Abstract
Introduction:
A consensus exists among vascular surgery societies that blunt thoracic aortic injury (BTAI) should be treated with thoracic endovascular aortic repair (TEVAR) in patients with favorable aortic anatomy. However, selecting the correct landing zones and endograft sizing remains challenging in patients with BTAI. To overcome these limitations, conformable endografts are often used for TEVAR, even in patients with BTAI. This study aimed to analyze the outcomes of these patients with respect to anatomical and technical factors.
Methods:
This retrospective study included all patients with BTAI who underwent TEVAR at a tertiary vascular center. The primary endpoint was aortic-related reintervention-free survival. Secondary endpoints were 30-day in-hospital mortality, postoperative complications, and radiological findings. Patients were divided into 2 groups: group 1, patients with a conformable endograft (cTAG Gore), and group 2, patients with a nonconformable endograft.
Results:
Between June 2001 and May 2019, 57 patients (50 men; mean age, 37.3 years) with BTAI received 30 (52.6%) conformable (Gore cTAG) and 27 (47.4%) nonconformable endografts. The 1-, 3-, and 5-year aortic-related reintervention-free survival was 100%, 100%, and 96.7% in group 1 and 81.4%, 81.4%, and 77.7% in group 2, respectively (p=0.03). Two postoperative type Ia endoleaks and 3 postoperative type II endoleaks were observed in group 2. The bird beak sign was noted more often in group 2 (70.4% vs 53.3, p=0.32).
Conclusions:
This study, in a small cohort of patients, demonstrated that the Gore cTAG, when properly selected according to the patient’s anatomy, appears to provide better outcomes after TEVAR for patients with BTAI than nonconformable devices. A bird beak sign occurred frequently in both groups and were not associated with poor outcomes. Further studies are required to confirm these findings.
Clinical Impact
This study in a small cohort of patients demonstrated that the Gore cTAG endoprosthesis, when properly selected to match the patient’s anatomy, appears to result in better outcomes after TEVAR in patients with BTAI than nonconformable devices. In addition, the BBS was common in both groups of patients and was not associated with a worse outcome.
Keywords
Introduction
Thoracic injuries remain a common finding in trauma patients presenting to emergency departments worldwide. According to a study from a large trauma center, major thoracic vessel injuries are rare in polytrauma 1 ; however, they usually have tragic consequences, with approximately 70% of victims dying at the accident scene. 2
Blunt thoracic aortic injury (BTAI) is the most common cause of traumatic death after head injury. 2 BTAI is defined as a tear in the aorta resulting from a combination of shear and stretch forces, including deceleration, 3 increased intravascular pressure, 4 and entrapment of the aorta between the vertebrae and anterior chest wall. 5 The most common lesion location is the aortic isthmus, 6 which is due to the fact that this part of the great vessel is a transition from the unfixed aortic arch to the fixed descending thoracic aorta 7 and the relatively lower tensile strength of this region.8,9
The consensus among vascular surgery societies is that BTAI should be treated via thoracic endovascular aortic repair (TEVAR) in patients with favorable aortic anatomy.6,10,11 However, TEVAR is associated with several complications. The selection of correct landing zones and endograft sizing remains a challenge in patients with BTAI. 12 Underestimation of the aortic diameter and sharper aortic arch angles are known risk factors for poor endograft apposition to the aortic arch, leading to long-term device-related complications.12,13 To overcome these limitations, conformable endografts are often used for TEVAR, even in patients with BTAI. A study by Farber et al 14 reported the long-term results of a Gore TAG-conformable (cTAG) endoprosthesis used in traumatic aortic transections. Freedom from all-cause mortality was 95 and 89% at 1 month and 5 years postprocedure, respectively. However, there is a lack of studies comparing the long-term outcome of the Gore cTAG endoprosthesis with other devices after TEVAR in patients with BTAI. This study aimed to analyze the outcomes of these patients with respect to anatomical and technical factors. Patients with BTAI who received the Gore cTAG endoprosthesis were compared with those who received nonconformable endografts.
Materials and Methods
Data Collection
In this retrospective study, all patients with BTAI were treated with TEVAR between June 2001 and May 2019 at a tertiary vascular center (Department of Vascular Surgery, University Hospital Regensburg, Regensburg, Germany). Data were obtained from a prospectively compiled database. The data were evaluated by one of the 2 principal investigators (LK and TB) and reviewed by the second principal investigator (TB). Patient consent was obtained upon hospital admission. The study was approved by the local ethics committee prior to data collection in accordance with the Declaration of Helsinki (approval number: 14-17-101).
Study Criteria
The indication for TEVAR was BTAI in all patients. BTAI was diagnosed using computed tomography angiography (CTA) and classified according to the Society of Vascular Surgery (SVS) blunt aortic injury grading system. 6 Based on the CTA findings, each case was discussed by an interdisciplinary trauma board (radiologists, trauma surgeons, vascular surgeons, and anesthesiologists), considering the competing priorities of other injuries. TEVAR was generally performed within 24 hours for BTAI grade II to IV injuries. Endografts were selected to achieve 10% to 20% oversizing relative to the native aorta. The proximal landing zone was in the aortic arch or just below and was classified according to the Ishimaru zones of the aorta. 15 Preoperative absolute contraindications to left subclavian artery (LSA) coverage without revascularization were identified: the presence of the posterior inferior cerebellar artery; a previous left internal mammary coronary artery bypass; an occluded, absent, atretic right vertebral artery; or left arm hemodialysis access. For comparative analysis, patients were classified into 2 groups: those with conformable endografts (all patients received Gore cTAG) (group 1) and those with nonconformable endografts (group 2).
Procedural Details
All procedures were performed under general anesthesia. Vascular access was achieved through surgical dissection of the common femoral artery or an ultrasound-guided percutaneous approach using 2 ProGlide devices (Abbott Vascular). After securing access using an 8-French sheath, heparinization was initiated. A floppy guidewire and marker flush catheter were used for the initial angiography of the aortic arch. After exchanging for a stiff wire (Lunderquist, Cook Medical), the endograft was delivered. An additional angiogram was obtained via a buddy catheter through the ipsilateral groin or contralateral femoral access with the device in place to ensure appropriate positioning. The LSA was covered to obtain a proximal landing zone or to achieve better apposition with the lesser curvature of the aortic arch. According to the guidelines 11 carotid-subclavian bypass was the first-line method for revascularization, in hemodynamic unstable patients chimney grafts were used. Requirements for the chimney grafts were a take-off angle of the left subclavian artery from the aortic arch of <75° and a distance between the left vertebral artery and the left subclavian artery orifice of > 40mm. Postdeployment balloon angioplasty was performed selectively when incomplete apposition of the graft at the proximal landing zone was observed. After device deployment, complete angiography was performed to exclude technical failures. Hemostasis was achieved using ProGlide devices or vascular sutures at the puncture site. The choice of the type of endograft depended on the surgeon’s preference. The first Gore cTAG was used in September 2004. Postoperatively, all patients received antiplatelet therapy and were monitored in the intensive care unit. All patients underwent a follow-up CTA before discharge. Follow-up visits included clinical examinations and CTA 6 months postoperatively and annually thereafter.
