Abstract
Clinical Impact
Ultrasound visualization of vascular closure devices during endovascular access closure leads to a significant decrease in overall and major post interventional access site complications. This non-invasive and often readily available imaging technique could therefore lead to an important decrease in morbidity and subsequent overall health care costs when added to the standard intervention protocol. With the increasing use of endovascular techniques to treat peripheral artery disease, the addition of ultrasound-techniques in closure of endovascular access sites could potentially have a large clinical impact, both on patient outcomes as well as financial outcomes.
Keywords
Introduction
Background
Peripheral artery disease (PAD) describes the condition of partial or complete obstruction of peripheral arteries, leading to a decrease in blood flow to the extremities.1,2 This can result in intermittent claudication complaints. In the more severe cases, PAD can lead to critical limb ischemia (CLI), ultimately leading to limb loss. 3
Patients with PAD worldwide are increasingly treated using endovascular techniques. 4 Most frequently, the access site for these procedures is the common femoral artery (CFA) or superficial femoral artery (SFA). After endovascular procedures, reliable closure of the puncture site is important, as vascular complications after the procedure are associated with higher morbidity and mortality. 5 Different vascular closure devices (VCD) with several techniques are available to establish secure closing. These devices have shown to be equally effective and safe as manual compression but result in a shorter hospitalization time and time to hemostasis.6,7 Furthermore, VCDs are associated with increased patient satisfaction and decreased incidence of combined adverse cardiovascular events and hematomas.6,7
Vascular Closure Devices
The most commonly used VCD worldwide in femoral percutaneous endovascular procedures is the Angio-Seal hemostatic puncture closing device (Terumo Interventional Systems, Somerset, New Jersey). 8 This VCD consists of 3 biodegradable components: a co-polymer anchor placed intravascularly at the access site, a collagen plug placed in the extravascular tissue tract, and a suture connecting anchor and collagen plug. 9 Despite numerous studies showing the safety and efficacy of the VCD in PAD,9 –12 several cases of minor and major complications have been described after closing of the femoral access site. 13 These complications consist of hematoma, pseudoaneurysms, recurrent wound bleeding, arterial dissection, and arterial occlusion.8,14,15 In the most severe cases, arterial occlusion led to severe lower limb ischemia. 8 These post-surgery complications do not only cause morbidity and mortality in patients, but also form a financial burden for the local hospital as well as the national health care system due to increased hospitalization time, additional therapy, and permanent sequalae.5,16,17 The VCD implantation failures leading to a significant number of complications could potentially be prevented by ultrasound guidance. This imaging technique is able to visualize the Angio-Seal footplate and thus could be of assistance in prevention of VCD deployment failures14,15 due to misplacement. Therefore, the aim of this study is to investigate if the use of ultrasound during Angio-Seal vascular closure can decrease vascular access complications after endovascular treatment in patients with PAD.
Methods
Trial Design and Participants
All consecutive endovascular procedures for PAD between 2017 to 2018 and 2020 to 2022 were included in this retrospective study. In 2019, clinicians started using the ultrasound during Angio-Seal placement. As of 2020, the use of ultrasound was actively reported in the radiology reports and performed as standard of care. Therefore, the procedures in year 2019, considered as the transition year, were not included in this study. This study protocol was approved by the institutional review committee. Furthermore, the Medical Ethical Committee approved a waiver of consent.
Patient Selection Procedure
Study data were retrieved from Hospital Information System (HiX, Chipsoft, Amsterdam, the Netherlands) and the Picture Archiving and Communication System (PACS). All visual data found in PACS were collected and analyzed by 5 interventional radiologists.2 –4,10,11 All other variables were collected in a manual search of patient dossiers in HiX (author 1). The inclusion criteria were endovascular treatment for PAD of the lower extremities between 2017 and 2018 or 2021 and 2022 with femoral access and the use of Angio-Seal for hemostasis at femoral access site. Exclusion criteria were missing documentation on closure guided by ultrasound in the years 2020 to 2022, other access sites than femoral access, acute limb ischemia, and use of other VCD. It was assumed that in case of lacking information on the use of ultrasound in 2017 and 2018, no ultrasound was used during the placement of the Angio-Seal as this was standard of care in that period of time.
