Abstract
Objective:
To compare the in vitro feasibility and fenestration characteristics of 2 in situ fenestration (ISF) techniques—re-entry catheter (REC-ISF) and electrified guidewire (EW-ISF)—in commercially available thoracic stent-grafts.
Methods:
Forty in vitro fenestrations were performed in RelayPro and Valiant endografts using either REC or EW (n=10 per graft per technique). The grafts were completely submerged in a saline bath at 37°C, mimicking physiological temperature. All fenestrations were sequentially balloon dilated with 2 mm and 6 mm plain balloons. Outcomes fenestration geometry and fabric damage, and puncture and dilatation success, defined as success in penetrating the graft and success in advancing the balloons, respectively. The fenestration area was measured immediately after dilatation and after 24 hours to evaluate elastic recoil.
Results:
Initial puncture succeeded in 60% of REC-ISF cases in RelayPro and 70% in Valiant, whereas EW-ISF achieved 100% puncture success across both grafts (p<0.05). In the Valiant graft, balloon dilatation was successful in 9/10 cases with EW-ISF, but in 0/10 cases with REC-ISF (p<0.001), as the 2 mm balloons could not be advanced within the fenestration holes. For both methods, the dilatation was, however, successful in all fenestrations in the RelayPro. In this graft, post-dilatation fenestration areas were similar between techniques (6.0 vs 6.01 mm2; p=0.809), but tearing occurred more frequently with EW-ISF (80% vs 0%, p<0.001). The REC-ISF produced elliptical, weft-aligned fenestrations; EW-ISF yielded variable shapes aligned with the warp. After 24 hours, REC-ISF fenestrations showed greater surface recoil than EW-ISF in RelayPro (23.1% vs 5.0%; p=0.037).
Conclusion:
The EW-ISF showed high technical feasibility across both analyzed grafts, producing consistent, easily reproducible, and dimensionally stable fenestrations. In contrast, REC-ISF demonstrated material-dependent technical limitations under the constraints of this model. These findings support the use of EW-ISF as an off-the-shelf ISF option in emergency settings when other fenestration tools (ie, excimer laser) are unavailable, even if a head-to-head comparison is warranted.
Clinical Impact
This paper presents a proof-of-concept evaluation of two newly described off-the-shelf techniques for in situ fenestration (ISF) in thoracic stent-grafts: electrified guidewire ISF (EW-ISF) and re-entry device ISF (RED-ISF). The findings demonstrate a clear technical advantage of EW-ISF, which achieved higher puncture success, more reproducible fenestration geometry, and greater dimensional stability compared with RED-ISF. As such, EW-ISF emerges as a more reliable off-the-shelf option when dedicated fenestration tools—such as excimer lasers—are unavailable. These results may broaden endovascular treatment possibilities in time-critical or anatomically challenging scenarios, ultimately enhancing procedural success and branch preservation during emergency thoracic aortic interventions.
Keywords
Introduction
In situ fenestration (ISF) of aortic stent-grafts has gained importance in emergency thoracic endovascular aortic repair to preserve flow to the left subclavian artery or other supra-aortic branches. 1 Traditionally, ISF has been performed using laser (L-ISF) or needle-based (N-ISF) techniques. The N-ISF has primarily been studied in vitro using percutaneous transhepatic cholangiography needles, with variable success.2,3
Newer approaches use a dedicated balloon-stabilized needle device. 4 In contrast to needle devices, L-ISF also facilitates an antegrade approach to target vessels and is therefore also usable for visceral vessels. 5 While research on in situ laser fenestrations, which demonstrate favorable mechanical long-term results, 6 is promising, the clinical availability of large excimer laser systems is limited. Therefore, research on alternative in situ fenestration methods is of interest.
Recently, peripheral re-entry catheters (RECs) have emerged as an off-the-shelf alternative for N-ISF, particularly in settings where dedicated, balloon-stabilized needle systems for retrograde access are unavailable.7–9 Most RECs are originally designed for peripheral arterial re-entry and incorporate a terminal curved, retractable needle and a single-lumen design. 10 Despite their potential, the use of RECs for ISF has not been systematically investigated, and no comparative data are available regarding other ISF modalities. A further novel technique, electrified wire ISF (EW-ISF), has recently been described in vitro, 11 with limited early in vivo application.12,13 Both REC-ISF and EW-ISF offer potential advantages, including lower cost and immediate availability compared to L-ISF, although supporting evidence remains limited.
Due to the flexibility of EW-ISF, it may be a method that also enables antegrade in situ fenestration.
The aim of this study was to assess and compare the in vitro feasibility of REC-ISF and EW-ISF, evaluate the characteristics of the resulting fenestrations, and quantify post-dilatation dimensions and elastic recoil.
Materials and Methods
Materials and Puncture Technique
Two polyester thoracic stent-grafts that have been described to work well as platforms for in situ fenestration in the case of the Relay graft and for physician on-bench modification in the case of the Valiant were tested: RelayPro (Terumo Aortic, Sunrise, Florida; multifilament, warp-backed weave) and Valiant (Medtronic, Minneapolis, Minnesota; monofilament 4/4 twill weave). Their fabric characteristics have been previously analyzed with scanning electron microscopy.14,15 Retrograde ISF was carried out with either a GoBack re-entry catheter (Upstream Peripheral Technologies, Caesarea, Israel; marketed as BeBack, Bentley Innomed, Hechingen, Germany) or a 0.018’’ Astato 30 electrified guidewire (Asahi Intecc, Aichi, Japan).
For REC-ISF, the catheter was introduced through a 7 F steerable sheath (Destino Twist, Oscor Inc, Palm Harbor, Florida) and supported by a 0.018’’ guidewire. The sheath tip was aligned perpendicular to the graft surface, and the re-entry needle was advanced through the graft fabric (Figure 1). The guidewire was advanced while retrieving the REC to provide support for further ballooning as in real-life conditions.
