Abstract
Objective:
To report 1-year outcomes of real-world clinical data from patients with de novo or restenotic iliac artery lesions treated with the GORE VIABAHN Endoprosthesis with PROPATEN Bioactive Surface (VSX) device.
Methods:
Devices were implanted to treat de novo atherosclerotic or restenotic iliac artery lesions. The primary endpoint was freedom from device-related serious adverse events (SAEs) at 1 year. Additional endpoints included descriptive analyses and Kaplan-Meier estimates of primary patency, primary-assisted patency, and secondary patency; freedom from major amputation, freedom from target lesion revascularization (fTLR); and changes in Rutherford classification and the Ankle Brachial Index (ABI).
Results:
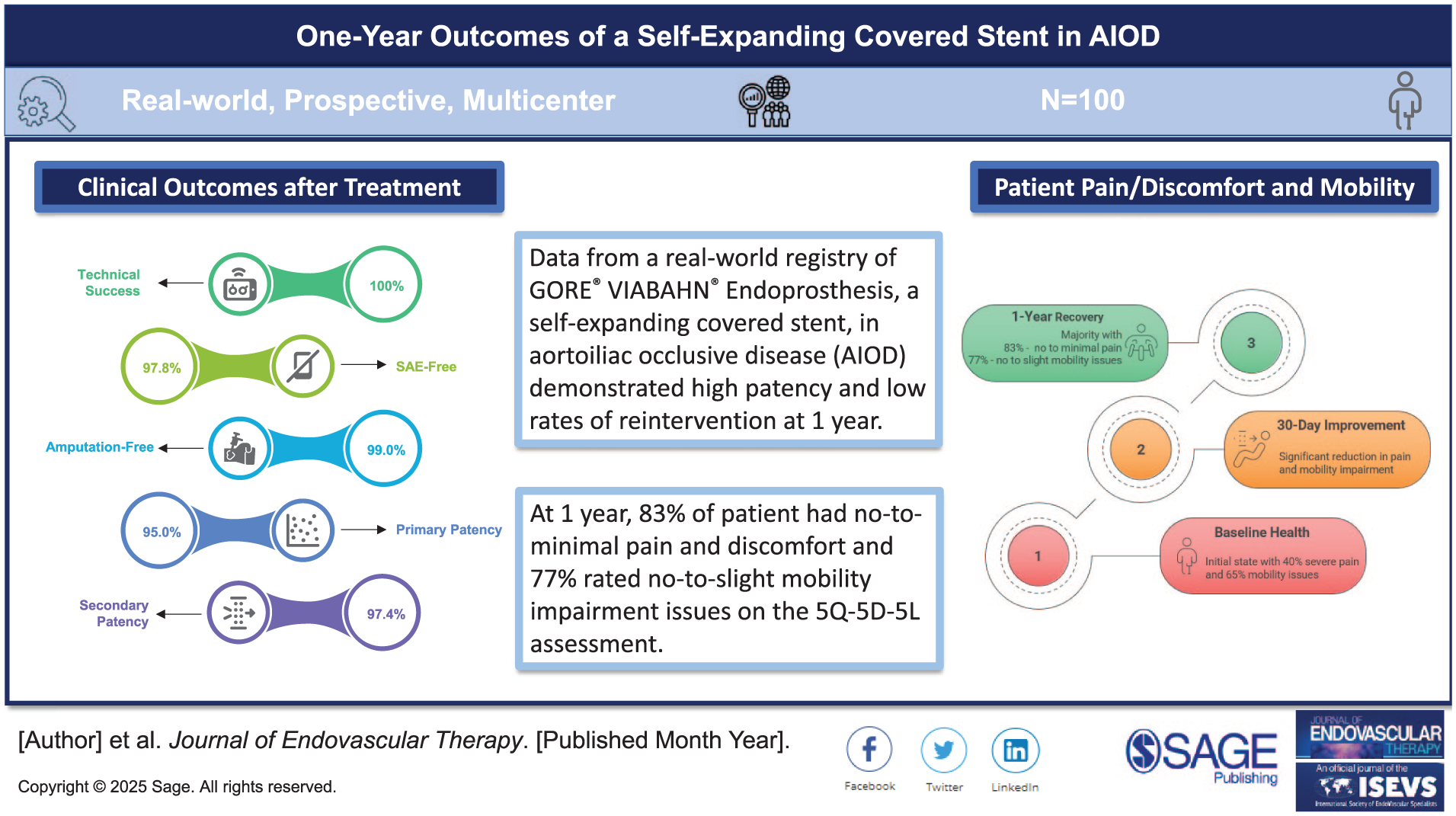
The 100 patients were treated, mean age 66.9 years and 75.0% male, had a mean (±SD) lesion length of 102±60.9 mm and 62.2% were chronic total occlusions; 70.6% were classified as Trans-Atlantic Inter-Society Consensus Document (TASC) C/D, and 20 lesions were across the inguinal ligament. Maximum stenosis was 88.6±17.9%. Technical success was 100%. At 1 year, there were no device-related deaths, 97.8% of patients were free from device-related SAEs, and 99.0% were free from major amputations. Estimates of lesion primary patency, primary-assisted patency, and secondary patency were 95.0% (confidence interval [CI]=87.1, 98.1), 96.3% (CI=89.0, 98.8), and 97.4% (CI=90.1, 99.4), respectively; fTLR was 96.2% (CI=90.1, 98.5). Patients with restenotic lesions and patients <60 years old had lower primary patency, primary-assisted patency, and fTLR rates. Change from baseline to 1 year in the mean ABI was 0.56±0.17 to 0.88±0.15. The rate of asymptomatic patients (Rutherford 0) was 1.0% at baseline and 63.2% at 1 year; patients with intermittent claudication decreased from 52.0% to 35.1%, and critical limb-threatening ischemia from 37.0% to 1.8%.
Conclusions:
The European registry 1-year outcomes demonstrated the safety and effectiveness of the VSX device in the treatment of iliac lesions evidenced by high patency, no device-related deaths, and low numbers of major amputation and reintervention procedures.
Clinical Impact
The European registry reflecting a ‘real-world’ setting of patients treated with the GORE® VIABAHN® Endoprosthesis with PROPATEN Bioactive Surface for aortoiliac occlusive disease demonstrated that the self-expanding covered stent was a safe and effective treatment option. At 1 year, 83% of patients had no-to-minimal pain and discomfort and 77% of patients rated no-to-slight mobility impairment issues (5Q-5D-5L assessment). There were no device-related deaths, only 1 amputation, and few reintervention procedures. This study revealed that patients younger than 60 years old or those treated for restenosis had greater risk for target lesion revascularization, device- or limb-related serious adverse events.
