agreed that better VP devices have to be made available for the aorto-iliac segment and 63% agreed this is the case for the crural segment.
Contraindications for Use of VP
Both in the online surveys and interviews, there was wide variation in terms of what participants thought might constitute a contraindication to using VP, with no agreement reached in either round. The 2 most common contraindications identified were (1) subintimal crossing (63% agreement that this is a contraindication) and (2) presence of circumferential thrombus (48% agreement). None reached agreement above 70%, as per the prespecified criteria. In qualitative interviewing, it was felt that frailty and a life-expectancy of less than 1 year might be a clinical (but not lesion-based) contraindication. Those interviewed were in agreement that presence of thrombus and subintimal crossing are the 2 instances where use of VP should be performed with caution, but do not necessarily constitute a contraindication. Almost all participants (101/103) found that rotational and/or directional atherectomy should not be used in the subintimal space.
Barriers Regarding the Use of VP
There was wide agreement (>70% in both cases) that the high cost of each individual device and lack of high-quality randomised evidence regarding clinical and cost-effectiveness of VP devices are the 2 main barriers behind more widespread use. Overall, 95% of participants expressed that there is a need for randomised effectiveness-driven research for all devices as well as for combination(s) of treatments, that is, VP followed by a definitive treatment. There was high-level agreement (71%) that high cost is a barrier regardless of whether the device is re-imbursed or not. Of interest, participants from both Europe and the United States, as well as other regions found that cost of VP devices is a key criterion regarding whether they select to use VP or not, regardless of reimbursement. This was corroborated in interviews (unanimous agreement).
Best Imaging Practices to Assess Whether VP Is Required and Plan a Subsequent Endovascular Procedure
There was high-level agreement that computed tomographic angiography (CTA) is required for all aorto-iliac lesions (93%) and for all femoropopliteal calcified lesions. Duplex ultrasonography was deemed the most appropriate imaging first-line modality in femoral (71%), popliteal, (98%), and crural (93%) lesions. A total 71% agreed that, if cost was not an issue, all lesions should be assessed with a combination of CTA, duplex, and diagnostic angiography in 2 views prior to using VP, and almost all agreed (102/103) that a combination of CTA and duplex is necessary to assess a lesion preoperatively in the aorto-iliac and femoropopliteal segment if considering VP use. Finally, the use of intravascular ultrasound was found (68%) to be a useful adjunct in planning and after using VP; the latter in particular was supported by all those interviewed (100%). In total, 91% agreed that inflow-outflow needs to be assessed on a case-by-case basis using CTA, duplex, and intra-procedural angiography.
Choice of VP Modality
Calcified lesions
In calcified lesions, there was high-level agreement (>75% in all cases) that IVL should be the first-line VP modality in aortic, iliac, common femoral, superficial femoral, profunda, and popliteal lesions. No agreement was reached for crural calcified lesions—this was identified as an area for future research by all those interviewed. The high-level agreement on use of IVL in calcified lesions persisted even for eccentric calcification. There was 50% agreement that rotational atherectomy should be considered in selected patients with calcified common femoral artery disease.
In-stent restenosis or occlusion
(a) Thrombo-aspiration was deemed as the most appropriate VP device for iliac stents (80%), but not for femoropopliteal stents (only 60%).
(b) Rotational atherectomy was deemed as the most appropriate VP device for femoropopliteal stents (73%). Participants did not express preference regarding the exact rotational atherectomy device.
Long lesions or occlusions
No agreement could be reached regarding optimal use of VP for long lesions (≥20 cm) or chronic calcified total occlusions ≥20 cm. There was near unanimous agree ment (98%) that improved device designs are required for such lesions, both in the free-text entries and interviews.
Thrombotic lesions
There was near unanimous agreement (98%) and strong views expressed in interviews (by all 11 interviewees) that endovascular thrombo-aspiration should be attempted as the first step in iliac and femoropopliteal lesions with thrombotic components. Interviewees expressed the need for improved device designs regarding crural thrombus, unanimously. Following use of endovascular thrombo-aspiration, survey participants and all interviewees expressed that the lesion should be re-assessed and a case-by-case decision should be made. The use of filters failed to reach high-level agreement (53%); interviewees also expressed unanimously that filters should only be considered on a case-by-case basis, depending on lesion characteristics; however, no one was aware of any relevant evidence in this area (filter use).
Definitive Treatment
Regarding definitive treatment following successful VP, based on current data, the following high-level agreement was reached per anatomical segment:
Iliac arteries—use of covered stents, both for common/external iliac arteries (71%)
Common femoral artery (and profunda)—use of drug-coated balloon angioplasty (73%)
Superficial femoral artery—use of drug-coated balloon angioplasty (73%)
Crural arteries—use of PBA (70%); of note, drug-coated balloons for crural arteries are not available across all 19 countries represented in this work.
