Abstract
Objective:
Knowledge of hostile factors and their influence on long-term seal in the iliac landing zone is limited. Currently endorsed clinical practice guidelines lack structural evidence on how the iliac landing zone should be assessed in the pre-, intra-, and postoperative phases. The goal of this study was to obtain an international, expert-based consensus on the definition of a hostile iliac landing zone, on how to size and plan stent-grafts to optimize sustainable distal seal, and on the postprocedural follow-up protocol.
Methods:
Delphi consensus methodology was used, involving a panel of international vascular surgeons experienced in endovascular aneurysm repair (EVAR). The first round consisted of open-ended and multiple-choice questions to explore current practices, with subsequent rounds refining statements through a 4-point Likert scale. Consensus was defined as >75% agreement or disagreement, and the analysis included stability testing and strength of consensus.
Results:
The study engaged 77 international vascular surgeons, reflecting diverse geographic locations and hospital affiliations. Consensus was achieved on critical preoperative planning elements for EVAR, including a clear definition for a hostile iliac landing zone. The importance of computed tomography angiography for postoperative follow-up imaging was emphasized, including evaluating distal seal length and recommending specific timing for follow-up computed tomography scans and intervention strategies for diminishing iliac seal.
Conclusions:
This international expert-based Delphi consensus establishes a comprehensive set of consensus-driven recommendations focused on the definition and management of hostile iliac landing zones in EVAR. The key recommendation of this study is the definition of a hostile iliac landing zone as short (<15 mm), wide (>24 mm), or conical (>10% diameter difference along the landing zone). Although consensus was achieved on several critical aspects, the study also reveals ongoing debates and considerations that warrant further exploration, including how to tackle diminishing seal without a type IB endoleak.
Clinical Impact
This Delphi consensus introduces a standardized definition of a hostile iliac landing zone as short (<15 mm), wide (>24 mm), or conical (>10% diameter difference), clinicians now have a clearer framework for assessing complex anatomies. This study provides a comprehensive set of consensus-driven recommendations focused on the definition and management of hostile iliac landing zones in EVAR which gives guidance where current guidelines lack specificity, particularly for distal iliac sealing. The study also reveals ongoing debates and considerations that warrant further exploration, including how to tackle diminishing seal without a type IB endoleak.
Keywords
Introduction
Endovascular aneurysm repair (EVAR) has emerged as the preferred therapeutic option for treating abdominal aortic aneurysms, especially in high-risk patients. 1 The Achilles’ heel of EVAR is the occurrence of type IA and IB endoleaks with the subsequent need for lifelong follow-up and reinterventions. 2 Durability of EVAR remains a concern, especially in patients with a longer life expectancy. Also, cost-effectiveness of EVAR is greatly impacted by the need for secondary interventions. Sustainable seal of the main body and limbs is crucial to obtaining a durable result.
Challenging anatomy of the infrarenal aortic neck has been addressed in numerous studies (including a Delphi consensus), specifically focusing on the concept of a “hostile neck.”3–6 These studies highlight the importance of accurate sizing and planning, but mostly focus on the infrarenal neck. The criteria for the proximal landing zone are also well defined in the instructions for use of commercially available endografts. For the distal (iliac) landing zone, however, there is less knowledge of hostile factors and their influence on long-term seal. 2 Currently endorsed clinical practice guidelines lack structural evidence on how the iliac landing zone should be assessed in the pre-, intra-, and postoperative phases.7–9
The goal of this study was to obtain an international, expert-based consensus on the definition of a hostile iliac landing zone, on how to size and plan stent-grafts to optimize sustainable distal seal, and on the postprocedural follow-up protocol.
Materials and Methods
Study Design
The Delphi consensus methodology, which was used in this study, is a recognized and well-established approach for attaining consensus among a panel of experts on subjects lacking empirical evidence. 10 In this study, a panel received a series of statements on the landing zone of the common iliac artery, and feedback from the participants was incorporated through several rounds until consensus was reached.
Before the panel was selected, a steering committee was assembled comprising clinical researchers and vascular surgeons with extensive experience in EVAR and Delphi consensus. The steering committee was responsible for the design of the study, including selection of panelists, formulation of the questions and statements and their reformulation throughout the rounds, as well as being responsible for the statistical analysis, interpretation of the results, and composition of this report.
The first round of this Delphi consensus consisted of open-ended and multiple-choice questions aiming to investigate the current practices and preferences of the panelists. These questions were carefully selected by the steering committee based on knowledge deficits in the current guidelines and available literature.7,9 Panel members had the opportunity to suggest additional statements and to comment on all questions in the first round. The steering committee then formulated a list of statements with a 4-point Likert scale (fully agree, agree, disagree, fully disagree) for the second round.
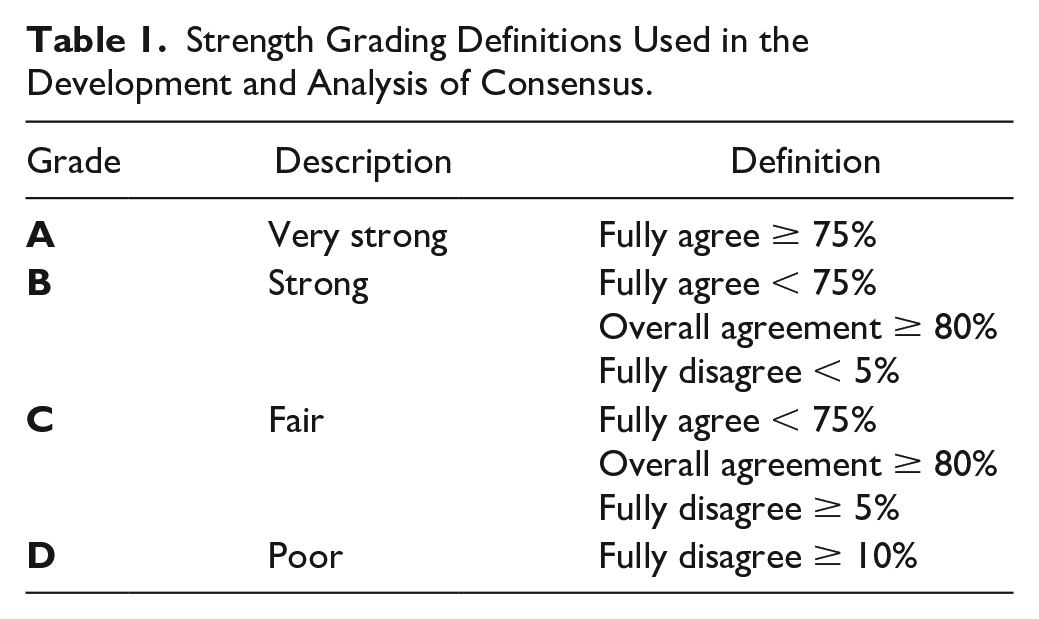
Agreement (fully agree, agree) of ≥75% of the panelists was considered a consensus to agree, and disagreement (disagree, fully disagree) of ≥75% of the panelists was considered negative consensus. Panelists who disagreed with a statement were asked to elaborate why they disagreed. Statements that did not reach 75% consensus were evaluated, including the reasons for disagreement. The steering committee rephrased these statements to aim for consensus in the third round. If consensus was not probable at the end of the second round, the statement was withdrawn. Consensus was set as a closing criterion for the statements. All statements from the second round that reached consensus were repeated in the third round to test for group stability. Consensus strength was graded based on the percentage of full and overall agreement (Table 1).
