Abstract
Background:
Paclitaxel-based devices improve femoropopliteal outcomes; however, the optimal strategy for de novo lesions by lesion complexity remains uncertain. This study compared long-term outcomes of full-coverage drug-eluting stent (DES) implantation and drug-coated balloon (DCB) angioplasty stratified by the Trans-Atlantic Inter-Society Consensus II (TASC II) classification.
Methods:
This retrospective, single-center study included 419 patients (507 limbs) with de novo femoropopliteal lesions treated between 2019 and 2021. Limbs were stratified into TASC II A/B (186 limbs) and C/D (321 limbs) groups and treated with DES (107 limbs) or DCB (400 limbs). The primary endpoint was 3-year primary patency, defined as freedom from target lesion revascularization (TLR) or duplex ultrasound-detected restenosis (peak systolic velocity ratio ≥2.5). Secondary endpoints included 3-year freedom from TLR and amputation-free survival (AFS).
Results:
In TASC II A/B lesions, 3-year primary patency was comparable between DES and DCB (80.0% vs 76.9%, p=0.432), as were freedom from TLR (84.2% vs. 88.0%, p=0.959) and AFS (74.3% vs 78.3%, p=0.834). In TASC II C/D lesions, DES achieved higher 3-year primary patency (85.1% vs 50.3%, p<0.001) and freedom from TLR (90.2% vs 66.7%, p=0.008) than DCB, while AFS did not differ significantly (90.2% vs 66.7%, p=0.074). Within the DCB cohort, 3-year primary patency was lower for C/D than A/B lesions (50.3% vs 76.9%, p<0.001), whereas DES outcomes were consistent across strata (85.1% vs 80.0%, p=0.828). The AFS was comparable between the 2 strategies across both TASC II strata. Regarding restenosis patterns, 3-year reocclusion rates were similar in A/B (DES 20% vs DCB 15%, p=0.544), whereas they were higher with DES in C/D (60% vs 29%, p=0.044). On multivariable analysis, below-the-knee runoff ≤1 (hazard ratio [HR]=1.96) and TASC II D classification (HR=2.57) were independent predictors of restenosis in DCB-treated C/D lesions, whereas ischemic wounds (HR=6.61) and extensive eccentric calcification (HR=14.3) predicted restenosis in DES-treated A/B lesions.
Conclusion:
Full-coverage DCB and DES yielded favorable outcomes in TASC II A/B lesions, supporting a stentless strategy in selected cases. For TASC II C/D lesions, DES provided superior long-term patency and lower reintervention rates, highlighting the importance of lesion complexity-based device selection.
Clinical Impact
This study demonstrates that lesion complexity, as defined by the TASC II classification, is a decisive factor in device selection for femoropopliteal interventions. In de novo TASC II A/B lesions, full-coverage drug-coated balloon angioplasty and drug-eluting stent implantation achieved comparable 3-year outcomes, supporting a stentless approach in suitable cases. In contrast, the drug-eluting stent strategy provided superior long-term patency in complex TASC II C/D lesions, emphasizing the value of full-lesion coverage and optimized lumen expansion. These findings offer lesion-specific guidance that may refine treatment algorithms, improve durability of revascularization, and support individualized therapy in peripheral artery disease.
This is a visual representation of the abstract.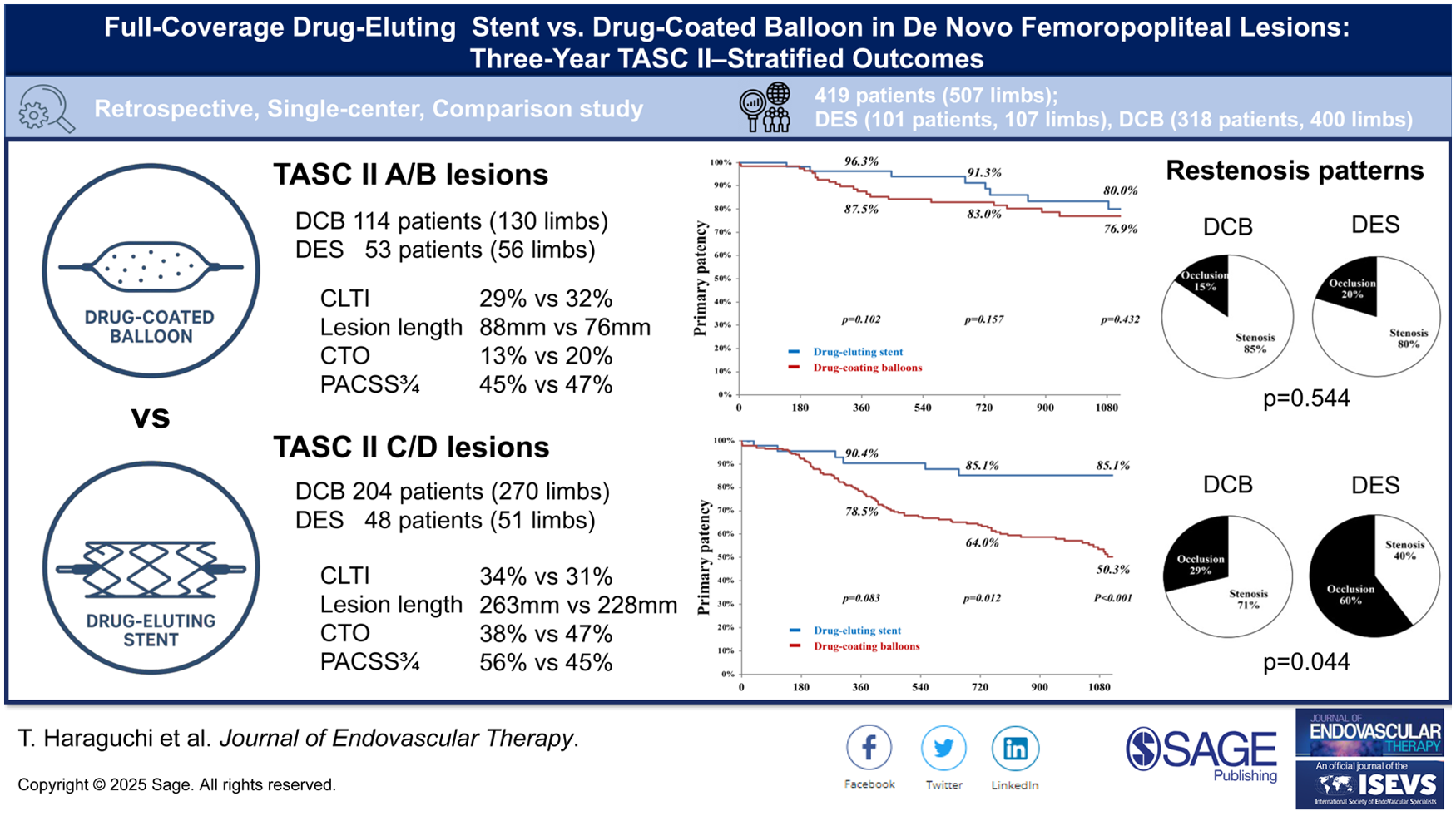
Introduction
The Trans-Atlantic Inter-Society Consensus (TASC II) classification is commonly employed to inform clinical decision-making regarding the choice of principal invasive treatment modalities and device selection. 1 Paclitaxel-based devices have demonstrated superior long-term outcomes compared with conventional non-drug interventions, and current guidelines support their use with a Class IIa recommendation. 2 For patients with TASC II A or B femoropopliteal lesions, drug-coated balloon (DCB) angioplasty is recommended following optimal plain balloon angioplasty, without routine stent implantation. Conversely, selective implantation of drug-eluting stents (DESs) is advised when balloon angioplasty yields suboptimal angiographic results in the femoropopliteal segment. This paradigm has led to a growing preference for stentless treatment strategies; however, studies in which DCB angioplasty alone was predominantly used have reported higher restenosis rates in complex lesions associated with multiple risk factors for recurrence. 3 Consequently, combined approaches incorporating adjunctive stent implantation have been more frequently adopted for complex or long lesions. 4
Among DES platforms, the Eluvia stent (Boston Scientific, Marlborough, Massachusetts) has demonstrated favorable long-term outcomes, with clinical results reported to surpass those of bare-nitinol and drug-coated stents.5 –7 Spot stenting has been identified as a predictor of restenosis, supporting the concept of full-lesion coverage.8,9 Moreover, larger post-procedural lumen area is associated with improved long-term patency in DES treatment, 10 and trials employing standardized vessel preparation have reported lower restenosis rates with DES compared with DCB. 11
However, previous comparative studies between DES and DCB have been limited by heterogeneous lesion complexity, variable procedural techniques, and inclusion of previously treated lesions, which are known predictors of restenosis.3,8,12 To date, no study has systematically applied standardized vessel preparation and compared the long-term outcomes of full-coverage DES vs DCB exclusively in de novo femoropopliteal lesions stratified by TASC II classification. This study aimed to evaluate and compare the 3-year clinical outcomes of full-coverage DES and DCB in patients with de novo femoropopliteal lesions, stratified by lesion complexity according to the TASC II classification.