Computer Tomography Analysis and Measurements
All studies were segmented and measured using the center lumen line in iNtuition software (TeraRecon, ver.4.4.14.254). The aortic arch angle and index were determined as described by Wojciechowski et al. 16 Aortic arch branching patterns have been reported by naming the vessels in order from the most proximal. The aortic arch type was determined according to the method described by both Madhwal et al 17 and Casserly et al 18 due to the reported significant differences. 19 Diameter and length were measured using the center-lumen line. Aortic injuries were categorized according to the classification proposed by Azizzadeh et al 20 and included in the SVS Guidelines. 6 Data on complications such as endoleaks, bird beak sign (BBS), intramural thrombus, and stent graft collapse rates were obtained using the first postprocedure and last available CTA. The BBS was defined as poor apposition of the stent to the inner curvature of the aortic arch. If present, further measurements were performed as described by Ueda et al. 21
Study Endpoints
The primary endpoint was aortic-related reintervention-free survival. This was defined as the number of days from the index procedure to the first aortic-related revision procedure or death. The secondary endpoints were 30-day mortality, in-hospital mortality, postoperative complications, and radiological findings. We defined the 30-day mortality as death within 30 days of surgery. In-hospital mortality was defined as death during the hospital stay. The postoperative complications included bleeding or hematoma related to the index procedure, perioperative stroke, peripheral ischemia, and LSA coverage-related ischemia. In addition, endoleaks, intramural thrombus, endograft collapse, and BBSs were noted. The last available status data were used for patients who were unavailable during the follow-up period. The follow-up index was calculated for each patient to increase the validity of the study. 22
Statistical Analysis
Statistical analyses were performed using TIBCO Software Inc. (2017) Statistica version 13b. All continuous variables are represented with the mean ± standard deviation or median (interquartile range). Categorical variables are presented as frequencies and percentages and were analyzed using the Chi-square test or Fisher’s exact test, where appropriate. The Kaplan-Meier life table method was used to assess reintervention-free survival. Comparisons were performed using the Mantel-Cox log-rank test. The subgroup analysis compared reintervention-free survival between patients with and without BBS after TEVAR. Statistical significance was set at p<.05.
Results
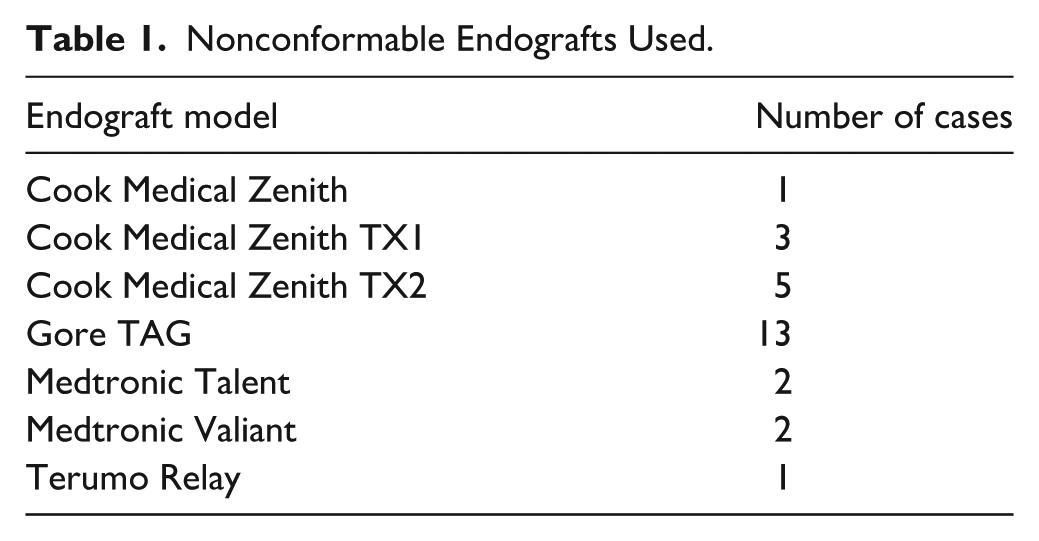
Between June 2001 and May 2019, 57 patients (50 men; mean age: 37.3 years) received 30 (52.6%) conformable (Gore cTAG) and 27 (47.4%) nonconformable endografts. The endografts used in group 2 are shown in Table 1.
Nonconformable Endografts Used.
Preoperative Risk Factors and Trauma Details
A BTAI was an indication in all cases. Grade I BTAI was present in 1 (1.8%) patient, grade II BTAI in 11 (19.3%) patients, grade III BTAI in 32 (56.1%) patients, and grade IV BTAI in 13 (22.8%) patients. Preoperative risk factors and trauma details were similarly distributed among groups, except for chronic kidney disease. A comparison of preoperative risk factors and trauma details is presented in Table 2.
Preoperative Risk Factors and Trauma Details.
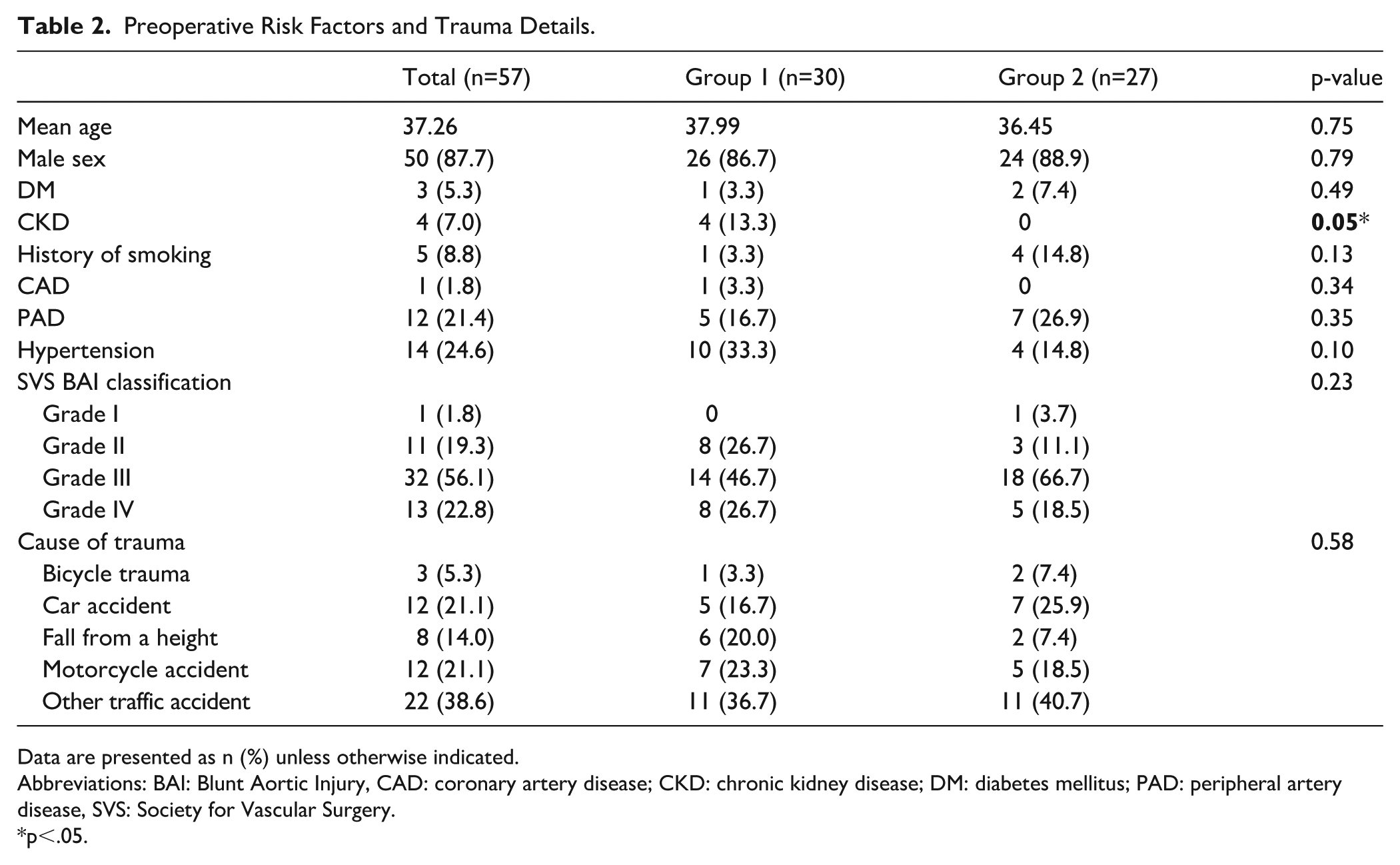
Data are presented as n (%) unless otherwise indicated.