Strength Grading Definitions Used in the Development and Analysis of Consensus.
Expert Panel
The steering committee identified a panel of international vascular surgeons experienced in the field of EVAR. These panel members were selected either based on previous publications in the field or from suggestions from the steering committee. An attempt was made to gather a heterogeneous group of vascular surgeons with different transatlantic nationalities who were practicing in academic and teaching hospitals. Experts who did not respond to a round were excluded from participating in subsequent rounds. The steering committee members were not part of the expert panel.
Data Collection
The Delphi consensus was conducted via online surveys, which were completed and collected using REDCap (Vanderbilt University, Nashville, TN, USA). Nonresponding panelists received reminder emails after 7 and 14 days subsequently. Identity of the participants was only known to the steering committee, and all responses were analyzed in aggregate in compliance with privacy laws. Panelists could withdraw from the Delphi procedure at any time. Institutional Review Board Approval was not required because no patient data were processed for this study. The study complied with European privacy legislation.
Stability Testing
Statistical analysis for stability testing was conducted using SPSS 28.0 statistical software (IBM, Armonk, NY, USA). Group stability was primarily tested using Pearson χ2 testing to conform to the methods of Dajani et al. 11 After this, cross tabs were used to identify the percentage of overlap between agree versus do not agree the respondents’ answers in rounds two and three to support our group stability values. p values were considered statistically significant with an α of <.05.
Results
Expert Panel
The steering committee invited 127 experts to participate in round 1, of whom 82 (65%) agreed to participate and responded to the first questionnaire. Of these 82 participants, 80 (98%) responded to the second round, and 77 (94%) to the third round, and their responses were included in the final analysis. Fifty-four panelists were based in Europe, 19 in Northern America, and four in Asia and New Zealand, representing 17 different countries. Of the 77 panelists, 81% were working in an academic center and 16% in a teaching hospital. Most of the panelists worked in a center where >100 elective procedures may take place annually, and 10 to 30 urgent EVARs are performed annually. The panelists were a mean age of 50.8 ± 8.3 years, and 12% were women (Table 2).
Characteristics of Participating Panelists in the DECIDE Study (n=77).
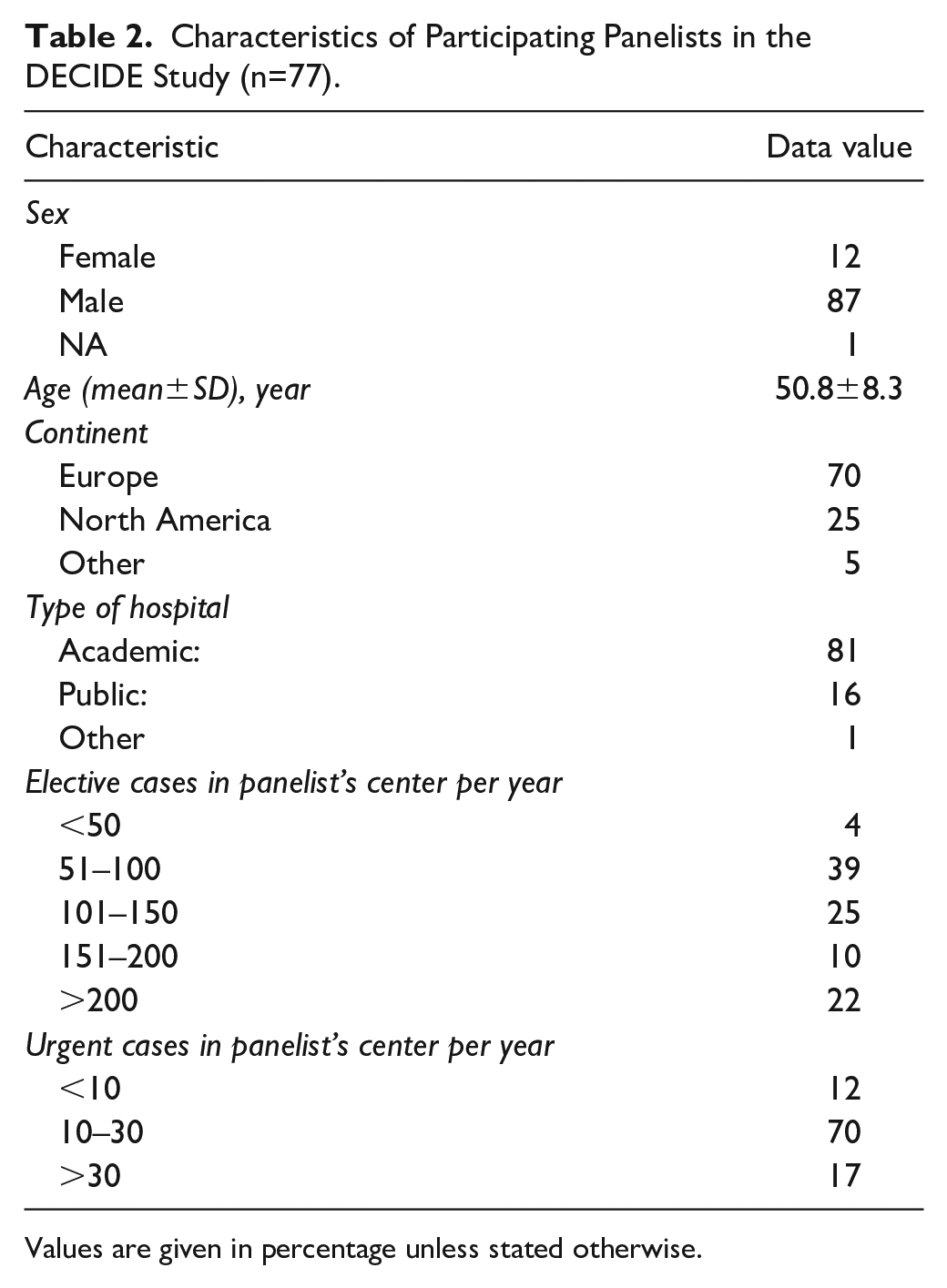
Values are given in percentage unless stated otherwise.