Materials and Methods
Study Population
This retrospective, observational, single-center study was conducted at our institution from January 2019 to December 2021. A total of 563 patients (745 limbs) with chronic lower extremity arterial disease (LEAD) and de novo lesions treated with DES or DCB were included in the analysis. Patients were excluded for prior endovascular intervention; lesions confined to the isolated popliteal artery or involving the popliteal P2-P3 segment; combination therapy (DES and DCB within the same treatment course or concomitant use of DES/DCB with other devices); and bailout stenting for flow-limiting dissection after DCB angioplasty. Combination therapy (97 patients, 157 limbs), isolated or P2-P3 popliteal lesions (41 patients, 73 limbs), and bailout stenting (6 patients, 8 limbs) were excluded.
Lesion complexity was classified according to the TASC II criteria, and patients were stratified into 2 groups: TASC II A/B and TASC II C/D. The study protocol was reviewed and approved by the institutional ethics committee of our institution and was conducted in accordance with the principles of the Declaration of Helsinki. Given the retrospective design, the requirement for written informed consent was waived.
Endovascular Procedures and Medical Therapy
Patients presenting with symptomatic femoropopliteal lesions with ≥70% diameter stenosis and classified as Rutherford category 2 to 6 were considered eligible for endovascular intervention. Vascular access was obtained via the common femoral artery on the ipsilateral or contralateral side of the target lesion using a 6-Fr sheath. An initial bolus of 5000 IU of unfractionated heparin was administered, with additional doses titrated to maintain an activated clotting time of ≥250 seconds. Lesion crossing was performed using a 0.014 or 0.018 inch guidewire. In chronic total occlusions (CTOs), fluoroscopy-guided crossing was attempted first; if unsuccessful, intraplaque wiring was performed under intravascular ultrasound (IVUS) guidance. The IVUS-guided CTO crossing workflow comprised (i) delineation of intraplaque entry point and planned re-entry site; (ii) real-time IVUS-guided guidewire manipulation to maintain an intraplaque route and avoid subintimal or extraluminal tracking, with selective guidewire escalation and microcatheter maneuvers as necessary; (iii) mapping of hard plaque, including dense fibrous and calcified components, to inform guidewire escalation and microcatheter support; and (iv) confirmation of sustained intraplaque passage before lesion preparation and definitive therapy (DES or DCB). Favoring intraplaque crossing is associated with superior primary patency compared with subintimal strategies and with fewer procedure-related complications and less aneurysmal degeneration after DES implantation.8,13,14
In IVUS-guided cases, reference diameters and minimum lumen area (MLA) were measured, and the external elastic membrane diameter of the reference vessel was used to inform balloon and stent sizing; in cases without IVUS guidance, vessel sizing and device selection were determined angiographically using quantitative vascular analysis. Balloon type and size were selected according to lesion morphology and anatomical location on angiography or IVUS. When successful vessel preparation was achieved, DCB angioplasty was generally performed; when preparation was unsuccessful, a DES-first strategy was implanted. For soft plaque lesions at high risk of complications, particularly distal embolization, where DCB therapy was considered unsafe or unlikely to succeed, a DES-first strategy was adopted. With DES diameters limited to 6 and 7 mm, lesions with small reference vessels in which DES implantation would clearly result in oversizing were instead treated with DCB. Accordingly, device selection reflected lesion morphology and reference vessel diameter. During pre-dilation and post-dilation, if procedure-related pain suggested over-dilation, balloon upsizing or additional pressure escalation was deliberately avoided to mitigate rupture risk.
The DCB angioplasty was performed with complete lesion coverage using the operator’s choice of device (low-dose DCBs: Lutonix [Becton Dickinson, Franklin Lakes, New Jersey] and Ranger [Boston Scientific, Marlborough, Massachusetts]; high-dose DCB: IN.PACT Admiral [Medtronic, Santa Clara, California]). When residual stenosis >50% was observed after DCB angioplasty, adjunctive post-dilatation with an appropriately sized plain balloon was performed unless bailout stenting was deemed necessary. For stent-based therapy, a fluoropolymer-based DES (Eluvia; Boston Scientific) was used; polymer-free drug-coated stents (Zilver PTX; Cook Medical, Bloomington, Indiana) were not employed. The DES implantation was followed by post-dilatation using a balloon of equal diameter to ensure full stent expansion. Atherectomy devices were not used during the study period due to restrictions imposed by domestic reimbursement policies. Hemostasis was achieved via manual compression or vascular closure devices.
Post-procedurally, dual antiplatelet therapy (DAPT) with aspirin 100 mg and clopidogrel 75 mg once daily was recommended for 1 month in the DCB group and 3 months in the DES group, in accordance with the product labeling. In cases judged by the operator to be at high risk of thrombotic occlusion, the DAPT duration was individualized and could be extended up to 3 months (DCB) and 6 months (DES). Thereafter, antiplatelet therapy was de-escalated to single antiplatelet therapy. When oral anticoagulation was indicated, both groups received an antiplatelet agent for 1 month, after which aspirin monotherapy was continued concomitantly with anticoagulation. Lipid-lowering therapy targeted a low-density lipoprotein cholesterol level of <55 mg/dL and a ≥50% reduction from baseline. 2 If these targets were not met on admission laboratory testing, high-intensity statin therapy with atorvastatin or rosuvastatin (up to 40 mg/d and 20 mg/d, respectively) was initiated or uptitrated. When further statin uptitration was not feasible or adverse effects occurred, ezetimibe 10 mg once daily was prescribed.
Study Endpoints and Definitions
The primary endpoint was 3-year primary patency, assessed using Kaplan-Meier estimates and defined as the absence of clinically-driven target lesion revascularization (TLR) or binary restenosis. Binary restenosis was defined as a peak systolic velocity ratio ≥2.5 on duplex ultrasound evaluation. The TLR was defined as a reintervention for ≥75% diameter stenosis on angiography within 5 mm proximal or distal to the target lesion, in the presence of recurrent clinical symptoms following the index procedure.
Secondary endpoints included 3-year freedom from TLR rate, amputation-free survival (AFS), defined as freedom from target limb major amputation and all-cause mortality, and restenosis patterns.
Patients were followed at 1, 6, and 12 months post-procedure and every 6 months thereafter, with additional visits prompted by any clinical deterioration. The follow-up protocol included measurement of the ankle-brachial index and lower-limb duplex ultrasonography at each visit. When ultrasonographic findings were inconclusive, computed tomography angiography, digital subtraction angiography, or both were performed to adjudicate endpoint status.
The LEAD severity was classified using the Rutherford scale, distinguishing moderate-to-severe intermittent claudication (classes 2-3) from chronic limb-threatening ischemia (CLTI), with or without tissue loss (classes 4-6). 15 Lesion complexity was graded according to the TASC II classification. 1 Calcification severity was assessed using the Peripheral Artery Calcification Scoring System (PACSS), 16 and dissection severity was evaluated using the National Heart, Lung, and Blood Institute classification system. 17 Successful vessel preparation was defined as <50% diameter stenosis and dissection without flow-limiting dissection. 18
Statistical Analysis
Continuous variables are presented as means±standard deviations and were compared using either the unpaired t-test or the Mann-Whitney U test, as appropriate. Categorical variables are expressed as counts (percentages) and were analyzed using the chi-squared test.