Abbreviations: BAI: Blunt Aortic Injury, CAD: coronary artery disease; CKD: chronic kidney disease; DM: diabetes mellitus; PAD: peripheral artery disease, SVS: Society for Vascular Surgery.
p<.05.
Procedural Details
The complete LSA ostium coverage was performed in 35 (61.4%) patients, whereas LSA revascularization was performed in 13 (22.8%) patients. This approach was significantly more often in group 2 versus group 1—77.8% vs 46.7%. Although proximal landing zone locations were similar. Left subclavian artery revascularization was performed via carotid-subclavian bypass in 9 (15.8%) patients. In addition, 4 (7.0%) patients underwent the chimney procedure. No significant differences in procedural details were observed between the groups. Comparisons of the procedural details are shown in Table 3.
Procedural Details.
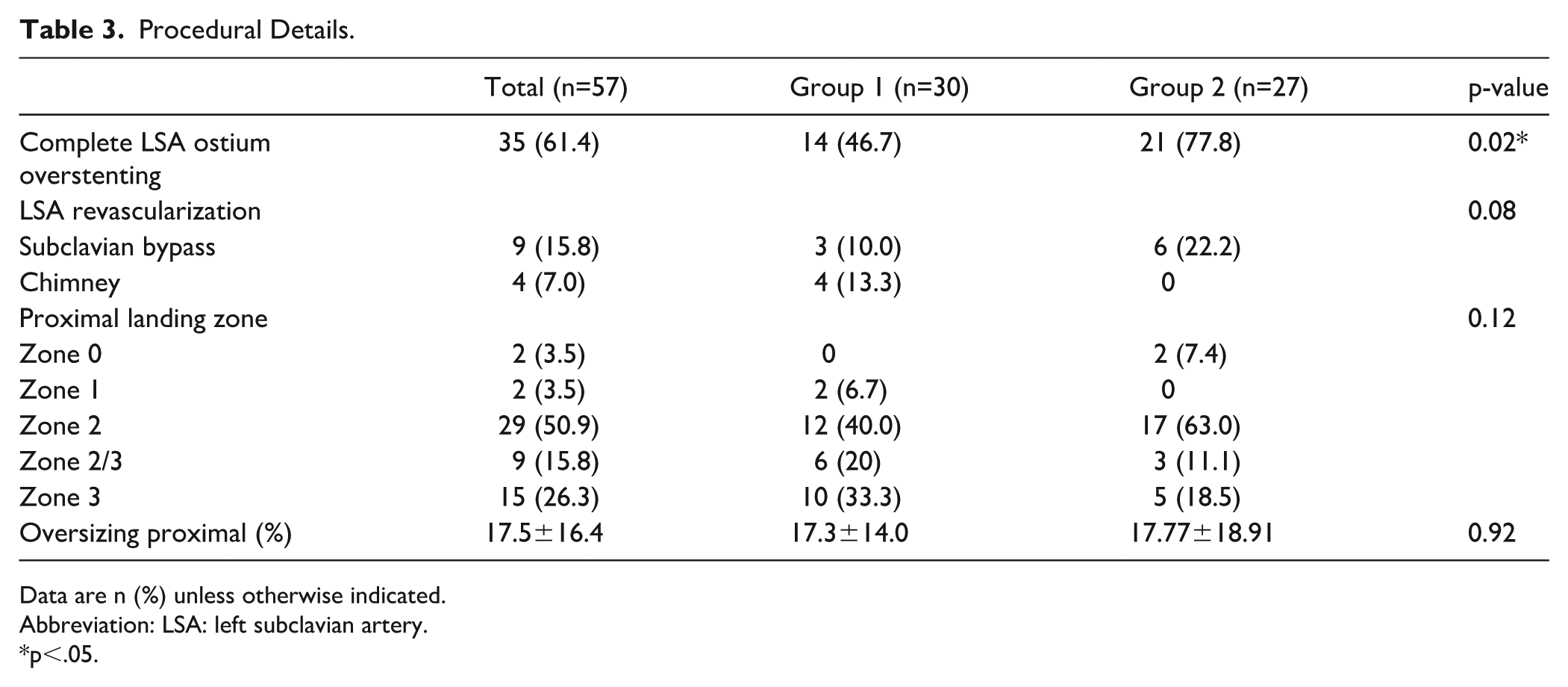
Data are n (%) unless otherwise indicated.
Abbreviation: LSA: left subclavian artery.
p<.05.
Anatomical Measurements
In both groups, the most common branching pattern was described as (from proximal to distal): brachiocephalic trunk, left common carotid artery (CCA), and left subclavian artery (LSA). There were no significant differences in anatomical measurements between the groups. The anatomical measurements are presented in Table 4.
Anatomical Measurements.
