Abstract
Purpose:
Iliac branch devices (IBDs) are indicated for the preservation of internal iliac artery (IIA) perfusion. Data on bridging stent choice in IBD are limited. The aim of this meta-analysis was to assess Advanta V12/iCast outcomes when used as bridging stent during IBD procedures.
Methods:
The English medical literature was systematically searched through PubMed, Scopus, and Cochrane Library (last search: 03.01.2025). The Preferred Reporting Items for Systematic Reviews and Meta-analyses statement was followed, and a predefined protocol was registered to PROSPERO. Randomized controlled trials and observational studies (2000–-2025) reporting on the Advanta V12/iCast-related outcomes were eligible. The ROBINS-I tool and Grading of Recommendations Assessment, Development and Evaluations (GRADE) were used to assess the risk of bias and quality of evidence. Primary outcomes were technical success, stenosis/occlusion, endoleak Ic/IIIc, and reintervention rates during follow-up. Data on the outcomes of interest were synthesized using proportional meta-analysis.
Results:
From 2698 study reports, 10 observational studies (510 targeted IIAs, 467 IIAs bridged with Advanta V12/iCast) were included. According to ROBINS-I, eight studies were of low and two were of moderate quality. Technical success was 98% (95% confidence interval (CI) = 93%–100%; I2 = 0%; certainty: very low). Regarding the 30-day outcomes, the occlusion/stenosis rate was 2% (95% CI = 0%–9%; I2 = 0%), the endoleak Ic/IIIc rate was 0.3% (95% CI = 0%–3%; I2 = 0%) and the reintervention rate was 1% (95% CI = 0%–3%; I2 = 0%). The mean follow-up among studies was 23.4 months (95% CI = 14.2–32.7). During follow-up, the occlusion/stenosis rate was 4% (95% CI = 1%–11%; I2 = 19%; certainty: very low), the endoleak Ic/IIIc overall rate was 5% (95% CI = 3%–8%; I2 = 0%; certainty: very low), and the reintervention rate was 5% (95% CI = 3%–9%; I2 = 0%; certainty: very low).
Conclusions:
The Advanta V12/iCast balloon-expandable covered stent showed low occlusion/stenosis, endoleak, and reintervention rates at 30 days and follow-up and appeared to be a reliable bridging stent choice in IBDs.
Clinical Impact
The available literature on iliac branch devices does not provide adequate evidence supporting bridging stent selection. This systematic review and meta-analysis on the use of Advanta V12/iCast as bridging stent showed a high technical success rate of 98%, along with low occlusion/stenosis, endoleak Ic/IIIc and reintervention rates at 30 days, which were maintained below 5% during the total follow-up. These results reflect on the reliability of Advanta V12/iCast when used as bridging stent in iliac branch devices. However, the low quality of evidence should be considered upon result interpretation.
Introduction
Iliac artery aneurysms are reported in up to 7% of all aortoiliac aneurysms, with 12%–48% of them being bilateral.1,2 Aneurysmal distal landing zones in the common iliac arteries could compromise the durability of a successful endovascular aortic aneurysm repair.3–5 The development and evolution of iliac branch devices (IBDs), since their initial introduction 20 years ago, permitted the preservation of flow in the internal iliac artery (IIA) during endovascular aortic aneurysm repair. 6 Endovascular preservation of the IIA is associated with a decreased risk of pelvic ischemia, when compared with embolization and distal sealing to the external iliac artery (EIA).7–9
Of the three available off-the-shelf IBDs in Europe (Cook ZBIS, Artivion E-Iliac, Gore IBE), only one (Gore IBE) has a dedicated bridging component. The incorporation of the IIA in all three devices can be though achieved using a self-expanding or balloon-expandable bridging covered stent. 10 However, as IBD may be vulnerable to both internal iliac branch occlusion and endoleaks, the choice of the bridging covered stent could play a significant preventive role. Finite evidence based on studies over a decade ago showed low occlusion rates of balloon-expandable covered stents, 10 including the Advanta V12/iCast (Getinge/Atrium Medical Corporation, Merrimack, New Hampshire). The good long-term outcomes of the Advanta V12/iCast led to its increased use.4,11
The aim of this analysis was to systematically search and analyze the 30-day and follow-up outcomes of the Advanta V12/iCast balloon-expandable covered stent as bridging component in IBDs.
Materials and Methods
Eligible Studies
The Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines were applied for the conduction of this systematic review and meta-analysis (Figure 1). 12 The predefined methodology, including the study type, inclusion criteria, and objectives, were registered to the PROSPERO. Prospective and retrospective observational cohort studies and any randomized control trial, published in English from 2000 to 2025, reporting on patients managed with IBDs using the Advanta V12/iCast balloon-expandable covered stent (Getinge/Atrium Medical Corporation) as bridging stent for the revascularization of the IIA or its main branches were eligible. Studies had to report on more than ten patients managed with IBDs for aortic aneurysms extending to the iliac arteries or isolated iliac aneurysms and, if this criterion was fulfilled, studies were further required to report on at least five Advanta V12/iCast bridging stents. The Advanta V12 is the exact same product as the iCast balloon-expandable covered stent, under different branding; the earlier is available in Europe, whereas the latter is available in the United States. The patients could have been treated with customized or any commercially available off-the-shelf IBDs.