The cumulative incidence of study endpoints at 3 years (up to 1120 days) was estimated using the Kaplan-Meier method, and differences between groups were assessed using the log-rank test. Statistical significance was defined as a 2-sided p<0.05. Independent predictors of primary patency were identified using Cox proportional hazards regression analysis. Variables with a p<0.10 in univariable analysis were included in the multivariable model.
All statistical analyses were performed using SPSS Statistics, version 30.0 (SPSS Inc, Chicago, Illinois).
Results
Baseline Characteristics
A total of 419 patients (507 limbs) were included; 101 patients (107 limbs) received full-coverage DES and 318 patients (400 limbs) underwent DCB. Baseline characteristics by treatment strategy are summarized in Tables 1 and 2. In TASC II A/B (186 limbs), most baseline demographics were comparable between groups. Chronic kidney disease was more frequent in the DES group (81% vs 60%, p=0.008), whereas the proportion on hemodialysis was similar (21% vs 22%, p=0.842). Distal reference diameter and lesion length were not different (5.3±0.9 vs 5.1±0.9 mm, p=0.192; 7.76±45 vs 88±55 mm, p=0.157). The mean total DES length was 99±45 mm, and the mean DCB length was 102±52 mm. Vessel preparation was frequent in both (89% DES vs 96% DCB, p=0.073). In TASC II C/D (321 limbs), groups were generally similar; however, DCB cases more often involved the popliteal segment (58% vs 25%, p<0.001), had longer lesions (263±66 vs 228±54 mm, p<0.001), and had smaller distal reference diameters (4.3±1.1 vs 5.0±1.0 mm, p<0.001). The mean total DES length was 225.1±51.8 mm, while the mean DCB length was 266±70 mm. Vessel preparation remained common (94% DES vs 99% DCB, p=0.014). Post-dilatation was performed in all DES cases and in a minority of DCB cases (100% vs 19%, p<0.001).
Baseline Patient and Lesion Characteristics.
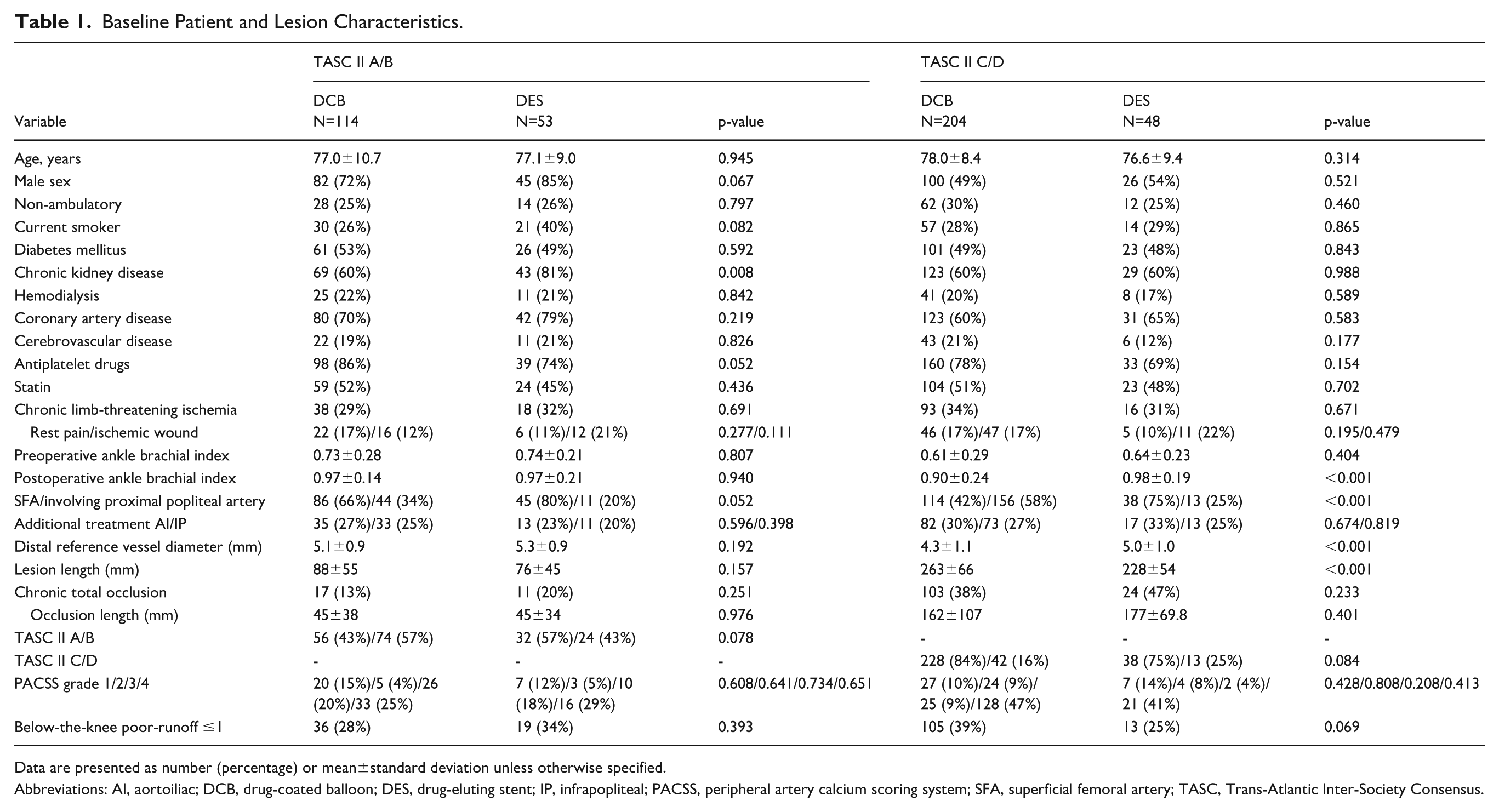
Data are presented as number (percentage) or mean±standard deviation unless otherwise specified.
Abbreviations: AI, aortoiliac; DCB, drug-coated balloon; DES, drug-eluting stent; IP, infrapopliteal; PACSS, peripheral artery calcium scoring system; SFA, superficial femoral artery; TASC, Trans-Atlantic Inter-Society Consensus.
Baseline Procedure Characteristics.