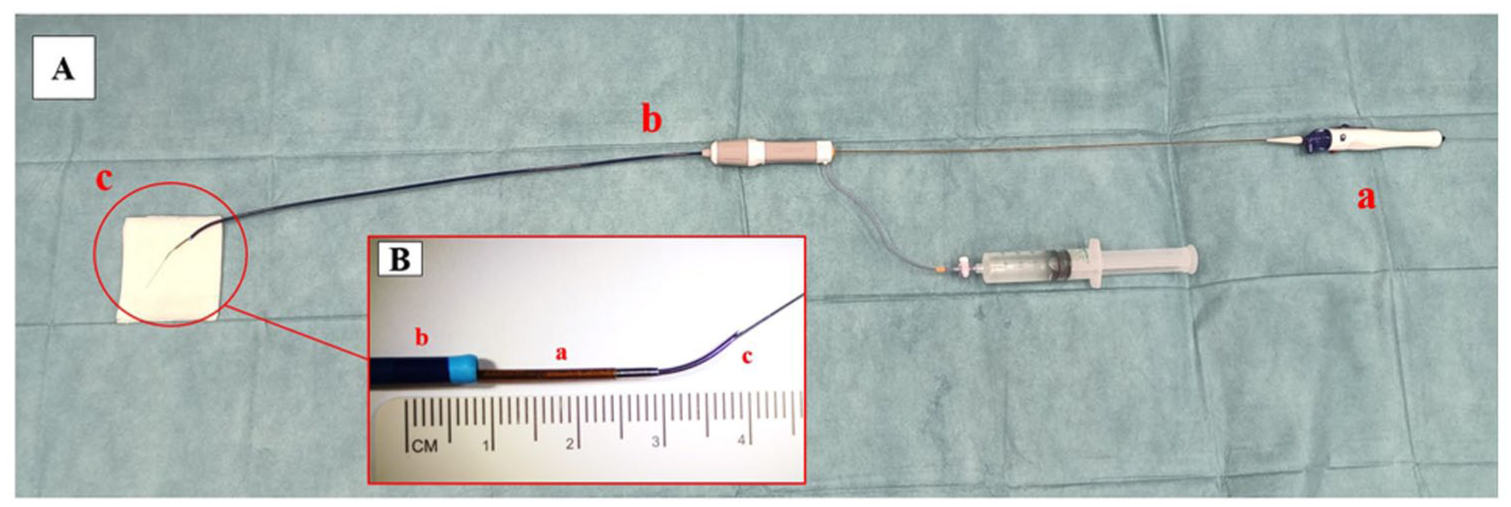
Experimental setting for re-entry catheter in situ fenestration (A): a: re-entry catheter; b: steerable sheath; and c: needle. Particulars of re-entry catheter setting (B): a: re-entry catheter; b: steerable sheath; and c: needle and supporting guidewire.
The EW-ISF was performed according to the same method previously well described in the literature, by electrification of a high-load tip 0.018’’ guidewire (Astato 30). 11
The graft samples were submerged in a saline bath at 37°C in a metal container. The electric circuit was organized with the neutral electrode pad connected to the bottom of the saline bath container. The wire was positioned perpendicularly to the graft surface and supported with a Berenstein catheter (Impress 5F angiographic catheter, Merit Medical Systems, South Jordan, Utah) inside a 7F steerable sheath (Destino Twist, Oscor Inc). The sheath and the catheter were constantly flushed with a 5% glucose solution. The hydrophilic coating was removed from the proximal end of the guidewire via a scalpel blade over approximately 2 cm in length to obtain proper de-insulation. The graft puncture was obtained by gently advancing the wire through the fabric while applying an electric burst to the wire proximal end with a monopolar cautery connected to the electrosurgical unit (VIO 300D monopolar cautery, Erbe Elektromedizin GmbH, Tübingen, Germany). The device was set in “cut mode” at 180 W power (Figure 2).
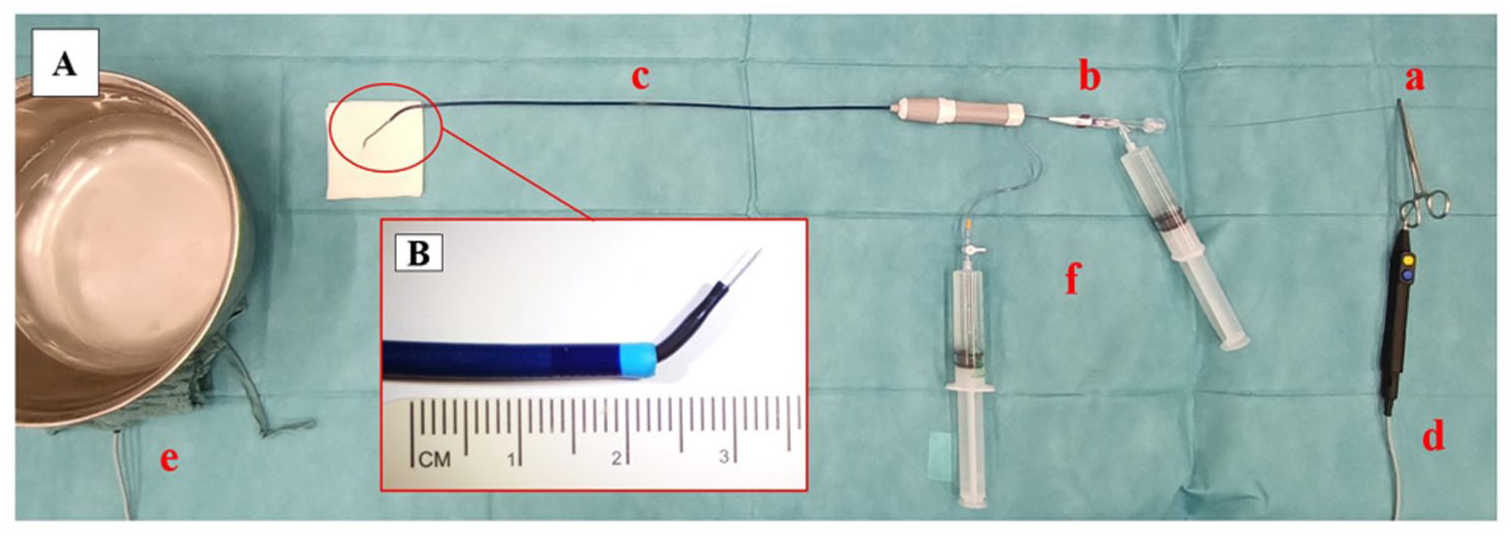
Experimental setting for electrified wire in situ fenestration (A): a: 0.018 guidewire; b: supporting 5F catheter; c: 7F steerable sheath; d: monopolar cautery at the level of de-insulated wire; and e: saline bath container and indifferent electrode. Particulars of electrified wire setting (B): a: 0.018 guidewire; b: supporting 5F catheter; and c: 7F steerable sheath.
