Abstract
Objectives:
Starting patients on hemodialysis (HD) with an arteriovenous fistula (AVF) and minimizing catheter contact time are major unmet needs for end-stage renal disease (ESRD) patients. Percutaneous arteriovenous fistulas (pAVFs) could address these needs, but current devices often result in flow loss to the brachial vein, requiring reinterventions for target vein maturation and initial clinical use. This study evaluated the technical feasibility and safety of a novel implant-based pAVF, designed to replicate surgical AVF anatomy by directing flow exclusively into the superficial venous system of the upper arm.
Methods:
The VENOS-1 trial (NCT05757726) is a prospective, single-arm, open-label early feasibility study. The primary effectiveness outcome was procedural success and primary safety outcome measures were serious adverse device events and major reinterventions at 6 weeks. Secondary outcomes included physiologic maturation, defined as brachial artery flow ≥500 mL/min and venous outflow diameter ≥5 mm, reintervention rate, and functional use.
Results:
Ten HD patients were enrolled and average follow-up was 15.9 months. Mean age was 45.1 years (range=27–61) and the mean body mass index was 24.3 kg/m2 (SD±9.2). Procedural success was 100%, with no serious adverse events or major reinterventions at 6 weeks. At 6 weeks, median brachial artery flow was 977 ml/min (interquartile range [IQR]±300), and cephalic vein diameter increased to 6.3 mm (SD±1.0 mm). Physiologic maturation was achieved in all patients without reintervention, on average by day 18 (range=13–40). Four subjects underwent ligation of the median basilic vein after reaching unassisted physiologic maturation to facilitate 2-needle inline cannulation of the upper-arm cephalic vein. Two percutaneous reinterventions were needed to maintain or restore patency across 10 subjects in the first 12 months. Access circuit primary patency at 1 year was 80%.
Conclusion:
This first-in human study demonstrates that an implant-based technique is effective and safely creates an AVF for HD. By directing arterial flow into the superficial venous system, the device creates hemodynamic conditions that facilitate physiologic maturation without adjunctive procedures, an observation that will require confirmation in larger studies.
Clinical Impact
This first-in-human study demonstrates that an implant-based percutaneous arteriovenous fistula (AVF) can consistently achieve physiologic maturation without need for flow diversion from the deep venous system. Flow conditions in the superficial venous system drives maturation and creates physical exam findings in the cephalic vein needed for cannulation. This novel approach addresses limitations of earlier percutaneous arteriovenous fistula technologies by simplifying access creation and optimizing hemodynamics in the cannulation zone. Clinicians may benefit from expanded access options that reliably result in physiologic maturation and lowers procedural complexity, which together could reduce catheter exposure. If validated in larger studies, this technique has the potential to shift dialysis access practice by offering an additional access option that creates reliable maturation for initiating hemodialysis with an AVF.
Keywords
Introduction
Central venous catheters (CVCs) for chronic hemodialysis (HD) vascular access are a major source of morbidity and healthcare expenditures. Shortening or altogether avoiding CVC contact time by creating long-term HD vascular access remains a critical objective across health care systems. 1 There is broad consensus that a mature arteriovenous fistula (AVF) is the preferred dialysis access due to its durability and improved patient outcomes. 2 Nevertheless, over 80% of patients in the United States initiate HD with a CVC notwithstanding decades of concerted effort to avoid CVCs, reflecting persistent barriers to permanent vascular access creation. 3 Among patients who receive AVFs, delayed and incomplete maturation often necessitates additional interventions, adding to and prolonging catheter dependence. 4
Percutaneous arteriovenous fistula (pAVF) technologies have the potential to mitigate some of these challenges by addressing various procedural barriers. In addition, a broader provider pool mitigates geographic disparities in access surgeon availability and delivery of care. However, the full potential of pAVF can only be realized if these solutions simultaneously ensure rapid and reliable maturation and cannulation without additional procedures. Commercially available thermal-based pAVF devices successfully demonstrated the feasibility of AVF creation; however, observational studies reveal significant limitations, including high reintervention rates and difficulty in cannulating pAVF with lower-flow in the superficial venous system, both of which have limited broader adoption.5,6
In this context, we introduce a novel implant-based pAVF, designed to closely replicate the anatomy and physiology of surgical AVFs. This innovative pAVF system is designed to optimize flow within the superficial venous system, which is necessary for both maturation and cannulation. Flow within the cannulation zone of the cephalic vein drives the physical examination findings that are needed for reliable cannulation for HD. A potential benefit of a percutaneously-delivered implant-based AVF is that it can avoid inflammation associated with surgical exposure, while also protecting the endothelium in the juxta-anastomotic segment from turbulent blood flow—two well-known causative factors for juxta-anastomotic stenosis. 7
This article details the 1-year outcomes from the VENOS-1 trial, a first-in-human, early feasibility study aimed at evaluating the clinical effectiveness and safety of a novel implant-based pAVF system.