In interviews, strong views were expressed regarding the lack of clinical/cost-effectiveness randomised data to support choice of subsequent treatment in any anatomical area.
Supplemental Appendix 4 lists the reporting of all survey and qualitative data as per the Consensus-Based Checklist for Reporting of Survey Studies (CROSS) checklist.
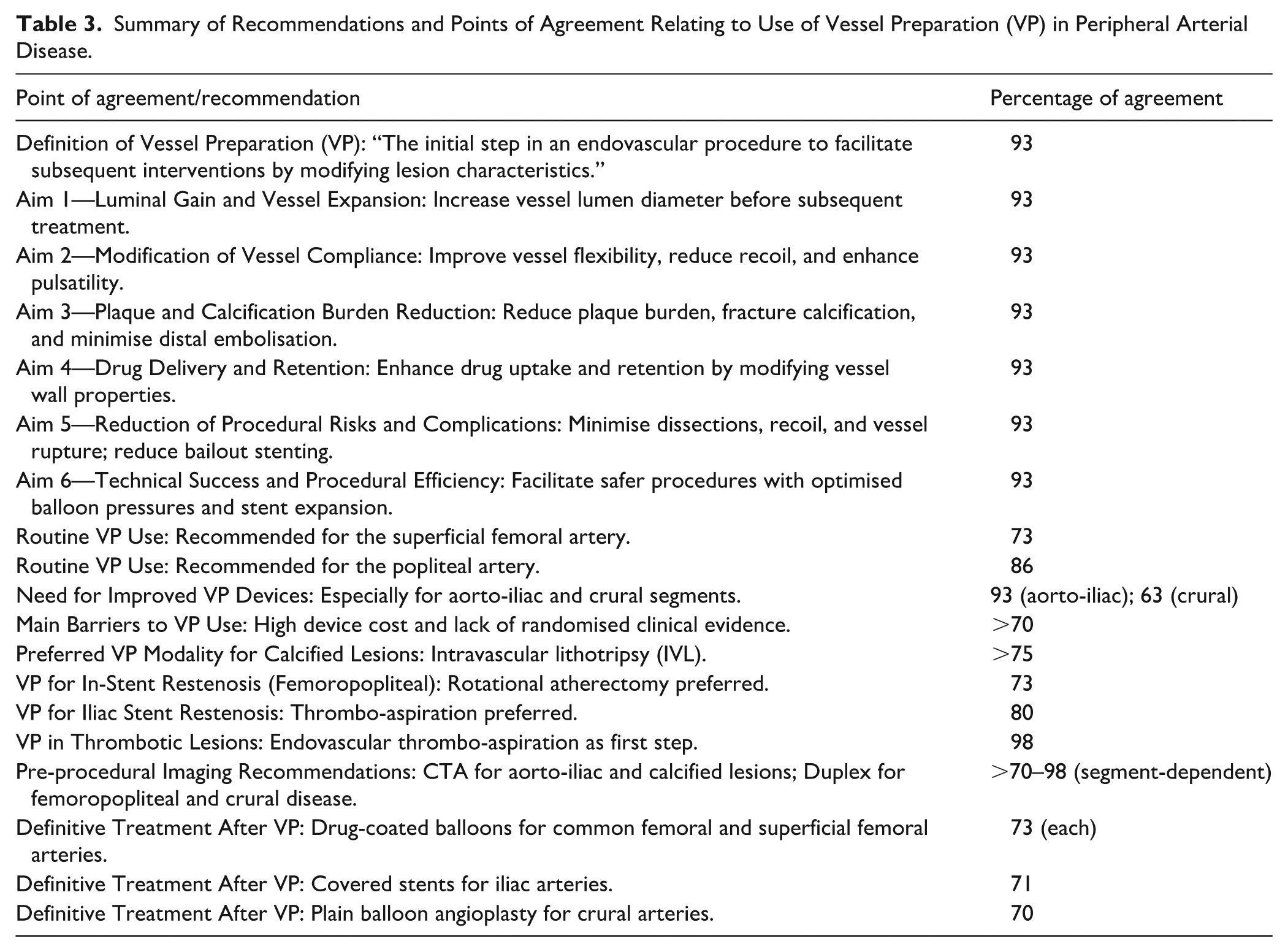
Table 3 provides a summary of the recommendations and points of agreement with relevant percentages of agreement/consensus amongst participants.
Summary of Recommendations and Points of Agreement Relating to Use of Vessel Preparation (VP) in Peripheral Arterial Disease.