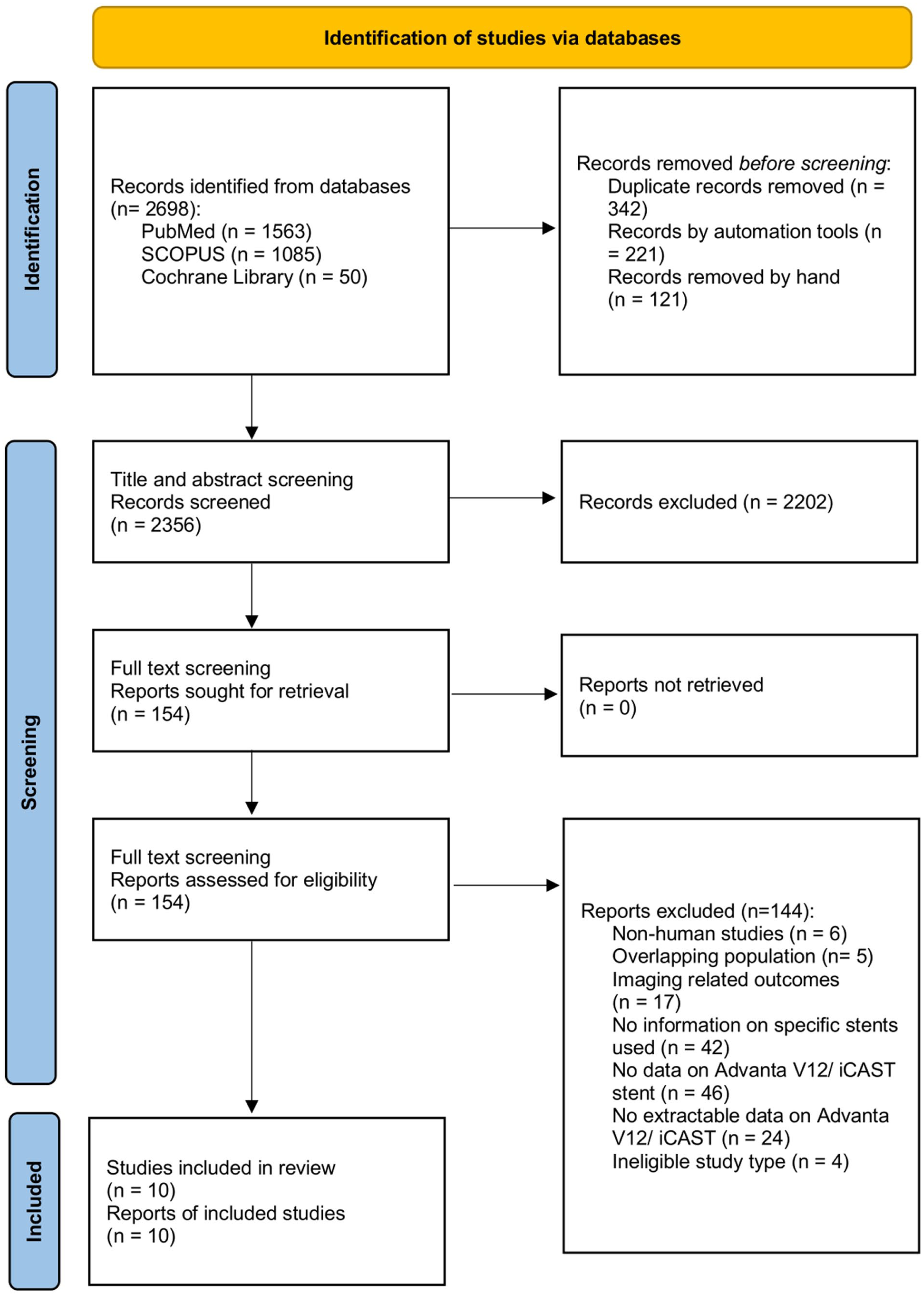
PRISMA 2020 flow chart depicting the study selection process. Ten studies have been included in the present systematic review and meta-analysis on Advanta V12/iCast balloon-expandable covered stent outcomes when used as bridging stent in iliac branch devices.
Studies reporting on patients managed with surgeon-modified endografts, fenestrated devices or any other complex technique, than IBDs, to revascularize the IIA were excluded. The proximal extend of coverage, in terms of infra-renal or supra-renal aortic coverage (eg, using fenestrated or branched endovascular aortic repair) and landing into the IIA trunk or any main branch of it (eg, superior or inferior gluteal artery) was not a criterion for exclusion. Studies reporting exclusively on other balloon-expandable stents as well as studies that did not provide extractable data on Advanta V12/iCast were also excluded. Case reports were not eligible. Among studies with potential overlapping populations, the most recently published study or the one presenting with the largest cohort was included in the analysis.
Ethical Considerations and Approval
This study complied with the Declaration of Helsinki and no approval by the ethics committee was required due to study design.
Search Strategy
The search end date was set for January 3, 2025. A systematic search of the English medical literature using the Medline, SCOPUS, and Cochrane databases was performed according to the PICO (Patient; Intervention; Comparison; Outcome [Supplementary Table 1]) model. 13 The following search terms, including Expanding Medical Subject Heading (MeSH terms), were used in various combinations (Supplementary Table 2): “abdominal aortic aneurysm,” “aortoiliac aneurysm,” “common iliac artery aneurysm,” “internal iliac artery aneurysm,” “endovascular aortic aneurysm repair,” “bridging stent,” “Advanta,” “iCast.” An extensive review of the available studies was conducted after abstract and subsequently full-text assessment by two authors. Any discrepancy was resolved after discussion with a third author.
Data Extraction
Data extraction was performed using a standardized Excel file, which included general study characteristics, as the year of publication, study nature, timespan, participating centers, and baseline information, such as number of patients, age, sex, underlying aortic disease (abdominal aortic aneurysm [AAA] or iliac aneurysm), landing zone of the bridging stent (IIA or gluteal artery), type of IBD, number of IIAs bridged using the Advanta V12/iCast. Information on 30-day and follow-up outcomes, including technical success, occlusion/stenosis, endoleak types Ic and IIIc, and reinterventions, was extracted. From studies which including also cases where the IIA was bridged using another bridging stent than the Advanta V12/iCast, only the data/cases reporting on the use of the Advanta V12/iCast were included for analysis. Data extraction was performed by two authors (GA and PN) and any discrepancy was resolved after discussion with a third author (TK).
Quality Assessment
The risk of bias was assessed using the ROBINS-I tool for nonrandomized observational studies. ROBINS-I evaluates seven domains of potential risk of bias, providing a score (“low,” “moderate,” “serious,” or “critical”), to achieve the maximum transparency and reliability of the conducted evaluation. 14 In addition, the Grading of Recommendations Assessment, Development and Evaluations (GRADE) was applied to evaluate the overall quality of evidence for each one of the main outcomes, by assessing seven key components, including the risk of bias, inconsistency, indirectness, imprecision, large effect, dose response, and confounder control. 15 Both assessments were performed by two independent investigators, and any discrepancy was resolved after discussion with a third author.
Definitions
As none of the included studies provided a specific definition for bridging stent-related technical success and relying on the available reporting standards, the technical success for bridging stent performance was defined as the composite of successful IIA catheterization and bridging stent deployment with a patent intended target vessel and absence of type Ic or IIIc endoleak, extending beyond 30 days. 16 The definitions of technical success of the included studies are presented in Table 1.
The Definition of Technical Success Per Study Included in a Systematic Review and Meta-Analysis of Ten Studies Reporting on Advanta V12/iCast Balloon-Expandable Covered Stent Outcomes When Used as a Bridging Stent in Iliac Branch Devices.

Abbreviations: IIA, internal iliac artery; EIA, external iliac artery; NA, not available; CIA, common iliac artery.
Outcomes
The primary outcomes were the technical success, occlusion/stenosis, endoleak Ic/IIIc, and reintervention rates of target vessels (either the IIA or superior gluteal artery) bridged with the Advanta V12/iCast during follow-up. Secondary outcomes were the stenosis/occlusion, endoleak Ic/IIIc, and reintervention at 30 days.