Materials and Methods
VENOS-1 is a prospective, single-arm, single-center, open-label, early feasibility study designed to evaluate the safety and feasibility of the Velocity pAVF System (Venova Medical, Inc., Los Gatos, CA). The study was conducted at the Sanatorio Italiano Hospital in Asunción, Paraguay, which has 2 decades of experience performing early feasibility trials for cardiovascular devices. VENOS-1 was conducted following the Declaration of Helsinki and Ethical Guidelines for Medical and Health Research Involving Human Subjects. The study was approved by the Research Ethics Committee of the Paraguayan Institute of Social Studies (IPES) and registered at ClinicalTrials.gov prior to study enrollment (NCT05757726).
Device Description
The Velocity pAVF system creates an AVF in the proximal forearm between the proximal radial artery (PRA) and cubital perforating vein (CPV) with an endovascular implant (Figure 1), allowing for upper-arm cannulation of the cephalic vein for patients requiring HD. The implant is an expanded polytetrafluoroethylene (ePTFE)-covered nitinol frame with a fenestration for inflow, creating an end-to-side connection between the CPV and the PRA, while simultaneously preserving unobstructed blood flow through the radial artery. The venous end of the implant is 11 mm in length, diverting outflow from the multiple adjacent deep veins and delivering the totality of the access flow volume into the superficial venous system of the upper arm. The implant has a tapered configuration and comes in 2 different sizes, with device selection based on PRA and CPV diameter. The sizing matrix accommodates PRA diameters between 2.0 and 4.0 mm and CPV diameters between 2.0 and 5.0 mm.
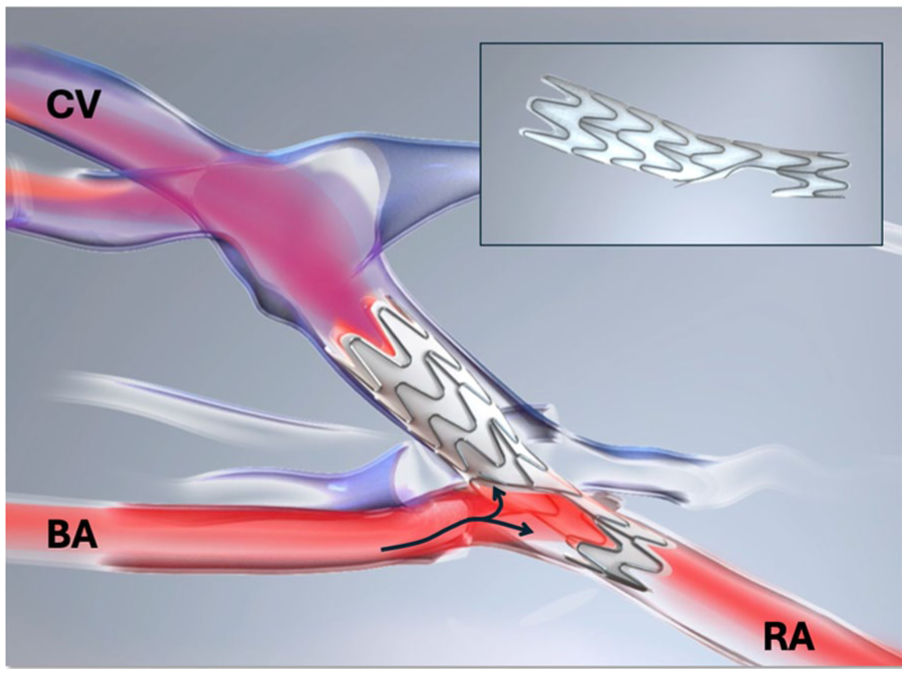
The implant is an ePTFE-encapsulated nitinol frame (inset) that creates an end-to-side connection between the cubital perforating vein (CPV) and proximal radial artery (PRA) in the proximal forearm. The direction of arterial flow is left to right through the brachial artery (BA) into the implant fenestration and unobstructed flow is preserved through the RA.
All procedures were performed under an axillary block. Study subjects received acetyl salicylic acid (ASA) 81 mg and clopidogrel 75 mg prior to the procedure and were instructed to continue the dual antiplatelet medications for 3 months. Under ultrasound guidance, the cephalic vein was punctured retrograde near the antecubital fossa with a 21 g needle and translumenal access was obtained through the CPV into the PRA, followed by introduction of a 0.018″ guidewire (Nitrex, 80 cm, Medtronic Inc., Minneapolis, USA). The Velocity pAVF delivery system is a 2.7 mm outer diameter device, which is advanced sheath-less over the 0.018″ guidewire. The device is positioned exclusively under ultrasound guidance. After confirming the nose cone is against the radial artery wall, a thumb slider is used deploy the venous end of the implant. The delivery system is advanced to deploy the arterial end of the implant, which is housed in the nose cone. The delivery system and guidewire are removed. The access site is managed with light manual pressure for ~5 minutes to achieve hemostasis, followed by an application of a band-aid. An animation of the procedure is shown in Supplemental Video 1.