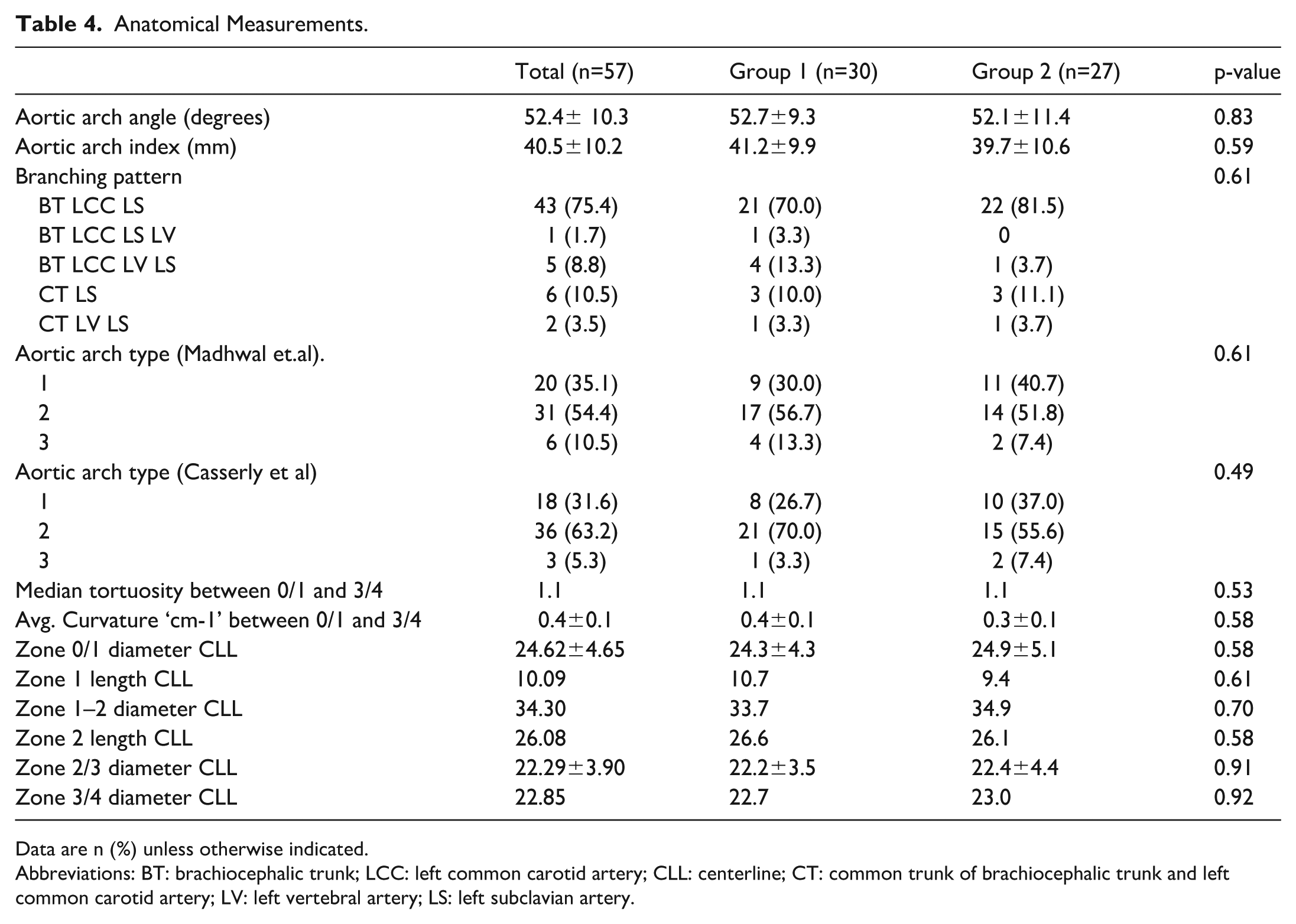
Data are n (%) unless otherwise indicated.
Abbreviations: BT: brachiocephalic trunk; LCC: left common carotid artery; CLL: centerline; CT: common trunk of brachiocephalic trunk and left common carotid artery; LV: left vertebral artery; LS: left subclavian artery.
30-day Results
The 30-day results are listed in Table 5. In the early period, 2 of 57 patients died, resulting in a 30-day mortality rate of 3.5%. The cause of death was unrelated to the aorta. Deaths were caused by multiorgan failure due to extensive injuries. The overall median hospital stay was 24 days, which was similar in both groups.
30-day Results.
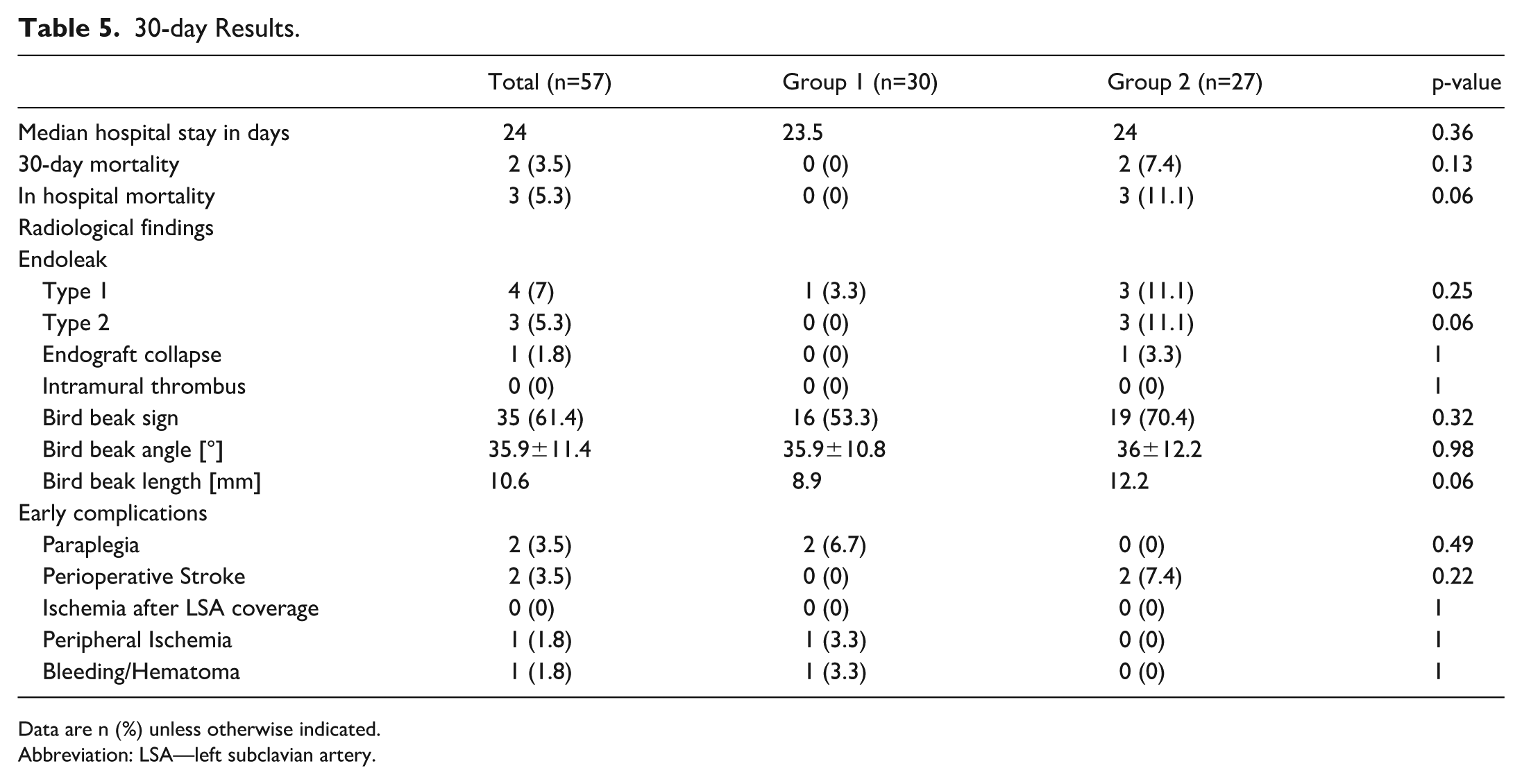
Data are n (%) unless otherwise indicated.
Abbreviation: LSA—left subclavian artery.
There were 3 type Ia endoleaks observed in patients who underwent prior nonconformable stent graft implantation. Two cases (#1 and #2) had proximal landing zone in zone 2 and distal in zone 4 and a standard branching pattern (brachiocephalic trunk, left common carotid and left subclavian artery), the BBSs have been observed. The case #1 had grade IV injury at the inner curvature, the aortic arch was type 1, with angle of 64.1 and arch index 49,8. The case #2 grade III injury at the descending aorta, the aortic arch was categorized as type 2 according to Madhwaland and type 3 according to Casserly, with angle of 41.7 and arch index of 36.2. The case #3 suffered from grade III injury. The patient had common ostium of bracho-cephalic trunk and left common carotid artery. The arch was type 2 with an angle of 73 degrees and arch index of 63.3. The proximal landing zone was in zone 3 and there was no BBS observed. These endoleaks have been successfully sealed with ballon angioplasty and Palmaz-stent implantation at the proximal landing zone. The patients with endoleaks type 2 did not require reintervention. A case of endograft collapse that required additional vascular procedures was also observed in group 2. One type Ib endoleak after conformable endograft implantation needed distal extension. There were no signs of an intramural thrombus on the first postoperative angiographic CT scan. The BBS was observed more often in group 2 (70.4% vs 53.3%; p=0.32). The angle and length measurements were similar between the 2 groups.