Introduction
Endovascular approach is recommended by the Society for Vascular Surgery (SVS) and European Society for Vascular Surgery (ESVS) as the first-line revascularization therapy for most patients with aortoiliac occlusive disease (AIOD).1,2 Primary stenting, preferably with self-expanding stents, is recommended in all lesions; in Trans-Atlantic Inter-Society Consensus Document II (TASC) C or D, covered stents are advised due to higher patency rates.2,3 The SVS guidelines recommend covered stents in instances of severe calcification at risk of vessel rupture. 1 In patients with chronic limb-threatening ischemia (CLTI) due to moderate or severe AIOD, the endovascular-first approach is supported by both the SVS and the ESVS1,4,5 depending on the patient’s history of prior interventions. Treatment algorithms are further complicated by disease complexity, severity of symptoms, lesion size, or complexity.2,4,5
Both bare metal and covered stents have been demonstrated to successfully treat AIOD.6–8 Studies of covered stents have reported better long-term patency, particularly in longer, heavily calcified iliac lesions.2,9,10 Balloon-expandable-covered stents were reported to have higher patency rates in patients with TASC II C/D lesions,2,9 although another failed to confirm this observation. 11 The various guidelines do not provide strong recommendations on stents1,4,5 selection in the common and external iliac arteries. The ESVS guidelines 2 suggest that self-expanding stents may be preferable, based on the lower risk of restenosis and target lesion revascularization compared to balloon-expandable stents. 12 The role of self-expanding covered stents in the treatment algorithm of AIOD is greatly unknown.
This registry aimed to collect real-world clinical data for up to 5 years in multiple locations in Europe, in patients who were treated with the GORE VIABAHN Endoprosthesis with PROPATEN Bioactive Surface (hereafter the VSX device; W. L. Gore & Associates, Flagstaff, Arizona). The Heparin Bioactive Surface on the VSX device provides thromboresistance. The registry recruited patients into different cohorts, depending on the anatomical location and indication. The aim of this article is to report the 1-year outcomes of patients with de novo or restenotic lesions in the iliac arteries.
Methods
Study Design
The observational, prospective, single-arm, multicenter, post-market clinical follow-up registry captured real-world use of the VSX device in patients with de novo or restenotic lesions in the iliac arteries. The VSX device is commercially available and is indicated for the treatment of de novo or restenotic lesions found in iliac arteries.
The study protocol was reviewed and approved by the institutional review board or ethics committee at each study site in compliance with applicable country laws and regulations. Patients provided written informed consent before undergoing any study procedures and were considered enrolled at the time of consent with the exception of emergency procedure, in which consent was obtained afterwards. The study was conducted according to the International Conference on Harmonization Good Clinical Practice, the Declaration of Helsinki guidelines, and in compliance with ISO 14155 (International Organization for Standardization). The study is registered with ClinicalTrials.gov (NCT04907240) and was sponsored by W. L. Gore & Associates.
The complete eligibility criteria of patients with de novo or restenotic lesions in the iliac arteries are shown in Supplemental Table 1. The inclusion criteria included patients 18 years or older with de novo or restenotic lesions in the iliac arteries who signed informed consent, were suitable for endovascular treatment, and willing to adhere to local follow-up requirements. Exclusion criteria included but were not limited to: lesions where full expansion of an angioplasty balloon catheter was not achievable during pre-dilatation or was insufficient to allow passage of the delivery system, lesions involving a major side branch that may be covered by the endoprosthesis, lesions not suitable for treatment with the available VSX device sizes, lesions that required treatment with an altered endoprosthesis or delivery system, hypersensitivity to or unable to tolerate heparin or antiplatelet therapy, pregnant or breast-feeding female at time of informed consent signature, and life expectancy <12 months due to comorbidities. Only patients who met all of the inclusion criteria and none of the exclusion criteria were included. No population vulnerable to undue influence or increased risk due to participation was included in this registry. Patients in the iliac cohort were scheduled for follow-up visits at 30 days and annually up to 5 years.
Endpoints and Measures
The primary endpoint of the registry was freedom from VSX device-related serious adverse events (SAEs) through 12 months. Additional endpoints included primary patency, freedom from major amputation (above the level of the ankle of the treated limb), freedom from target lesion revascularization (fTLR), secondary patency, survival, and device-related thrombosis. Primary patency was defined as blood flow through the target lesion without the need for repeat surgical or endovascular procedures. Primary-assisted patency was defined as blood flow through the original treated lesion with the use of an additional or secondary surgical or endovascular procedure prior to occlusion. Secondary patency was defined as blood flow through the original treated lesion regardless of the use of an additional or secondary surgical or endovascular procedures after occlusion. The manner of patency assessment imaging was left to the individual investigator’s standard practice/discretion. Freedom from TLR was defined as no surgical or percutaneous revascularization procedure of the original treated lesion. Endpoint definitions are provided in Supplemental Table 1. Changes from baseline were assessed for the Rutherford classification, Ankle Brachial Index (ABI), and in the 5-level EQ-5D version (EQ-5D-5L) Mobility and Pain and Discomfort domains.
Statistical Method
The analyses of the primary endpoint and secondary endpoints were descriptive in nature. No formal hypotheses were tested. A patient-based binomial proportion was summarized as a percent with a 95% confidence interval (CI) along with a separate Kaplan-Meier (KM) analysis for the primary endpoint, freedom from VSX stent graft-related SAE at 12 months. Secondary endpoints were summarized as proportions or percents and analyzed using time-to-event methods (eg, KM). All safety data were summarized using the intent-to-treat population. The denominator reported reflects the number of patients for which data are available for a specific data point.
Subgroup analyses were performed on the following variables: disease classification (occlusion vs stenosis), treatment course (de novo vs restenotic), implant location (external iliac artery [EIA] vs common iliac artery [CIA]/CIA+EIA), sex at birth (female vs male), age (<60 vs ≥60 years old), or single vs dual antithrombotic (excludes heparin, excludes patients with no antithrombotic regimen at discharge and patients with ≥3 antithrombotic medications). Kaplan-Meier curves were created for each subgroup and compared via the log-rank test to identify statistically significant differences. Due to the exploratory nature of the registry, significance was set at the alpha=0.05 level, and no adjustments were made for multiple comparison tests. All analysis was conducted in SAS v9.4 (SAS Institute Inc, Cary, North Carolina).