Study Protocol
Subjects were eligible for participation if they were HD dependent with a CVC and had anatomy suitable for a surgical brachial-cephalic AVF. A complete list of inclusion and exclusion criteria is presented in Supplemental Table 1. Prior to enrollment, subjects underwent a history and physical examination, clinical labs and an upper extremity vein mapping using duplex ultrasound (DUS). Following the procedure, subjects underwent a clinical examination of the upper extremity and a DUS on day 1, 2 weeks, 6 weeks, 12 weeks, 6 months, and 1 year after the procedure.
The dialysis access DUS was performed according to a standardized protocol with a vascular 12 to 15 MHz broadband linear array transducer (EPIQ-5, Phillips Healthcare, Amsterdam, Netherlands). Spectral Doppler imaging was used to measure the pulse wave velocity of the brachial artery 5 cm proximal to the implant, followed by a volumetric flow calculations in duplicate. Angle of insonation was standardized at 60°. Measurements of the cephalic and basilic vein diameters and depth were captured at a distance of 3 and 6 cm beyond the radial artery interface. All measurements were performed in duplicate, and the reported diameter is the average across all 4 measurements at the 2 sites.
Outcome Measures
The primary effectiveness endpoint was procedural success, defined as successful deployment of the pAVF implant between CPV and PRA with DUS confirmation of arterialized flow in the cephalic vein. Arterialized flow was defined as a pulsatile waveform assessed with pulse wave velocity sampled 1 cm central to the venous end of the implant at the completion of the procedure. The primary safety endpoints were serious adverse device events and major reinterventions at 6 weeks. Serious adverse device events were defined as any serious adverse event (SAE) that reasonably could be caused by the device or the procedure. Major reintervention was defined as any open surgery, thrombectomy or thrombolysis in the index limb following pAVF creation.
Secondary endpoints included time to physiologic maturation, defined as DUS demonstrating upper-arm outflow vein diameter of ≥5 mm and brachial artery blood flow of ≥500 mL/min. 8 Additional secondary endpoints included functional maturation, defined as 2-needle cannulation of the AVF on 3 consecutive sessions. Access circuit primary patency was defined as the interval from time of access creation until the first occurrence of any reintervention to maintain or reestablish patency or until access abandonment. The access circuit was defined as the inflow radial artery, implant, cannulation segments, and venous outflow through the innominate vein. Interventions to facilitate cannulation, such as superficialization, transposition, or accessory outflow vein ligation, are not included in access circuit patency determination. Cumulative patency was defined as the time of access creation until the final abandonment of the access.
Statistical Analysis
Descriptive statistics were utilized to summarize patient demographics, baseline characteristics, and outcomes. Continuous variables are reported as mean±standard deviation (SD) or median with interquartile range (IQR), depending on distribution. Categorical variables are presented as frequencies and percentages. Time-to-event outcome measures were evaluated using Kaplan-Meier methodology, with graphical representations provided as survival curves. Survival analyses were performed using Stata v 11.2.
Results
Ten subjects were enrolled in October 2023. The mean age was 45.1 years (range=27–61) and the mean body mass index was 24.3 kg/m2 (SD±9.2). Etiology of kidney failure was hypertension in 4/10 (40%) and diabetes in 6/10 (60%). After an axillary block with 2% lidocaine, the mean PRA diameter was 3.0±0.5 mm (range=2.1–4.3 mm, SD) and the CPV diameter, was 4.0±0.7 mm (range=3.2–4.9 mm). The primary effectiveness endpoint of procedural success was achieved in all patients. Intra-procedure DUS confirmed outflow from the PRA with no signs of arterialized blood flow into the deep venous system in any subject. There was no evidence of pseudoaneurysm or peri-implant hematoma on intraprocedural imaging in any subject. A representative diagnostic transfemoral angiogram is shown in Figure 2 (Supplemental Video 2).