After application of the inclusion and exclusion criteria, included patients were divided into 2 study groups (Figure 1). Group 1 (non–ultrasound-guided Angio-Seal, NG-Seal) consists of all selected patients treated in the period 2017 to 2018 found eligible according to the inclusion and exclusion criteria. Group 2 (ultrasound-guided Angio-Seal, US-Seal) consists of all selected patients treated in the period 2020 to 2022 with registered use of ultrasound techniques during placement of the Angio-Seal in HiX. Patient demographics and treatment characteristics are summarized in Table 1.
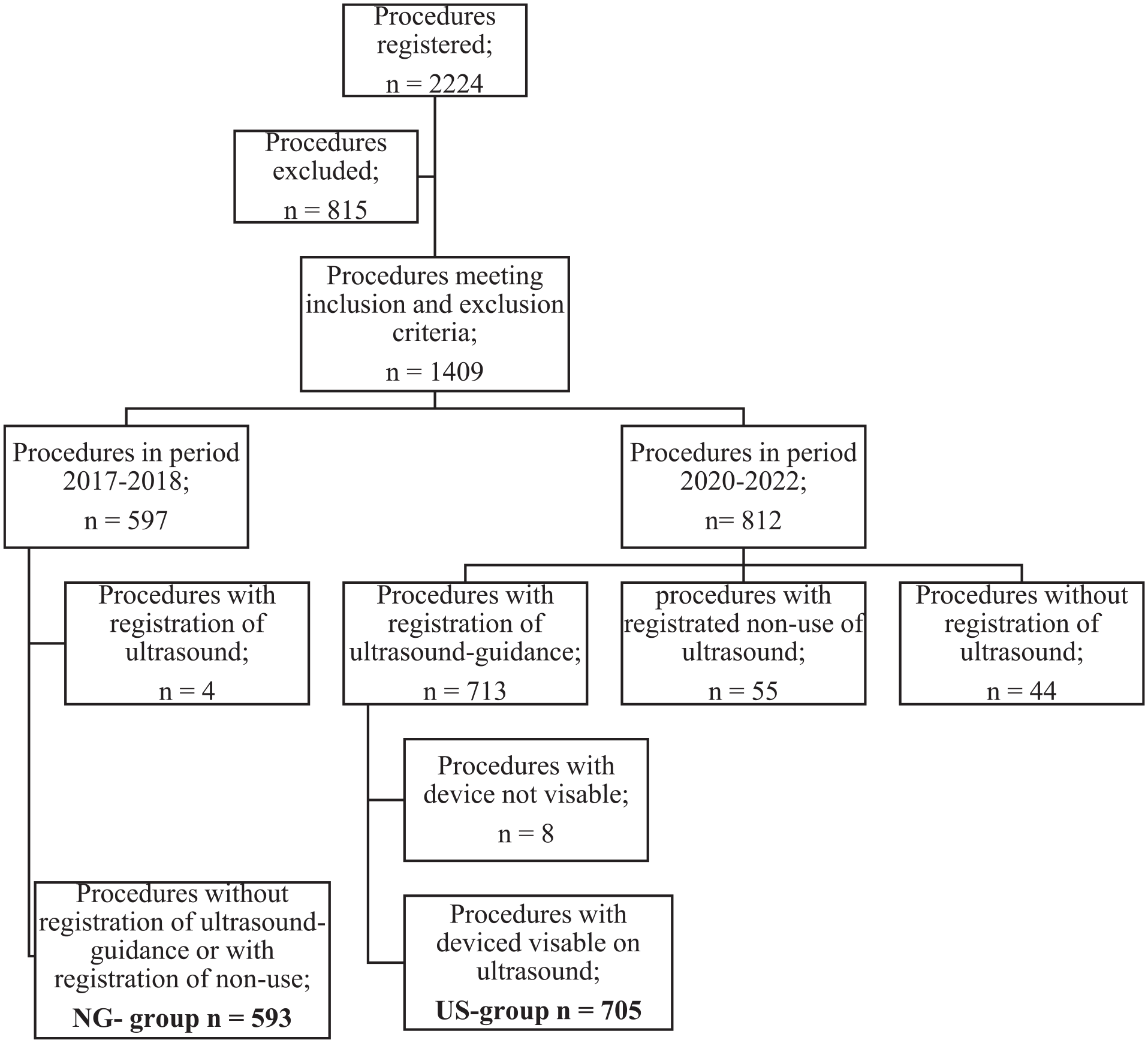
Flow chart study population.
Study Population Characteristics.