Discussion
This work represents the first international effort to provide a structured, expert-derived definition of VP in endovascular PAD treatment, encompassing all relevant anatomical segments—aortic, iliac, femoral, popliteal, and crural vessels. Despite the increasing use of VP in clinical practice, there has, until now, been no consistent definition of what VP entails or consensus on its primary objectives; at the same time, European guidance advocates considering VP when intervening in those with PAD, 8 without, however, defining what VP refers to. Through a modified Delphi methodology approach involving more than 100 experienced interventionists from 19 countries across all relevant specialties, this work delivers a foundational definition of VP as “the initial step in an endovascular procedure to facilitate subsequent interventions by modifying lesion characteristics.” Furthermore, it describes/defines 6 core aims (luminal gain, improved compliance, plaque modification, drug uptake enhancement, complication reduction, and procedural efficiency) thus providing, for the first time, a unified framework that can guide clinicians, researchers, and policymakers.
In the absence of standardised terminology and agreed-upon goals, previous studies have reported inconsistent outcomes and variable use of VP strategies across regions and institutions, as showed in a review by this group. 7 By clearly defining the purpose and desired effects of VP, this work lays the groundwork for more uniform clinical application and improved interpretation of future research outcomes. It also serves as a valuable resource for clinicians seeking to understand which imaging modalities and definitive therapies are appropriate before and after VP, with specific guidance offered per anatomical segment.
This work also highlights key areas for future research. An attempt to develop a femoropopliteal segment treatment algorithm, including VP, has been made, 13 but was not based on randomised evidence. This is also reflected in this Delphi consensus, as the description of device preferences is useful but is based on expert opinion rather than evidence-based recommendations. There is a pressing need for high-quality, independently-funded randomised controlled trials (RCTs) that evaluate not only the clinical effectiveness of VP devices but also their cost-effectiveness in different arterial segments. Trials exploring combinations of VP with the so-called “definitive therapies”—such as DCBs or various stent types—are especially important. In addition, the development of a core outcome set for PAD interventions, including VP-specific endpoints, is essential to ensure consistency and comparability across studies. Furthermore, trials should be as pragmatic as possible, as the vast majority of randomised research evaluating modern PAD technologies use strict clinical and anatomic inclusion criteria that exclude most patients from taking part. 14
The high cost of VP devices remains a major barrier to widespread adoption. While they may reduce long-term health care costs by lowering the need for reintervention, 15 this hypothesis remains unproven without robust RCT data. Until then, uptake will vary depending on local reimbursement policies and institutional budgets.
Regarding the contraindications for VP, no agreement could be reached. Concerns were raised about the use of atherectomy in the subintimal space, probably due to the higher risk of perforation and when thrombus is present. However, many physicians suggested the use of mechanical or aspiration thrombectomy in cases of thrombotic disease. In this context, the presence of thrombus cannot be considered a contraindication for all VP devices. Finally, during the in-person interviews, frailty was considered a relative contraindication due to the prolonged procedures that VP may require. Overall, there are no clear contraindications for VP, and decisions should be based on the complexity of the individual clinical scenario.
Study Limitations
This work has several limitations. First, the evidence supporting VP technologies remains limited, with few high-quality, independently-funded RCTs available. As such, recommendations are primarily based on expert opinion rather than robust comparative data. Given the lack of high-quality evidence, the Delphi method primarily reflects expert opinion, which is influenced by individual preferences, and consensus does not necessarily imply correctness. Furthermore, this Delphi consensus cannot substitute for randomised evidence or the development of formal guidelines. Second, the lack of a standardised core outcome set for PAD and VP interventions limits the consistency and comparability of future studies. In addition, while the consensus process included a diverse expert panel, patient involvement was limited, and real-world applicability may vary across health care systems. Finally, the high cost of VP devices and associated resource variability may impact global implementation.
Conclusion
In summary, this work identified, for the first time, a formal definition and set of aims for VP in PAD intervention. It offers a practical guide to imaging and treatment selection, identifies current limitations, and sets the stage for future trials and clinical guidelines.