Selection of Statements
The first round consisted of 25 questions about current clinical practice. From this, 37 statements were constructed with an aim for consensus with a 4-point Likert scale, and submitted in the second round (Tables 3–5). After the second round, three statements were modified and resubmitted in the third round, two statements were withdrawn, and 32 statements were resubmitted without modification (Supplementary Table S1).
Statements in the Delphi Consensus Concerning the Planning and Preoperative Evaluation of the Iliac Landing Zone in Endovascular Aneurysm Repair.
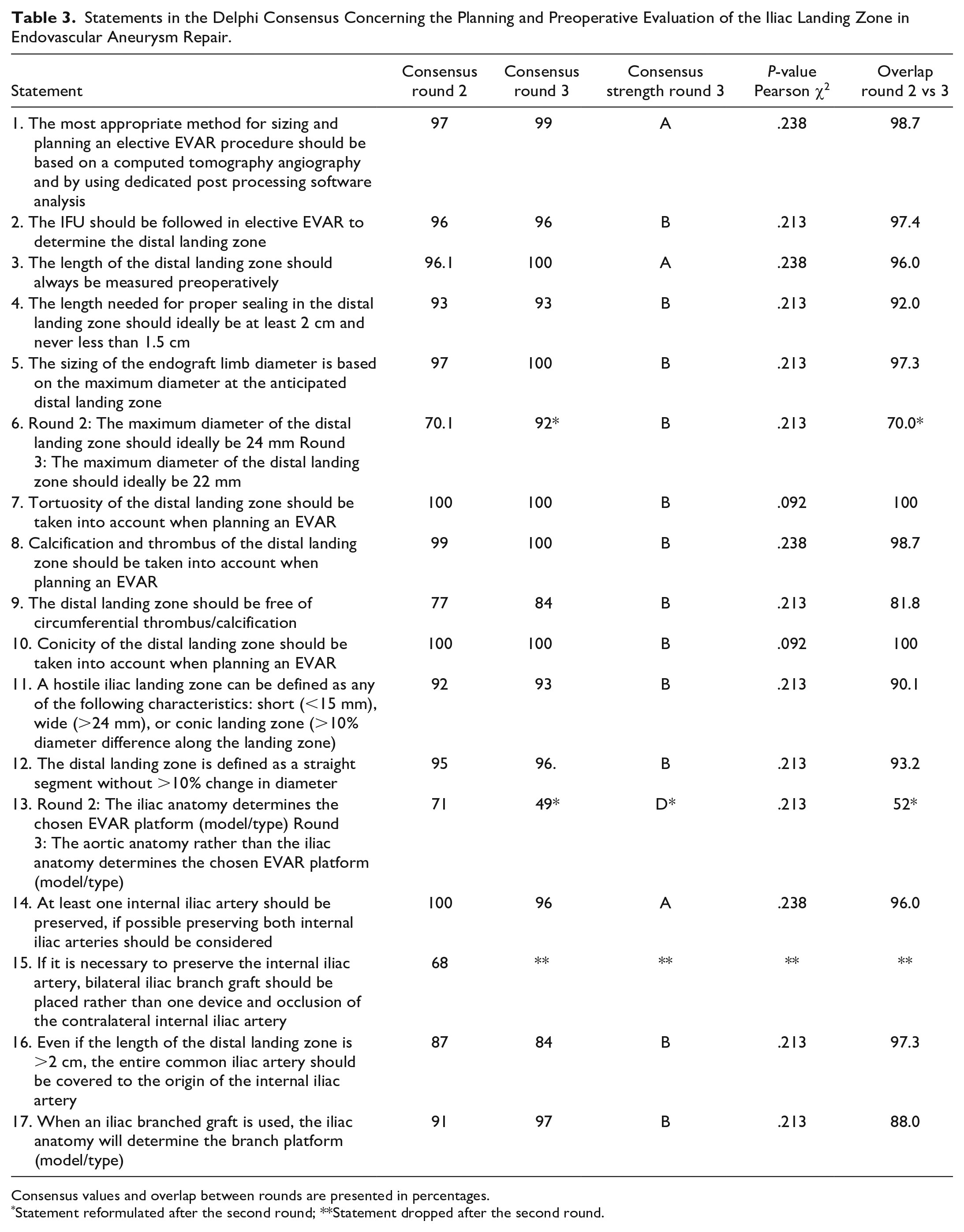
Consensus values and overlap between rounds are presented in percentages.
Statement reformulated after the second round; **Statement dropped after the second round.
Statements in the Delphi Consensus Concerning the Intra-Operative Management of the Iliac Landing Zone in Endovascular Aneurysm Repair.