For each technique, 10 fenestrations were initially created in each graft (40 in total). A different device (EW or REC) was employed for each graft.
Puncture success was defined as graft traversal on the first attempt without the need for fenestration device repositioning; failures prompted sheath/device repositioning and further attempts.
Ballooning Scheme
After successful perforation, all fenestrations were sequentially balloon dilated with a 2 × 20 mm Sterling plain balloon (PB) (Boston Scientific, Marlborough, Massachusetts) and a 6 mm Admiral Xtreme PB (Medtronic). Each balloon was inflated at nominal pressure for 10 seconds. The ballooning scheme was performed as described in the literature. 11
Balloon passage and inflation were performed by 1 experienced operator; a single rescue attempt was permitted by a second operator if initial dilatation failed. Damage to the balloon tip was promptly addressed by replacing the device before further use.
Dilatation success after puncture was defined when dilatation was achieved either at the first or second attempt. The inability of either of the 2 sequentially used balloons to cross the fenestration hole was rated as a failure.
Fenestration Assessment
Fenestrations were assessed with a Dino-Lite Edge digital microscope (×40 magnification) and Dino-Capture 3.0 software (both IDCP, Glasgow, UK) after puncture, immediately after dilatation, and 24 hours later. Horizontal (weft) and vertical (warp) diameters and surface area were measured; shape and fenestration features (tearing, bulging, shredding, fraying) were recorded (Figure 3). The following definitions were applied:
Tearing: full-thickness rip extending from the fenestration edge.
Bulging: >50% circumferential thickening of the rim.
Shredding: graft shreds partially obstructing the fenestration opening.
Fraying: ≥ 5 damaged fibers encircling the orifice.
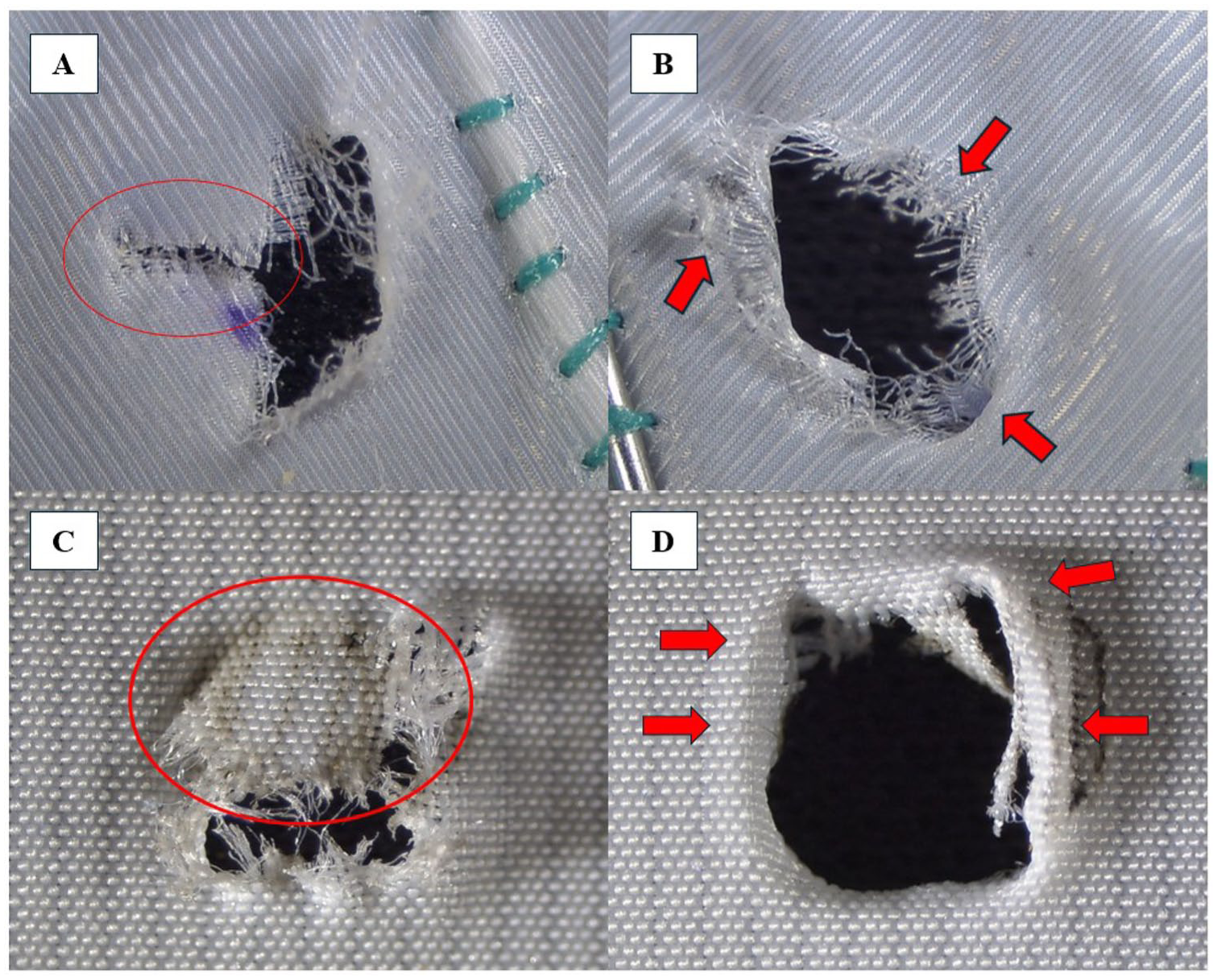
Demonstration of fenestration features after balloon dilation. Tearing after electrified wire in situ fenestration in the Valiant (A), fraying after electrified wire in situ fenestration in the Valiant (B), shredding after electrified wire in situ fenestration in the RelayPro (C), bulging after electrified wire in situ fenestration in the RelayPro (D). Red ovals and arrows mark the specific features.