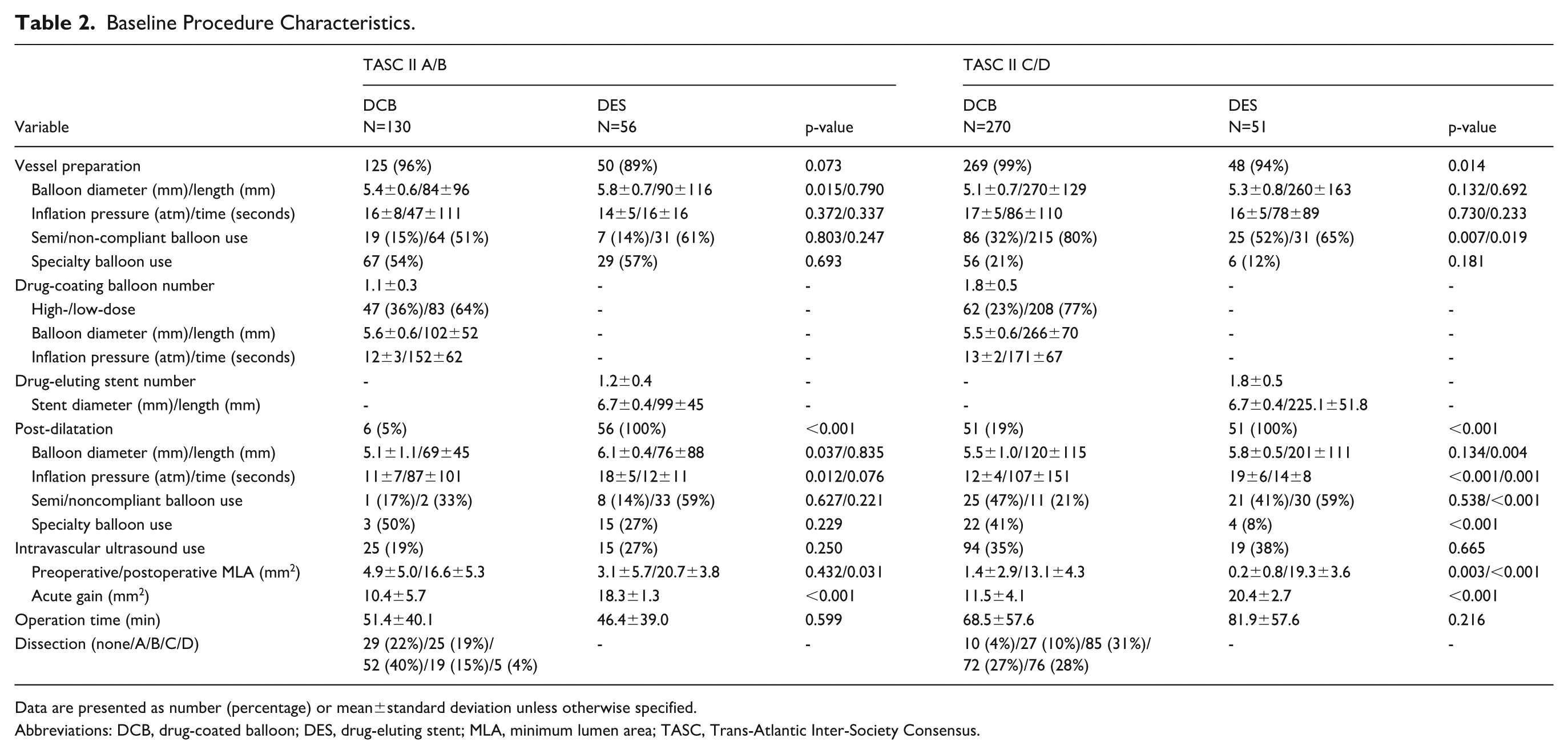
Data are presented as number (percentage) or mean±standard deviation unless otherwise specified.
Abbreviations: DCB, drug-coated balloon; DES, drug-eluting stent; MLA, minimum lumen area; TASC, Trans-Atlantic Inter-Society Consensus.
Clinical Outcomes
Primary patency at 3 years was comparable between DES and DCB in TASC II A/B (80.0% vs 76.9%, p=0.432), whereas in the TASC II C/D, it was significantly higher with DES (85.1% vs 50.3%, p<0.001) (Figure 1). Freedom from TLR at 3 years was similar in TASC II A/B (84.2% vs 88.0%, p=0.959), but in TASC II C/D, DES consistently outperformed DCB (90.2% vs 66.7%, p=0.008) (Figure 2). The AFS did not differ between groups through 3 years. Clinical outcomes are presented in Table 3.
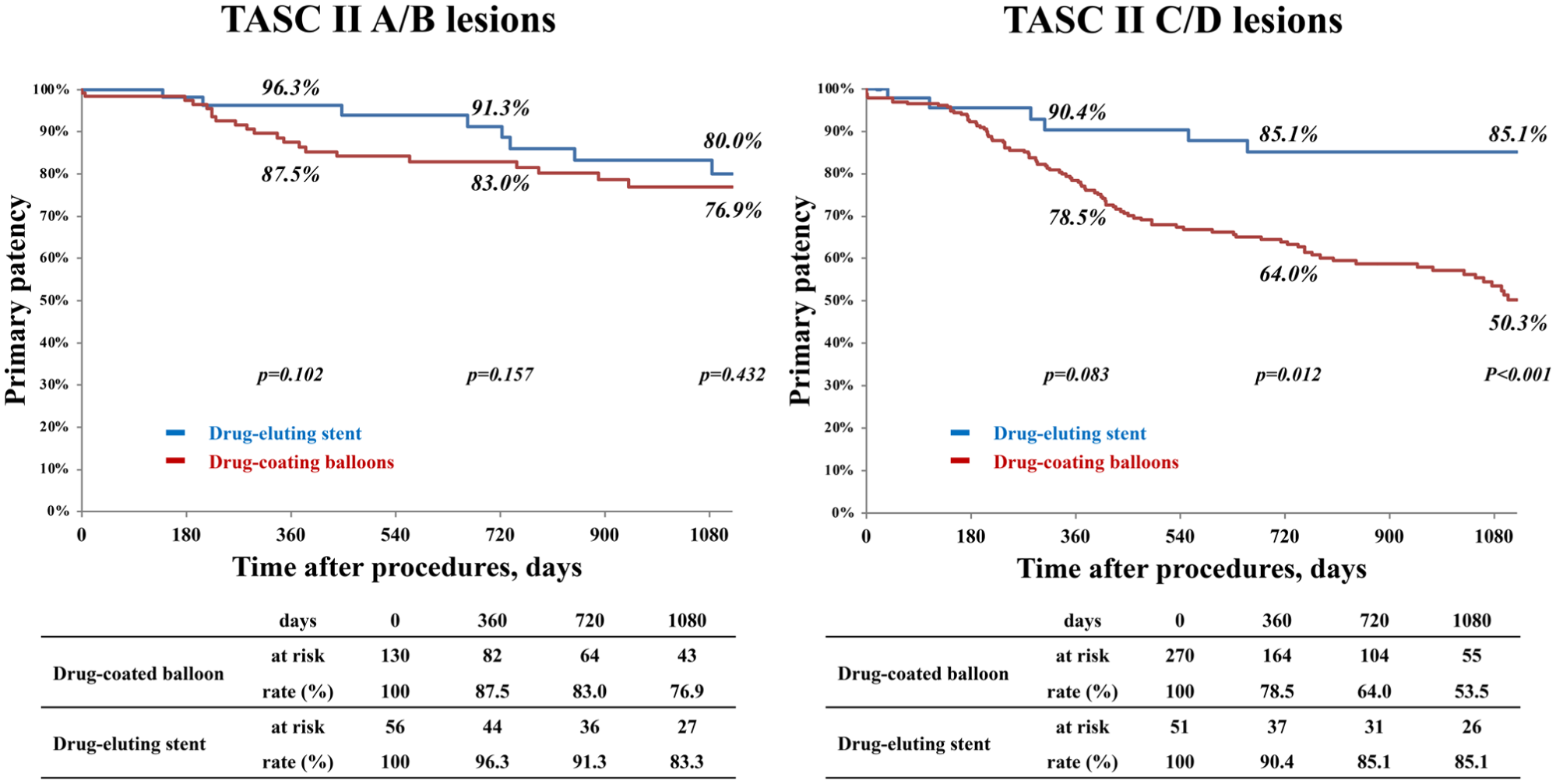
Primary patency for DES vs DCB strategies in TASC II A/B and C/D lesions. DES, drug-eluting stent; DCB, drug-coated balloon; TASC II, Trans-Atlantic Inter-Society Consensus II.