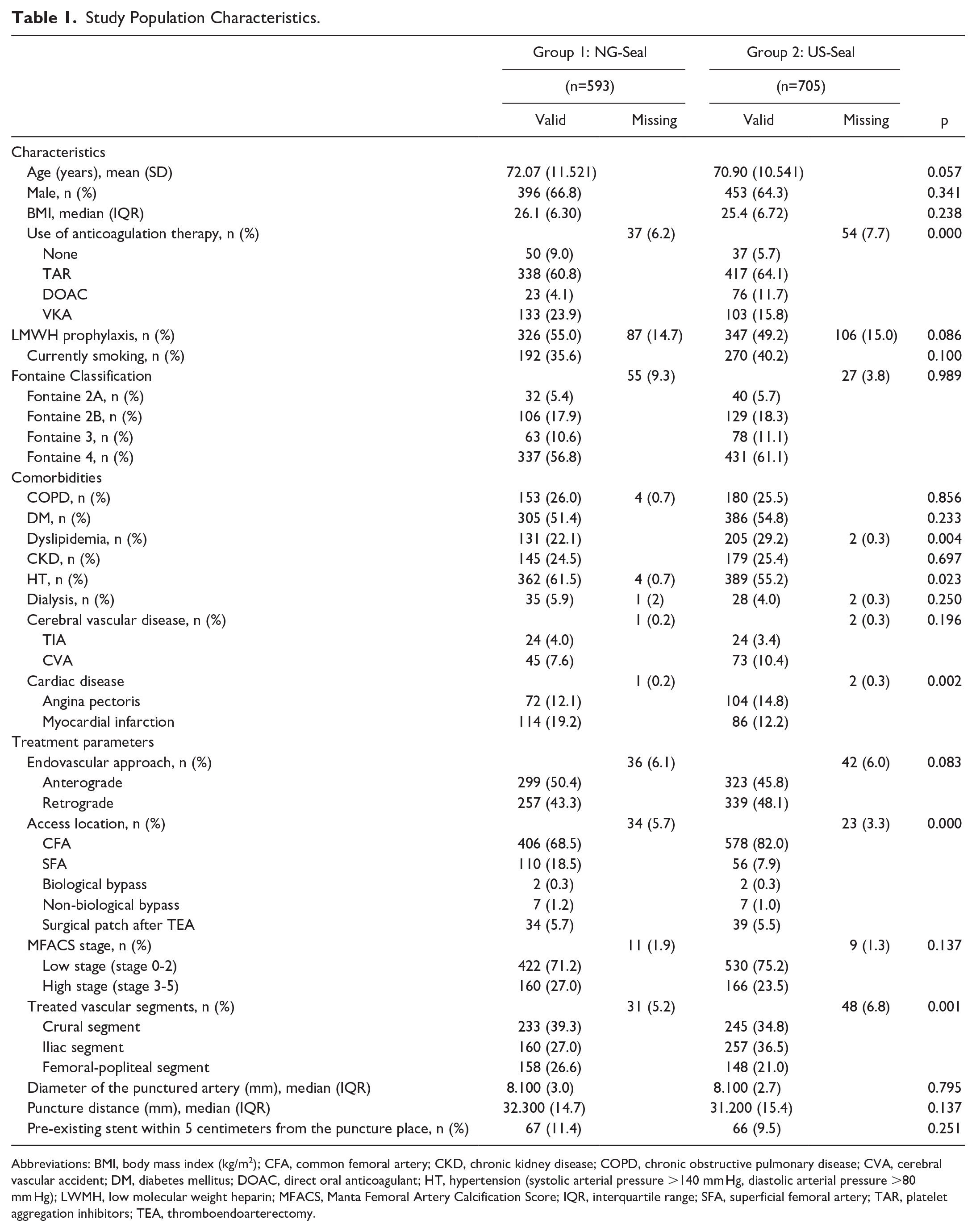
Abbreviations: BMI, body mass index (kg/m2); CFA, common femoral artery; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; CVA, cerebral vascular accident; DM, diabetes mellitus; DOAC, direct oral anticoagulant; HT, hypertension (systolic arterial pressure >140 mm Hg, diastolic arterial pressure >80 mm Hg); LWMH, low molecular weight heparin; MFACS, Manta Femoral Artery Calcification Score; IQR, interquartile range; SFA, superficial femoral artery; TAR, platelet aggregation inhibitors; TEA, thromboendoarterectomy.