Fabric recoil was calculated as the absolute and relative change in fenestration diameters and area between the immediate and 24 hour measurements.
Statistical Analysis
The normality of the continuous variables was assessed using the Shapiro-Wilk test. Continuous variables were presented as median and interquartile range, whereas categorical variables were expressed as frequency and percentage. The Mann-Whitney U test and the Fisher’s exact test were applied for comparison of the independent continuous variables and categorical variables, respectively. Inter-observer agreement was calculated using the intraclass correlation coefficient (ICC) for absolute agreement and expressed with a 95% confidence interval (CI). A p-value<0.05 was considered significant. All statistical data analysis was performed with software SPSS Statistics for Windows (IBM release 24, Chicago, Illinois).
Results
Puncture Feasibility and Technical Success
In the RelayPro group, initial graft puncture was successful in 6/10 (60%) cases using the REC-ISF, compared to 10/10 (100%) with EW-ISF (p=0.043). In the Valiant group, the initial puncture succeeded in 7/10 (70%) cases of REC-ISF, while EW-ISF succeeded in all attempts (p=0.211). Dilatation success after puncture was achieved in all EW-ISF procedures. In contrast, REC-ISF resulted in 100% success in RelayPro grafts but failed entirely in Valiant grafts (0/10), despite multiple attempts (p<0.001). The diagram in Figure 4 shows the study setup and flow. Table 1 summarizes puncture success and overall technical outcomes for each technique and graft type.
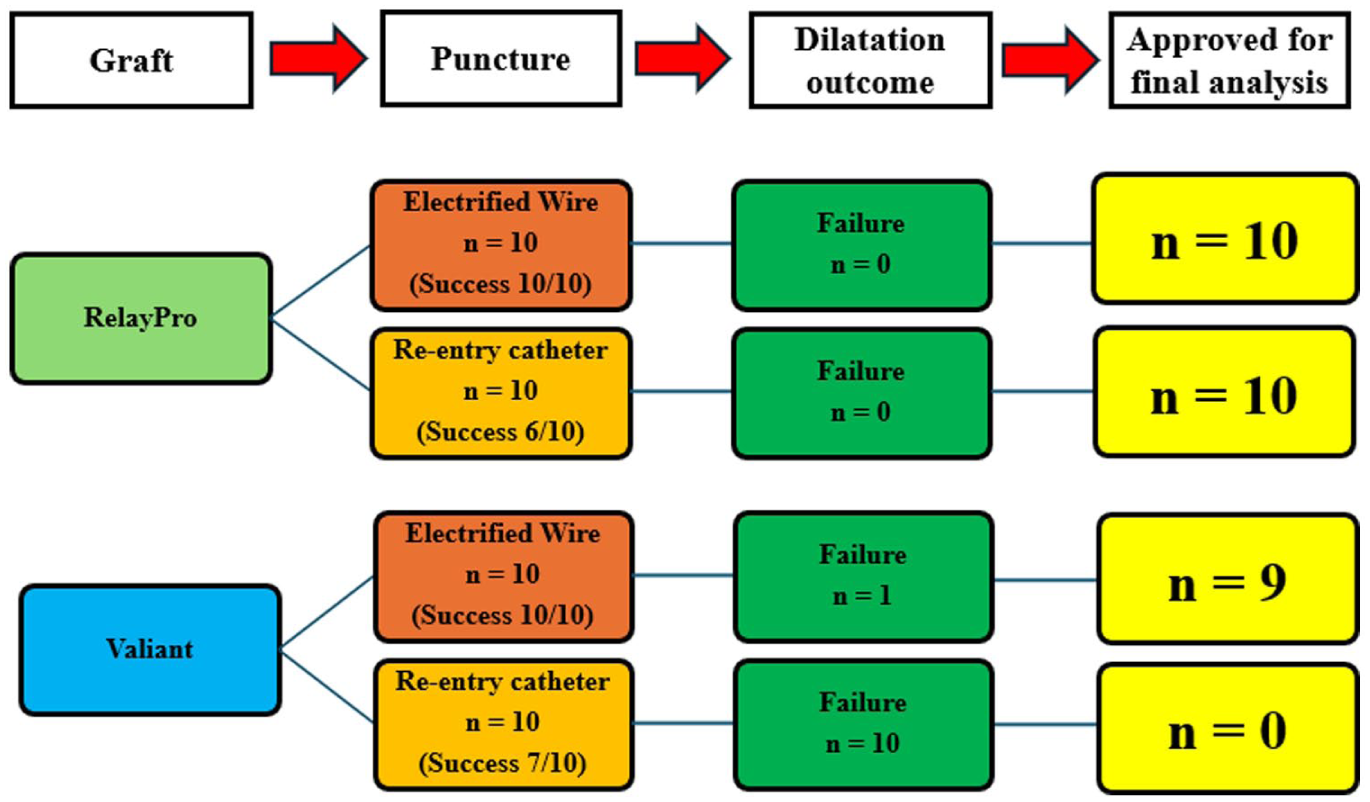
Study flow with outcomes after graft puncture and balloon dilatation.
Fenestration Efficacy and Technical Success.