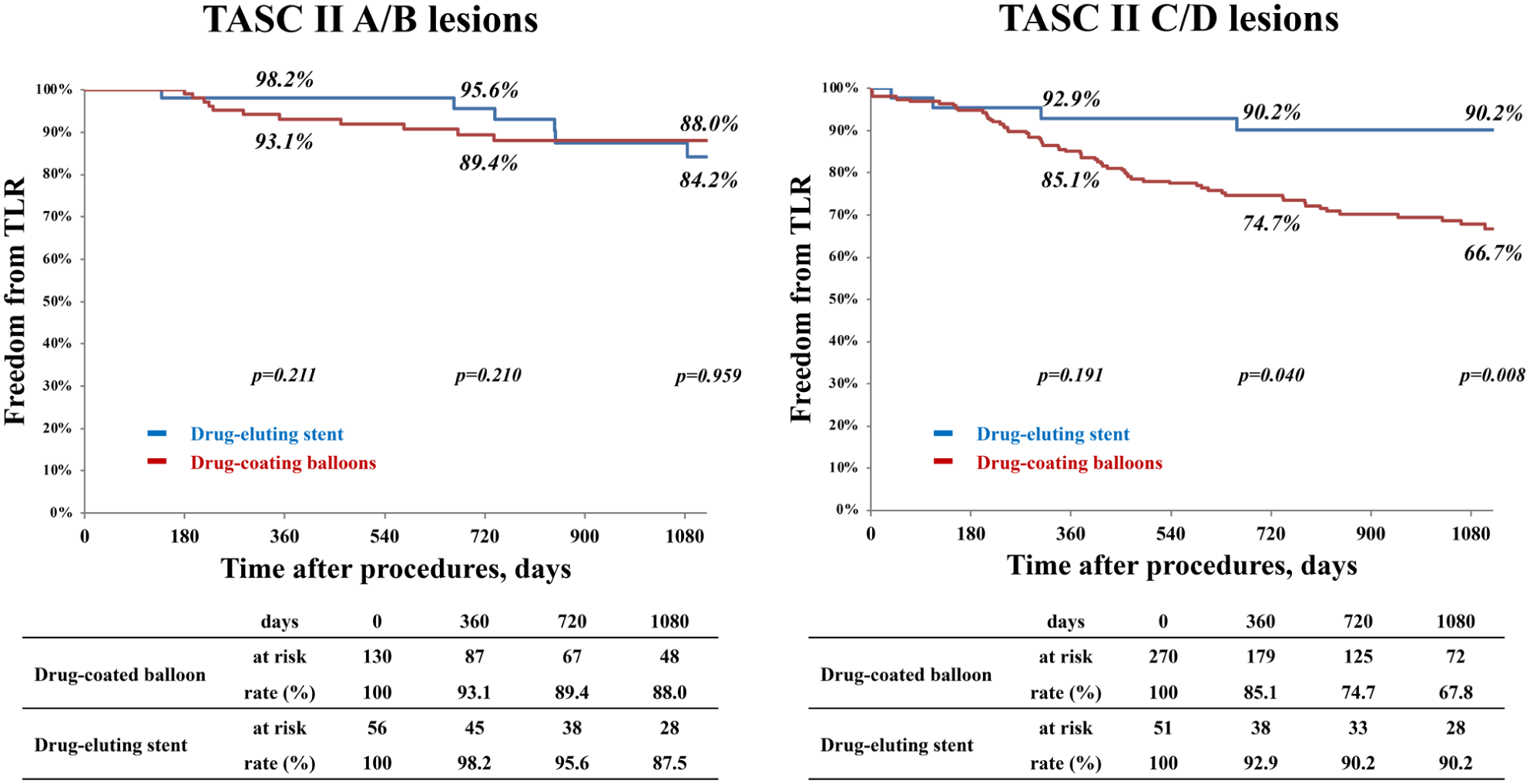
Freedom from TLR for DES vs DCB strategies in TASC II A/B and C/D lesions. TLR, target lesion revascularization; DES, drug-eluting stent; DCB, drug-coated balloon; TASC II, Trans-Atlantic Inter-Society Consensus II.
Study Outcomes.
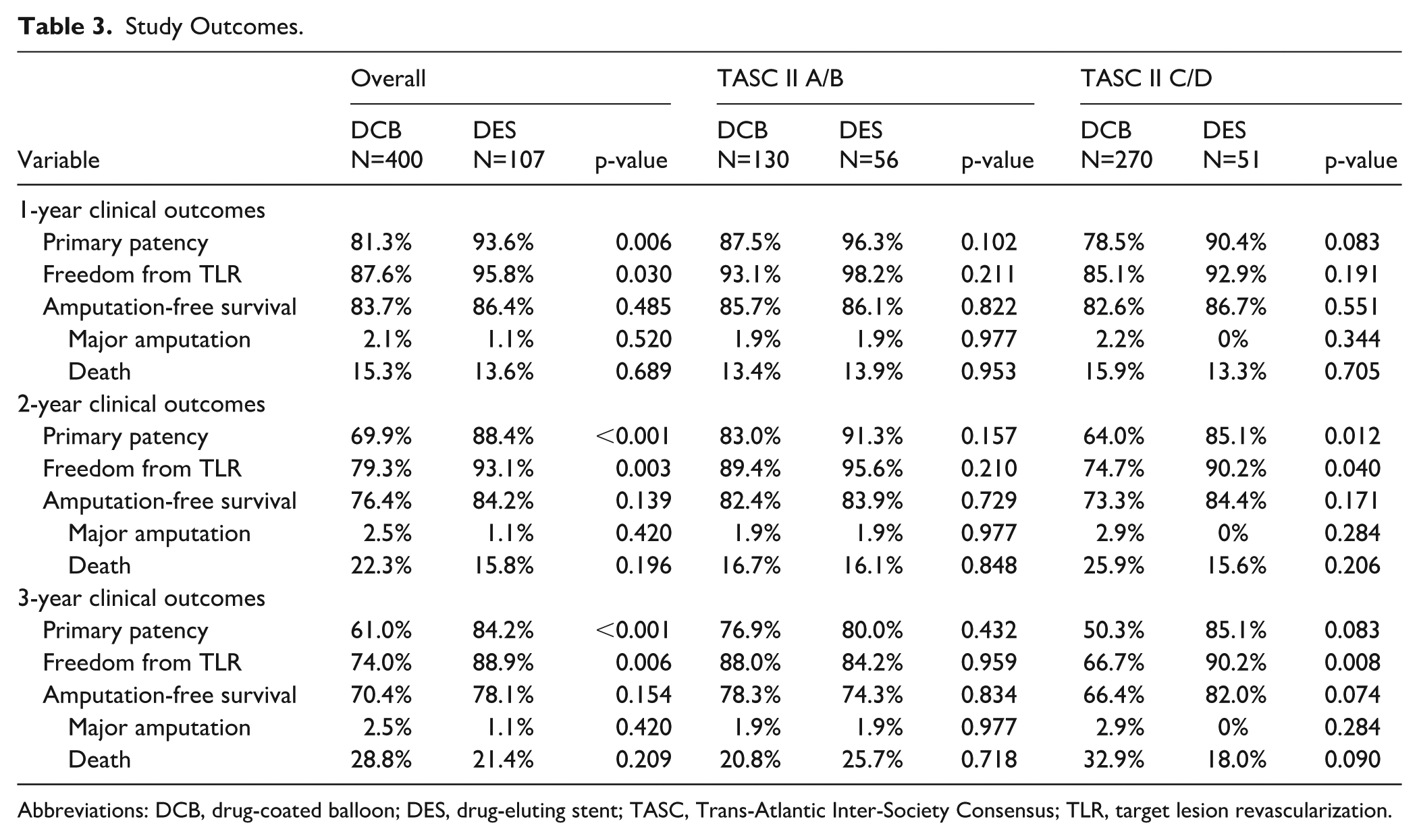
Abbreviations: DCB, drug-coated balloon; DES, drug-eluting stent; TASC, Trans-Atlantic Inter-Society Consensus; TLR, target lesion revascularization.
Figure 3 shows primary patency by treatment within each cohort. In the DCB cohort, 3-year primary patency was significantly worse in TASC II C/D than A/B across time, whereas DES outcomes were similar across complexity strata. Clinical outcomes by treatment within each TASC II category are summarized in Supplemental Table 1.