Study Variables and Outcomes
The primary outcomes of this study were vascular access complications registered in HiX and confirmed in PACS. Complications registered were bleeding, hematoma equal or larger than 5 cm at access site, pseudoaneurysm at access site, stenosis or occlusion at access site, infection at access site, dissection or occlusion at access site, and punction-related death. Complication rates were divided into minor complications, major complication, and overall complications (major and minor complications combined). The minor complication rate included complications without additional treatment, complications with additional manual compression and compression bandaged needed and prolonged immobilization longer than 2 hours, conservative treatment after infection at access site, and antibiotic treatment after infection at access site. The major complications were defined as the need for additional surgical, endovascular, or percutaneous (eg, thrombin injection) treatment or punction-related death.
Furthermore, data on potential risk factors were collected. The risk factors history of cerebral vascular or cardiac disease, hypertension (HT), use of oral anticoagulation, current smoking behavior, sex, and endovascular approach (antegrade vs retrograde) were collected from patient dossiers in HiX. The diameter of the punctured artery (in mm) and the distance between the skin access and the punctured arterial wall (in mm) were measured on computed tomography (CT) in PACS. Femoral arterial calcification was scored on CT using the MANTA Calcification Score (MFACS).18,19 The MFACS was further divided into the compound variables such as low MFACS (stage 0, 1, and 2) and high MFACS (stage 3, 4, and 5). This subdivision was made, as the majority of the included procedures was classified as MFACS stage 0 to 4 and only the minority of the procedures was classified as severely calcified. Other potential risk factors identified on the CT were treated segments, access location, and pre-existing stent within 5 cm from the puncture place. Definitions of all obtained variables can be found in Appendix A.
Statistical Analyses
Sample size calculation was based on the complication rate of the Angio-Seal as mentioned in the literature. Currently, this rate varies from 2.5% to 4.45%.9,20,21 To detect a decrease in the complication rate of 60% with a standard error of 0.05 and a power of 80% (5% complication rate without ultrasound; 2% complication rate with ultrasound), a total of 1176 femoral vascular access closures must be included.
All obtained variables are presented as frequencies with percentages for categorical variables, as mean standard deviation for normally distributed continuous variables and as median±interquartile range for not-normally distributed continuous variables. For comparison of the continuous data between the 2 treatment groups, either the independent t-test or the Mann-Whitney U-test was used. In the case of categorical data, treatment groups were compared using the Pearson’s chi-square test.
Different complication rates were compared between the US-Seal group and the NG-Seal group and presented in Table 2. Multivariable binary logistic regression was used to obtain odds ratios (ORs) and their 95% confidence intervals (CIs) for the complication rate in ultrasound-guided placement of the Angio-Seal (Table 3). This multivariate analysis tested the OR for all potential predictors for vascular complications with a significance of p≤0.2 in the univariate analysis (Appendix B, Appendix C, Appendix D). This statistical analysis was performed for all complications combined, as well as minor and major complications separately. In order to visualize the differences in complication rates between the subgroups of low and high MFACS, Table 4 was added.
Complication Rates.

No need for further treatment, need for manual compression and compression bandage needed and prolonged immobilization, conservative treatment, or antibiotics.
Additional endovascular treatment, additional surgical treatment, additional transfusion, and punction-related death.
Multivariate Binary Logistic Regression of Overall, Minor, and Major Complications.

Minor and major complications combined.
No need for further treatment, need for manual compression and compression bandage needed and prolonged immobilization, conservative treatment, or antibiotics.
Need for additional endovascular treatment, additional surgical treatment, additional transfusion, and punction-related death.
Abbreviations: CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; CVA, cerebral vascular accident; HT, hypertension (systolic arterial blood pressure 140 mm Hg, diastolic arterial blood pressure >80 mm Hg); MFACS, Manta Femoral Artery Calcification Score; IQR, interquartile range; TIA, transient ischemic attack.
Subgroup Analysis.