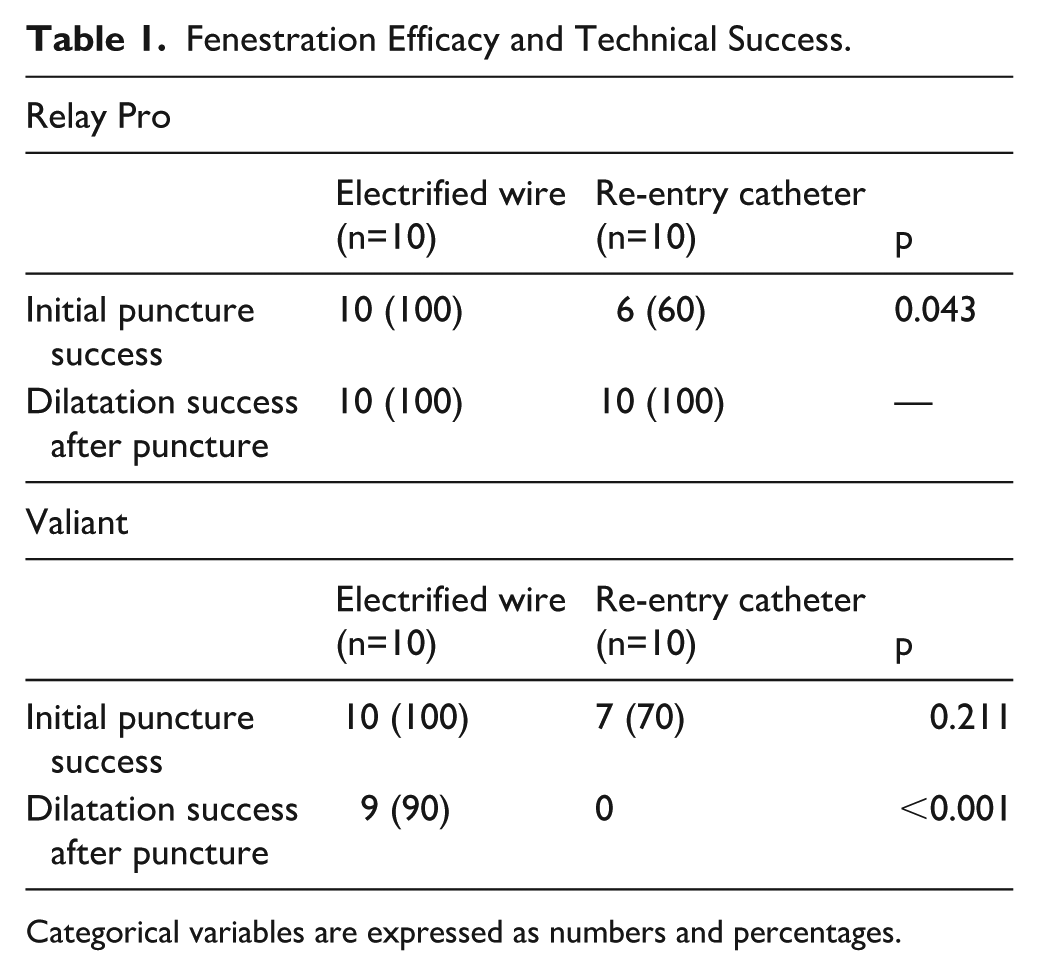
Categorical variables are expressed as numbers and percentages.
Qualitative Assessment of Fenestrations
After REC-ISF, initial fenestrations appeared punctiform with no clearly visible orifice; polyester yarns were laterally displaced without significant fabric disruption. In contrast, EW-ISF consistently produced visible holes, albeit with irregular contours and a surrounding “penumbra” of blackened fabric.
During balloon dilatation, REC-ISF required substantial forward pressure and tension, often deforming the balloon tip (Figure 5), requiring the device substitution. The EW-ISF allowed smoother balloon passage and less mechanical resistance.
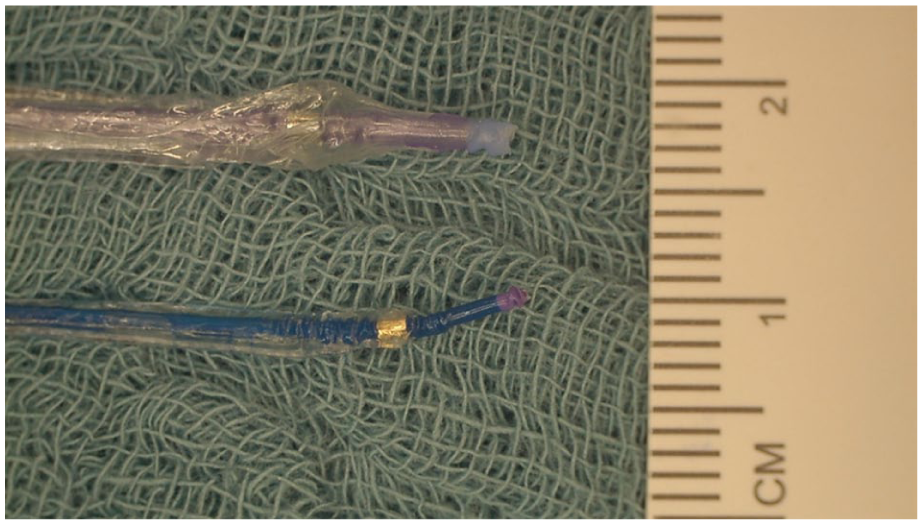
Damage to the tip of the 6 mm (above) and 2 mm plain balloon after 1 attempted dilatation of an in situ fenestration performed with a re-entry catheter.