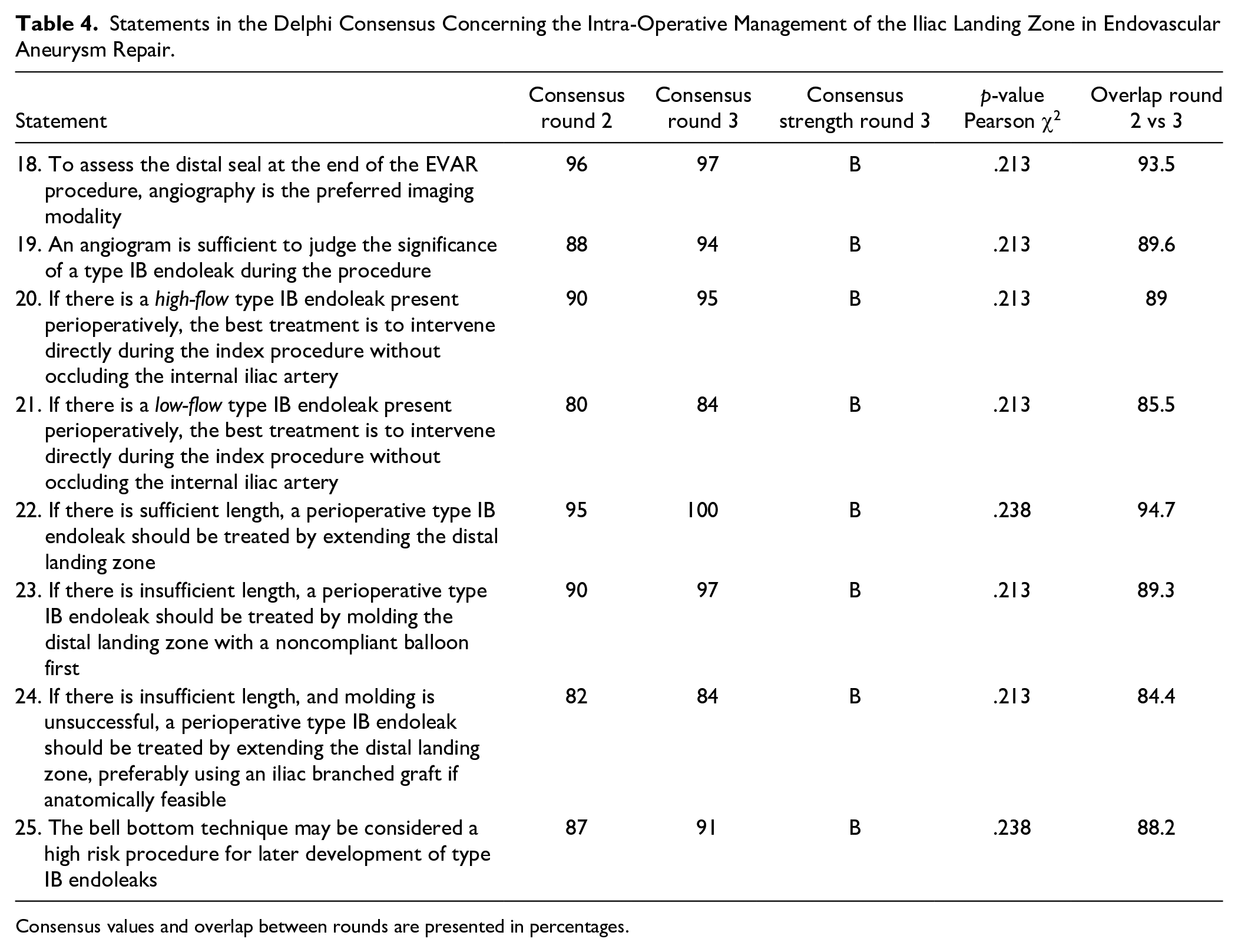
Consensus values and overlap between rounds are presented in percentages.
Statements in the Delphi Consensus Concerning the Postoperative Follow-Up of the Iliac Landing Zone in Endovascular Aneurysm Repair.
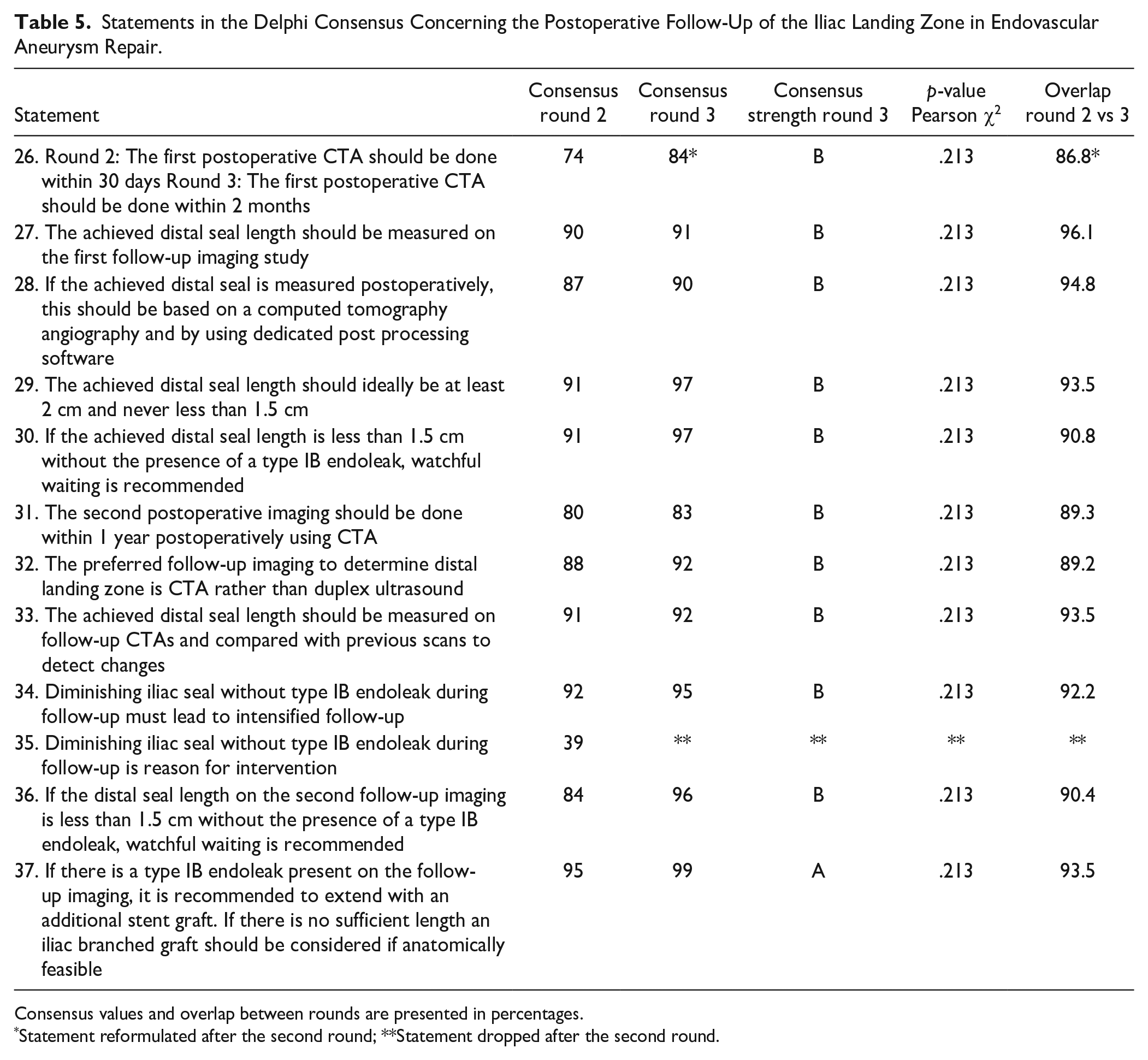
Consensus values and overlap between rounds are presented in percentages.