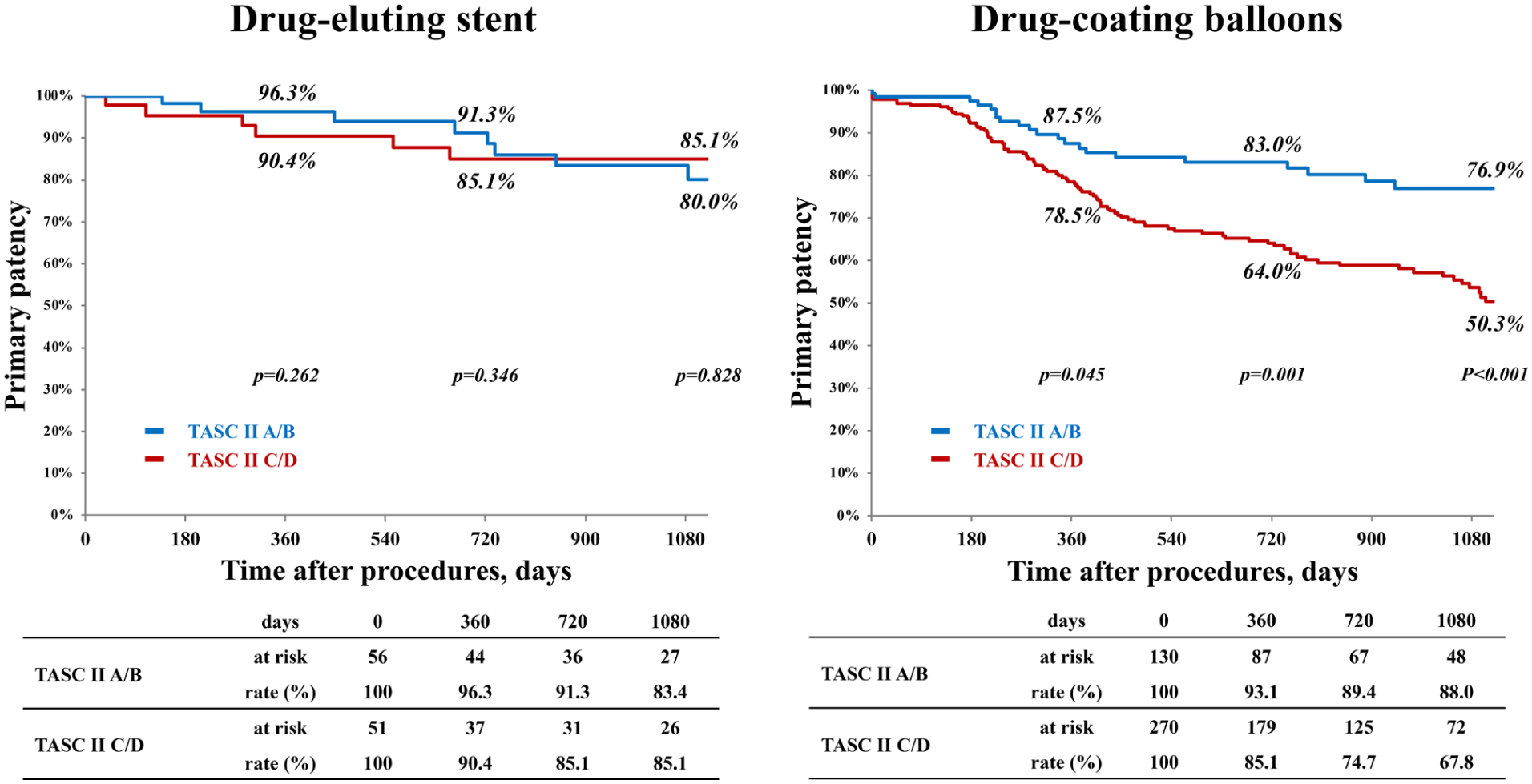
Primary patency for TASC II A/B vs C/D lesions treated with DES and DCB strategies. TASC II, Trans-Atlantic Inter-Society Consensus II; DES, drug-eluting stent; DCB, drug-coated balloon.
Predictors of Patency Loss
Multivariable analysis is shown in Table 4. In TASC II A/B treated with DCB, statin use was a protective patency factor (hazard ratio [HR]=0.32; 95% confidence interval [CI]=0.13-0.78), whereas proximal popliteal involvement conferred higher risk (HR=2.45; 95% CI=1.03-5.86). In TASC II C/D treated with DCB, below-the-knee runoff ≤1 (HR=1.96; 95% CI=1.30-2.95) and TASC II D classification (HR=2.57; 95% CI=1.60-4.13) were independent predictors of patency loss. In DES-treated TASC II A/B, ischemic wounds (HR=6.61; 95% CI=1.32-33.1) and PACSS grade 2 (HR=14.3; 95% CI=2.7-90.0) were associated with increased risk. No independent predictors were identified for DES-treated TASC II C/D.
Predictors of Patency Loss.
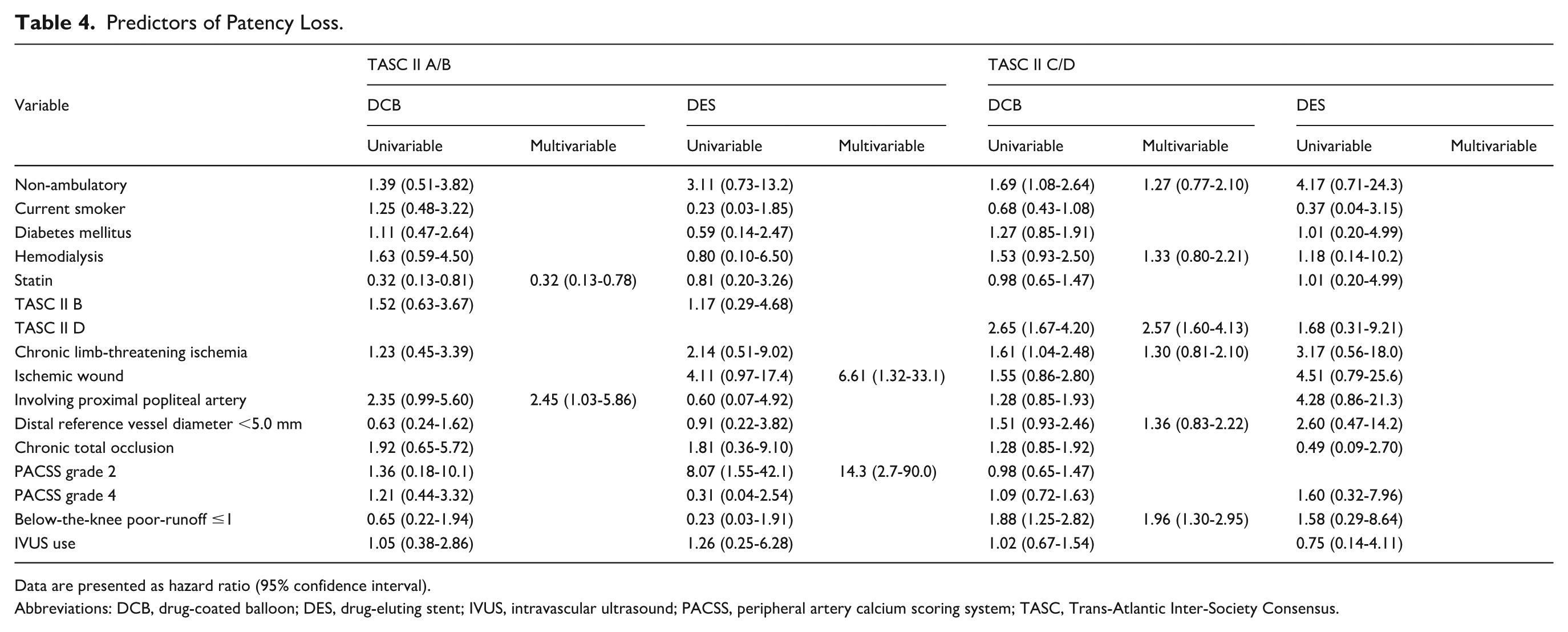
Data are presented as hazard ratio (95% confidence interval).
Abbreviations: DCB, drug-coated balloon; DES, drug-eluting stent; IVUS, intravascular ultrasound; PACSS, peripheral artery calcium scoring system; TASC, Trans-Atlantic Inter-Society Consensus.
Supplemental Table 2 summarizes predictors of patency loss in the overall cohorts. In the overall DCB cohort, TASC II D classification (HR=2.91; 95% CI=1.84-4.62), proximal popliteal involvement (HR=1.49; 95% CI=1.01-2.19), and below-the-knee runoff ≤1 (HR=1.65; 95% CI=1.14-2.39) were independent predictors. In the overall DES cohort, ischemic wounds (HR=4.30; 95% CI=1.03-18.0) and a stent-to-distal reference vessel diameter ratio ≥1.3 (HR=8.23; 95% CI=1.75-38.7) predicted patency loss.