Statistical Analysis
Data regarding the outcomes of interest were extracted as event counts, while incidence proportion along with 95% confidence intervals (CIs) was calculated for each individual study and synthesized through meta-analysis of single proportions. Only integer proportions are reported, complying with our data qualitative and quantitative characteristics. Between-study heterogeneity was statistically assessed through tau squared and I2 statistics, which were presented through forest plots, along with individual studies and syntheses’ effect estimates. A generalized linear mixed model method based on logit transformation through the “metaprop” function was used to calculate the summary proportions along with their 95% CIs. 27 The method was preferred due to small sample size, as well as event count of included studies. Due to probable intrinsic heterogeneity between individual studies samples, irrespective of heterogeneity statistics’ values, the random-effects model was chosen to be presented. A maximum likelihood estimator was used for tau squared and Knapp and Hartung method was used to calculate CIs for the overall estimate. Publication bias was assessed through Egger’s test and visualized in funnel plots, if at least 10 studies were included in the quantitative synthesis. No imputation methods were applied for missing values. A continuity correction of 0.5 was only applied for individual study results. When presented as a summary, categorical data regarding patients’ characteristics were reported as cumulative proportions with 95% CIs (calculated through the same meta-analytical methods as the main outcome-related results), whereas continuous data were expressed as weighted means and corresponding 95% CIs (calculated by an inverse variance weighting-based method through “metamean” function). In addition, when medians with range or interquartile range were reported, conversion to mean values was applied.28,29 Statistical analyses and corresponding plots were performed with RStudio (Version 2024.12.0+467; Integrated Development Environment for R, Posit Software, PBC, Boston, Massachusetts).
Results
Study Selection and Risk of Bias
The systematic search yielded 2698 study reports. After study selection, 10 studies were included in the qualitative and quantitative synthesis (Figure 1).17–26 All included studies were of observational nature, with nine being retrospective,18–26 and one prospective. 17 The main characteristics of the included studies are shown in Table 2.
Main Characteristics of Studies Included in a Systematic Review and Meta-Analysis of 10 Studies Reporting on Advanta V12/iCast Balloon-Expandable Covered Stent Outcomes When Used as a Bridging Stent in Iliac Branch Devices.

ROBINS-I tool 14 risk of bias assessment revealed important sources of bias among the included studies, classifying 8 out of 10 at serious17–22,24,25 and 2 at moderate risk.23,26 Bias was mainly attributed to inadequate reporting and control of confounding, patient selection, handling of missing data, and postinterventional status related classification of intervention (Supplementary Figure 1). GRADE revealed a “very low” quality of evidence for all primary outcomes, a finding consistent with the observational nature of included studies and risk of bias assessment. 15 GRADE and “Summary of evidence” are provided in Supplementary Tables 3 and 4.
Patients and Target Vessels
In total, 474 patients were included (449 males [95%; 95% CI = 92%–97%]; Table 3). The mean age was 72.8 (95% CI = 71.5–74.2) years. The indication for treatment was stated in nine studies,17–22,24–26 including cases of common iliac artery (CIA) aneurysms with or without aneurysmal involvement of the abdominal aorta,17–20,22,25,26 along with patients needing extension of previous abdominal aortic repair due to type Ib endoleak (Table 3). 21 One study set an indication for repair in cases with aneurysmal involvement of the IIA or isolated IIA aneurysms. 24 Baseline anatomic characteristics are presented in Table 4. The mean aortic diameter was 51 mm (95% CI = 44–57 mm; reported in five studies),17–20,25 whereas the mean CIA diameter was 35 mm (95% CI = 32–38 mm; reported in six studies),17,18,20,23,25,26 with the largest aortic or iliac common or internal diameter setting the indication for repair. A CIA aneurysm with concomitant AAA was identified in 314 cases (85%; 95% CI = 73%–92%) and an isolated CIA aneurysm was present in 61 cases (15%; 95% CI = 8%–27%).17–22,25,26
The Indication for Repair and Number of Patients, Devices and Stents of Studies Included in a Systematic Review and Meta-Analysis of 10 Studies Reporting on Advanta V12/iCast Balloon-Expandable Covered Stent Outcomes When Used as a Bridging Stent in Iliac Branch Devices.
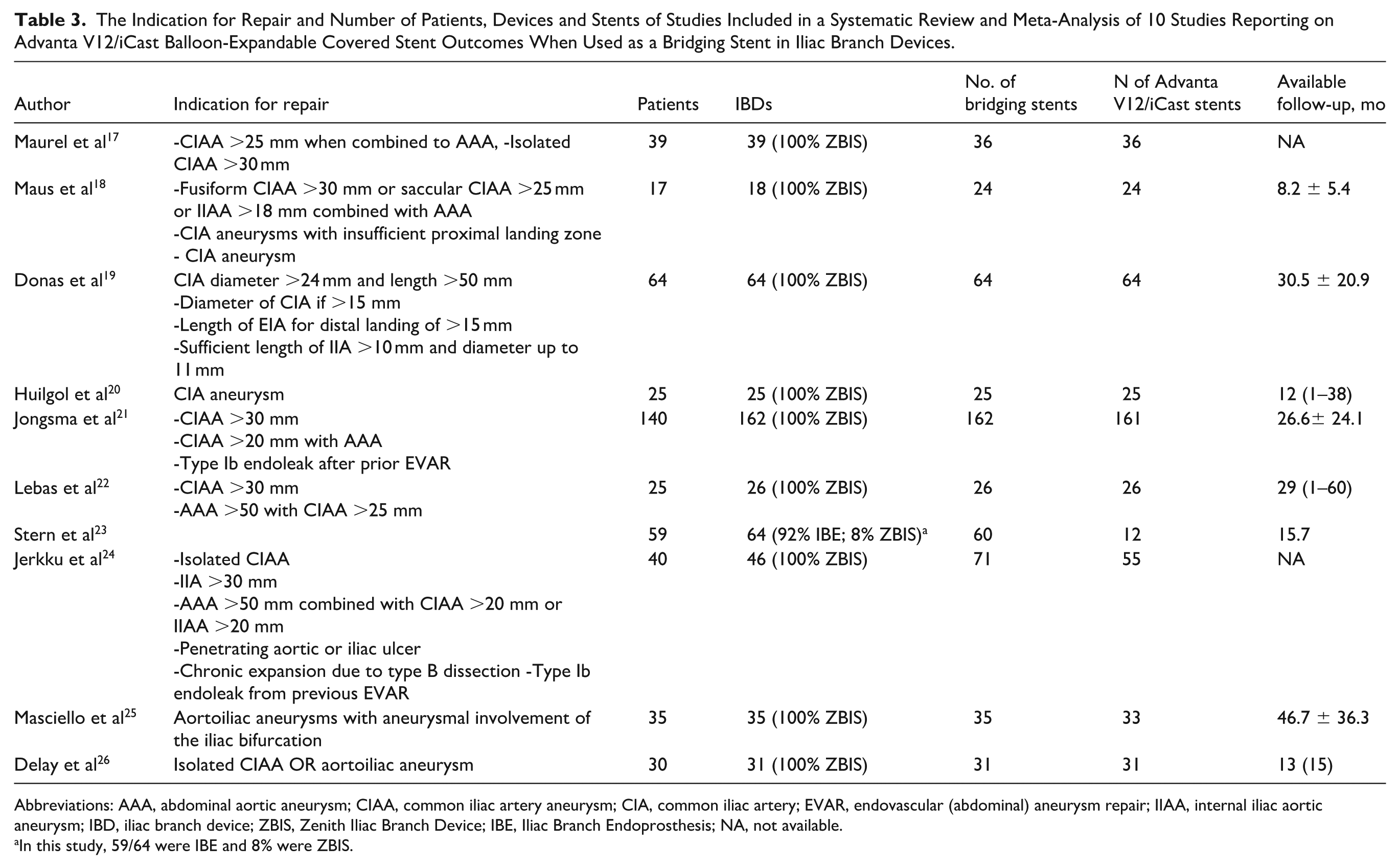
Abbreviations: AAA, abdominal aortic aneurysm; CIAA, common iliac artery aneurysm; CIA, common iliac artery; EVAR, endovascular (abdominal) aneurysm repair; IIAA, internal iliac aortic aneurysm; IBD, iliac branch device; ZBIS, Zenith Iliac Branch Device; IBE, Iliac Branch Endoprosthesis; NA, not available.