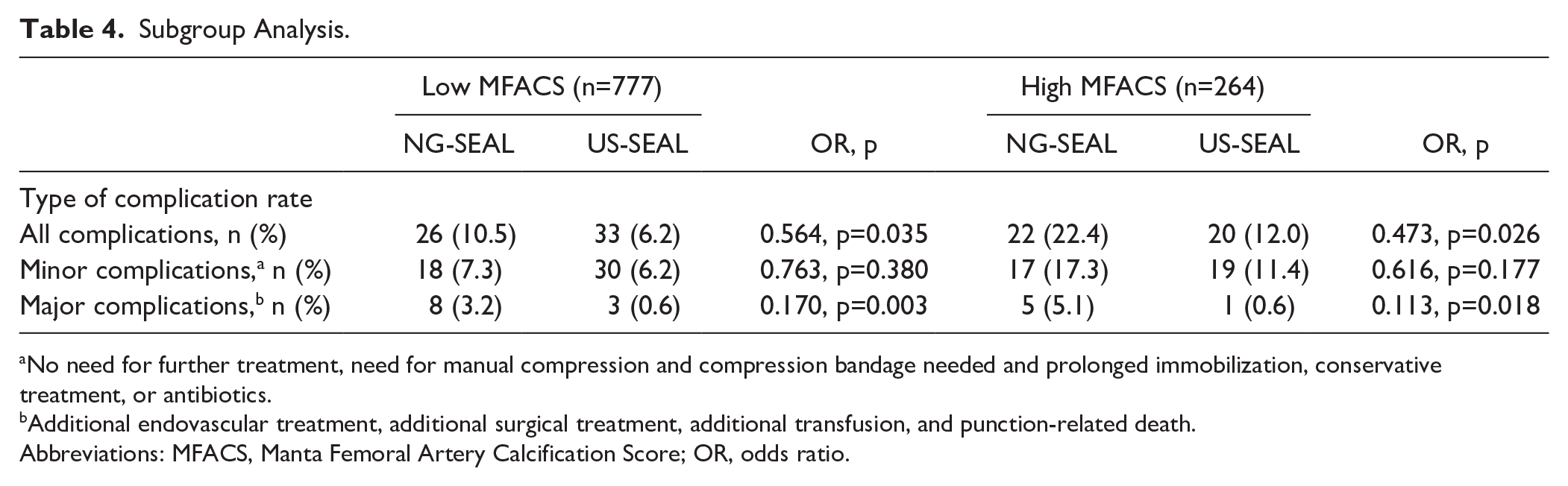
No need for further treatment, need for manual compression and compression bandage needed and prolonged immobilization, conservative treatment, or antibiotics.
Additional endovascular treatment, additional surgical treatment, additional transfusion, and punction-related death.
Abbreviations: MFACS, Manta Femoral Artery Calcification Score; OR, odds ratio.
Procedural Details
The complete procedure was performed by 2 professionals (2 radiologists, a radiologist and a resident/fellow interventional radiology or radiologist and vascular surgeon/vascular surgery resident/fellow). The procedural introduction sheath was exchanged for the 6 or 8 Fr Angio-Seal sheath. Hereafter, all steps were done under continuous ultrasound guidance in the long axis. The position of the sheath was visualized using ultrasound, and the Angio-Seal-carrier was advanced into the sheath. The sheath was withdrawn over the carrier until the anchor was deployed in the center of the arterial lumen as close to the anterior wall as possible to prevent anchoring of the device against the posterior wall. Then, the anchor was locked against the sheath and pulled against the anterior vessel wall. Hereafter, the Angio-Seal deployment was completed following standard procedure.
Results
Study Population
In the period January 2017 until December 2018 and January 2020 until December 2022, 2224 punctures were performed. Eventually, 1298 procedures were analyzed in 826 patients (Figure 1) and subsequently divided into the NG-Seal group (n=593) and the US-Seal group (n=705).
The baseline characteristics of the treated patients are summarized in Table 1. The US-Seal group showed a higher use of oral anticoagulants (p=0.000) and a lower percentage of comorbidities dyslipidemia (p=0.004), cardiac disease (p=0.002), and HT (p=0.023). The US-Seal group showed a higher number of procedures with the CFA as access location, whereas the SFA was the access location in the majority of the procedures within NG-Seal group (p=0.000). In the NG-Seal group, most procedures were performed in the crural segment, whereas the majority of the procedures in the US-Seal group was performed in the iliac segment (p=0.001).
Table 2 presents the overall complication rates and the complication rates divided into major and minor complications. The US-Seal showed significantly lower complication rates for overall complication rate (7.5% vs 12.6%, p=0.002) as well as the minor complication rate (7.0% vs 9.9%, p=0.001) and major complication rate (0.6% vs 2.7%, p=0.001).