Restenosis Patterns
At 3 years, reocclusion rates in TASC II A/B were similar between groups (DES 20% vs DCB 15%, p=0.544). In TASC II C/D, reocclusion was more frequent with DES than with DCB (60% vs 29%, p=0.044) (Figure 4).
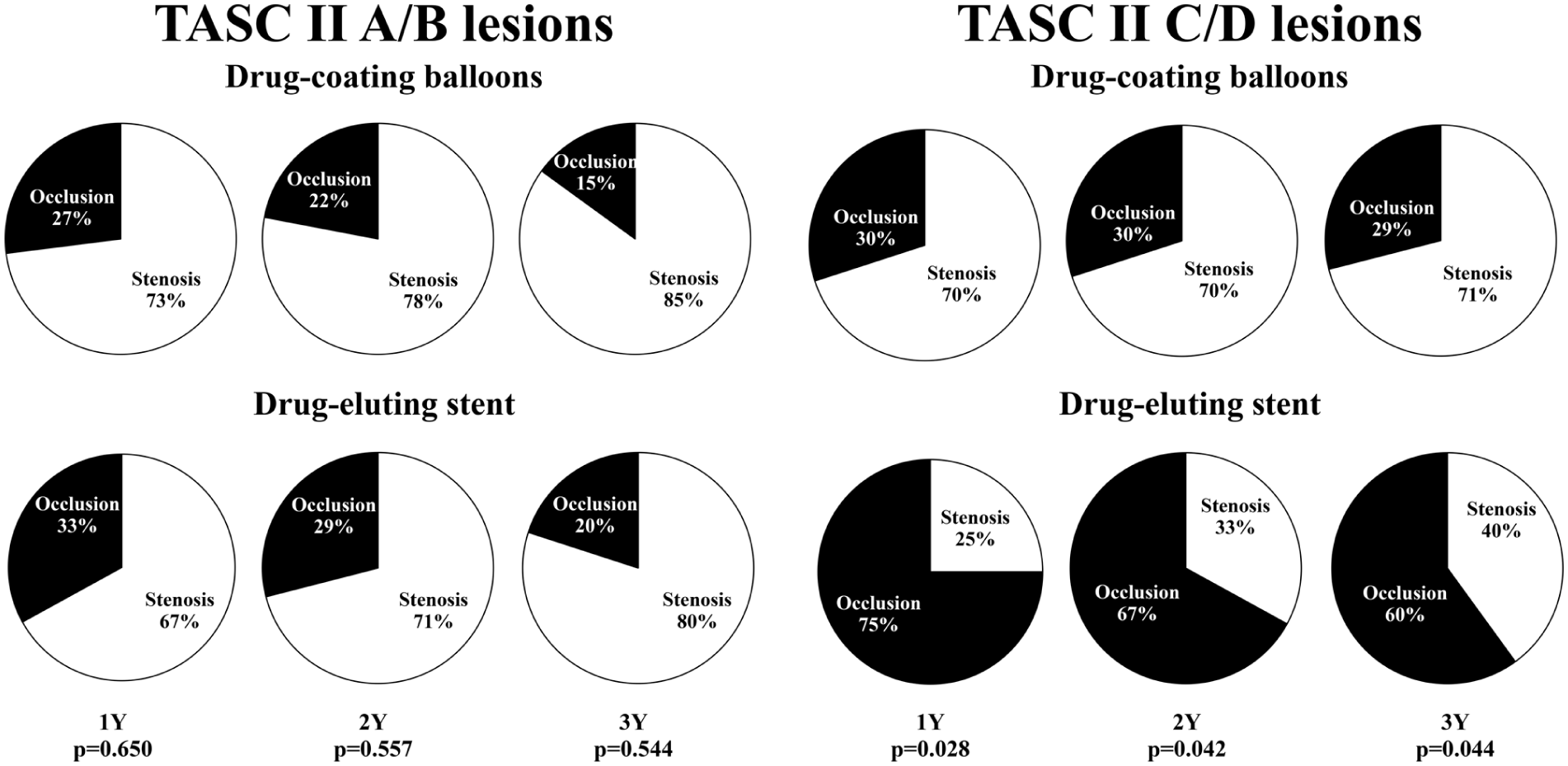
Restenosis patterns for DES vs DCB in TASC II A/B and C/D lesions. DES, drug-eluting stent; DCB, drug-coated balloon; TASC II, Trans-Atlantic Inter-Society Consensus II.
Discussion
This retrospective study compared long-term outcomes of full-coverage DES and DCB in de novo femoropopliteal lesions, stratified by TASC II classification. Lesion complexity emerged as a key determinant of device performance. In TASC II A/B lesions, both devices achieved high 3-year primary patency and freedom from TLR, with no significant differences, supporting the selective use of a stentless approach in suitable cases. In contrast, in TASC II C/D lesions, DES consistently achieved superior patency and TLR outcomes despite near-universal vessel preparation in both groups. The AFS and all-cause mortality were comparable between treatment strategies in both complexity categories, suggesting no adverse survival trade-off for either approach. In the TASC II C/D cohort, AFS numerically favored DES over DCB, with the between-group difference driven primarily by mortality rather than major amputation. This trend may partly reflect residual confounding due to modest baseline imbalances, since DES recipients tended to be younger, more frequently ambulatory, and less often on hemodialysis or presenting with CLTI, which could have biased the composite endpoint in favor of DES.
These results reaffirm that lesion complexity and arterial morphology are essential factors in device selection for femoropopliteal interventions.1,2 The comparable performance of DCB and DES in TASC II A/B lesions further supports the feasibility of a stentless strategy in suitable cases, which preserves future treatment options. The favorable outcomes with DES in TASC II C/D lesions may be attributed to consistent full-lesion coverage and routine post-dilatation, both of which optimize stent expansion and increase the post-procedural lumen area. In addition, the polymer-based sustained drug release of the Eluvia platform may confer prolonged antiproliferative effects in long or complex lesions.5,8,11,19 On the contrary, DCB performance may be limited in heavily calcified or diffusely diseased segments due to insufficient drug penetration or paclitaxel retention in the arterial wall.20,21
A notable observation in this study was that the 3-year primary patency rate of DES exceeded that reported in previous trials.5,22,23 Earlier studies identified spot stenting, CTO, and prior revascularization as independent predictors of 1-year restenosis following DES implantation. 8 The superior results observed here may reflect the exclusive inclusion of de novo lesions and the consistent use of a full-coverage DES strategy, which minimizes the negative effects associated with focal stenting. The CTO has also been linked to higher restenosis rates with DCB therapy. 3 Prior studies on DCB-treated femoropopliteal CTOs indicate that intraluminal angioplasty, compared with subintimal approaches, is associated with superior outcomes, potentially through achieving a larger MLA and reducing flow-limiting dissections.14,24 In our cohort, predominant use of IVUS-guided intraplaque crossing may have contributed to the relatively favorable outcomes in the DCB group. Nevertheless, TASC II D classification emerged as an independent predictor of restenosis in the DCB group, emphasizing the potential advantage of full-coverage DES in highly complex lesions. The TASC II C/D cohort exhibited more adverse anatomical features than A/B lesions, which influenced both procedural and clinical outcomes. Device utilization also differed by lesion complexity, with DES used in 30.1% (56/186) of TASC II A/B lesions and 15.9% (51/321) of C/D lesions. This pattern likely reflects anatomical constraints, including smaller distal reference vessel diameter and longer lesion length in C/D lesions, both of which limit available stent sizes and lengths, thereby reducing the feasibility of a full-coverage DES approach. In the DCB group, post-procedural dissections were more severe in TASC II C/D than in A/B lesions. The IVUS assessment revealed that acute luminal gain was largely comparable across TASC II classifications in both treatment groups; however, post-procedural MLA was smaller in C/D lesions. These findings suggest that final lumen area, rather than the magnitude of acute luminal gain, may be the key determinant of long-term patency.