Statement reformulated after the second round; **Statement dropped after the second round.
Planning and Preoperative Evaluation
There were 17 statements on preoperative planning (Table 3) in which experts agreed that sizing and planning elective EVAR procedures should be based on a computed tomography (CT) angiography (CTA) with dedicated postprocessing software. The experts agreed that instructions for use should be respected to determine the distal landing zone, emphasizing the importance of measuring length and basing endograft diameter on the maximum diameter at the anticipated distal landing zone. There was consensus on the ideal length and diameter; namely, that the distal landing zone should ideally be at least 20 mm and never <15 mm and that the diameter should ideally be <22 mm. Experts agreed on the importance of tortuosity, calcification, thrombus, and conicity when planning EVAR. In addition, the consensus highlighted the need to cover the entire common iliac artery to the origin of the internal iliac artery, with the choice of iliac branched graft determined by iliac anatomy.
The before mentioned statements resulted in an accepted definition for a hostile iliac landing zone: a hostile iliac landing zone can be defined as any distal landing zone with any of the following characteristics: short (<15 mm), wide (>24 mm), or conical (>10% diameter difference along the landing zone).
Intraoperative Phase
All intraoperative statements are shown in Table 4. Completion angiography was the preferred imaging method to assess the distal seal at the end of an EVAR procedure and sufficient to judge the significance of a type IB endoleak during the procedure. The experts also agreed that the best treatment in both high-flow and low-flow type IB endoleaks is to intervene immediately during the index procedure. When sufficient length remains in the distal common iliac artery, an intraoperative IB endoleak should be treated by extending the distal landing zone, and when the length is insufficient, it should be treated by molding with a noncompliant balloon first. When the length is insufficient and molding is unsuccessful, an intraoperative type IB endoleak should be treated by extending the distal landing zone, preferably using an iliac branched graft if anatomically feasible. The bell-bottom technique is considered a high-risk procedure for later development of type IB endoleaks.
Postoperative Phase
The full set of postoperative statements and outcomes are shown in Table 5. Key recommendations that reached consensus for imaging follow-up were that the first postoperative CTA should be done within two months after EVAR and should include measurement of the distal seal length. The distal seal between the endograft limb and iliac artery should be ideally at least 20 mm, but not <15 mm. The second postoperative CTA should be within 1 year, and the distal seal should be compared with previous scans to detect changes. If there is diminishing seal without endoleaks, intensified follow-up and imaging is recommended.
Discussion
This Delphi consensus has established the novel definition of a hostile iliac landing zone as short (<15 mm), wide (>24 mm), and/or conical (>10% diameter difference along the landing zone). This provides a clear framework for identifying challenging anatomies and harmonizing reporting in future research.
Iliac distal landing zone problems, such as type IB endoleaks, have been vastly underreported in the scientific literature,2,12 even though type I endoleaks are the leading cause of late rupture after EVAR.13,14 Even though current international guidelines represent a commendable effort to summarize the sometimes limited literature, they acknowledge that some recommendations are based on low level evidence. When guidelines are derived from lower levels of evidence, it can be helpful to consider the insights of key experts in the field. A lot is still unknown, and although this Delphi consensus sheds some light on missing information, further research is needed to fill in the blanks in our knowledge regarding gray areas of evidence, as detailed below.
Preoperative Considerations
Consensus was achieved on various preoperative aspects, such as the use of CTA with dedicated postprocessing software for sizing and planning, which is in line with literature and the European Society for Vascular Surgery (ESVS) and Society of Vascular Surgery (SVS) guidelines.9,15,16 It would be optimal, though, to have a structured protocol with consideration of anatomical factors, such as tortuosity, calcification, and thrombus, to highlight a unified approach to planning to prevent complications in challenging iliac landing zones.17,18 In the current ESVS guidelines no clear cut-off values for safe landing zones are given in the recommendations. Instead the recommendation is that EVAR outside the IFU’s is not recommended in elective setting, something that is supported with low level evidence. 9 In this Delphi consensus a diameter of >24 mm has been defined as hostile, and consensus was reached that the maximum diameter of an ideal landing zone should be <22 mm. This is not mirrored in the current IFUs included in the ESVS guidelines, which give a range of 18 to 25 mm as a maximum diameter of the common iliac artery. 9 Though these diameters are found to be acceptable in the IFU, the experts believe there is a significant risk in exceeding 22 mm diameter. This is in line with literature showing that landing in common iliac arteries with a diameter >22 mm is prone to type IB endoleaks.19,20 The IFUs also consider a length of 10 to 20 mm as a safe landing zone whereas the consensus by the experts was that <15 mm is considered a hostile landing zone. 9 The guidelines suggest an iliac fixation length of 20 mm, with some evidence indicating that a length greater than 20 mm may reduce the risk of proximal stent graft migration.9,17 However, this is not an official recommendation due to the lack of high level evidence.
Intraoperative Management
In the interaoperative phase, a clear preference for angiography to assess the distal seal was shown, which was consistent with the current practices of a large portion of our Delphi panel.
However, it is important to acknowledge that cone-beam CT has gained attention worldwide as an intraoperative imaging modality, particularly for its ability to detect endoleaks and assess the circumferential seal. Some panelists also mentioned the use of intravascular ultrasound (IVUS), a contrast-free alternative occasionally employed for intraoperative imaging. 21 While angiography remains the predominant method among our panelists, there is growing recognition that cone-beam CT and possibly IVUS offer valuable complementary options, which may eventually play a larger role in intraoperative management when availability increases.