In this study, 59/64 were IBE and 8% were ZBIS.
Anatomic Baseline Characteristics of Patients Included in a Systematic Review and Meta-Analysis of Ten Studies Reporting on Advanta V12/iCast Outcomes When Used as a Bridging Stent in Iliac Branch Devices.
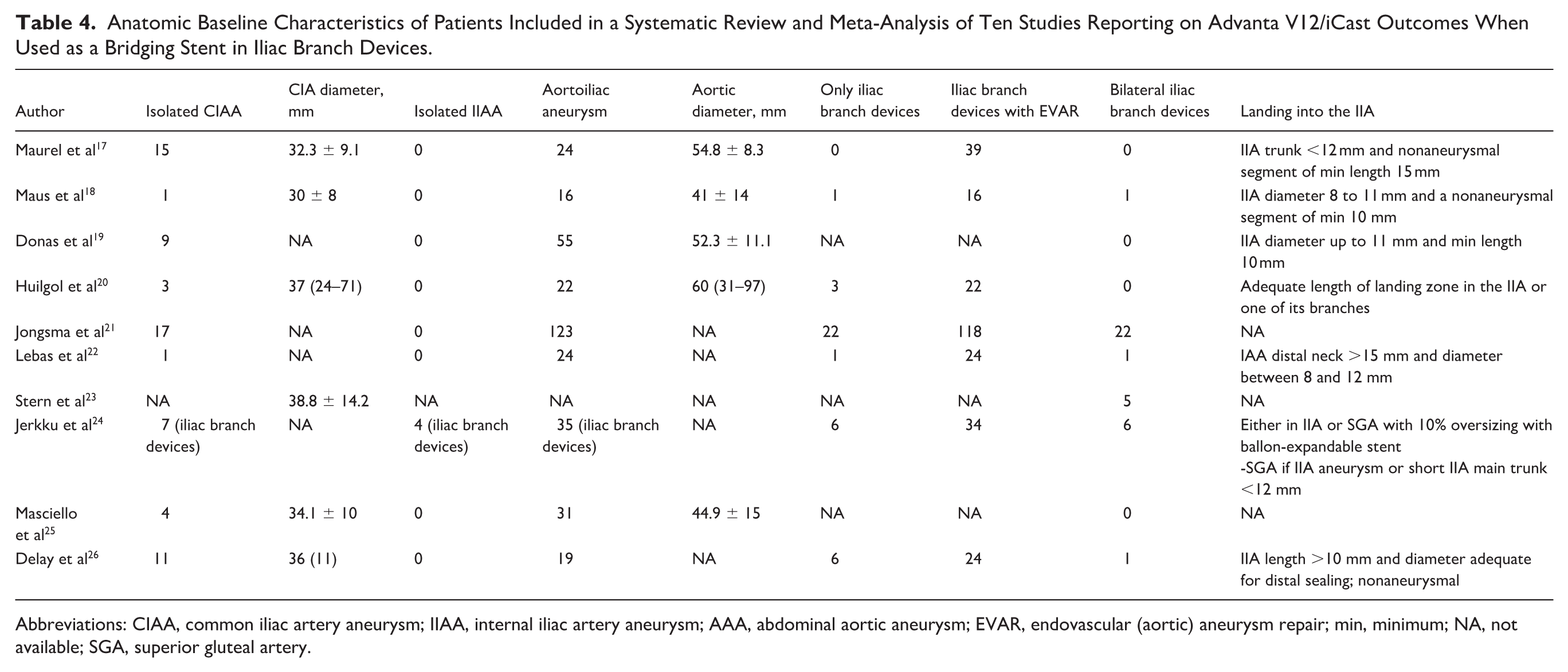
Abbreviations: CIAA, common iliac artery aneurysm; IIAA, internal iliac artery aneurysm; AAA, abdominal aortic aneurysm; EVAR, endovascular (aortic) aneurysm repair; min, minimum; NA, not available; SGA, superior gluteal artery.
In nine studies, the Zenith Branch Iliac Endovascular Graft (ZBIS; Cook Medical, Bloomington, Indiana) in combination to an Advanta V12/iCast balloon-expandable covered stent was implanted.17–22,24–26 In one study, 59 Gore Excluder Iliac Branch Endoprosthesis devices (IBE; W. L. Gore & Associates, Newark, Delaware) were used along with five ZBIS; all (IBE and ZBIS) bridged using an Advanta V12/iCast bridging stent. 23 No dedicated limb for the IBE devices was used in that cohort. 23 Masciello et al 25 used both ZBIS and IBEs, but the Advanta V12/iCast stent was used for bridging only along with ZBIS devices. In total, 451 out of 510 IBDs were ZBIS (88%) and 59 were IBE (12%).
Iliac branch device implantation was performed along with an endovascular aortic repair in 277 cases (90%; 95% CI = 79%–95%), while it was solely implanted in 39 cases (10%; 95% CI = 5%–21%).17,18,20–22,24 Thirty-six patients were treated with bilateral IBD (3%; 95% CI: 1%–11%).17–25 In total, 510 IBDs were implanted, and 534 stents were used for bridging. Among them, 467 (99% [83%–100%]) were Advanta V12/iCast. Six studies reported on cohorts where the Advanta V12/iCast was explicitly used,17–20,22,26 whereas in two studies, they represented most of the implanted stents.21,25
Advanta V12/iCast-Related Outcomes
Thirty-Day Outcomes
Results on 30-day outcomes were extractable in nine studies.17–20,22–26 The estimated cumulative technical success was 98% (95% CI = 93%–100%; I2 = 0%), with very low certainty according to GRADE (Figure 2). As technical success was the only outcome of interest, for which all ten studies were included in the synthesis, a funnel plot was created, and Egger’s test was calculated (Supplementary Figure 2). A considerable publication bias was present.