Results
Patient Characteristics
In the iliac cohort of the global registry, 100 patients were enrolled and treated between October 2021 and June 2023 across 19 sites in Europe (Belgium, France, Germany, Greece, Italy, the Netherlands, Spain, Sweden, and the United Kingdom). The demographics and lesion characteristics of patients in the iliac cohort are described in Table 1. Patients had a mean age of 66.9 years old, 75.0% were male at birth, and 41.0% were current smokers. The total of 112 lesions had a mean length of 102.0 mm, 78.6% were de novo, 62.2% were chronic total occlusions, and the maximum percent diameter stenosis was 88.6%. Chronic limb-threatening ischemia was present in 37.0% of the patients, and 70.6% of the lesions were classified as TASC II C/D.
Patient Demographics, Medical History, and Lesion Characteristics.

The denominator reflects the number of patients for which data are available for a specific variable or outcome.
Abbreviations: TASC II = Trans-Atlantic Inter-Society Consensus Document on Management of Peripheral Arterial Disease II; VSX = GORE VIABAHN Endoprosthesis.
Ten patients did not have an evaluation.
Procedural Outcomes and Disposition
In 100% of patients, target vessel access was successfully obtained, and the device was successfully implanted. Procedural information is provided in Table 1. The mean (±SD) duration of the procedure was 144.9±89.2 minutes. The median blood loss was 50 mL (range=0, 1100). Five patients required a blood transfusion; all 5 had additional procedures performed at the time of device implantation. A total of 134 devices were implanted. The majority of placements were in the EIA only (73; 67.6%); 20 (18.5%) were in both the CIA and EIA. Of the 20 CIA+EIA-treated lesions, only 1 case was a TASC IIA. The 1 case involving a TASC II A was treated in the target limb only; lesion length was 54 mm with 60% diameter stenosis. A 10 mm × 10 cm VSX and a GORE VIABAHN VBX Balloon-Expandable Endoprosthesis (11 mm × 3.9 cm) were used. In addition, a common femoral artery endarterectomy was performed. The mean diameter and length of the device were 7.6±0.9 mm and 9.0±3.5 cm, respectively. The additional placement of a non-VSX stent in the target vessel was required for 38 patients. Access was percutaneous in 58.0% of patients. Pre-dilation before stent deployment was performed in 45.0% of patients. Of the 55 cases that were not pre-dilated, 23 were chronic total occlusion, and the remaining 32 were stenotic. The VSX device configurations included 12 bilateral placements, of which 9 were (in combination with) kissing stents or covered endovascular reconstruction of the aortic bifurcation (CERAB) configurations, and 20 VSX devices were implanted across the inguinal ligament. The VSX device was used as a bailout in 31/98 (31.6%) patients, of which 14 of these patients required the addition of a non-VSX stent during the procedure. Of note, 1 patient entered the trial as asymptomatic (Rutherford=0). The originally planned procedure was an abdominal aortic aneurysm. An Endurant IIs stent graft system (Medtronic, Minneapolis, Minnesota) was implanted, and the VSX device was used as a bailout in the iliac.
At discharge, of patients with data reported, 98.0% (97/99) were administered antiplatelet or anticoagulant therapies (Table 1). All but 1 patient (98/99) was discharged home; discharge disposition was not available for 1 patient.
The 1-month follow-up visit was completed for 93 (95.9%) of the 97 patients eligible for a 30-day evaluation. At 1 year, 90/100 patients were ongoing in the study; 6 patients withdrew consent, 3 died (all deaths were non-lesion-related), and 1 had an open surgery bypass of the VSX device.
Safety Outcomes
Freedom from stent-graft-related SAEs through 1 year was achieved in 97.8% (88/90) of patients (95% CI=92.2, 99.7). Two patients experienced a device-related SAE. The first patient, with Factor V Leiden disorder, had a restenotic lesion. A VSX device across the inguinal ligament and an Advanta V12 device (Advanta V12 balloon-expandable-covered stent; Getinge Maquet, Rastatt, Germany) were implanted. On day 11 post-procedure, the patient had thrombosis of the iliac tract, inclusive of the VSX device and the superficial femoral artery Patency was restored following surgical intervention. The second patient had a de novo lesion; VSX and Absolute Pro (Absolute Pro Vascular Self-Expanding Stent System; Abbott Cardiovascular, Abbott Park, Illinois) devices were implanted. On day 29, this patient had thrombosis of the right common and external iliac arteries that included the VSX device. The thrombosis resolved on day 42, and patency was restored following surgical intervention. Both patients were receiving antithrombotic medications, and neither required amputation.
In the subgroup analyses, freedom from a device-related SAE was significantly lower in patients <60 years old (vs ≥60 years old; p=0.003) and freedom from an SAE on a study limb was significantly lower in patients with restenotic lesions versus de novo (p=0.043; Table 2).
Subgroup Analyses for Primary and Secondary Endpoints by Lesion Level.