Early complications were rare and did not differ significantly between the groups. A paraplegia was observed in 2 patients (group 1), and was associated with concomitant injuries such as spinal epidural hematoma and thoracic vertebral fractures. In 2 patients with LSA, infarction of the posterior inferior cerebellar artery occurred (group 2). No ischemia was observed in the upper extremities. In group 1, 1 case of peripheral ischemia was caused by concomitant popliteal artery dissection. In addition, 1 patient who experienced traumatic retroperitoneal bleeding required packing.
Long-term Outcomes
The median follow-up time was 4.3 years. The 31 patients had a follow-up available for 4 years or more. During the follow-up period, 4 patients died, resulting in an overall death rate of 7%. The median (Q25-Q75) follow-up index was 0.71 (0.16–0.94). There were no statistical difference between group 1 and group 2 (p=0.25)—0.62 (0.16–0.90) and 0.83 (0.15–0.98) accordingly. The follow-up index values’ distribution is presented in Figure 1. The later mortality was caused by septic shock in the intensive care unit and massive hemorrhage from the esophagus at 43 and 257 days after the index procedure.
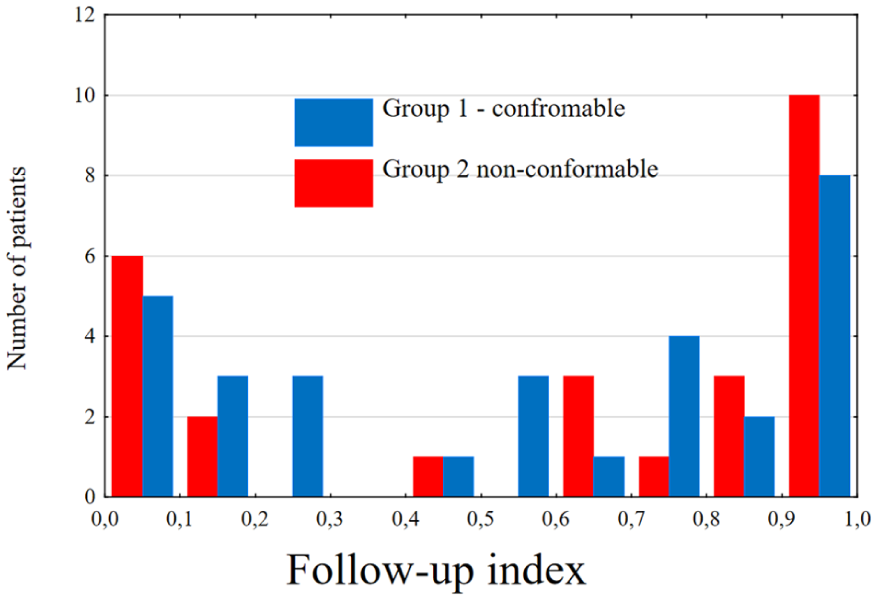
Follow-up index distribution in both groups.
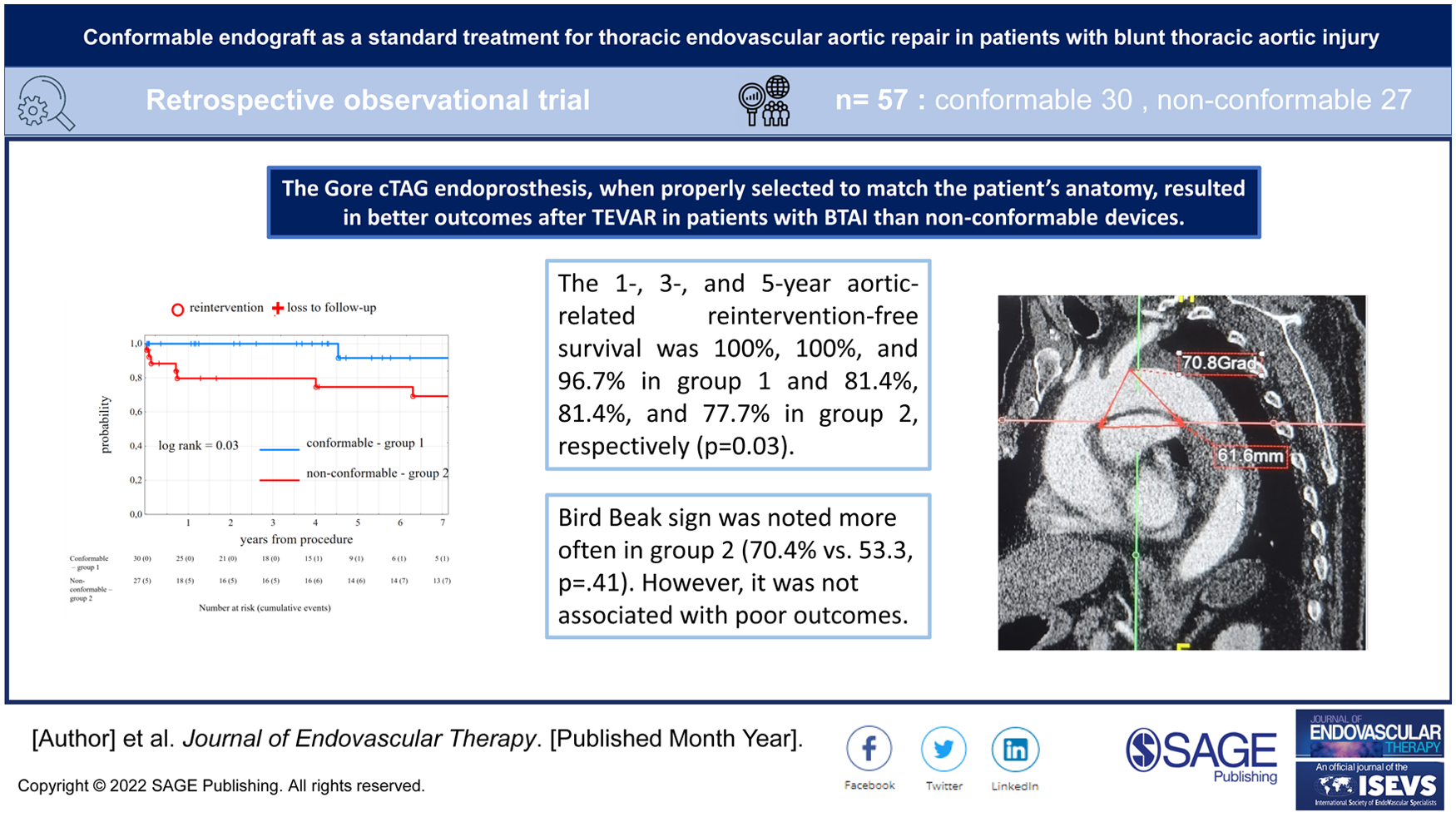
At 1, 3, and 5 years, the overall aortic reintervention-free survival rates were 91.2%, 91.2%, and 87.7%, respectively. The 1-, 3-, and 5-year aortic reintervention-free survival rates were 100%, 100%, and 96.7% in group 1 and 81.4%, 81.4%, and 77.7% in group 2, respectively (p=0.03; Figure 2).