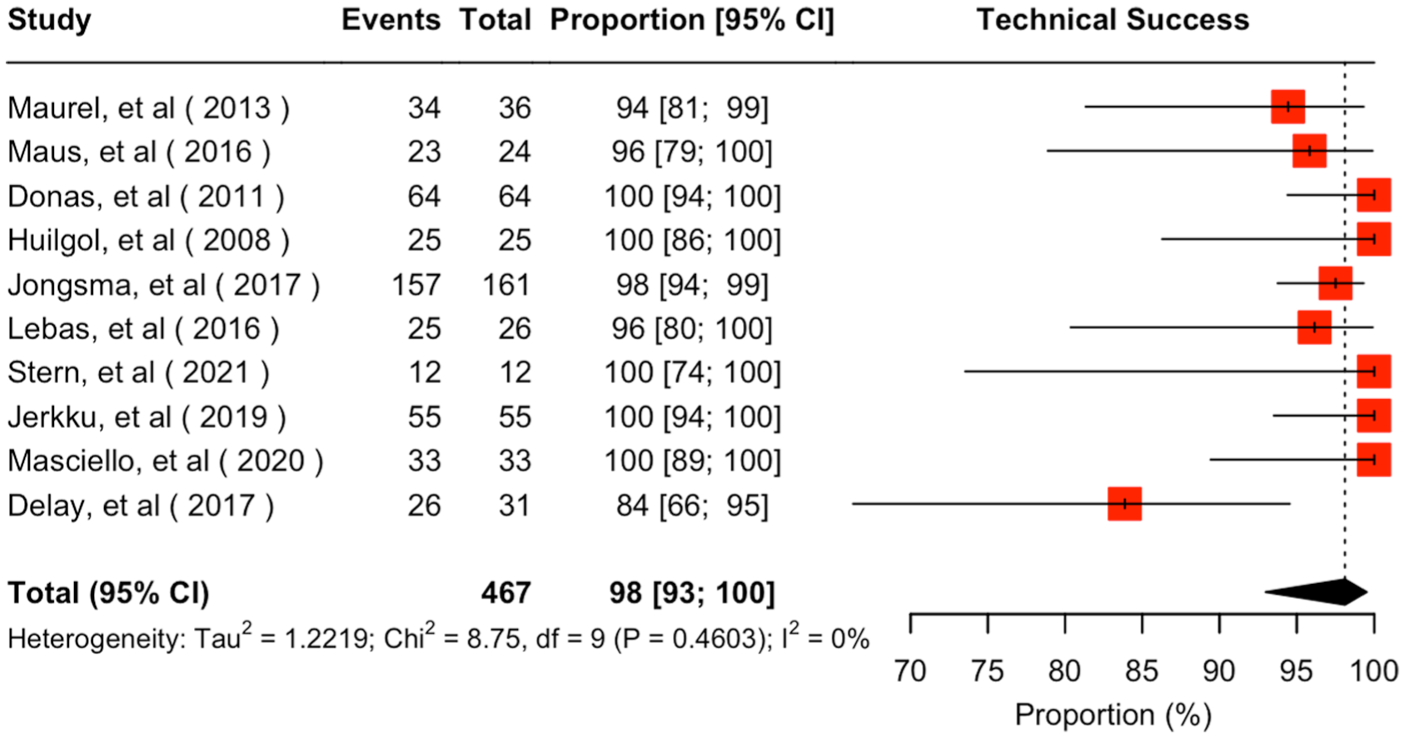
Forest plot for technical success of Advanta V12/iCast when used as a bridging stent in iliac branch devices. CI, confidence interval.
The estimated occlusion/stenosis rate at 30 days was 2% (95% CI = 0%–9%; I2 = 0%; Supplementary Figure 3). Three out of nine patients suffering from IIA occlusion presented buttock claudication and one presented both buttock claudication and stage I colic ischemia.17,19,22 The remaining five occlusion cases were associated to intraoperative IIA thrombosis, for which no revascularization was attempted. 26 Among these patients, one presented with colic ischemia and required hemicolectomy. 26
The estimated endoleak Ic/IIIc rate at 30 days was 0.3% (95% CI = 0%–3%; I2 = 0%; Supplementary Figure 4). There was only one case of endoleak Ic reported in Maus et al 18 that required a reintervention, where an additional Advanta V12/iCast balloon-expandable covered stent was placed. This endoleak Ic reintervention, along with an IBD occlusion, reported in Donas et al, 19 which was managed with revascularization and relining, led to an estimated 30-day reintervention rate of 1% (95% CI = 0%–3%; I2 = 0%; Supplementary Figure 5).