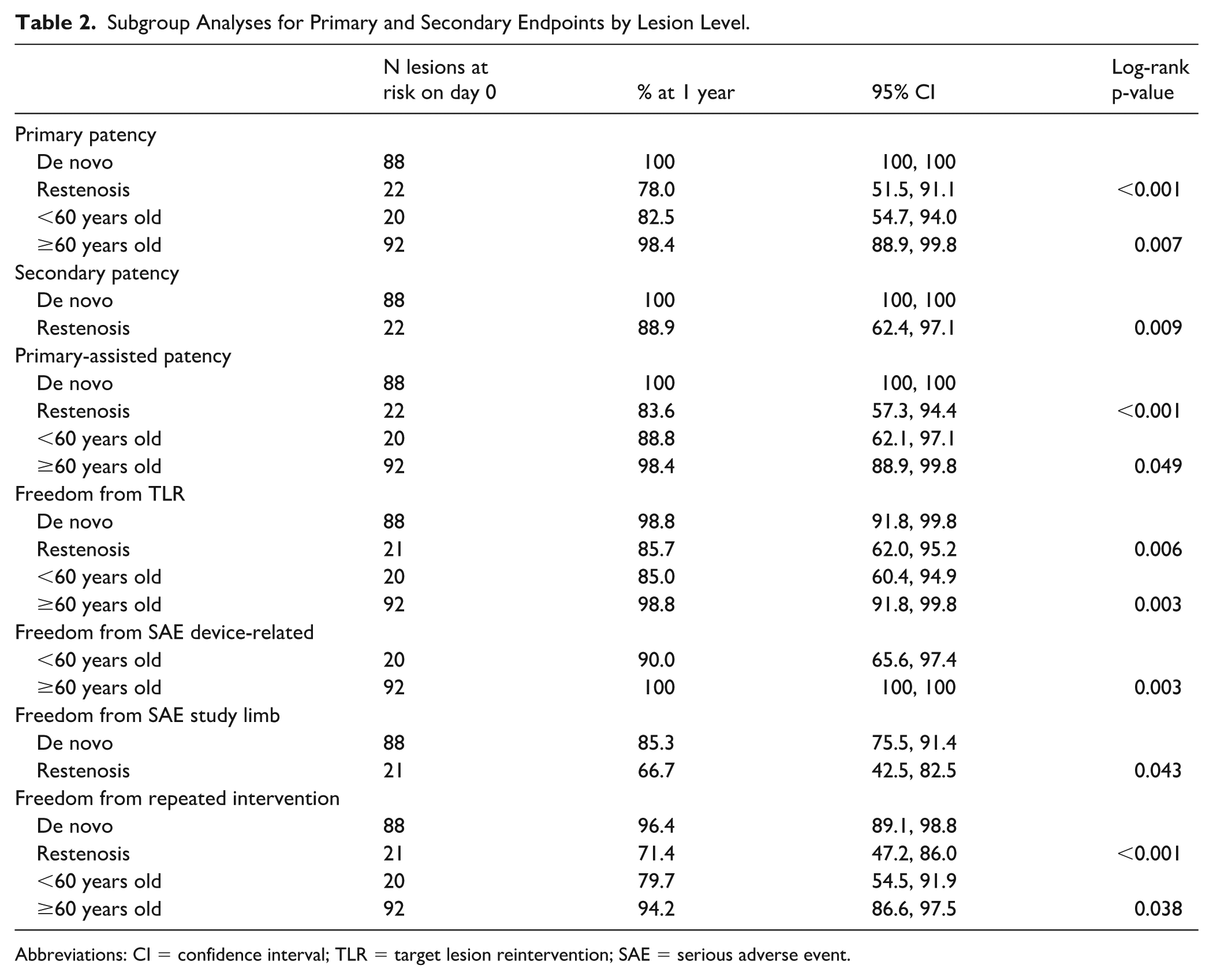
Abbreviations: CI = confidence interval; TLR = target lesion reintervention; SAE = serious adverse event.
Over 1 year, 36/100 patients (36.0%) had a total of 61 SAEs, and 17 patients (17.0%) had a total of 24 non-SAEs. Of the 36 SAEs, 3 were device-related and occurred in 2 patients; 6 were procedure-related SAEs and occurred in 6 patients; and 29 were disease-related SAEs and occurred in 19 patients.
The KM estimate of freedom from major amputation by lesion at 1 year was 99.0% (95% CI=93.4, 99.9) (Figure 1, Panel A). The 1 amputation event was of the lower right leg in a patient with a previous below-the-ankle amputation on the study limb who presented at baseline, with a restenotic lesion and a Rutherford classification of 5. The amputation occurred on study day 56 due to infection, inadequate vascularization, and gangrene.
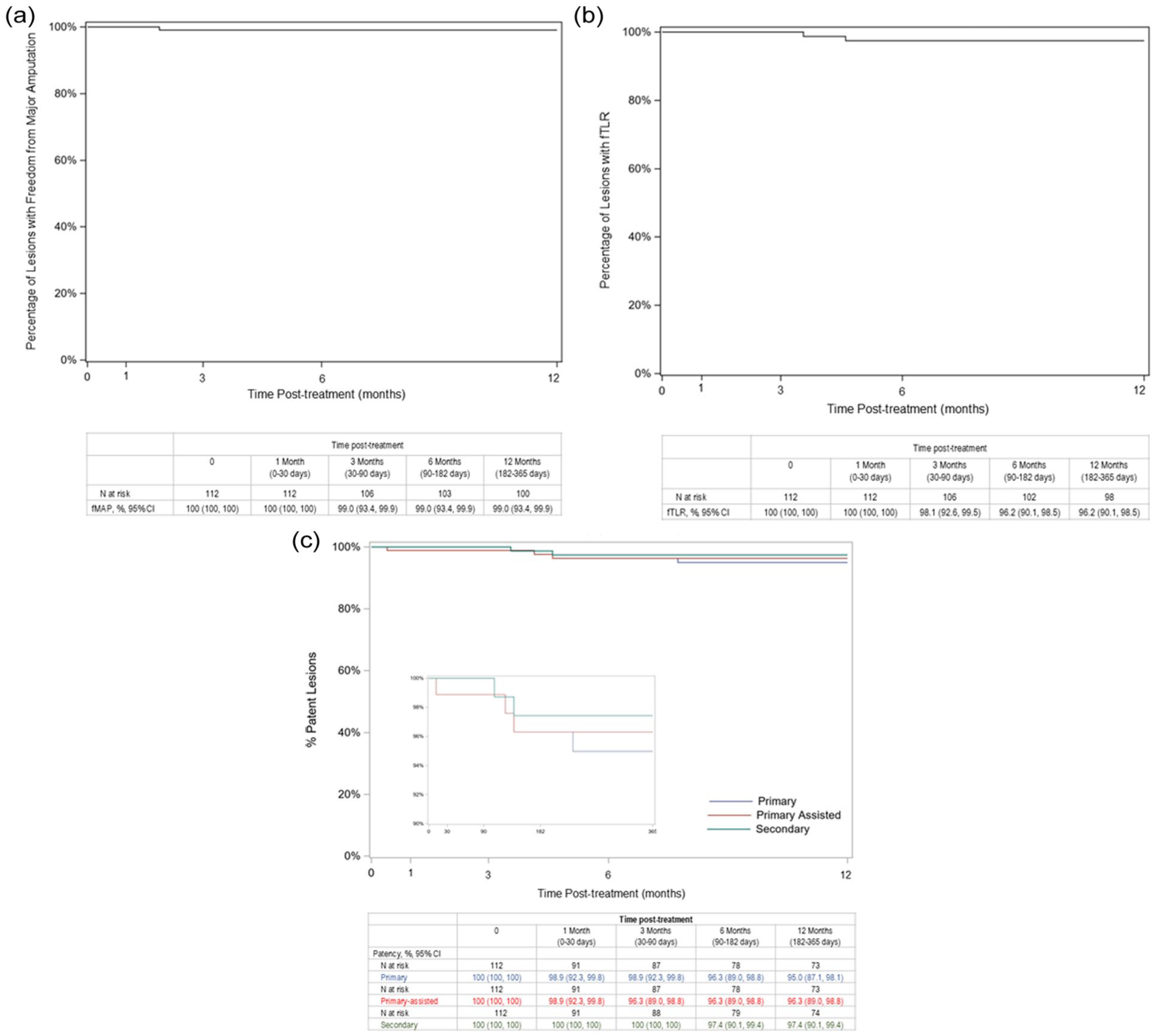
Kaplan-Meier estimates of major amputation (Panel A) by lesion over 12 months. Freedom from target lesion reintervention (Panel B). Primary patency, primary-assisted patency, and secondary patency (Panel C). The inset shows these patency estimates on a scale from 90 to 100%. Abbreviations: CI = confidence interval; fMAMP = freedom from major amputation; fTLR = freedom from target lesion reintervention. Standard error does not exceed 10%.