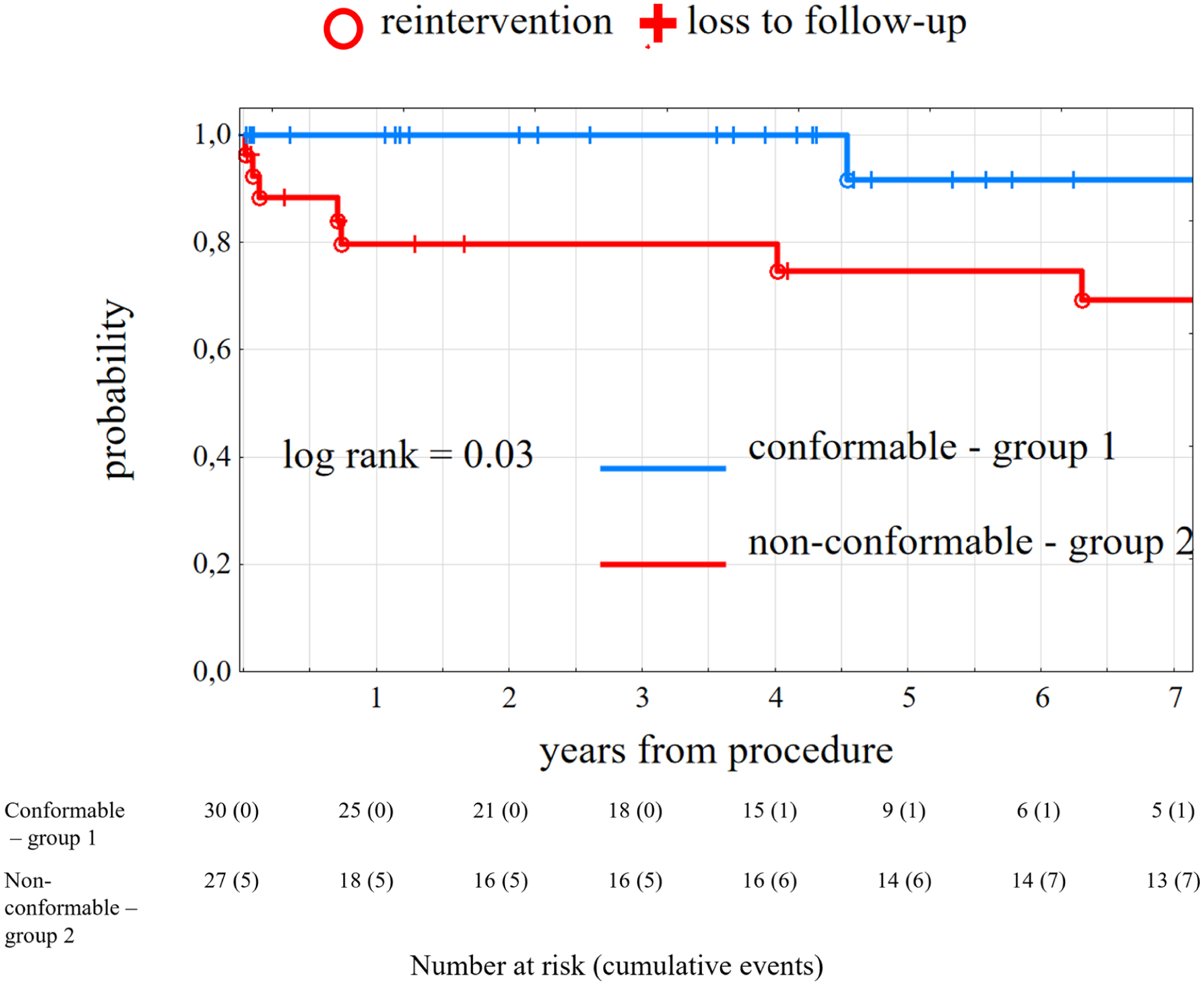
Aortic related reintervention free survival in both groups.
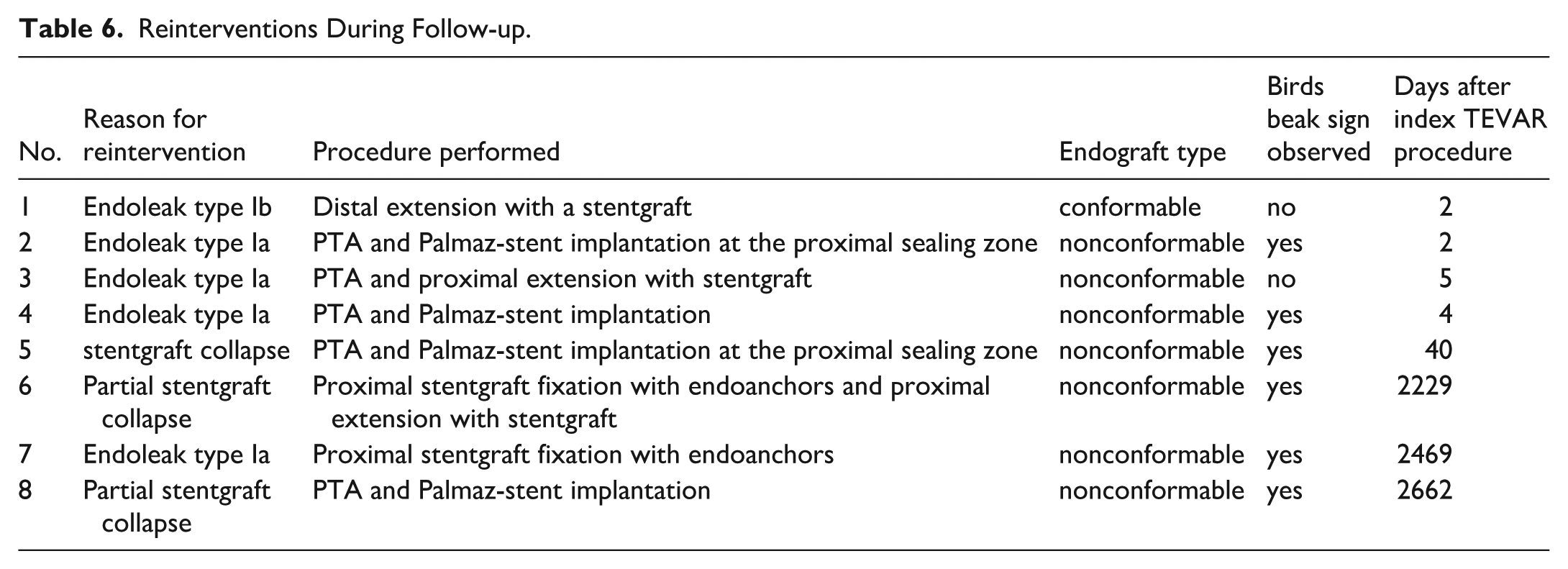
Reinterventions
During the follow-up there have been 8 aortic related reinterventions observed. The 1 happened in group 1 and 7 in group 2. The detailed description of each reintervention can be found in Table 6. The comparison of anatomical data between patients with and without reintervention showed no significant differences. However, there was a tendency for bigger aortic arch diameter in zones 1 and 2 and nonconformable endograft use with LS overstenting was more frequent in reintervention group. In addition, most of the patients who needed reintervention in follow-up (87.5%) had aortic arches type II according to Madhwal et al, compared with 49% of patients with uneventful follow-up. The parameters are summarized in Table 7.