Multivariate Binary Regression of Vascular Access Complications
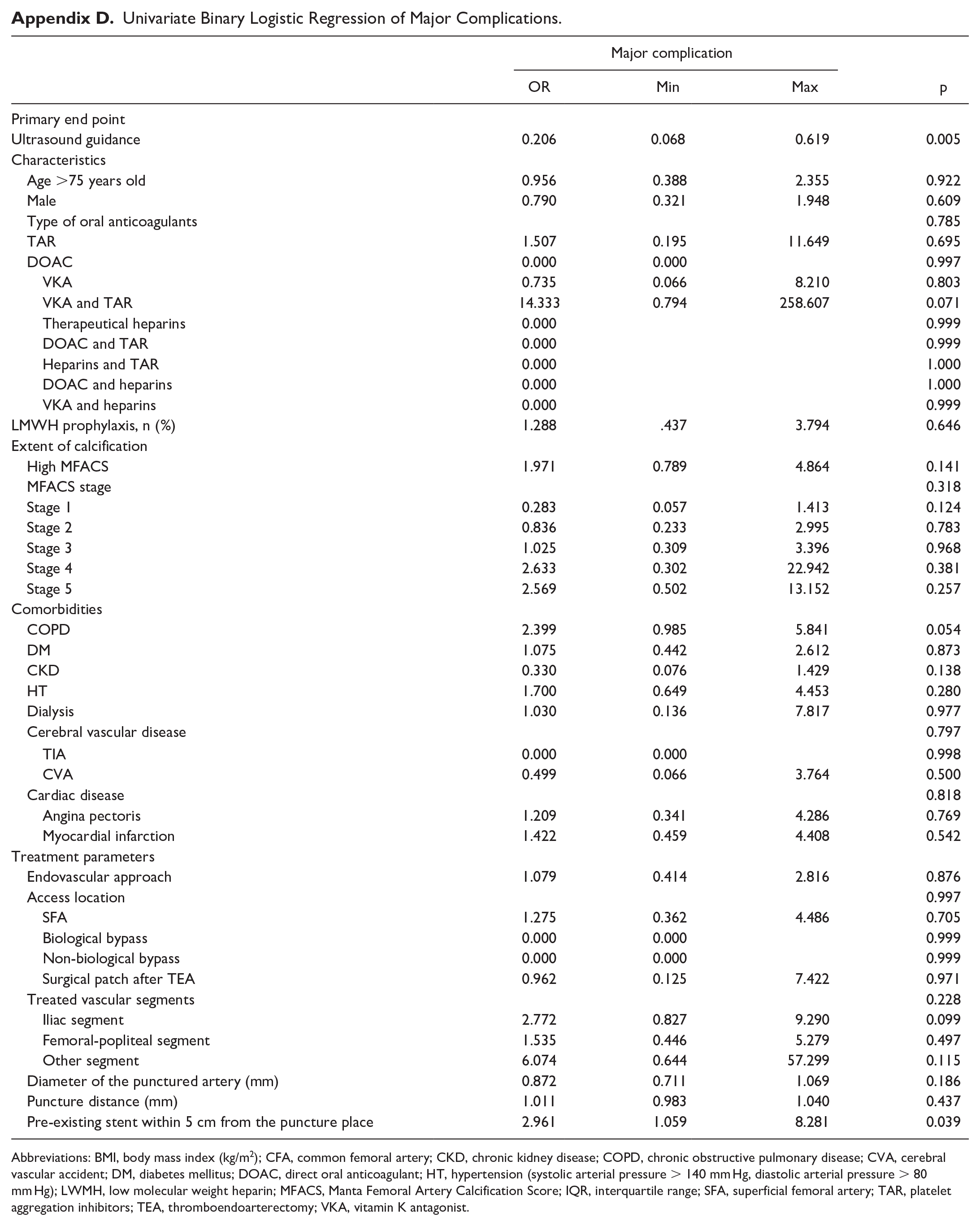
The univariate analysis of the association between all vascular access complications and potential risk factors reveals a significant result for MFACS high vs low (OR=2.059; p=0.000) and HT (OR=1.543; p=0.028). Cerebral vascular disease (p=0.122), sex (p=0.132), myocardial infarction (p=0.098), endovascular approach (p=0.111), and diameter of the punctured artery (p=0.10) were included in the multivariate analysis based on a p-value<0.20 (Appendix B). Multivariate binary logistic regression (Table 3) shows a significant advantage in terms of less overall complications in group 2 with OR of 0.605 (95% CI=0.408-0.896; p=0.012).
Multivariate binary logistic regression (Table 3) shows an OR of 0.210 (95% CI=0.070-0.635; p=0.005) for major complications in the ultrasound-guided test group. Multivariate binary logistic regression (Table 3) shows an OR of 0.696 (95% CI=0.459-1.056; p=0.088) for minor complications in the ultrasound-guided test group.