Follow-up Outcomes
Eight studies provided extractable results for a mean follow-up of 23.4 (95% CI = 14.2–32.7) months.18–23,25,26 The estimated occlusion/stenosis rate was 4% (95% CI = 1%–11%; I2 = 19%) with very low certainty according to GRADE (Figure 3). Endoleak Ic/IIIc overall estimated rate was 5% (95% CI = 3%–8%; I2 = 0%; Figure 4), demonstrating very low certainty according to GRADE. Twenty reinterventions were performed during follow-up, deriving an overall estimated rate of reintervention at 5% (95% CI = 3%–9%; I2 = 0) with very low certainty according to GRADE (Figure 5). Reinterventions were performed mainly due to type Ic (12 cases)18–22 and type IIIc endoleaks (6 cases).19,21,22,25 Two reinterventions were performed due to an IBD occlusion.18,19
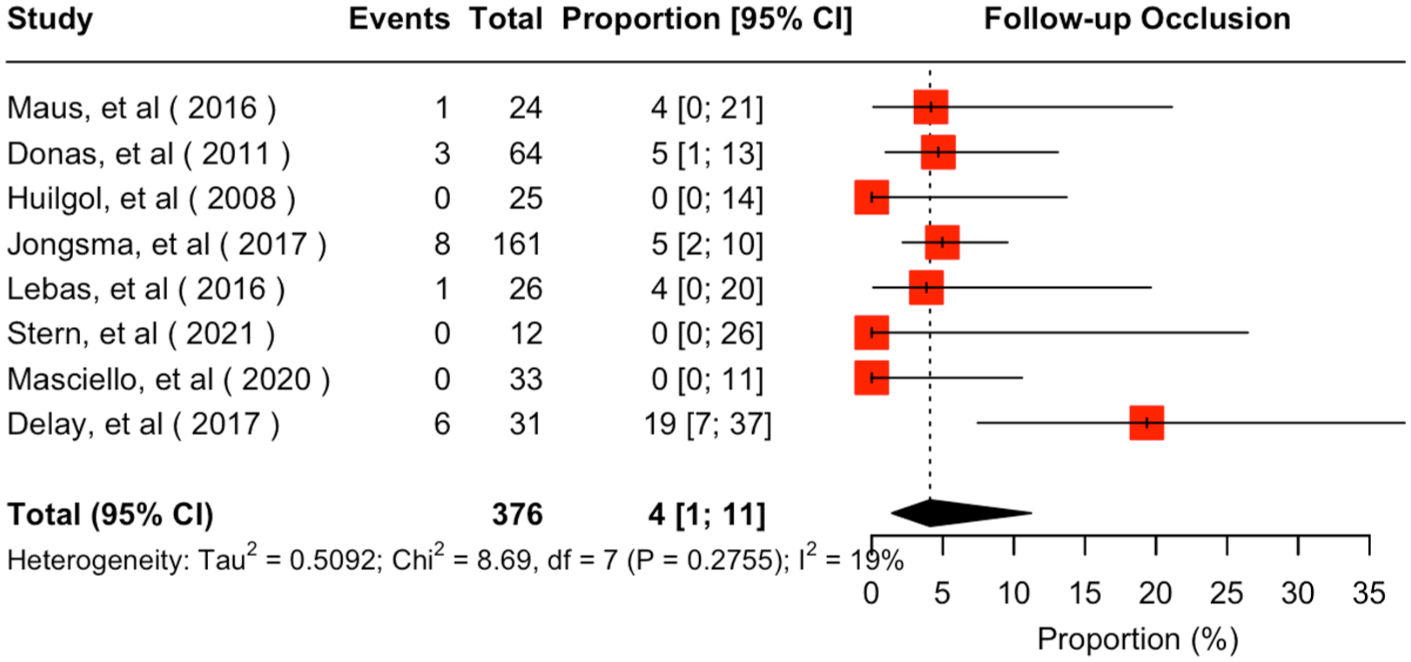
Forest plot for follow-up occlusion rate of Advanta V12/iCast when used as a bridging stent in iliac branch devices. CI, confidence interval.
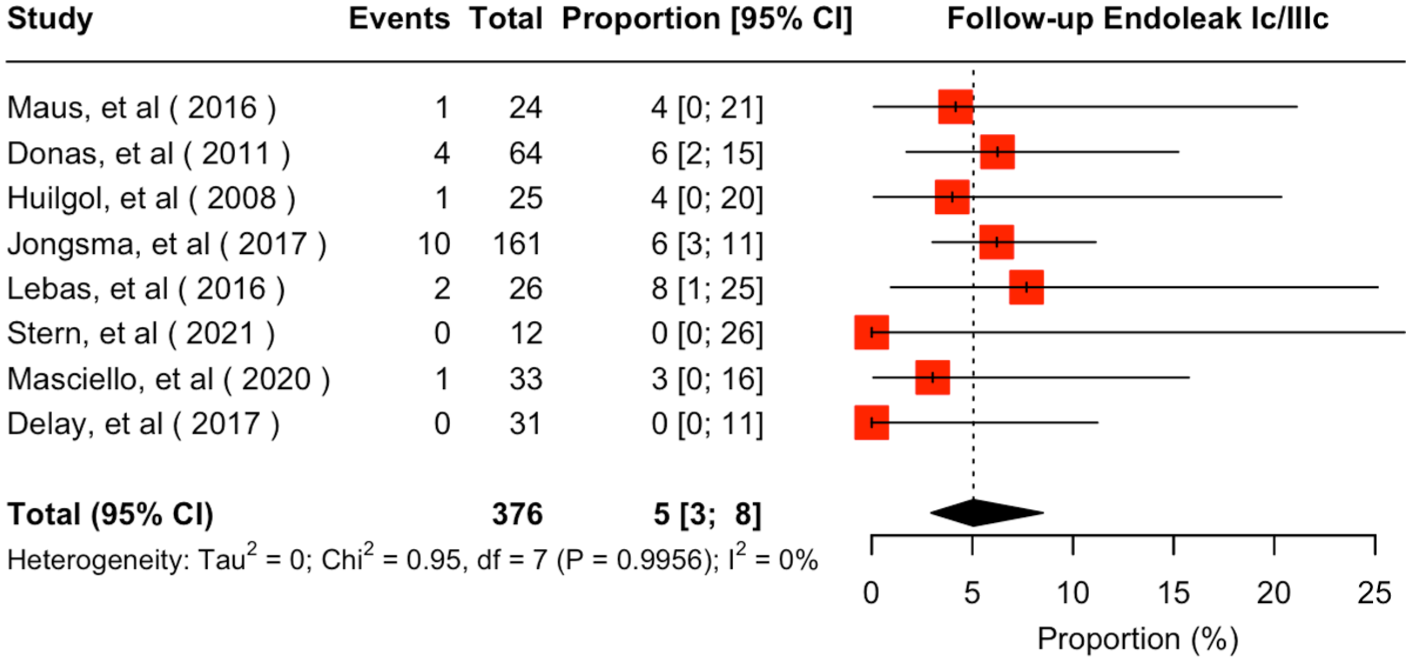
Forest plot for follow-up endoleak Ic/IIIc rate of Advanta V12/iCast when used as a bridging stent in iliac branch devices. CI, confidence interval.