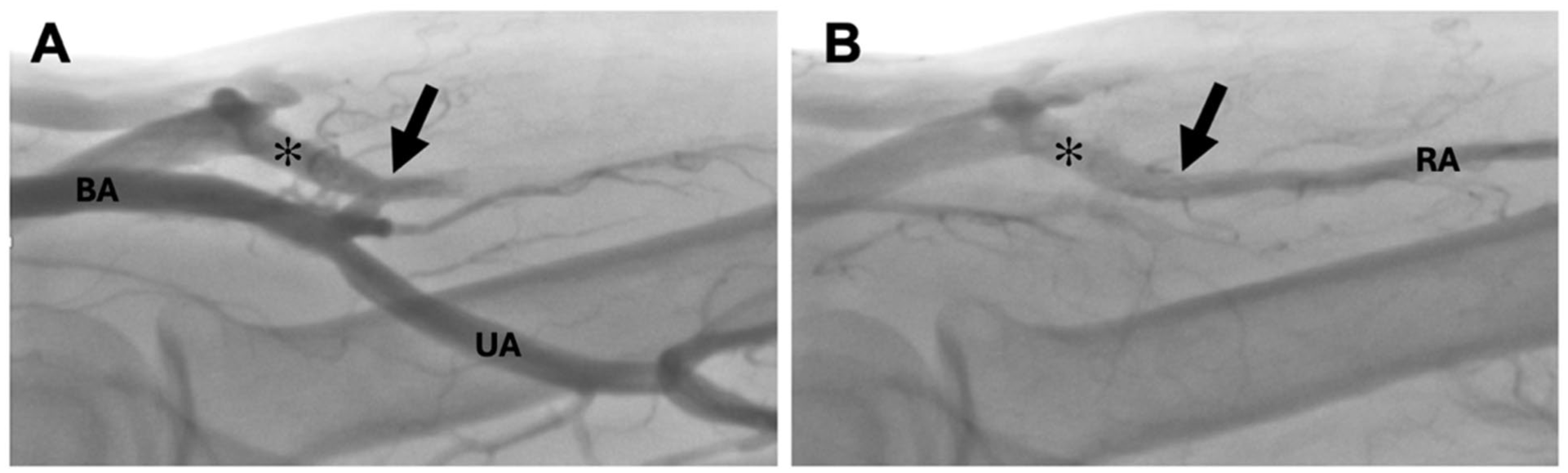
Angiogram demonstrating a proximal forearm fistula from the radial artery (RA) to the cubital perforating vein (✻), without flow into the deep venous system. The arrow denotes the location of the implant. (A) Early antegrade filling of the pAVF. (B) Late retrograde flow from the radial artery into the CPV, demonstrating an open end-to-side connection. Brachial artery (BA), ulnar artery (UA).
Brachial artery flow volume (Qa) (median [IQR]), a key predictor of maturation and cannulation, increased progressively from 744 mL/min (478 mL/min) at day 1 to 977 mL/min (300 mL/min) at day 42 (Figure 3). There was a corresponding increase in upper-arm cephalic vein diameter from 3.4 mm (0.3 mm) at baseline to 6.2 mm (0.6 mm) on day 42 (Supplemental Figure 2). The composite outcome measure of physiologic maturation was achieved in all subjects within the first 6 weeks (Figure 4). The median time to achieve physiologic maturation was 18 days. Physiologic maturation was unassisted in all subjects, with no reinterventions needed from the date of the procedure through eclipsing the physiologic maturation threshold.
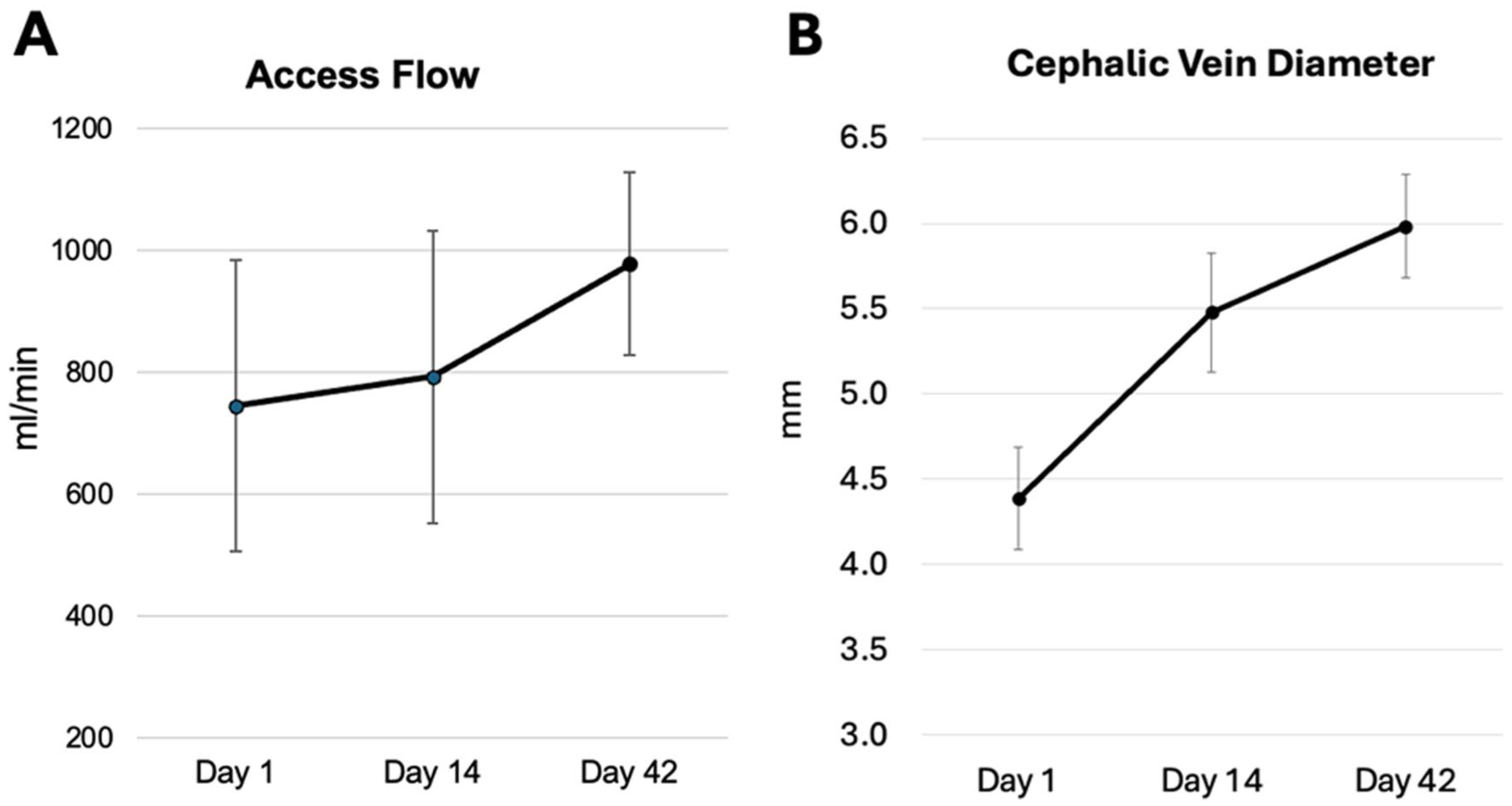