Subgroup analysis comparing the effects of ultrasound guidance between cases with low and high MFACS (Table 4) shows persistent statistical significance of the advantage in terms of overall complications and lower complications in both the low and high MFACS groups.
Discussion
Main Findings
As PAD is increasingly treated with endovascular techniques using VCDs, the importance for a reliable closure of the vascular access site is of great importance to prevent health costs. This study shows that (1) ultrasound guidance during the placement of the Angio-Seal device decreases the overall amount of overall vascular access complications during the first 3 months after vascular access from 12.6% to 7.5%, and major complications from 2.6% to 0.6%; (2) furthermore, ultrasound guidance is a significant predictor for major and overall complications; and (3) in addition, a high degree of femoral calcifications defined as MFACS stage 3 to 5 can be identified as an individual risk factor for the overall complication rate and minor complication rate after femoral vascular closure. However, ultrasound guidance leads to a significant decrease in both major as well as overall complications when looking at the groups with high and low MFACS separately.
This beneficial role of ultrasound guidance during placement of the VCD can be explained by the fact that this imaging technique is able to visualize the footplate of the Angio-Seal during deployment of the device. Complications are often caused by wrongly placed VCDs or direct failure during deployment.8,14,15 This technical failure is more common in severely calcified vascular access sites due to the inability to secure the VCD in these femoral arteries.18,22 This could explain the role of high femoral calcification score as individual predictor for vascular access complications. In addition, ultrasound guidance facilitates the real-time diagnosis of potential complications such as arterial occlusion and/or Angio-Seal dislodgement. It also allows for visualization of the foot plate, which aids in the manipulation of the foot plate away from the posterior wall, if necessary. Furthermore, endovascular bail-outs for Angio-Seal complications have been described in the literature.8,23 Moreover, if necessary, the prompt diagnosis of complications enables timely and adequate surgical consultation. The decrease in minor complication rate did not reach statistical significance. As 95.3% of the minor complications were classified as a hematoma at access site within 3 months of the procedure, a possible explanation could be that the minor complication rate in this study was mainly intraprocedural-driven, instead of driven by post-procedural device failure. This explanation is supported by the previous literature showing a procedural hematoma risk during endovascular procedures, regardless of post-interventional VCD deployment failure. 5
This study specifically looked at the difference in complication rates of ultrasound-guided and blind placement of the Angio-Seal VAD in PAD patients. Owing to the novelty of this concept, limited literature is available and the currently available data are subject to heterogeneity, resulting in a wide variety of definitions, outcome measurements, and type of procedures studied. 24 However, the significant decrease in seal-related complication rates and negative predicative role of ultrasound guidance is in concordance with the results of previous studies comparing ultrasound-guided placement of different VCDs with blind placement.14,18,22 Literature also reports the increased vascular complication rate seen in patients with MFACS equal or above stage 3. 18
Several studies investigated the complication rate of conventional blind placement of the Angio-Seal extensively. The overall complication rate of 12.3% as seen in this study is in line with this earlier conducted research5,18,24 However, several of these studies also show lower complication rates after placement of this specific seal.9,25 –28 This could be explained by the large variety in study design and their retrospective character, making these studies subject to bias and decreasing their comparability.
Important limitations to this study are the potential influences of measurement bias and performance bias as a consequence of its retrospective nature. Furthermore, no randomization was performed, potentially leading to a selection bias and residual confounding. Nevertheless, the combined influence of these biases is expected to be small, since primary outcomes were well and all VCDs were placed by highly trained clinicians.
There were several imbalances in patient characteristics between the investigated groups. We attempted propensity score matching to ensure a balanced comparison between the ultrasound-guided and non–ultrasound-guided groups. However, due to the large number of potential confounders, achieving a balanced matched set was not feasible. Instead, we adjusted for potential confounders by including factors associated with the outcome in a univariate analysis in our multivariate logistic regression model.
Another potential limitation is that in our health center, there is always a colleague available for assistance during the Angio-Seal deployment; this might not be the case in other health care centers. The learning curve of the ultrasound-guidance procedure was not evaluated, but based on our practical experience, it is expected to be steep for physicians with ultrasound experience.
In conclusion, this study shows the beneficial role of ultrasound in the prevention of mainly major vascular access complications during placement of the Angio-Seal in patients undergoing endovascular treatment for PAD. Furthermore, vascular access complications tend to be more common in patients with severe arterial calcification. Based on our findings, we recommend using ultrasound guidance as standard of care for endovascular closure using the Angio-Seal closure device in patients with PAD.