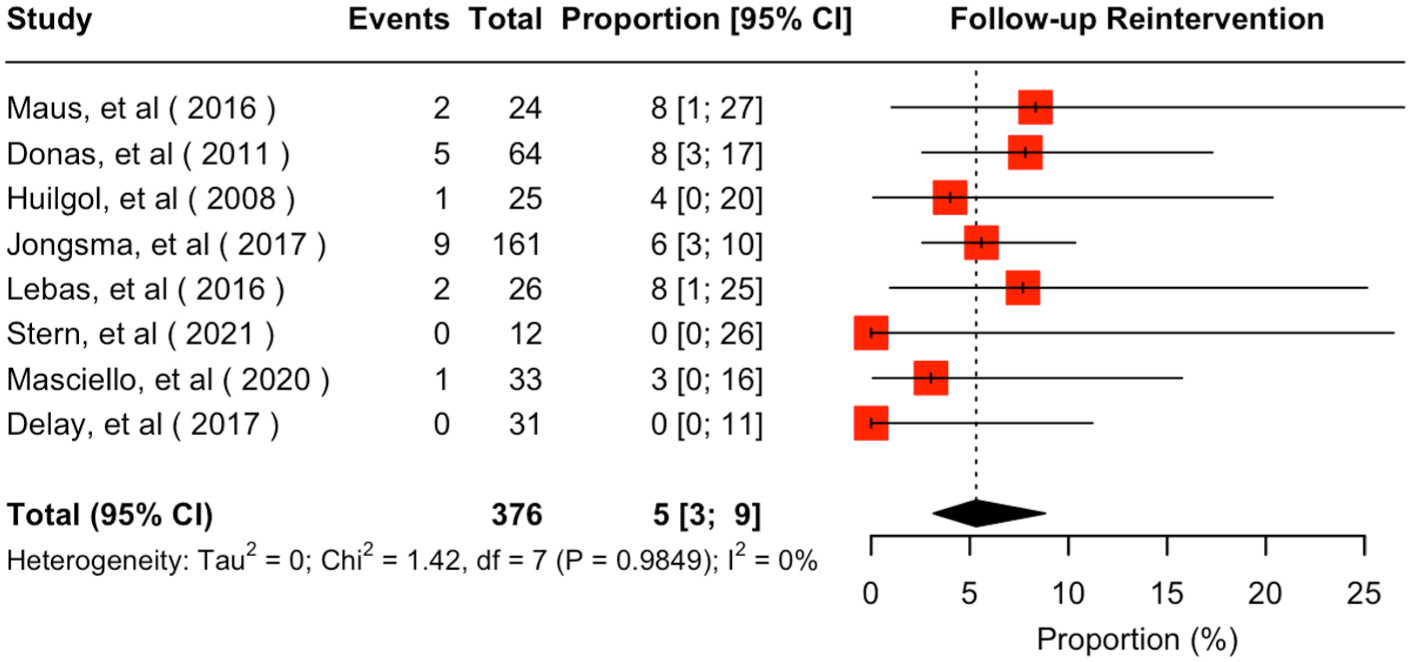
Forest plot for follow-up reintervention rate of Advanta V12/iCast when used as a bridging stent in iliac branch devices. CI, confidence interval.
Discussion
Iliac branch devices maintain the perfusion of the IIA and its main branches to prevent clinical consequences related to its occlusion.7,30 The Advanta V12/iCast balloon-expandable covered stent has been proven to be highly efficient, when used in complex aortic repair, including both branches and fenestrations. 31 This effectiveness is also replicated in IBD procedures, demonstrating low incidence of adverse events.19,21 This systematic review and meta-analysis revealed a high technical success rate of 98%, along with low rate of 30-day adverse outcomes. Follow-up outcomes showed an overall low bridging stent-related occlusion/stenosis and endoleak Ic/IIIc rate (of 4% and 5%, respectively), with a low proportion of 5% requiring any reintervention.
Internal iliac artery preservation during endovascular procedures represents common practice as it decreases the risk of adverse events, including buttock claudication, sexual dysfunction, and colic or spinal cord ischemia.7,30 Both the Society of Vascular Surgery (SVS) and the European Society for Vascular Surgery (ESVS) recommendations support the use of IBDs as a first-line management option during endovascular aortoiliac aneurysm repair.32,33 However, none of the available guidelines addresses the question of bridging stent selection and its potential impact on the durability of the repair. Besides from the Internal Branch Endoprosthesis (IBE; W. L. Gore & Associates, Flagstaff, Arizona), which is combined with a specific internal iliac limb component, information on bridging stent selection and their specific characteristics is provided neither by industry nor from vascular societies. Previous studies, that attempted comparisons of self-expanding versus balloon-expandable covered stents in IBDs, presented controversial findings and failed to prove superiority of the one choice over the other.10,34 However, they highlight the fact that balloon-expandable covered stents represent the first choice in most cases, with the Advanta V12/iCast being used as bridging stent in approximately 70% of them.10,34,35
The overall estimated technical success was 98% (95% CI = 93%–100%), being placed among the highest levels of previously reported experience regarding IBDs, ranging between 93% and 98%.35–37 The low technical failure rate may be associated with the tractability, low profile, and minimal unmounting rates of the Advanta V12/iCast during operative maneuvers, even in unfriendly IIA anatomies, such as in case of ostial stenoses, high tortuosity, and steep IIA orificial angles. 10 The Advanta V12/iCast’s low profile seems to permit advancement and accurate deployment into the target vessel using small sheaths while the low incidence of 30-day adverse events, including 2% of occlusion/stenosis and 1% reintervention rate seems to confirm that the Advanta V12/iCast covered stent may be a safe solution during complex endovascular procedures.31,35,36
Follow-up patency of the IIA bridged with the Advanta V12/iCast covered stent remained high, with the occlusion/stenosis rate being estimated at 4% at 24 months leading to a very high patency rate in IBD cohorts, especially when compared with the previously published literature.35–37 In case of an IIA bridging stent occlusion or stenosis, a reintervention is mainly indicated when symptoms are present and the benefit of operation compensates the associated risks.7,30,38 Accurate positioning and increased radial force, along with increased pullout resistance and considerable flexibility, could have been characteristics that led to low incidence of adverse events, including a low occlusion and reintervention rate during follow-up.10,39 The high conformity and adaptiveness of the Advanta V12/iCast balloon-expandable covered stent have led to its implementation in stent-combining techniques, applying more than one balloon-expandable or both balloon-expandable and self-expanding covered stents to achieve bridging in demanding anatomic conditions, where an extensive coverage was needed between device branch and distal target vessel landing zone.24,40
During follow-up, the endoleak Ic/IIIc incidence remained low, with a cumulative estimated rate of 5%. This rate can be considered a more than acceptable “drawback” introduced by the nature of IBD or IIA occlusion consequences. 41 In fact, the follow-up endoleak rate after IBD has been reported to reach approximately 12%–13% according to previous systematic reviews.35–37 Despite that the vast majority of early target-vessel-related type Ic and IIIc endoleaks have been proposed to resolve spontaneously up to 30 days after the procedure, persistent or new type Ic or IIIc endoleak during follow-up seems to be the leading etiology for IBD reinterventions.4,42,43 This fact is, also, confirmed in the current cohort in which 18 out of 20 IIA-related reinterventions were performed due to either a Ic or IIIc endoleak during follow-up, with only one of them being persistent, whereas the remaining were newly observed endoleaks.21,35,36
Most patients included in this meta-analysis were treated with the ZBIS device bridged with Advanta V12/iCast balloon-expandable covered stent. Results are therefore highly representative of this combined use and support previous evidence that suggest a favorable synergy.11,35 An alternative device implanted in our cohort was IBE. The combination of IBE with other bridging stents may be viable, with the existing evidence though suggesting inferiority compared with the dedicated solution. 44 Completing the triad of off-the shelf available devices, the E-liac Stent Graft System (JOTEC GmbH, Hechingen, Germany) has, also, been combined with the Advanta V12/iCast in the available literature, proving adequacy, but not being sufficient to reach further conclusions. 45 Until a dedicated solution for bridging the IIA exists, the Advanta V12/iCast seems to be a reliable solution for IIA preservation regardless the applied IBD.