(A) Access flow assessed with duplex ultrasound volumetric calculation in the brachial artery. (B) Cephalic vein diameter changes in response to intralumenal flow conditions. Values are median±IQR.
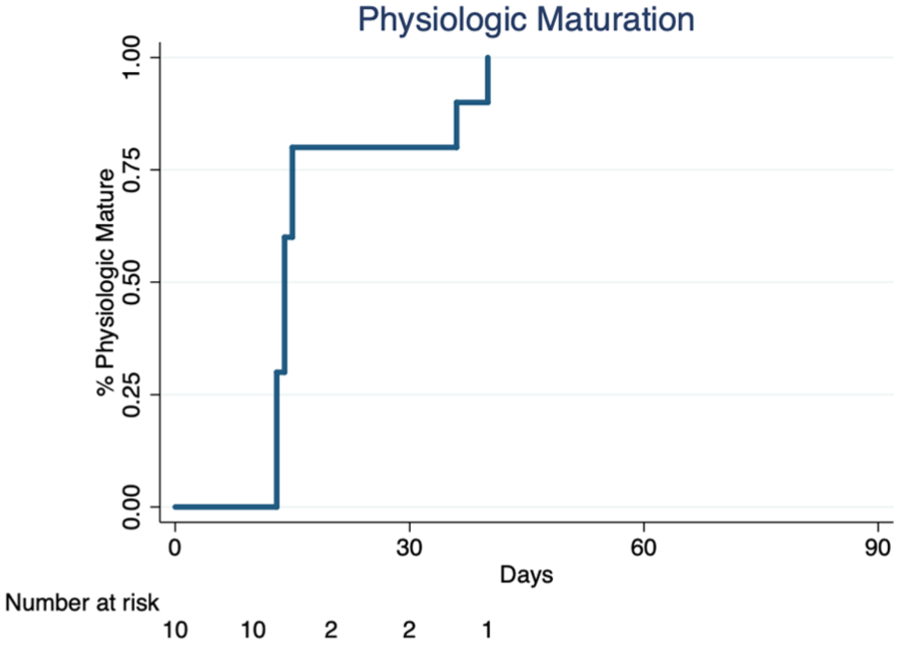
Cumulative time-to-event curve for Physiologic Maturation. All study participants achieved physiologic maturation within 6 weeks and no reintervention was needed in any patient to reach this outcome measure.
Subjects were followed up on average for 15.9 months (median=17.5, range=4–18 months). One subject exited the study early due to catheter-associated sepsis at day 65 that colonized the implant, which was subsequently removed. There were no infections in the remainder of subjects who were followed up through study termination. Four subjects had split venous outflow through both basilic and cephalic veins and underwent ligation of the medial basilic vein to direct the flow toward the cephalic vein to facilitate inline cannulation. Nine subjects achieved functional maturation with 2-needle cannulation of the upper-arm cephalic vein, without the need for special training on cannulation technique (Figure 5).