Three deaths occurred; none were device related. The causes of death were sepsis (day 211), hypercalcemia (day 327), and a non-viable target limb (day 338).
The KM estimate of fTLR at 1 year was 96.2% (95% CI=90.1%, 98.5%) (Figure 1, Panel B). A total of 13 repeat interventions were performed in 9/100 patients (9.0%) for reasons of total vessel occlusion (n=5) or restenosis (n=1), or “other” (n=7). Of the “other” reintervention reasons, 3/7 were repeat interventions in the study device and were for rethrombosis of the right iliac (the target limb), and 2 (in the same patient) were for necrosis. The remaining 4/7 “other” reinterventions were not on the study device but were for reasons of disease-related amputation, compartment, debridement of foot abscess, and “other” iliac artery. Only one of the repeat interventions had a new VSX device implanted in the iliac. An open surgery bypass of the VSX device was conducted to address a total occlusion (day 107) in the previously mentioned patient with Factor V Leiden disorder. The patient was removed from the study following bypass surgery. The patient had prior reinterventions for occlusion on days 12 and 82, for which patency was restored after each reintervention.
In subgroup analyses, the fTLR and freedom from repeat intervention (fRI) were significantly lower in patients with restenotic lesions versus de novo (p≤0.006) and in patients <60 years old vs ≥60 years old (p≤0.038).
Patency and Clinical Outcomes
At 1 year, the KM estimates of lesions with primary patency, primary-assisted patency, and secondary patency were 95.0% (95% CI=87.1%, 98.1%), 96.3% (CI=89.0%, 98.8%), and 97.4% (95% CI=90.1%, 99.4%), respectively (Figure 1, Panel C).
Subgroup analyses (Table 2) revealed that primary patency, primary-assisted patency, and secondary patency were significantly lower in patients with restenotic lesions (vs de novo; p≤0.009). Primary patency and primary-assisted patency were lower for patients <60 years old (vs ≥60 years old; p≤0.049).
Rutherford classification
The change of Rutherford classification from baseline to 1 year is illustrated in Figure 2. At baseline, 37.0% of patients had CLTI (Rutherford classification 4 to 6). At 30 days, 53.8% (42/78) of patients were asymptomatic, and the percentage of patients with CLTI was reduced to 11.6%. At the 1-year follow-up, 63.2% (36/57) of patients were asymptomatic and only 1 patient had CLTI (Rutherford classification 4).
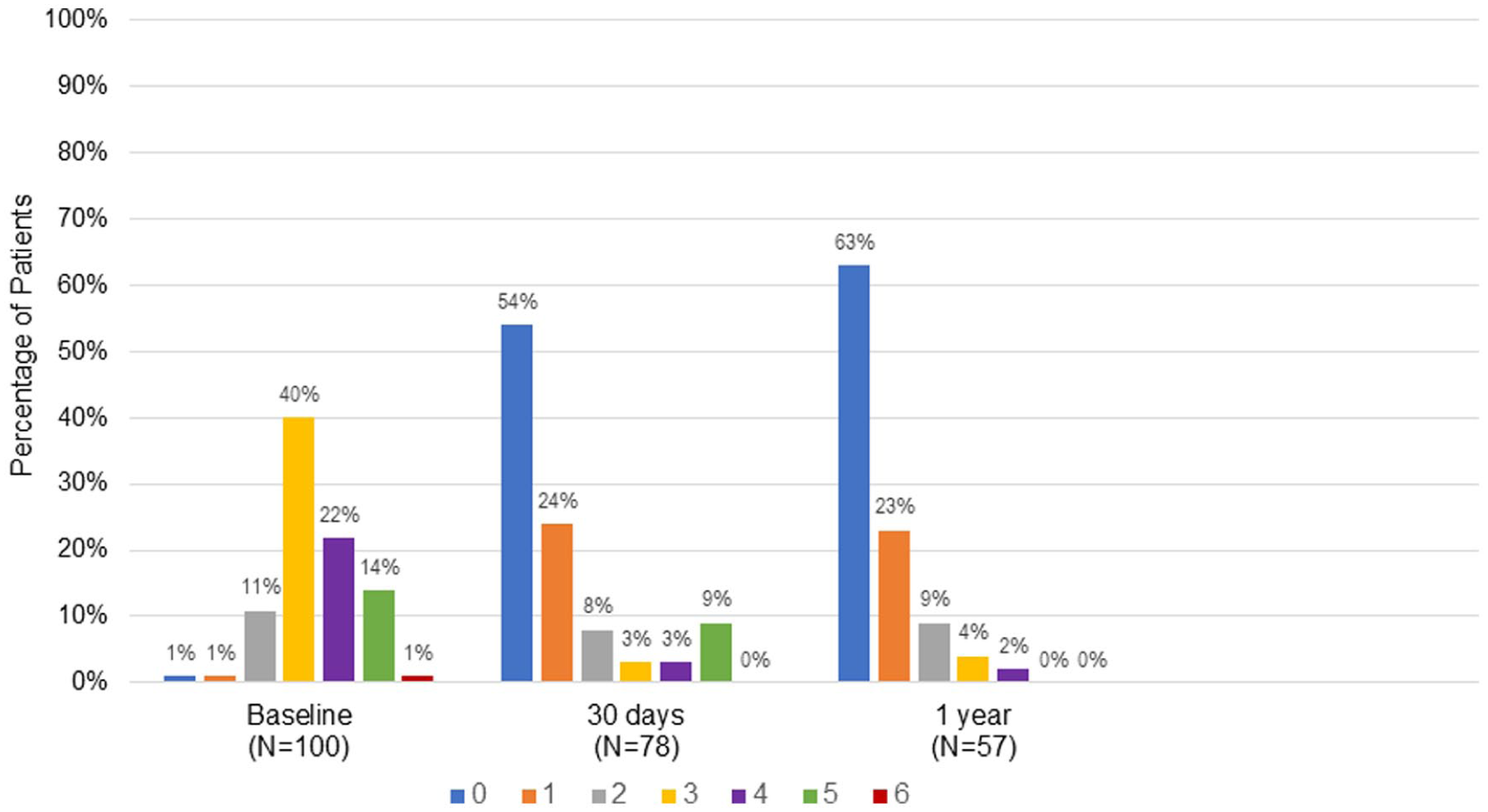
Rutherford classification from baseline to 1-year follow-up. At baseline, 10 patients were not evaluated. At baseline, 37% (n=37) of patients were in CLTI categories. By the 30-day follow-up, the number of patients in the CLTI category dropped to 12% (n=9), and by the 1-year follow-up, only 1 patient (2%) was classified as CLTI. Abbreviations: CLTI = chronic limb-threatening ischemia which is the summation of the number of patients in the Rutherford Classification of 4, 5, and 6.