Supplemental Material
sj-docx-1-jet-10.1177_15266028261424732 – Supplemental material for Use of Vessel Preparation in Endovascular Peripheral Arterial Disease (PAD) Interventions: A Global Qualitative Analysis
Supplemental material, sj-docx-1-jet-10.1177_15266028261424732 for Use of Vessel Preparation in Endovascular Peripheral Arterial Disease (PAD) Interventions: A Global Qualitative Analysis by Athanasios Saratzis, Lorenzo Patrone, Eric A. Secemsky, Anahita Dua, Hany Zayed, Giovanni Torsello, Isabelle Van Herzeele and Konstantinos Stavroulakis in Journal of Endovascular Therapy
Supplemental Material
sj-docx-2-jet-10.1177_15266028261424732 – Supplemental material for Use of Vessel Preparation in Endovascular Peripheral Arterial Disease (PAD) Interventions: A Global Qualitative Analysis
Supplemental material, sj-docx-2-jet-10.1177_15266028261424732 for Use of Vessel Preparation in Endovascular Peripheral Arterial Disease (PAD) Interventions: A Global Qualitative Analysis by Athanasios Saratzis, Lorenzo Patrone, Eric A. Secemsky, Anahita Dua, Hany Zayed, Giovanni Torsello, Isabelle Van Herzeele and Konstantinos Stavroulakis in Journal of Endovascular Therapy
Supplemental Material
sj-docx-3-jet-10.1177_15266028261424732 – Supplemental material for Use of Vessel Preparation in Endovascular Peripheral Arterial Disease (PAD) Interventions: A Global Qualitative Analysis
Supplemental material, sj-docx-3-jet-10.1177_15266028261424732 for Use of Vessel Preparation in Endovascular Peripheral Arterial Disease (PAD) Interventions: A Global Qualitative Analysis by Athanasios Saratzis, Lorenzo Patrone, Eric A. Secemsky, Anahita Dua, Hany Zayed, Giovanni Torsello, Isabelle Van Herzeele and Konstantinos Stavroulakis in Journal of Endovascular Therapy
Supplemental Material
sj-docx-4-jet-10.1177_15266028261424732 – Supplemental material for Use of Vessel Preparation in Endovascular Peripheral Arterial Disease (PAD) Interventions: A Global Qualitative Analysis
Supplemental material, sj-docx-4-jet-10.1177_15266028261424732 for Use of Vessel Preparation in Endovascular Peripheral Arterial Disease (PAD) Interventions: A Global Qualitative Analysis by Athanasios Saratzis, Lorenzo Patrone, Eric A. Secemsky, Anahita Dua, Hany Zayed, Giovanni Torsello, Isabelle Van Herzeele and Konstantinos Stavroulakis in Journal of Endovascular Therapy
Footnotes
Acknowledgements
None.
ORCID iDs
Ethical Considerations
The East Midlands Research Ethics Committee provided confirmation that this project does not require ethical approval from a National Health Service (NHS) Committee as it does not involve patients or use of patients’ data/information. No payments were made to those taking part in the project. The University of Leicester (United Kingdom) ethical approval committee approved this work in December 2024 (Reference: 2195).
Author Contributions
A.S., K.S.: Conception, design, analysis, reporting, funding, data collection.
H.Z., I.V.H., G.T., A.D., E.A.S.: data collection, reporting, external review, analysis.
VPAD collaborators: data collection, qualitative interviewing, approval, critical review, reporting.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly funded by Shockwave Medical Ltd., Boston Scientific Ltd., Medtronic Ltd., Philips Ltd., who covered teleconferencing costs, software costs, and travel, but did not take part in data analyses or pay any salaries; funders did not have any input in collecting, maintaining, or analysing data (no funding reference[s] available). The work was delivered by the Research Collaborative for Peripheral Arterial Disease, an independent entity. All authors and collaborators have reviewed, edited, and approved this manuscript and have not been paid to take part in this work.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Athanasios Saratzis: Honoraria and lecture fees/consulting for Shockwave, Abbott, Cook; educational grant support from Cook; research funding from Shockwave, Abbott, Boston Scientific, Angiodroid. Konstantinos Stavroulakis: Consulting for Phillips, Shockwave, Terumo, Boston Scientific, received Honoraria from Medtronic, Abbott, Cook, Bentley, Biotronik; research grants from Boston Scientific. Lorenzo Patrone: Speaker/proctor/consultant for Abbott Medical, Angiodroid, Asahi, BD, Bentley, Biotronik, Cook Medical, Shockwave, Terumo. Giovanni Torsello: Research funding and speaker honoraria from Boston Scientific, WL Gore, Cook, and Medtronic. Hany Zayed: Speaker/Proctor/Consultant for Abbott Medical, Boston scientific, Bentley, Cook Medical, Gore Medical, LimFlow and Cordis; institutional research grants from Abbott Medical. Eric Secemsky: Funding: NIH/NHLBI K23HL150290; Grants to Institution: BD, Boston Scientific, Cook, Medtronic, Philips; Consulting: Abbott, Asahi, BD, Boston Scientific, Cook, Cordis, Endovascular Engineering, Evident Vascular, Gore, InfraRedx, Medtronic, Philips, RapidAI, Rampart, R3, Shockwave, Siemens, SoniVie, Teleflex, Terumo, Thrombolex, VentureMed, Zoll. Anahita Dua: Speaker for Boston Scientific, Abbott, Gore, Medtronic, Cook. None of the collaborators hold shares in companies relevant to this work or have been paid to contribute to this work.
Data Availability Statement
Anonymised non-identifiable tabular data (no quotes) are available after contact with the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