Reinterventions During Follow-up.
Reintervention Subgroups Comparison.
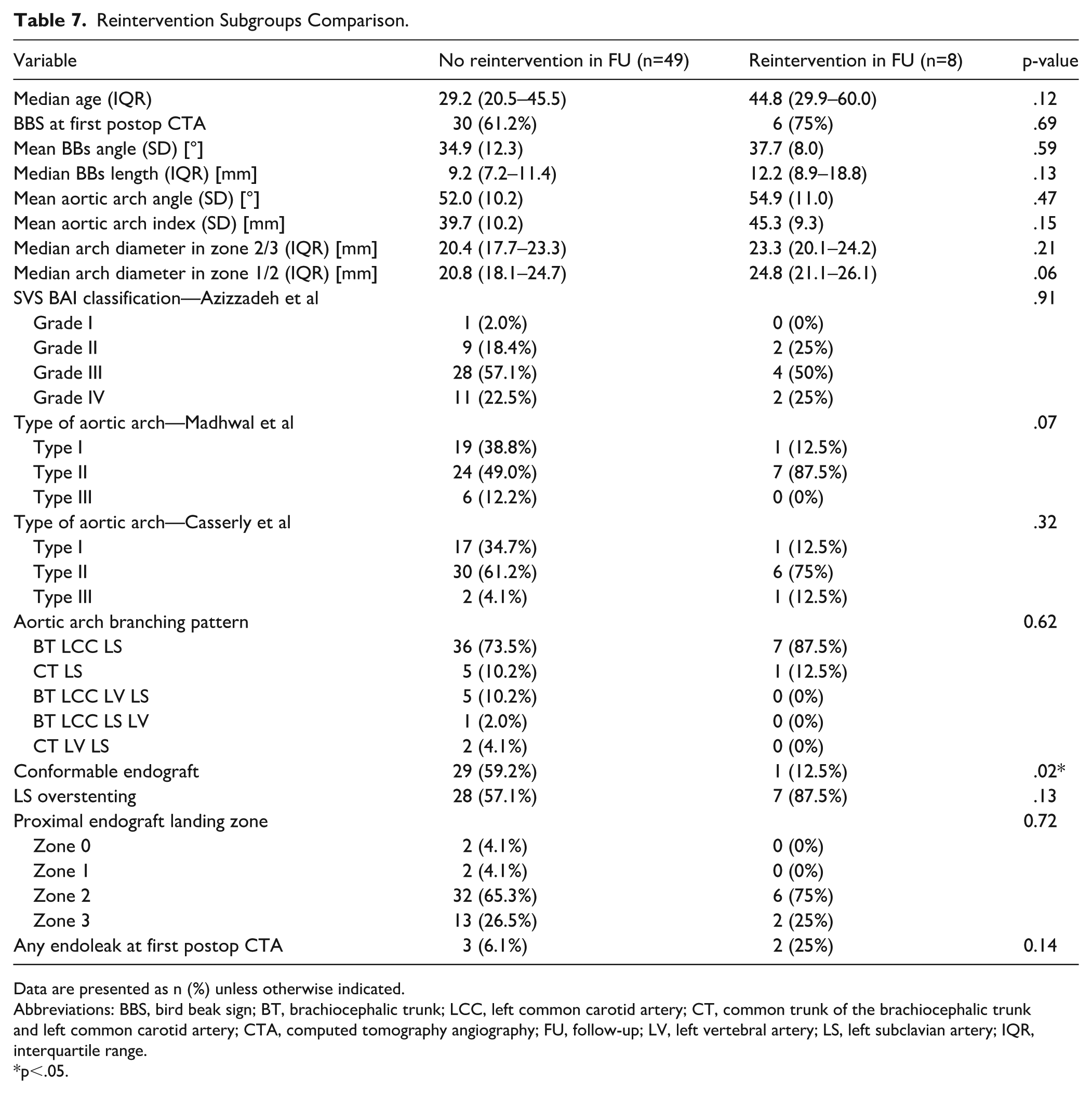
Data are presented as n (%) unless otherwise indicated.
Abbreviations: BBS, bird beak sign; BT, brachiocephalic trunk; LCC, left common carotid artery; CT, common trunk of the brachiocephalic trunk and left common carotid artery; CTA, computed tomography angiography; FU, follow-up; LV, left vertebral artery; LS, left subclavian artery; IQR, interquartile range.
p<.05.
Bird Beak Sign Analysis
Subgroup analyses based on BBS scores are presented in Table 8. The BBS was detected in 35 patients. The aortic arch index was significantly larger when BBS was not observed. In addition, there was a tendency for larger aortic arch diameter in that group.
Bird Beak Sign Analysis.
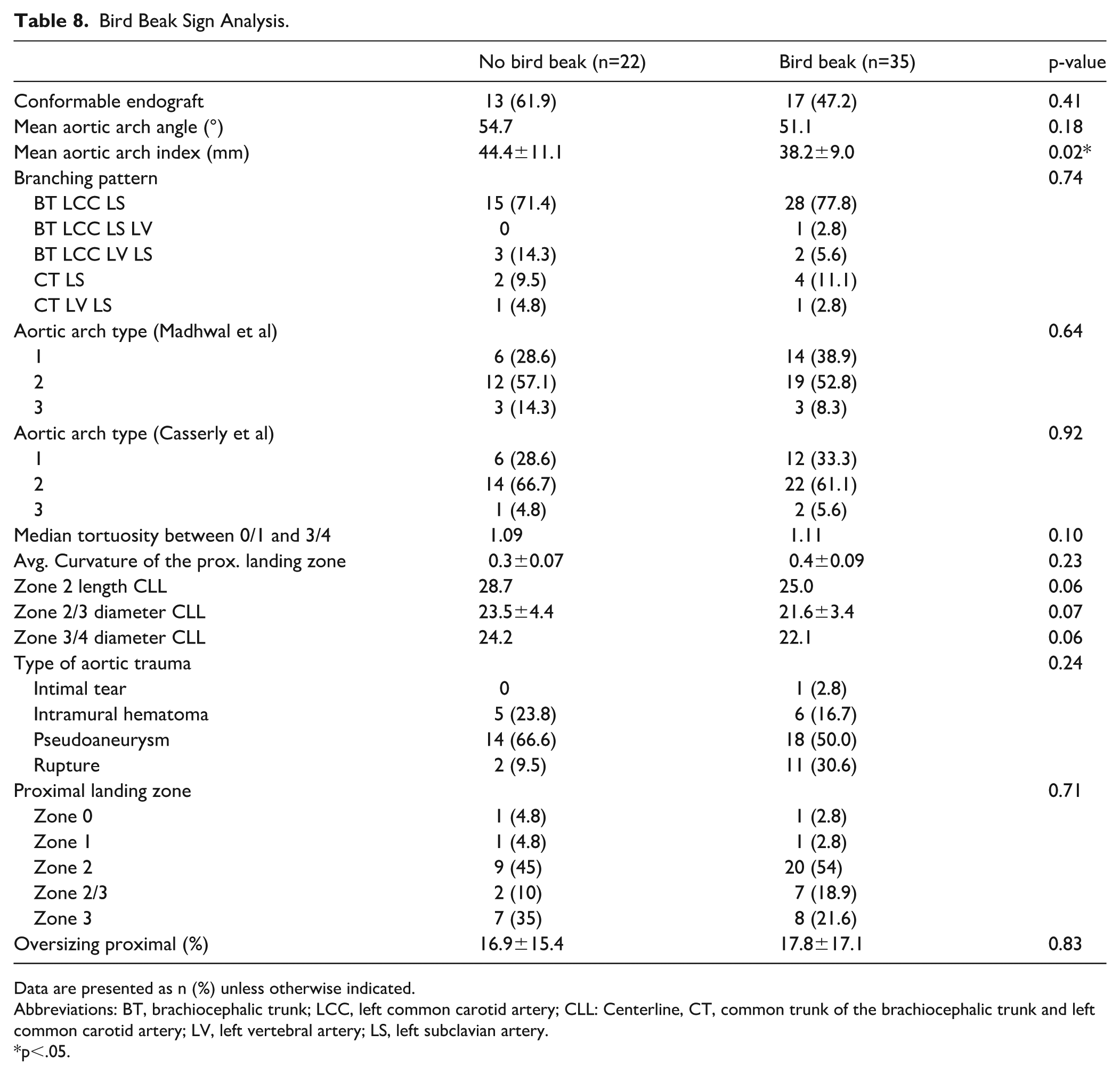
Data are presented as n (%) unless otherwise indicated.