Footnotes
Appendices
Univariate Binary Logistic Regression of Major Complications.
| Major complication | p | |||
|---|---|---|---|---|
| OR | Min | Max | ||
| Primary end point | ||||
| Ultrasound guidance | 0.206 | 0.068 | 0.619 | 0.005 |
| Characteristics | ||||
| Age >75 years old | 0.956 | 0.388 | 2.355 | 0.922 |
| Male | 0.790 | 0.321 | 1.948 | 0.609 |
| Type of oral anticoagulants | 0.785 | |||
| TAR | 1.507 | 0.195 | 11.649 | 0.695 |
| DOAC | 0.000 | 0.000 | 0.997 | |
| VKA | 0.735 | 0.066 | 8.210 | 0.803 |
| VKA and TAR | 14.333 | 0.794 | 258.607 | 0.071 |
| Therapeutical heparins | 0.000 | 0.999 | ||
| DOAC and TAR | 0.000 | 0.999 | ||
| Heparins and TAR | 0.000 | 1.000 | ||
| DOAC and heparins | 0.000 | 1.000 | ||
| VKA and heparins | 0.000 | 0.999 | ||
| LMWH prophylaxis, n (%) | 1.288 | .437 | 3.794 | 0.646 |
| Extent of calcification | ||||
| High MFACS | 1.971 | 0.789 | 4.864 | 0.141 |
| MFACS stage | 0.318 | |||
| Stage 1 | 0.283 | 0.057 | 1.413 | 0.124 |
| Stage 2 | 0.836 | 0.233 | 2.995 | 0.783 |
| Stage 3 | 1.025 | 0.309 | 3.396 | 0.968 |
| Stage 4 | 2.633 | 0.302 | 22.942 | 0.381 |
| Stage 5 | 2.569 | 0.502 | 13.152 | 0.257 |
| Comorbidities | ||||
| COPD | 2.399 | 0.985 | 5.841 | 0.054 |
| DM | 1.075 | 0.442 | 2.612 | 0.873 |
| CKD | 0.330 | 0.076 | 1.429 | 0.138 |
| HT | 1.700 | 0.649 | 4.453 | 0.280 |
| Dialysis | 1.030 | 0.136 | 7.817 | 0.977 |
| Cerebral vascular disease | 0.797 | |||
| TIA | 0.000 | 0.000 | 0.998 | |
| CVA | 0.499 | 0.066 | 3.764 | 0.500 |
| Cardiac disease | 0.818 | |||
| Angina pectoris | 1.209 | 0.341 | 4.286 | 0.769 |
| Myocardial infarction | 1.422 | 0.459 | 4.408 | 0.542 |
| Treatment parameters | ||||
| Endovascular approach | 1.079 | 0.414 | 2.816 | 0.876 |
| Access location | 0.997 | |||
| SFA | 1.275 | 0.362 | 4.486 | 0.705 |
| Biological bypass | 0.000 | 0.000 | 0.999 | |
| Non-biological bypass | 0.000 | 0.000 | 0.999 | |
| Surgical patch after TEA | 0.962 | 0.125 | 7.422 | 0.971 |
| Treated vascular segments | 0.228 | |||
| Iliac segment | 2.772 | 0.827 | 9.290 | 0.099 |
| Femoral-popliteal segment | 1.535 | 0.446 | 5.279 | 0.497 |
| Other segment | 6.074 | 0.644 | 57.299 | 0.115 |
| Diameter of the punctured artery (mm) | 0.872 | 0.711 | 1.069 | 0.186 |
| Puncture distance (mm) | 1.011 | 0.983 | 1.040 | 0.437 |
| Pre-existing stent within 5 cm from the puncture place | 2.961 | 1.059 | 8.281 | 0.039 |
Abbreviations: BMI, body mass index (kg/m2); CFA, common femoral artery; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; CVA, cerebral vascular accident; DM, diabetes mellitus; DOAC, direct oral anticoagulant; HT, hypertension (systolic arterial pressure > 140 mm Hg, diastolic arterial pressure > 80 mm Hg); LWMH, low molecular weight heparin; MFACS, Manta Femoral Artery Calcification Score; IQR, interquartile range; SFA, superficial femoral artery; TAR, platelet aggregation inhibitors; TEA, thromboendoarterectomy; VKA, vitamin K antagonist.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