Consensus was reached on immediate intervention for an intraoperative type IB endoleak, even when it is considered low flow. There is, however, no solid evidence that shows whether a low-flow type IB endoleak will or will not resolve without immediate intervention. 19 Future studies should provide more clarity about the urgency to intervene. In line with the recommendations of the current ESVS guidelines, the preferred method of treating a IB endoleak during the index procedure depends on the landing zone, where elongation without stenting over the internal iliac artery is preferred when the landing zone is suitable. 9 There is a cautionary note against the bell-bottom technique, in line with literature showing flared iliac limbs increase the risk of a type IB endoleak. 22
Postoperative Follow-Up
Postoperatively, the emphasis on CTA for follow-up imaging and criteria for evaluating distal seal length set a standard for long-term assessments. In the current ESVS guidelines, it is advised that iliac-related factors including sealing length should be included in the follow-up strategy. 9 The panelists agreed that changes in the distal landing zone are best viewed using CTA and should be compared with baseline findings during follow-up. This is supported by recent literature that shows how the iliac seal may diminish over time due to graft displacement and progression of disease in the iliac arteries, making it important to evaluate the sealing zone in detail, preferably using CTA.19,23 Exact evaluation of the distal sealing zone and modifications over time on cross-sectional CTA may be difficult to objectify. This opens the room to implementation of more accurate post processing analytic methods. Although follow-up schedules vary greatly in different centers, the panelists did agree that the first CTA should be done within two months and the second within the first year. 24 This is more in line with the guidelines of the SVS and the American Heart Association that advise obtaining a CT after 1 year, in contrast to the ESVS guidelines, which advise obtaining a CT within 5 years if the patient is deemed at low risk for complications.7–9
Consensus was also reached on how to best deal with a diminishing distal seal without clear type IB endoleaks. The guidelines advise treating type IB endoleaks promptly; however, they do not offer advice on how to manage a diminishing seal, merely that compromised sealing zones may be considered for intervention.7,9 The expert panel agreed that follow-up and imaging should be intensified when the seal diminishes. There was no consensus on when and how to intervene when the seal diminishes, something that is also lacking in the current guidelines. The implementation of intensified surveillance and how to manage this was not investigated in this study, but should be a topic for further research to improve tailored care for EVAR patients to be able to make recommendations at what stage an intervention is deemed most appropriate.
Overall, there is added value in structuralizing pre- and postoperative imaging and assessing the iliac landing zone. This Delphi consensus provides a basis for future guidelines and shows where solid evidence is lacking concerning the iliac landing zone. Level 1 evidence, consisting of randomized controlled trials, is challenging in this field. Larger retro- and prospective multicentre registries studies investigating the iliac landing zone and type IB endoleaks are needed to increase the knowledge. Until these studies are conducted, the advice given by the expert panel represents a practical source of reference for practicing physicians. A Delphi consensus approach highlights existing knowledge gaps rather than providing definitive conclusions. We emphasize that our findings are intended to guide future research and discussions, serving as a catalyst for the development of more robust, evidence-based guidelines as further empirical data becomes available.
Study Limitations
The findings from this study must be interpreted within the context of its limitations. First, the Delphi methodology may be subject to intrinsic shortcomings. Delphi studies have been criticized because the researchers choose the included items, thereby potentially introducing bias. To counteract this, the expert panel had the opportunity to modify and comment on statements or suggest additional ones. Second, the inclusion criteria for the experts meant a random selection was not feasible; thus, a large, preselected group of international experts proposed by the core team was invited, potentially introducing selection bias because they might not fully represent actual worldwide expertise. Because only 77 of our intended 127 panelist responded to all rounds, there was more homogeneity in our panel despite our efforts to create a diverse panel. Even though the non-response rate between the rounds was low, the initial refusal of participation in the first round might cause a selection bias.
Our primary focus was endoleaks and the failure of seal; hence, there is an absence of statements regarding iliac limb occlusion and patency, which represent an additional clinical concern. Although some statements did not reach consensus and were rejected from the final formulation, this may not be equivalent to the assumption they would not address clinically relevant questions. Therefore, consensus statements should only be considered as evidence in progress to be further investigated and confirmed by clinical studies, if possible, and need to be implemented in daily practice with proper clinical judgment. To mitigate this limitation, clinical practice guidelines from recognized scientific societies were consulted when available to ensure proposed statements were in agreement whenever possible. In addition, the statements concerning calcifications, thrombus and tortuosity were generic and did not address the different methods for quantifying these morphological parameters. Future studies may dive further into the grading of these parameters and their influence on sealing durability.
Conclusion
This international expert-based Delphi consensus establishes a comprehensive set of consensus-driven recommendations focused on the definition and management of hostile iliac landing zones in EVAR. The key recommendation of this study is the novel definition of a hostile iliac landing zone as short (< 15 mm), wide (>24 mm), or conical (>10% diameter difference along the landing zone). Although consensus was achieved on several critical aspects, the study also reveals ongoing debates and considerations that warrant further exploration, including how to tackle diminishing seal without a type IB endoleak.
Supplemental Material
sj-docx-1-jet-10.1177_15266028241295919 – Supplemental material for Transatlantic Delphi Consensus on the Common Iliac Artery Sealing Zone in Endovascular Aorto-Iliac Aneurysm Repair (the DECIDE Study)
Supplemental material, sj-docx-1-jet-10.1177_15266028241295919 for Transatlantic Delphi Consensus on the Common Iliac Artery Sealing Zone in Endovascular Aorto-Iliac Aneurysm Repair (the DECIDE Study) by Maria-Annette Kooijman, Mario D’Oria, Luca Bertoglio, Isabelle Van Herzeele, Ross Milner, Jean-Paul P.M. de Vries and Richte C.L. Schuurmann in Journal of Endovascular Therapy
Footnotes
Authors’ Notes
The authors would like to thank all members of the DECIDE Study Expert Panel for their invaluable contributions, insights, and dedication to advancing our understanding of best practices in EVAR. Their expertise and commitment were instrumental in shaping the study’s findings, and we are deeply appreciative of their time and efforts.
DECIDE—study collaborative study group (in alphabetical order)
1. Gautam Agarwal (Email: gagarwal@augusta.edu): Department of Surgery, Division of Vascular and Endovascular Surgery, Medical College of Georgia, Augusta University, Augusta, GA, USA.
2. Michele Antonello (Email: michele.antonello.1@unipd.it): Division of Vascular and Endovascular Surgery, Department of Cardiac, Thoracic, Vascular Sciences and Public Health, University of Padua, Padua, Italy.