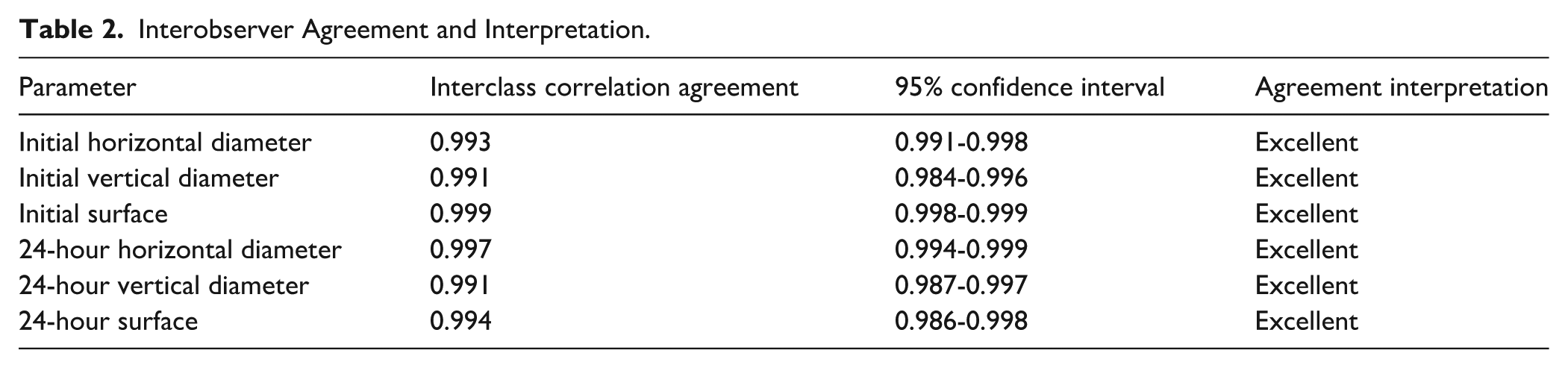
Morphologically, REC-ISF created elliptical fenestrations in 9/10 RelayPro cases, while EW-ISF yielded more variable shapes (p=0.040). Fabric tearing occurred in 8/10 (80%) EW-ISF cases but in none of the REC-ISF fenestrations (p<0.001). Fraying, shredding, and bulging showed no significant differences between groups (Figure 6). Inter-observer agreement was excellent (ICC>0.9) for all measured parameters (Table 2).

Fenestrations after balloon dilatation in the current experimental setting.
Interobserver Agreement and Interpretation.
Geometrical Parameters After Balloon Dilation
Due to failed REC-ISF dilatation in Valiant grafts, geometric comparison was limited to the RelayPro group. Post-dilatation fenestration areas were similar between REC-ISF and EW-ISF (6.00 mm2 vs 6.01 mm2; p=0.809). However, the orientation and dimensions of the fenestrations differed: REC-ISF produced wider horizontal (weft-aligned) diameters and shorter vertical (warp) axes compared to EW-ISF, which showed the opposite pattern (both p<0.001).
When comparing EW-ISF between RelayPro and Valiant grafts, no significant difference in fenestration area or shape distribution was observed. Shredding appeared more frequently in RelayPro, and shape heterogeneity was greater compared to Valiant, where fenestrations were mostly elliptical or round. Detailed measurements are provided in Table 3 (REC-ISF vs EW-ISF in RelayPro) and Table 4 (EW-ISF in RelayPro vs Valiant).
Comparison of Geometrical Parameters After Fenestration of RelayPro Endograft Using Electrified Wire and Re-entry Catheter.
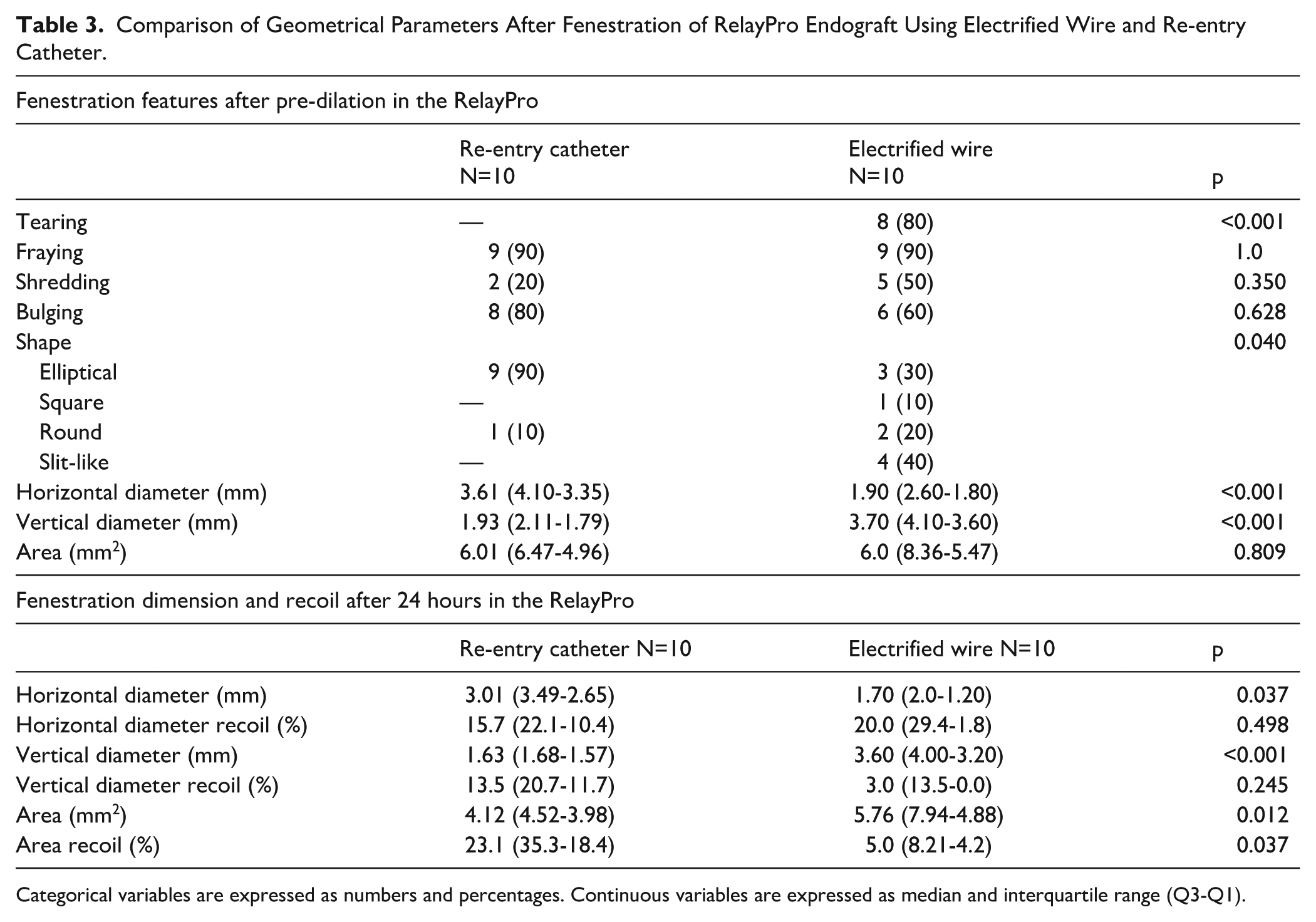
Categorical variables are expressed as numbers and percentages. Continuous variables are expressed as median and interquartile range (Q3-Q1).