Ankle Brachial Index
The changes in ABI scores from baseline to 1 year are illustrated in box-plot analyses shown in Figure 3. The mean±SD baseline ABI score was 0.56±0.17 from the 52 patients with a measurement recorded. At 30 days, in the 26 patients with a measurement, the mean change in the ABI score was 0.33±0.15 and was relatively consistent in the 22 patients at 1 year (0.31±0.17); the mean score at 1 year was 0.88±0.15.
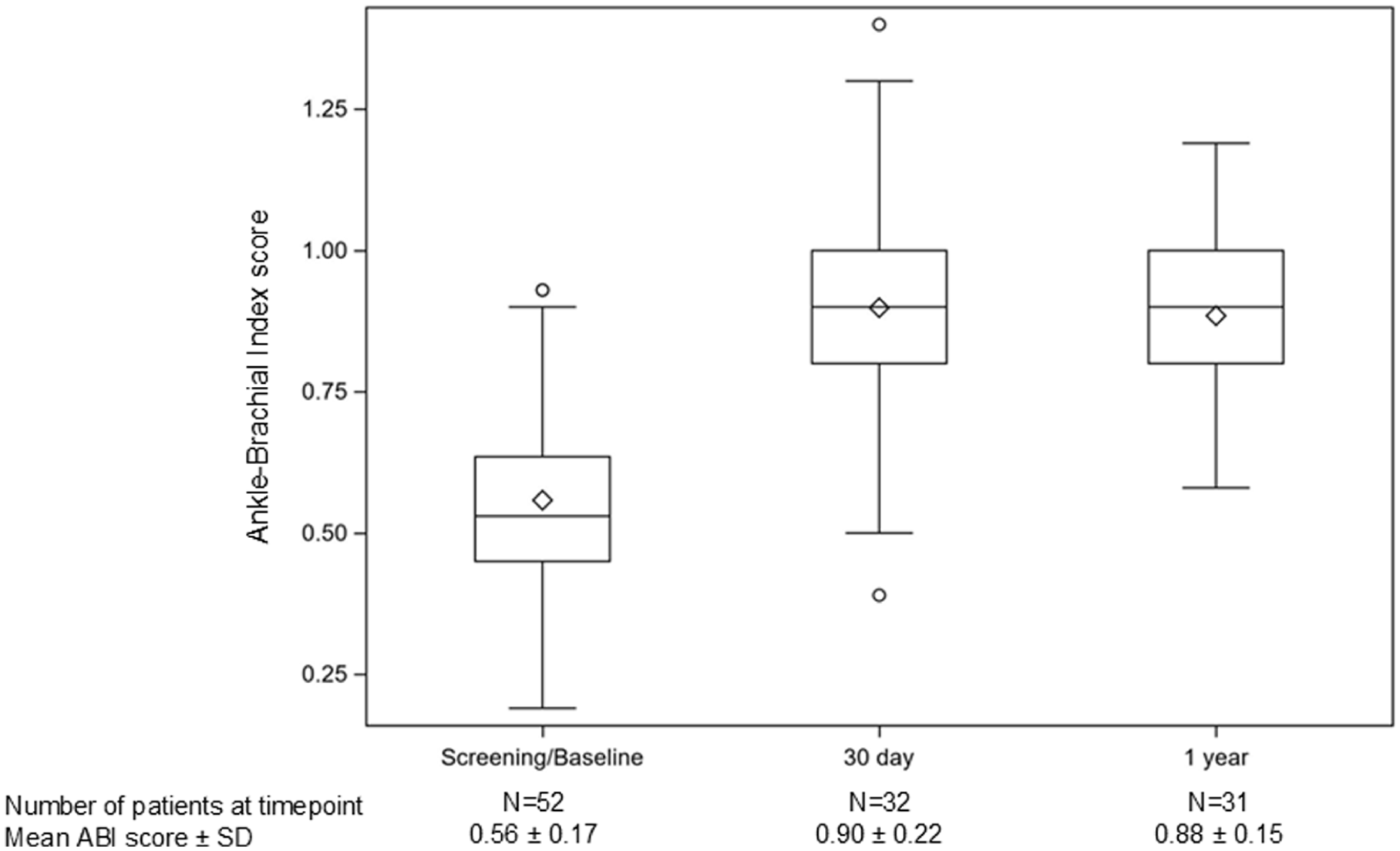
ABI—box plots. The diamond within the box is the mean, the line in the box is the median, the box’s lower and upper values represent the 25th and 75th percentiles, the ends of the whiskers are 1.5 times the interquartile range (75th percentile less 25th percentile), and the circles outside of the ends of the whiskers are outliers. Abbreviations: ABI = Ankle Brachial Index; SD = standard deviation.