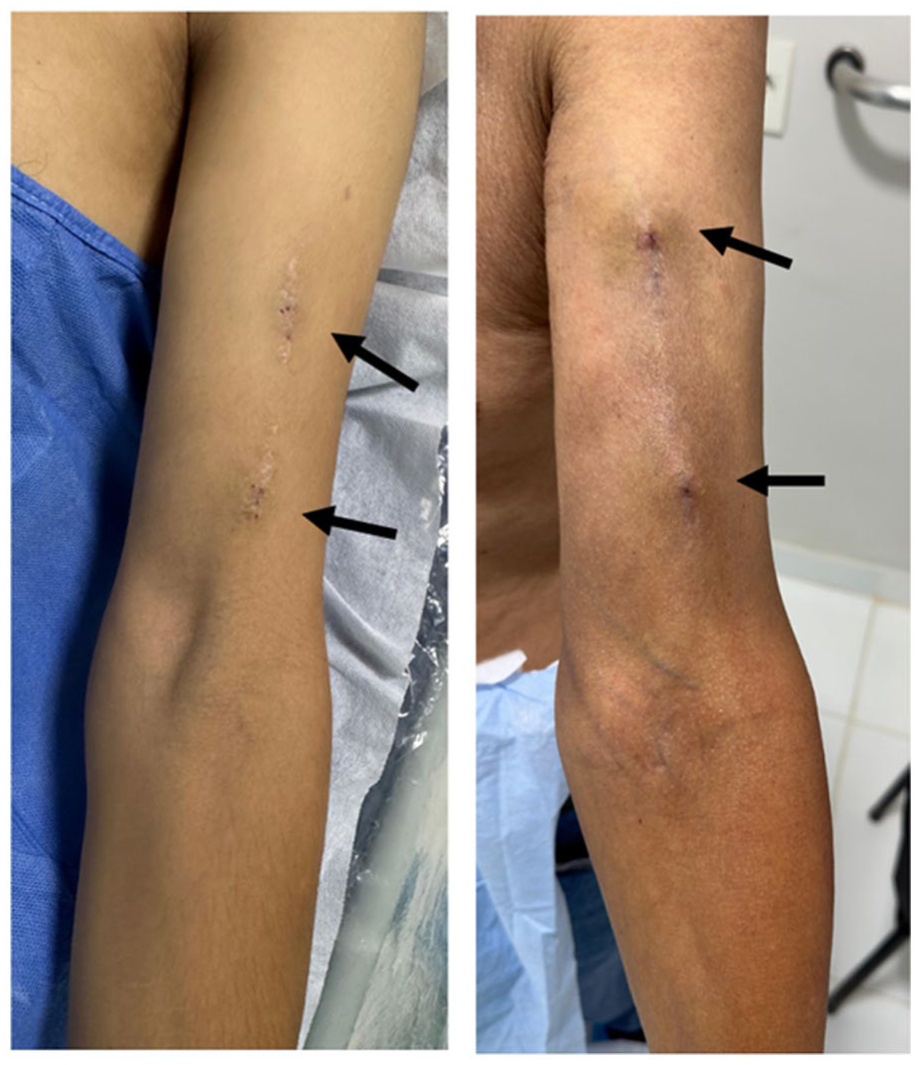
Cephalic vein cannulation sites 1-year post-procedure. A single outflow, normal flow AVF simplifies cannulation. Inline cannulation of upper-arm cephalic vein was achieved in 9 of 10 subjects.
The pAVF access provided sustained clinical use over the duration of follow-up. There was a single balloon angioplasty of the venous end of the implant at 224 days in 1 subject, otherwise all other subjects have had uninterrupted functional use of the pAVFs throughout follow-up. The total number of reinterventions to maintain or restore access patency for all subjects was 2 over the course of the study, for an average of 0.2 reinterventions-per-patient-year. The access circuit primary patency at 1 year was 80% and cumulative patency was 90%.
While the implant-based pAVF quickly reached the threshold for physiologic maturation, over the ensuing 12 months, Qa plateaued never exceeding 1400 ml/min (Figure 6). The radial artery remained patent through the duration of the study for all subjects. The flow in the radial artery was retrograde or bidirectional in all patients, with no instances of radial artery thrombosis or occlusion. There were no instances of hand ischemia or high output pAVF in any patient during follow-up.
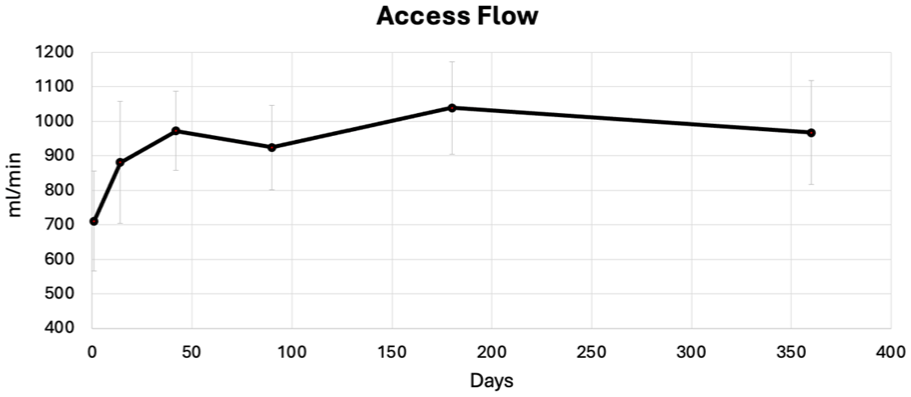
Access flow, measured in the brachial artery, rises quickly then plateaus over long-term follow-up. The implant has fixed resistance which protects against high-flow fistulas that lead to hand ischemia or cardiac complications.