3. Frederico Bastos Goncalves (Email: f.bastosgoncalves@nms.unl.pt): NOVA Medical School, Faculdade de Ciências Médicas, NMS, FCM, Universidade Nova de Lisboa, Lisboa, Portugal; Hospital de Santa Marta, Centro Hospitalar Universitário de Lisboa Central, Lisboa, Portugal; Hospital CUF Tejo, Lisboa, Portugal
4. Jonathan Bath (Email: bathj@health.missouri.edu): Division of Vascular Surgery, University of Missouri, Columbia, MO, USA.
5. Adam Beck (Email: awbeck@uabmc.edu): Division of Vascular Surgery and Endovascular Therapy, Department of Surgery, University of Alabama at Birmingham, Birmingham, AL, USA.
6. Xavier Berard (Email: xberard@yahoo.fr): Vascular Surgery Department, Bordeaux University Hospital, Bordeaux, France.
7. Colin Bicknell (Email: colin.bicknell@imperial.ac.uk): Imperial Vascular Unit, Imperial College Healthcare NHS Trust and Department of Surgery and Cancer, Imperial College London, London, UK.
8. James Black (Email: jhblack@jhmi.edu): Division of Vascular Surgery and Endovascular Therapy, Department of Surgery, The Johns Hopkins University School of Medicine, Baltimore, MD, USA.
9. Michel Bosiers (Email: michel.bosiers@insel.ch): Department of Vascular Surgery, University Hospital Bern, University of Bern, Bern, Switzerland.
10. Daniela Branzan (Email: daniela.branzan@medizin.uni-leipzig.de): Department of Vascular Surgery, University Hospital Leipzig, Leipzig, Germany.
11. Jacob Budtz-Lilly (Email: jacoblilly@clin.au.dk): Department of Cardiovascular Surgery, Division of Vascular Surgery, Aarhus University Hospital, Aarhus, Denmark
12. Caroline Caradu (Email: caroline.caradu@chu-bordeaux.fr): Department of Vascular Surgery, Bordeaux University Hospital, Bordeaux, France.
13. Stephen Cheng (Email: wkcheng@hku.hk): Division of Vascular Surgery, Department of Surgery, Queen Mary Hospital, The University of Hong Kong, Hong Kong, China.
14. Martin Czerny (Email: martin.czerny@universitaets-herzzentrum.de): Department of Cardiovascular Surgery, University Heart Centre Freiburg-Bad Krozingen, Germany & Faculty of Medicine, Albert Ludwigs University Freiburg, Freiburg im Breisgau, Germany.
15. Randall DeMartino (Email: DeMartino. Randall@mayo.edu): Division of Vascular and Endovascular Surgery, Mayo Clinic, Rochester, MN, USA.
16. Nuno Dias (Email: nunovdias@gmail.com): Vascular Center, Department of Thoracic Surgery and Vascular Diseases, Skåne University Hospital Malmö and Department of Clinical Sciences Malmö, Lund University, Malmö, Sweden.
17. Matthew Eagleton (Email: MEAGLETON@mgh.harvard.edu): Division of Vascular and Endovascular Surgery, Massachusetts General Hospital, Boston, Mass.
18. Wouter van den Eynde (Email: wouter_vandeneynde@hotmail.com): Department of Vascular and Thoracic Surgery, Imelda Hospital, Bonheiden, Belgium.
19. Mark Farber (Email: mark_farber@med.unc.edu): Division of Vascular Surgery, Department of Surgery, University of North Carolina, Chapel Hill, NC, USA.
20. Stefano Fazzini (Email: stefanofazzini@hotmail.it): Vascular Surgery Unit, Biomedicine and Prevention Department, University of Rome Tor Vergata, Rome, Italy.
21. Ciro Ferrer (Email: cferrer@hsangiovanni.roma.it): Vascular and Endovascular Surgery Unit, San Giovanni—Addolorata Hospital, Roma, Italy.
22. Bram Fioole (Email: Fiooleb@maasstadziekenhuis.nl): Department of Vascular Surgery, Maasstad Hospital, Rotterdam, The Netherlands.
23. Thomas Forbes (Email: thomas.forbes@uhn.ca): Division of Vascular Surgery, Peter Munk Cardiac Centre & University Health Network, University of Toronto, Toronto, ON, Canada.
24. Antonio Freyrie (Email: antonio.freyrie@unipr.it): Vascular Surgery, Department of Medicine and Surgery, University of Parma, Parma, Italy.
25. Mauro Gargiulo (Email: mauro.gargiulo2@unibo.it): Department of Vascular Surgery, University of Bologna, DIMEC, Bologna, Italy; Bologna Metropolitan Vascular Surgery Unit, IRCCS University Hospital S. Orsola, Bologna, Italy.
Bob Geelkerken (Email: r.geelkerken@mst.nl): Department of Vascular Surgery, Medical Spectrum Twente, Enschede, The Netherlands; Experimental Centre for Technical Medicine, University of Twente, Enschede, The Netherlands.
27. Stephan Haulon (Email: haulon@hotmail.com): Aortic Center, Department of Aortic and Vascular Surgery, Hôpital Marie Lannelongue, Groupe Hospitalier Paris Saint Joseph, Université Paris-Saclay, Gif-sur-Yvette, France.
28. Joost van Herwaarden (Email: J.A.vanHerwaarden@umcutrecht.nl): Department of Vascular Surgery, University Medical Center Utrecht, Utrecht, The Netherlands.
29. Jan Heyligers (Email: j.heyligers@etz.nl): Department of Surgery, Elisabeth-TweeSteden Ziekenhuis, Tilburg, The Netherlands.
30. Caitlin Hicks (Email: chicks11@jhmi.edu): Department of Surgery, Johns Hopkins University School of Medicine, Baltimore, MD, USA.
31. Andrew Holden (Email: AndrewH@adhb.govt.nz): Department of Interventional Radiology, Auckland Hospital, Auckland, New Zealand.
32. Tal Hörer (Email: Tal.horer@regionorebrolan.se): Faculty of Medicine and Health, Department of Cardiothoracic and Vascular Surgery and Department of Surgery, Örebro University, Örebro, Sweden.
33. Sabrina Houthoofd (Email: sabrina.houthoofd@uzleuven.be): Department of Vascular Surgery, University Hospitals Leuven, Leuven, Belgium
34. Tomasz Jakimowicz (Email: tomj@am.edu.pl): Department of General, Vascular and Transplant Surgery, Medical University of Warsaw, Warsaw, Poland.