Five-level EQ-5D version
Figure 4 illustrates the EQ-5D-5L scores from baseline to 1 year for the Mobility (Panel A) and the Pain and Discomfort Scores (Panel B) subscales. At baseline, mobility impairment was rated severe in 65.2% (30/46) of patients. At 30 days, only 8.9% (6/68) of patients rated severe impairment. At 1 year, mobility impairment was rated as severe for 7.0% (4/57) of patients; 77.2% (44/57) rated little or none.
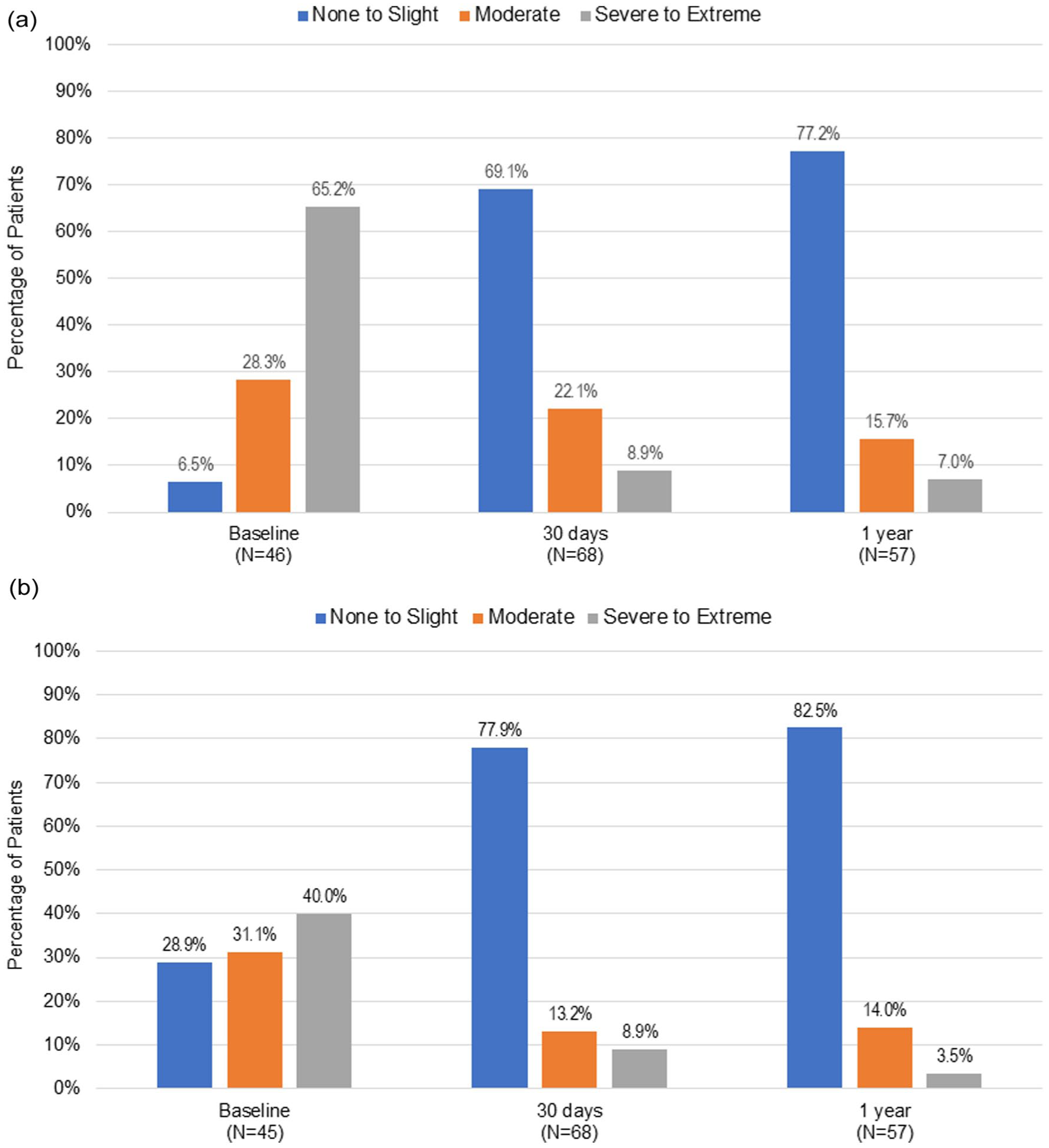
EQ-5D-5L mobility scores (Panel A). EQ-5D-5L pain and discomfort scores (Panel B).
The pain and discomfort rating at baseline was severe in 40% (18/45) of patients and slight or none in 28.9% (13/45) of patients. At 30 days, only 8.9% (6/68) of patients rated pain or discomfort as severe. At 1 year, 3.5% (2/57) of patients had a rating of severe; 82.5% (47/57) rated slight discomfort or none.
The mean (SD) increase in the overall EQ-5D-5L score from the baseline of 0.42 (0.36) to 1-year score was 0.88 (0.18).
Discussion
The 1-year outcomes of patients with de novo or restenotic lesions in the iliac arteries treated with the VSX device demonstrated safety and effectiveness in a “real-world” setting. There were no device-related deaths, only 1 major amputation, and a 9% reintervention rate. The VSX device had high estimated rates of primary, primary-assisted, and secondary patency (95%, 96%, and 97%, respectively). Clinically, the percentage of patients with CLTI at 1 year was reduced by 35% from baseline with nearly two thirds of patients rated as asymptomatic. The health status of the cohort significantly improved with nearly all patients having improvement by at least 1 Rutherford category. The percentage of patients with severe mobility impairment at baseline was 65%, and at 1 year, more than three quarters of the patients had little or no impairment. Similarly, 40% of patients at baseline rated severe pain and discomfort, and by 1 year, 83% of patients reported slight or no pain or discomfort.
The VSX device primary patency rates were consistent with results of studies in patients with AIOD treated with the GORE VIABAHN VBX Balloon-Expandable Endoprosthesis (W. L. Gore & Associates) and ranged from 95% to 97%.13–17 Primary patency of the VSX device was 95% at 1 year, and greater than that reported with other covered stent devices (84% and 92%)18–22 and balloon-expandable stents (94%). 23 In the aforementioned studies, there were lower percentages of patients with chronic total occlusion (ranging from 13% to 47%) and shorter lesion lengths (range 2.6 cm to 5.5 cm) relative to this study with 62% of patients having chronic total occlusion and a mean lesion length of 10 cm with some lesions extended proximally into the aorta while other lesions extended distally into the femoral artery. Thrombosis in the VSX device occurred in 4 patients within the first year after treatment. With so few thrombosis events, it is impossible to determine the contribution of smaller diameter sizes to loss of patency.