Discussion
The results of this first-in-human study demonstrate the effectiveness of an implant-based approach for creating pAVFs for HD vascular access. Beyond achieving 100% technical success and demonstrating a favorable safety profile, this method of access creation consistently delivered rapid and reliable physiologic maturation, minimal reintervention to the implant, functional use that did not require special cannulation techniques, and sustained patency through 12 months. These outcomes address critical needs necessary to optimize dialysis access performance and have substantial potential to address significant problems related to HD access creation, initial clinical use, and durability.
An implant-based approach offers several important advantages over traditional surgical AVFs. The standardized nature of the procedure ensures uniform cross-sectional area and outflow resistance, resulting in reproducible flow conditions across most patients. Establishing ideal flow conditions early in the life cycle of dialysis access is necessary for outward remodeling of the outflow vein, facilitating higher rates of physiologic maturation than seen in most surgical AVF series.
Placed into context against trials of surgical AVFs, VENOS-1 sets a notably higher standard for physiologic maturation by requiring a larger vein diameter that is achieved within a shorter timeframe (6 weeks) than most contemporary studies. In high quality observational trials, such as the Hemodialysis Fistula Maturation study, only 53% of surgical AVFs reached physiologic maturation by 6 weeks, despite using a lower threshold for vein diameter (4 mm). 9 Similar results have been reported in other high-quality prospective studies, including the randomized Patency I and II trials. 10 Not only did we observe uniform physiologic maturation across all subjects, but importantly, all were unassisted. This is in stark contrast to what is typically observed in surgical AVF studies, where balloon angioplasty in the first 6 months may be necessary in more than 40% of AVFs. 4
One of the advantages inherent in a pAVF is that it avoids common barriers to surgical access placement, such as referral delays, testing requirements, scheduling complexities, and operating room availability, all which lead to additional catheter exposure time. In addition, the simplicity and predictability of this approach enable expanding the provider pool to include interventionalists, potentially addressing regional disparities in dialysis access surgery availability and delivery of care. The full potential of pAVF technology, which is limited by “first-generation” devices requiring follow on “flow directing” procedures, frequent reinterventions, and specialized cannulation training, is actualized through the implant-based method. The design ensures a superficial outflow pathway, preventing diversion of flow into the deep venous system. This focused flow precisely replicates surgical fistula anatomy, preserving essential physical examination cues for cannulation while ensuring adequate superficial vein flow that meets the needs of the HD circuit. Consequently, inline cannulation is straightforward and free from risks of inefficient HD related to recirculation.
In addition, the implant-based method demonstrates exceptional durability by addressing the primary causes of AVF dysfunction. Surgical AVFs commonly experience juxta-anastomotic stenosis, driven by inflammation induced by turbulence or surgical trauma–or in the case of commercially available pAVF devices, by the thermal injury or barotrauma. Multiple clinical outcome studies have reported reintervention rates between 30% and 40% within 6 months in surgical AVF.11,12 In the Ellipsys pivotal trial, there was an average of 2.2 interventions per patient in the first 90 days. 13 In addition, recent large single-center study by Shahverdyan et al 14 reported juxta-anastomotic venous lesions of 50% in pAVFs as compared with 35% in the surgical comparator. In contrast, this study observed 2 percutaneous interventions in 10 subjects over the first 12 months, less than what is typically reported in surgical AVF trials. The remaining 4 procedures were cubital vein ligations, a minor subcutaneous procedure which consist of stab incision and ligating the accessory outflow. In surgical series of forearm AVFs, these types of procedures are needed in ~15% of subjects. 4 Cubital vein ligation does not have the magnitude of risks (radiation and contrast) or costs as angioplasty or coil embolization, which is commonly needed for first-generation pAVFs. And just as in surgical AVFs, ligating the accessory outflow vein can easily be performed at the time of the initial procedure.
An implant-based pAVF has the potential advantage of avoiding inflammation associated with open surgical exposure while also interrupting the effects of turbulence in the juxta-anastomotic segment—a common initiating factor for stenosis. 7 The PTFE-encapsulated implant shields the juxta-anastomotic endothelium from sensing turbulence in the short term, potentially interrupting the signaling transduction involved in endothelial activation. These potential advantages could help explain the reduced need for angioplasty observed in this study.
While this early feasibility study supports the overall safety profile of an implant-based pAVF, potential risks must be acknowledged. As with all endovascular implants, device and procedure-related complications may include access site injury, dissection, acute thrombosis, or implant migration, among others. None of these events were observed in this small early feasibility study. There are additional risks specific to this unique application of an endovascular implant to create an AVF. These include possible loss of future dialysis access options, loss of radial artery patency, and infection.