Limitations
Results should be interpreted cautiously due to the retrospective observational nature of included primary studies. Risk of bias assessment led to an assignment of serious bias in 8 out of 10 studies included in this analysis and highlights the gap in the literature and the need of higher quality evidence through prospective or retrospective studies, regarding the optimal choice for the IIA. As there was a prespecified eligibility definition on minimum numbers regarding both sample size and cases treated with the Advanta V12/iCast stents, findings may have been affected by the excluded studies. Due to lacking coherence among the included studies in terms of technical success definition, a definition for internal iliac branch–related technical success was created according to the available reporting standards. 16 Regardless this attempt to address definition variability, possible incoherent reporting between studies cannot be ruled out. Follow-up periods among studies varied considerably leading to possible bias introduction. Bridging stent selection was, mainly, operator-driven and the final decision was subject to objective parameters as anatomic suitability as well as subjective ones like preference, experience, and availability. Concurrent implantation of more than one bridging stent, could result in combination of the Advanta V12/iCast with a different stent in one study; that could, probably, introduce some bias. 24 Jerkku et al 24 reported the combination of more than one stent; however, data from this study were available only for the 30-day outcomes, without any event recorded. In addition, usage of different iliac branch devices; including the IBE and ZBIS, may have introduced bias regarding the main outcomes of the analysis and should be taken into consideration. The number of the included studies in most of the syntheses did not allow for further presentation of funnel plots and publication bias assessment. The low level of between-study heterogeneity, indicated by the low I2 values throughout meta-analysis results should be interpreted conservatively, due to low number of events and studies, as well as the nature of the analysis. 27 Indirectness between the population of interest and the primary studies’ population should be considered as a possible source of bias while lack of data extractability has limited their availability. 46
Conclusion
Advanta V12/iCast balloon-expandable covered stent showed low occlusion/stenosis, endoleak Ic/IIIc and IIA–related reintervention rates both at 30 days and mid-term follow-up, indicating that it is a reliable choice of bridging stent during IBD implantation. However, low quality of evidence should be taken under consideration when interpreting these results.
Supplemental Material
sj-docx-6-jet-10.1177_15266028251408018 – Supplemental material for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices
Supplemental material, sj-docx-6-jet-10.1177_15266028251408018 for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices by George Apostolidis, Konstantinos Dakis, José I. Torrealba, Giuseppe Panuccio, Konstantinos Spanos, Tilo Kölbel and Petroula Nana in Journal of Endovascular Therapy
Supplemental Material
sj-docx-7-jet-10.1177_15266028251408018 – Supplemental material for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices
Supplemental material, sj-docx-7-jet-10.1177_15266028251408018 for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices by George Apostolidis, Konstantinos Dakis, José I. Torrealba, Giuseppe Panuccio, Konstantinos Spanos, Tilo Kölbel and Petroula Nana in Journal of Endovascular Therapy
Supplemental Material
sj-docx-8-jet-10.1177_15266028251408018 – Supplemental material for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices
Supplemental material, sj-docx-8-jet-10.1177_15266028251408018 for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices by George Apostolidis, Konstantinos Dakis, José I. Torrealba, Giuseppe Panuccio, Konstantinos Spanos, Tilo Kölbel and Petroula Nana in Journal of Endovascular Therapy
Supplemental Material
sj-docx-9-jet-10.1177_15266028251408018 – Supplemental material for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices
Supplemental material, sj-docx-9-jet-10.1177_15266028251408018 for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices by George Apostolidis, Konstantinos Dakis, José I. Torrealba, Giuseppe Panuccio, Konstantinos Spanos, Tilo Kölbel and Petroula Nana in Journal of Endovascular Therapy
Supplemental Material
sj-tiff-1-jet-10.1177_15266028251408018 – Supplemental material for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices
Supplemental material, sj-tiff-1-jet-10.1177_15266028251408018 for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices by George Apostolidis, Konstantinos Dakis, José I. Torrealba, Giuseppe Panuccio, Konstantinos Spanos, Tilo Kölbel and Petroula Nana in Journal of Endovascular Therapy
Supplemental Material
sj-tiff-2-jet-10.1177_15266028251408018 – Supplemental material for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices
Supplemental material, sj-tiff-2-jet-10.1177_15266028251408018 for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices by George Apostolidis, Konstantinos Dakis, José I. Torrealba, Giuseppe Panuccio, Konstantinos Spanos, Tilo Kölbel and Petroula Nana in Journal of Endovascular Therapy
Supplemental Material
sj-tiff-3-jet-10.1177_15266028251408018 – Supplemental material for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices
Supplemental material, sj-tiff-3-jet-10.1177_15266028251408018 for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices by George Apostolidis, Konstantinos Dakis, José I. Torrealba, Giuseppe Panuccio, Konstantinos Spanos, Tilo Kölbel and Petroula Nana in Journal of Endovascular Therapy
Supplemental Material
sj-tiff-4-jet-10.1177_15266028251408018 – Supplemental material for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices
Supplemental material, sj-tiff-4-jet-10.1177_15266028251408018 for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices by George Apostolidis, Konstantinos Dakis, José I. Torrealba, Giuseppe Panuccio, Konstantinos Spanos, Tilo Kölbel and Petroula Nana in Journal of Endovascular Therapy
Supplemental Material
sj-tiff-5-jet-10.1177_15266028251408018 – Supplemental material for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices
Supplemental material, sj-tiff-5-jet-10.1177_15266028251408018 for Systematic Review and Metanalysis on the Advanta V12/iCast Outcomes as Bridging Stent in Iliac Branch Devices by George Apostolidis, Konstantinos Dakis, José I. Torrealba, Giuseppe Panuccio, Konstantinos Spanos, Tilo Kölbel and Petroula Nana in Journal of Endovascular Therapy
Footnotes
Acknowledgements
None
ORCID iDs
Ethical Considerations
This study complied with the Declaration of Helsinki and no approval by the ethics committee was required due to study design.
Author Contributions
GA: methodology; data collection; studies evaluation; statistical analysis; writing of the initial manuscript; critical review of the manuscript; KD: data collection; studies evaluation; critical review of the manuscript; JIT: critical review of the manuscript; GP: critical review of the manuscript; KS: critical review of the manuscript; TK: conceptualization; critical review of the manuscript. PN: conceptualization; methodology; writing of the initial manuscript; critical review of the manuscript; supervision.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tilo Kölbel is a consultant for Cook Medical and Getinge, and proctor for and has intellectual property with Cook Medical, receiving royalties, speaking fees, and research, travel, and educational grants. All authors declare no support from any organization for the submitted work; no financial relationships with any organizations that might have an interest in the submitted work; no other relationships or activities that could appear to have influenced the submitted work.
Data Availability Statement
Data can be provided by corresponding author upon request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