Geometrical Parameters After Fenestration of Valiant and RelayPro Endografts Using Electrified Wire.
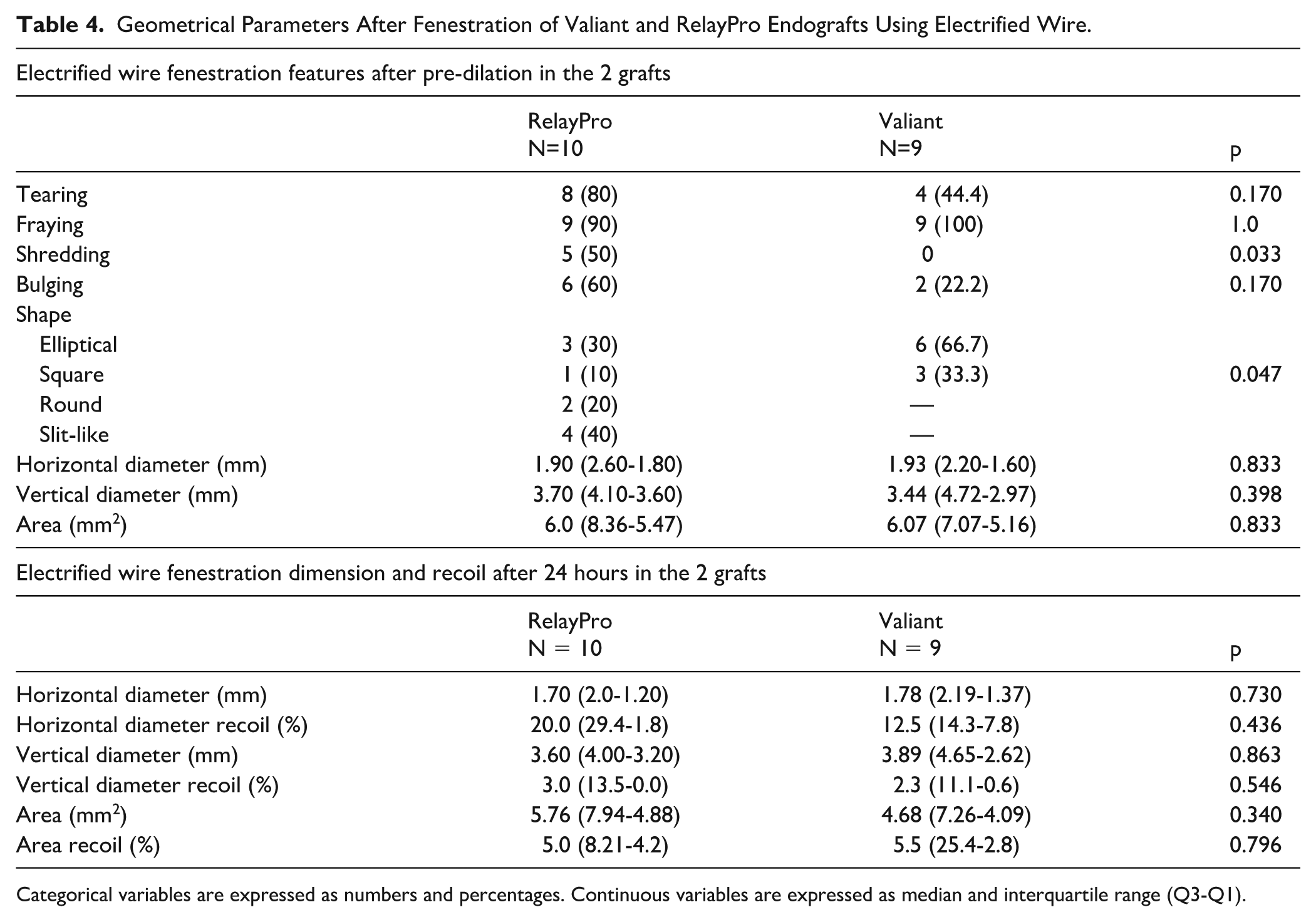
Categorical variables are expressed as numbers and percentages. Continuous variables are expressed as median and interquartile range (Q3-Q1).
Fenestration Recoil at 24 Hours
All fenestrations demonstrated measurable surface and diameter recoil at 24 hours.
In RelayPro grafts, REC-ISF fenestrations showed significantly greater surface recoil compared to EW-ISF (23.1% vs 5.0%; p=0.037). In addition, the fenestration area at 24 hours was significantly smaller after REC-ISF (p=0.012). When comparing EW-ISF between the 2 graft types, recoil magnitude did not differ significantly (RelayPro 5.0% vs Valiant 4.1%). These findings suggest that the REC technique may preserve graft elasticity, while EW-ISF induces more permanent deformation with less recoil.
Discussion
This in vitro study systematically compared 2 ISF techniques—REC-ISF and EW-ISF—in 2 commercially available thoracic stent-grafts. While both methods were technically feasible in the RelayPro device, substantial differences were observed regarding procedural success, fenestration quality, and biomechanical behavior, with important implications for clinical practice.
Feasibility and Procedural Performance
The EW-ISF showed consistent reliability, achieving 100% success in both initial puncture and balloon dilatation. This aligns with previous experimental findings, 11 supporting EW-ISF as a practical off-the-shelf approach for emergency or hybrid ISF procedures.