The velocity pAVF leverages unique anatomy not commonly applied in surgical practice—a PRA to perforating vein AVF. In the Vascular Quality Initiative (VQI) registry of over 52 170 first-time accesses, AVF’s utilizing similar anatomy was seen in only 361 patients (0.7%). 15 A high-level design objective was to create an additional access option for patients without taking an existing one away. The venous end of the implant is 11 mm long and resides entirely within the CPV. As it is delivered percutaneously, tissue planes in the antecubital fossa are not disturbed. In the event of acute failure, the access can be converted to a brachiocephalic AVF without undue difficulty.
Most infections in dialysis access are related to colonization from skin flora at the time of needle access. This implant is deep to the muscular fascia and remote to the cannulation zone. Therefore, the most likely cause of infection is hematogenous colonization, and based on the experience with PTFE stents in cephalic arch stenosis or other peripheral vascular interventions, intravascular implant colonization from hematogenous sources are a rare occurrence.
Finally, as with surgical AVF in the forearm, standard practice is to confirm normal ulnar circulation prior to the procedure. When this is true, radial artery occlusions are clinically silent events. One example, among many, is the experience with trans-radial access for coronary interventions. In the RAIL-TRAC study of 1945 patients, the incidence of radial artery occlusion was 17%. 16 Importantly, this was a clinically silent event in all. The observed low frequency and mild nature of these risks combined, coupled with promising early outcomes for pAVF function use, strongly supports further evaluation in a larger clinical study to fully characterize long-term safety and efficacy.
Several limitations should be considered when interpreting the results of this study. First, the study’s sample size was small, limiting the generalizability of the findings. Second, the study was conducted at a single center, which may introduce selection bias and further constrain external validity. Finally, the service population of the study center has resulted in demographic profile of enrolled patients that was relatively young compared with the broader end-stage renal disease (ESRD) population. This may potentially limit applicability of the results to the global ESRD population. These limitations highlight the preliminary nature of the findings and underscore the need for larger trials to confirm the observed outcomes.
Ultimately, the combination of optimized flow conditions in the superficial venous system, managing turbulence in the juxta-anastomotic segment, and streamlined procedural logistics offers a clear path toward reliable fistula maturation, improved clinical outcomes, and potentially reduced catheter dependency. If validated in larger studies, this promising approach has significant implications for health care economics by offering substantial cost savings to Medicare and other health care payers.
Supplemental Material
sj-docx-1-jet-10.1177_15266028251396313 – Supplemental material for A Novel Percutaneous Arteriovenous Fistula for Hemodialysis Access: One-Year Outcomes of the VENOS-1 First-In-Human Trial
Supplemental material, sj-docx-1-jet-10.1177_15266028251396313 for A Novel Percutaneous Arteriovenous Fistula for Hemodialysis Access: One-Year Outcomes of the VENOS-1 First-In-Human Trial by Robert Shahverdyan, Dirk M. Hentschel, Adrian Ebner, Erik van der Burg and Shant M. Vartanian in Journal of Endovascular Therapy
Supplemental Material
sj-docx-2-jet-10.1177_15266028251396313 – Supplemental material for A Novel Percutaneous Arteriovenous Fistula for Hemodialysis Access: One-Year Outcomes of the VENOS-1 First-In-Human Trial
Supplemental material, sj-docx-2-jet-10.1177_15266028251396313 for A Novel Percutaneous Arteriovenous Fistula for Hemodialysis Access: One-Year Outcomes of the VENOS-1 First-In-Human Trial by Robert Shahverdyan, Dirk M. Hentschel, Adrian Ebner, Erik van der Burg and Shant M. Vartanian in Journal of Endovascular Therapy
Footnotes
Acknowledgements
The authors thank MMC Medical International Services, Donna Merithew, and Laura Minarsch for their contribution in performing the study, collecting data, and reviewing the manuscript.
Ethical Approval and Informed Consent
This study was conducted following the Declaration of Helsinki and Ethical Guidelines for Medical and Health Research Involving Human Subjects. The study and all amendments were reviewed and approved by the Research Ethics Committee (CEI) of the Paraguayan Institute of Social Studies (IPES). All subjects signed a written informed consent when screened for participation in the study. The informed consent contained all the elements of informed consent specified in the US Code of Federal Regulations (21 CFR 50.25). The trial has been registered at ClinicalTrials.gov with NCT05757726.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was fully funded by Venova Medical, including all costs associated with study design, conduct, and data collection.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Ebner has no financial conflict to declare. Dr Shahverdyan and Dr Hentschel have consulting agreements as Scientific Advisory Board members for Venova Medical. Dr Vartanian and Mr van der Burg have equity holdings in Venova Medical, which sponsored this first-in-human study.
Data Availability Statement
The data supporting the findings of this study are available upon reasonable request from the corresponding author, subject to approval from Venova Medical.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