Abbreviations: BT, brachiocephalic trunk; LCC, left common carotid artery; CLL: Centerline, CT, common trunk of the brachiocephalic trunk and left common carotid artery; LV, left vertebral artery; LS, left subclavian artery.
p<.05.
Discussion
Current guidelines from both the European Society for Vascular Surgery10,11 and SVS 6 recommend TEVAR over open repair for the treatment of BTAI. Unfortunately, late complications and the need for reintervention remain concerns with TEVAR. This issue is particularly relevant in the context of BTAI because patients tend to be younger than the general aortic population and have a potentially long life expectancy. A recent systematic review analyzed the outcomes of 991 patients who underwent TEVAR after BTAI. The mean age was 34.5±16.5 years (range 16–89 years). Mortality and reintervention rates were low at 2.0 and 1.0%, respectively. However, the follow-up period was relatively short (29 months). 23 Our study had a mean follow-up of 52 months, which provides more data regarding long term results. Nevertheless, most of the reinterventions in our study happened during first 40 postoperative days (Table 6). The higher reported rate of early reinterventions might be associated with the available devices and unfavorable patients’ anatomy—not analyzed in the systematic review. 23 Another study revealed that patients with BTAI had more severe pulsatile deformation of the aorta and stress on the endograft during the cardiac cycle than those treated with TEVAR for aortic aneurysms or dissections. 24 Thus, after endovascular treatment with BTAI, patients developed a more demanding anatomy for the implanted endograft. 25 In such a hostile environment, the device is prone to failure over its lifetime, which is particularly long for a young cohort of patients with BTAI. Therefore, the lifelong follow-up of patients undergoing TEVAR after BTAI is essential. Most of the early reinterventions in our study group were required due to endoleak type IA. They were successfully sealed with Palmaz stent implantation and balloon angioplasty. There might be a group of patients, with particularly demanding aortic arch anatomy, which may benefit from such maneuver at the index procedure to prevent the proximal endoleak.
The anatomy of the aortic arch differs between older and younger patients. Redheuil et al 26 reported that the length and diameter of the aortic arch increased significantly with age. The length of the ascending aorta increases the most with age, leading to aortic dilation and a decreased aortic curvature. In our study, we primarily treated young patients with types II and III aortic arches. A sharper aortic arch angle is a risk factor of poor endograft apposition to the aortic arch wall, leading to long-term device-related complications.12,13 To overcome this problem, manufacturers offer new types and generations of devices for BTAI.24,27 Farber et al 14 provided insights into the performance of a conformable endograft, the Gore cTAG. No aortic ruptures, wireframe fractures, erosions, lumen obstructions, device compressions, or thrombus-related events were reported during 5 years of follow-up in the 50 patients. Our analysis revealed good results with TEVAR for BTAI using a conformable endograft. Aortic-related reintervention-free survival was significantly better, with 96.7% in group 1 and 77.7% in group 2. We believe that the unique endograft design of the Gore cTAG conforms to a wide range of anatomical variables for the treatment of BTAI. With better graft apposition, phenomena such as BBS, which can lead to subsequent endograft collapse, may be minimized.
The data on BBS scores after endovascular treatment with BTAI is controversial. Although conformable stent-grafts theoretically provide better apposition to the aortic wall, the present study found no statistical difference in BBS between conformable and nonconformable endografts. However, there is a trend toward longer (12.2 vs 8.9 mm) and more frequently observed (70% vs 53%) BBS in the nonconformable group. This could be explained by the fact that the cTAG can also be angled, resulting in better apposition to the inner curvature of the aortic arch. A recent systematic review examined the incidence of this phenomenon after endovascular repair of BTAI. 28 Eleven studies involving 408 patients were analyzed, with a mean follow-up of 8.2 years. The late reintervention rate was 2.1%, with 0.1% occurring after 5 years. BBS was identified in 38.7%.
Another study by García Reyes et al 12 analyzed the long-term outcomes of TEVAR, focusing on BBS and oversizing in 34 patients with BTAI. BBS was seen in 65%. In this study, noncompliant endografts were used to treat BTAI in 31 patients. We observed BBS in 60% of our patients, also in group 1. However, as in the abovementioned study, the reintervention rate did not increase in this group of patients. A meta-analysis by Marocco-Trischitta et al 13 investigated the incidence of BBS after TEVAR. Indications for treatment were trauma, aneurysm, or dissection. In contrast to our results, complications occurred in 14.7% (95% CI: 7.4%–27.3%) of patients with BBS and in 6.3% (95% CI: 2.5%–15.4%) of patients without BBS within a range of 0 to 72 months. Moreover there was significantly more LSA overstenting in group 2, which may contribute to more risk for graft related complications, if BBS occurs. The risk difference between patients with and without BBS was 8.2% (95% CI: 0.3%–16.1%, p=0.042; I2=69.0%). 13 The authors concluded that BBS appears to be associated with a high risk of type Ia endoleaks and endograft migration, warranting specific and long-term surveillance. 13 We believe, that this phenomenon is partly due to the anatomy of the aortic arch in the treated BTAI patients. Another reason could be the reduced radial force of the cTAG stent graft, which is low compared with other stent grafts. However, there is a trend toward a shorter bird beak in group 1 compared with group 2 and a lower reintervention rate was seen in group 1, although anatomical measurements showed no differences in arch morphology between the groups.
Limitations
The main limitations of this study were relatively small number of patients and its retrospective nature. The rarity of this pathology makes it impossible to perform a prospective study with a larger cohort at a single center. Secondly, there was a reduced completeness of follow-up. Owing to the long time elapsed since the index procedure, patients often changed their contact information, which prevented researchers from collecting up-to-date follow-up information. In addition, various nonconformable devices were used for BTAI treatment, with 1 specific type accounting for nearly half of all cases (Table 1).
Conclusions
The selection of the optimal device for urgent BTAI treatment is critical for long-term therapeutic success. This study in a small cohort of patients demonstrated that the Gore cTAG endoprosthesis, when properly selected to match the patient’s anatomy, appears to result in better outcomes after TEVAR in patients with BTAI than nonconformable devices. In addition, the BBS was common in both groups of patients and was not associated with a worse outcome. Further studies are needed to confirm these findings.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