In contrast, REC-ISF showed notable limitations in feasibility. The re-entry catheter’s curved needle was prone to sliding on the graft surface, and, particularly in the Valiant graft, all dilatation attempts failed despite high axial force and multiple efforts, in contrast with previous N-ISF experiences 3 using stiffer needle systems but in accordance with other experiences with a dedicated balloon-stabilized puncture device, where failed attempts have also been described, depending on angulation and catheter position.
In addition, significant deformation and damage to the dilation balloons were reported. 16 To achieve adequate fenestration sizes for bridging stent-graft deployment, EW and REC fenestrations will require a sequence of ballooning steps, eventually incorporating the use of cutting balloons. These findings are consistent with previous clinical experiences. Specifically, Nana et al 9 highlighted the necessity of sequential dilation using multiple low-profile coronary balloons and cutting balloons to achieve successful REC-ISF and attributed the difficulty in balloon advancement primarily to the angulation of the aortic arch. However, our results suggest that, beyond arch angulation, the puncture hole’s geometry and initial size may be the primary determinants of balloon passage resistance.
Morphology and Fabric Interaction
Qualitatively, REC-ISF produced precise, slit-like fenestrations with minimal visible disruption and no tearing. In contrast, EW-ISF created irregular fenestrations frequently accompanied by a characteristic thermally damaged “penumbra” and a higher incidence of tearing. These differences reflect the distinct underlying mechanisms: mechanical fiber displacement for REC-ISF vs thermal disruption for EW-ISF, echoing earlier comparisons of needle- and laser-based ISF techniques. 17 Interestingly, fenestration orientation varied by technique rather than graft type. The REC-ISF fenestrations consistently aligned horizontally (weft-aligned), whereas EW-ISF fenestrations predominantly aligned vertically (warp-aligned). Fenestration orientation may affect subsequent bridging stent alignment and sealing, 2 although the clinical relevance of this finding remains speculative. Despite morphological differences, post-dilatation fenestration areas did not differ significantly between techniques, emphasizing the importance of balloon sizing in determining final fenestration dimensions.
Elastic Recoil and Stability
One of the most clinically relevant findings of this study was the significant difference in elastic recoil behavior. The REC-ISF fenestrations exhibited substantial area reduction over 24 hours (23.1%), while EW-ISF fenestrations were significantly more dimensionally stable (5.0% in RelayPro, 4.1% in Valiant). These findings suggest that the REC technique may preserve graft elasticity, while EW-ISF induces more permanent deformation with less recoil. The REC-ISF primarily separates fibers without permanent disruption, preserving fabric elasticity. In contrast, EW-ISF—like laser techniques—likely induces thermal denaturation of polyester fibers, thereby reducing their recoil capacity. 17 The clinical implications of this observation are twofold. On one hand, greater recoil may enhance bridging stent fixation by creating a tighter seal, reducing the risk of IIIc endoleaks 18 and embolization 19 ; on the other hand, excessive recoil may compromise stent expansion, potentially increasing the risk of stenosis or bridging stent compression. Future in vivo studies are required to identify the optimal balance between sealing efficacy and stent expansion.
Clinical Implications
Procedurally, EW-ISF emerged as more practical and consistently successful across graft types. Its simplicity and dimensional stability support its use as a reliable off-the-shelf solution for emergent or hybrid thoracic ISF when laser equipment is unavailable. A head-to-head comparison with the standard of care (L-ISF) represents the next necessary step to assess this technique. In such a comparison, the described techniques may have to compete against protocols using a smaller 0.9 mm laser fiber that create fenestrations of a similar size and also need several dilatation steps (Figure 7). Larger 2.0 mm or even 2.3 mm fibers create larger fenestrations without the need for sequential dilatation steps. Also, other common endografts have to be incorporated in such research. Due to missing funding or sponsoring of our study, it could not be expanded.

Visual comparison of available puncture devices and relative in situ fenestration outcomes. A: puncture devices (from left to right): 2.0 mm laser fiber, 0.9 mm laser fiber, re-entry catheter, electrified wire. B: in situ fenestration with 2.0 mm laser. C: in situ fenestration with 0.9 mm laser. D: in situ fenestration with re-entry catheter. E: in situ fenestration with electrified wire.
The REC-ISF, despite its superior visual fenestration quality and limited fabric trauma, demonstrated poor feasibility and required high mechanical effort. In its current form, it may be best reserved for selective retrograde applications—such as left subclavian artery preservation in favorable arch anatomy.
For a comparatively hard-to-traverse fenestration, a stabilizing maneuver may be used in clinical practice for either fenestration technique but was not incorporated in our test setup. After creation of the fenestration and initial wire traversal, a snare catheter can be used to establish a through-and-through (externalized) wire. Subsequently, antegrade advancement of a balloon via femoral access over this through-and-through rail provides markedly improved support and trackability and reduces the risk of stent-graft displacement, as the device is pressed intraluminally against the aortic wall rather than the reverse. 20
Limitations
This study was performed in a controlled in vitro environment and cannot fully simulate in-vivo conditions such as pulsatile flow, arterial wall compliance, or stent-graft radial force. Only 2 stent-graft types were tested; results may not generalize to other fabric characteristics or brands. Recoil and balloon resistance were assessed visually and not mechanically quantified. In addition, no long-term fatigue testing was conducted to evaluate fenestration durability under cyclic stress. The results show poor feasibility of REC-ISF, but only 1 device was tested, and different RECs might produce differences in fenestrations. Similarly, different wires could be tested for same purpose.
Conclusion
The EW-ISF and REC-ISF both achieved successful in vitro fenestration in the RelayPro graft. The EW-ISF was technically easier, reproducible across grafts, and may offer improved dimensional stability for bridging stent placement due to reduced recoil. The REC-ISF, while producing cleaner orifices with less fabric damage, was challenging to execute and failed entirely in the Valiant graft.
Footnotes
Acknowledgements
None mentioned.
Ethical Approval
Not required.
Informed Consent
Not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