In DES-treated TASC II A/B lesions, ischemic tissue loss and extensive eccentric calcification were identified as independent predictors of restenosis. The emergence of long eccentric calcification as a predictor in TASC II A/B, but not in C/D, lesions may reflect the higher proportional burden of calcification relative to lesion length in shorter A/B lesions. Heavily calcified eccentric plaques may impair balloon angioplasty effectiveness and hinder symmetrical stent expansion, thereby increasing the risk of restenosis. 25 Certain multivariable predictors exhibited wide confidence intervals, indicating limited statistical precision; accordingly, these findings should be regarded as exploratory and hypothesis-generating and warrant validation in larger prospective cohorts.
Prior studies have suggested that DCBs are more frequently associated with restenosis, whereas DES is more often linked to reocclusion.5,8 Consistent with these reports, in the present study, DES-treated TASC II C/D exhibited a significantly higher reocclusion rate over 3 years, whereas in TASC II A/B, no significant between-strategy differences were observed, and restenosis predominated in both groups. These observations suggest that reocclusion after DES in TASC II C/D may be driven by thrombotic burden across long-stented segments, delayed endothelialization associated with sustained paclitaxel release over approximately 1 year, and limited scaffold conformability within tortuous, calcified plaque morphologies. Practical risk-mitigation strategies may include extension of dual antiplatelet therapy, selective adjunctive anticoagulation in high-risk profiles, meticulous pre- and post-dilatation to optimize stent expansion, and intensified duplex ultrasound surveillance during follow-up.
Finally, sirolimus-coated balloons warrant consideration. Recent data indicate favorable long-term durability and outcomes. A Japanese multicenter study in femoropopliteal disease reported high 1-year primary patency and freedom from TLR with an acceptable safety profile. 26 Similarly, extended follow-up series in femoropopliteal steno-occlusive disease have demonstrated maintained efficacy without emergent safety signals at 3 years. 27 Moreover, trials that included both femoropopliteal and below-the-knee lesions have shown consistent 3-year performance, suggesting a potential class effect across vascular beds while underscoring device-specific delivery chemistries and elution kinetics. 28 As these platforms gain wider adoption, rigorously designed head-to-head comparisons with paclitaxel-coated devices, particularly in complex TASC II C/D lesions, are needed to define comparative effectiveness and to identify lesion subsets most likely to benefit.
Limitations
Several limitations warrant consideration. First, this was a single-center retrospective study, which introduces inherent selection bias and limits the generalizability of the findings. Although lesion stratification was performed using the TASC II classification, unmeasured confounders, such as operator technique and device selection criteria, may have affected outcomes. Second, lost to follow-up, defined a priori as the inability to ascertain vital status and study outcomes for ≥12 months before the 3-year landmark despite attempts using ≥2 independent sources, was observed in 73/419 patients (17.4%), comprising 15/101 (14.9%) in the DES group and 58/318 (18.2%) in the DCB group. This degree of attrition in a retrospective design may influence the results; therefore, the findings should be interpreted with appropriate caution. Third, imaging guidance was nonuniform. In cases without intravascular imaging, vessel sizing was estimated angiographically using quantitative vascular analysis, which may introduce variability in luminal optimization. Prior studies suggest that IVUS-guided sizing improves acute luminal gain and reduces geographic miss.10,29,30 Accordingly, inconsistent imaging guidance, specifically the preferential use of IVUS in more complex lesions and the tendency to omit IVUS in simpler lesions, may have introduced variability in vessel sizing and selection bias and thereby contributed to differences in long-term outcomes. Fourth, although vessel preparation was performed in nearly all cases, the specific devices and techniques varied by operator, introducing procedural heterogeneity. Fifth, follow-up duplex ultrasound and clinical assessments were conducted according to institutional protocols, which may have introduced variability in restenosis detection. Sixth, this study excluded bailout stenting and atherectomy to reduce confounding and procedural heterogeneity and to isolate the intrinsic efficacy of DES and DCB strategies. Although this design strengthens internal validity, it may limit external generalizability because these adjunctive techniques are frequently employed in routine practice. In Japan, atherectomy use is constrained by domestic reimbursement policies and bailout stenting during DCB therapy is not reimbursed, factors that influence operator behavior and treatment selection, and caution is warranted when extrapolating these findings to healthcare systems in which atherectomy and reimbursed bailout stenting are routine. In such systems, operators may employ more aggressive vessel preparation and salvage flow-limiting dissections with provisional stenting; accordingly, DCB performance could differ from that observed in the present cohort. Seventh, isolated popliteal lesions and those involving the popliteal P2-P3 segments were excluded; therefore, the generalizability of full-coverage strategies with DCB or DES to these highly mobile segments cannot be established from our data. Lesions extending beyond the popliteal P1 segment were excluded because a prior report suggests that deploying DES across the P2-P3 segments may increase the risk of restenosis, 31 which could confound intergroup comparisons. In this cohort, lesions involving the P1 segment were more frequent in the DCB group; however, the previous literature has not identified popliteal lesions as an independent predictor of restenosis after DCB, 3 indicating that selection bias related to our enrollment criteria is likely limited. Moreover, although contemporary practice in the popliteal artery tends to favor motion-tolerant platforms (eg, biomimetic stents) rather than self-expandable DES, our findings suggest that a full-coverage DES strategy may achieve durable patency when confined to the proximal popliteal segment. Finally, some subgroup analyses were exploratory because of small sample sizes and limited statistical precision.
Conclusion
Device selection for de novo femoropopliteal lesions should be tailored to lesion complexity and arterial morphology within a TASC II-based framework. In complex C/D lesions, full-lesion-coverage DES with optimized expansion provides superior long-term patency and fewer reinterventions, whereas in A/B lesions, a selective leave-nothing-behind DCB strategy achieves outcomes comparable to DES at 3 years. Embedding lesion complexity into treatment algorithms may optimize patency, reduce repeat interventions, and allocate advanced devices to patients most likely to benefit. These recommendations should be validated in prospective, multicenter, randomized trials to refine TASC II-guided leave-nothing-behind vs full-lesion-coverage decision-making.
Supplemental Material
sj-docx-1-jet-10.1177_15266028261420088 – Supplemental material for Full-Coverage Drug-Eluting Stent Vs Drug-Coated Balloon in De Novo Femoropopliteal Lesions: Three-Year TASC II-Stratified Outcomes
Supplemental material, sj-docx-1-jet-10.1177_15266028261420088 for Full-Coverage Drug-Eluting Stent Vs Drug-Coated Balloon in De Novo Femoropopliteal Lesions: Three-Year TASC II-Stratified Outcomes by Takuya Haraguchi, Masanaga Tsujimoto, Yuhei Kasai, Yoshifumi Kashima, Katsuhiko Sato and Tsutomu Fujita in Journal of Endovascular Therapy
Supplemental Material
sj-docx-2-jet-10.1177_15266028261420088 – Supplemental material for Full-Coverage Drug-Eluting Stent Vs Drug-Coated Balloon in De Novo Femoropopliteal Lesions: Three-Year TASC II-Stratified Outcomes
Supplemental material, sj-docx-2-jet-10.1177_15266028261420088 for Full-Coverage Drug-Eluting Stent Vs Drug-Coated Balloon in De Novo Femoropopliteal Lesions: Three-Year TASC II-Stratified Outcomes by Takuya Haraguchi, Masanaga Tsujimoto, Yuhei Kasai, Yoshifumi Kashima, Katsuhiko Sato and Tsutomu Fujita in Journal of Endovascular Therapy
Footnotes
Acknowledgements
The authors thank all the members of the Sapporo Heart Center.
Ethical Considerations
The study protocol was reviewed and approved by the institutional ethics committee of Sapporo Heart Center and was conducted in accordance with the principles of the Declaration of Helsinki.
Consent to Participate
Given the retrospective design, the requirement for written informed consent was waived.
Consent for Publication
Not applicable. This study did not include any identifiable data from individual participants.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data sets are not publicly available due to the presence of potentially identifiable patient information, but de-identified data may be available from the corresponding author on reasonable request and with appropriate institutional approvals.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