The patient characteristics in this study included an average lesion length of 10 cm (in some cases, involved both the EIA and the CIA), 62% of the VSX device implantations in the EIA, and 70% of patients were classified as TASC II C/D. Technical success was achieved in all patients. The outcomes were consistent with VSX device studies that had patient characteristics with similar or lesser degrees of disease severity.24,25 In both studies of 93 and 110 patients, approximately 70% were TASC C/D, 11 and 30% had complete iliac occlusion, and the mean lesion length was shorter (7.0 cm and 9.4 cm). Technical success was achieved in all patients for both studies.
Placement of the VSX device across the inguinal ligament occurred in 20 patients, and 100% technical success was achieved in these patients. Results through 1 year were reassuring, as they were not different from those landing above the ligament. Similarly, another previous study of GORE VIABAHN devices (W. L. Gore & Associates) had placement across the inguinal ligament in 136/147 patients in an emergent setting and demonstrated 100% technical success and high rates of patency out to 3 years without evidence of stent fracture, stenosis, or kinking. 26 These data support the flexibility of the VSX device as a safe and effective surgical alternative to open surgery when placement in distal external iliac and common femoral arteries across the inguinal ligament is required.
Subgroup analyses showed that the VSX device performed consistently regardless of occlusion, common or EIA involvement, sex, and antithrombotic regimen for patency, fTLR, fRI, or device- or limb-related SAE outcomes. However, lesion type (de novo vs restenotic) and age (<60 vs ≥60) did result in differences. Patients with restenotic lesions had lower primary patency, primary-assisted patency, and secondary patency; more target lesion revascularization or repeated reintervention; and more device- or limb-related SAEs (all p≤0.043). These findings are consistent with a 5-year study of CERAB technique in patients with AIOD 27 where loss of primary patency was significantly greater in restenosis patients and younger patients (56 years old vs 63 years old). Younger patients in the current study had lower primary and primary-assisted patency, more target lesion revascularization or repeated interventions, and more device-related SAEs (all p≤0.049). This is consistent with a study of endovascular treatment for suprainguinal artery disease in which patients who were less than 60 years old were at higher risk of reintervention and major adverse limb events relative to older patients. 28 These findings highlight the need for follow-up in patients younger than 60 and those treated for restenosis.
Data from the VSX device studies indicate that the VSX device can be safely used both in the common and EIA. The availability of longer device lengths is an advantage of the VSX device over the balloon-expandable-covered stents in extensive disease, as fewer devices would be needed. Furthermore, the greater flexibility of the VSX device may be beneficial in the EIA, particularly when compared with the older balloon-expandable-covered stents.
Limitations
These study results from a post-market clinical follow-up registry are limited by the study design, which introduces variability in data collection and outcomes reporting (eg, reason for treatment, calcification, etc.). Furthermore, the patient population includes patients who have been treated with the VSX device in combination with other devices.
In this study, 38 patients required additional stenting, but the reason for additional stenting was not collected and location was not always consistently reported. This is recognized as a limitation of the real-world study design. Additional stenting is not a weakness of the stent but often the choice of the operator, who may want a balloon-expandable stent in the CIA, and a self-expanding in the EIA. This is a strength, as this type of study design tends to be more reflective of a real-world population. This registry is limited to 1 device, and outcomes could not be compared.
Clinically-driven TLR was not collected; instead, freedom from TLR was the endpoint and was defined as no surgical or percutaneous revascularization procedure of the original treated lesion was assessed. The observational registry and real-world design of the study meant that the manner of assessment (eg, imaging) was left to the individual investigator’s standard practice/discretion.
Conclusions
The 1-year outcomes from this prospective European registry reflecting a “real-world” setting demonstrated the safety and effectiveness of the VSX device as a treatment in patients with AIOD. This was evidenced by the absence of device-related deaths, only 1 amputation, and few reintervention procedures. Although SAEs were low in this cohort, patients younger than 60 years old or those treated for restenosis had greater risk for target lesion revascularization and device- or limb-related SAEs and emphasizing the need for continued surveillance.
Supplemental Material
sj-docx-1-jet-10.1177_15266028251399582 – Supplemental material for One-Year Clinical Outcomes of a Self-Expanding Covered Stent for Aortoiliac Occlusive Disease
Supplemental material, sj-docx-1-jet-10.1177_15266028251399582 for One-Year Clinical Outcomes of a Self-Expanding Covered Stent for Aortoiliac Occlusive Disease by Michel M.P.J. Reijnen, Peter Mezes and Konstantinos Tigkiropoulos in Journal of Endovascular Therapy
Footnotes
Acknowledgements
The authors thank the study participants and investigators of the VIABAHN Global Registry. Millie Hollandbeck (Phoenix, Arizona) for medical writing support, Eric Novak (W. L. Gore & Associates, Inc) for statistical support, and Chrystal Redding (W. L. Gore & Associates, Inc) for the technical review of the manuscript for assurance of data accuracy in accordance with good publication practice guidelines.
Authors’ Note
Data were presented in part at the Vascular Society LINC 2025 meeting, January 28–31, 2025; Leipzig, Germany, and at the 47th Charing Cross International Symposium, April 23–25, 2025; London, United Kingdom.
Ethical Consideration
The study protocol was reviewed and approved by the institutional review board or ethics committee at each study site in compliance with applicable country laws and regulations. The study was conducted according to the International Conference on Harmonization Good Clinical Practice, the Declaration of Helsinki guidelines, and in compliance with ISO 14155 (International Organization for Standardization).
Consent to Participate
Patients provided written informed consent before undergoing any study procedures and were considered enrolled at the time of consent with the exception of emergency procedure, in which consent was obtained afterwards.
Consent to Publication
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: W. L. Gore & Associates, Inc sponsored this study, and the medical writing support was provided by Millie Hollandbeck.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.M.P.J.R. received the research support, is a consultant, and received a speaker’s fee from W. L. Gore & Associates. P.M. has no conflicts of interest to report. K.T. has no conflicts of interest to report.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