35. Andrea Kahlberg (Email: kahlberg.andrea@hsr.it): Division of Vascular Surgery, IRCCS San Raffaele Scientific Institute, “Vita-Salute” University, Milan, Italy.
36. Manar Khashram (Email: manar.khashram@gmail.com): Department of Surgery, University of Auckland, New Zealand; Department of Vascular Surgery, Waikato Hospital, Hamilton, New Zealand.
37. Tilo Kölbel (Email: tilokoelbel@googlemail.com): German Aortic Center Hamburg, University Heart Center, University Hospital Hamburg Eppendorf, Hamburg, Germany.
38. Drosos Kotelis (Email: drosos.kotelis@insel.ch): Department of Vascular Surgery, Bern University Hospital, University of Bern, Bern, Switzerland.
39. Göran Lundberg (Email: goran.lundberg@sll.se): Department of Vascular Surgery, Karolinska Institute, Karolinska University Hospital, Stockholm, Sweden.
40. Thomas Maldonado (Email: Thomas. Maldonado@nyumc.org): Division of Vascular and Endovascular Surgery, NYU Langone Medical Center, New York, NY, USA.
41. Nicola Mangialardi (Email: nikmangialardi@gmail.com): Department of Vascular Surgery, San Camillo-Forlanini Hospital, Rome, Italy.
42. Kevin Mani (Email: kevin.mani@surgsci.uu.se): Department of Surgical Sciences, Section of Vascular Surgery, Uppsala University, Uppsala, Sweden.
43. Tara Mastracci (Email: tara.mastracci@nhs.net): Vascular Surgery, St. Bartholomew’s Hospital, London, UK.
44. Blandine Maurel (Email: blandine.maurel@chu-nantes.fr): CHU Nantes, l’institut du thorax, service de chirurgie cardio-vasculaire, Nantes, France; Université de Nantes, Nantes, France.
45. James McCaslin (Email: james.mccaslin@nhs.net): The Northern Vascular Centre, Freeman Hospital, Newcastle upon Tyne, UK.
46. Bernardo Mendes (Email: Mendes. Bernardo@mayo.edu): Division of Vascular and Endovascular Surgery, Mayo Clinic, Jacksonville, FL, USA.
47. Westley Ohman (Email: ohmanj@wustl.edu): Department of Surgery, Section of Vascular Surgery, Washington University School of Medicine, St. Louis, MO, USA.
48. Matteo Orrico (Email: dr.orrico.matteo@gmail.com): Department of Vascular Surgery, Azienda Ospedaliera San Camillo-Forlanini, Circonvallazione Gianicolense, Rome, Italy.
49. Kenneth Ouriel (Email: kouriel@namsa.com): North American Science Associates, New York, NY, USA.
50. Jean Panneton (Email: pannetjm@evms.edu): Division of Vascular Surgery, Eastern Virginia Medical School, Norfolk, VA, USA.
51. Gianbattista Parlani (Email: parlani.gianbattista@gmail.com): Vascular and Endovascular Surgery Unit, S. Maria della Misericordia Hospital, University of Perugia, Perugia, Italy.
52. Gabriele Piffaretti (Email: gabriele.piffaretti@uninsubria.it): Unit of Vascular Surgery, Department of Medicine and Surgery, University of Insubria School of Medicine, Varese, Italy.
53. Raffaele Pulli (Email: raffaele.pulli@unifi.it): Vascular and Endovascular Surgery—Department of Emergency and Organs Transplantation, “Aldo Moro” University of Bari School of Medicine, Bari, Italy.
54. Michel MPJ Reijnen (Email: mmpj.reijnen@gmail.com): Department of Surgery, Rijnstate, Arnhem and MultiModality Medical Imaging Group, TechMed Centre, University of Twente, Enschede, The Netherlands.
55. Timothy Resch (Email: Timothy.andrew.resch@regionh.dk): Department of Vascular Surgery, Rigshospitalet, Copenhagen, Denmark; Faculty of Health and Medical Sciences, University of Copenhagen, Copenhagen, Denmark.
56. Vicente Riambau (Email: vriambau@gmail.com): Department of Vascular Surgery, Cardiovascular Institute of Hospital Clínic, University of Barcelona, Barcelona, Spain.
57. Sonia Ronchey (Email: sonia.ronchey@gmail.com): Department of Vascular Surgery, San Filippo Neri Hospital, Rome, Italy.
58. Maria Antonella Ruffino (Email: mariaantonellaruffino@gmail.com): Department of Interventional Radiology, Imaging Institute of Southern Switzerland, Ente Ospedaliero Cantonale, Lugano, Switzerland.
59. Marc van Sambeek (Email: marc.v.sambeek@catharinaziekenhuis.nl): Department of Surgery, Catharina Hospital Eindhoven, Eindhoven, The Netherlands.
60. Salvatore Scali (Email: salvatore.scali@surgery.ufl.edu): Division of Vascular Surgery and Endovascular Therapy, University of Florida, Gainesville, FL, USA.
61. Andres Schanzer (Email: andres.schanzer@umassmemorial.org): Division of Vascular Surgery, Department of Surgery, University of Massachusetts Medical School, Worcester, MA, USA.
62. Geert Willem Schurink (Email: gwh.schurink@mumc.nl): Department of Vascular Surgery, Maastricht University Medical Center, Maastricht, The Netherlands.
63. Nicla Settembre (Email: nicla.settembre@yahoo.com): Department of Vascular Surgery, Nancy University Hospital, University of Lorraine, Vandoeuvre les Nancy, France.
64. Matthew Smeds (Email: matt.smeds@health.slu.edu): School of Medicine, Saint Louis University, Saint Louis, MO, USA; Division of Vascular and Endovascular Surgery, Department of Surgery, Saint Louis University, Saint Louis, MO, USA.
65. Glenn Wei Leong Tan (Email: glenn_tan@ttsh.com.sg): Vascular Surgery Service, Department of General Surgery, Tan Tock Seng Hospital, Singapore.
66. Ignace Tielliu (Email: i.f.j.tielliu@umcg.nl): Division of Vascular Surgery, Department of Surgery, University Medical Center Groningen, University of Groningen, Groningen, The Netherlands.
67. Nicola Troisi (Email: troisimd@gmail.com): Vascular Surgery Unit, Department of Translational Research and New Technologies in Medicine and Surgery, University of Pisa, Pisa, Italy.
68. Yamyme Tshomba (Email: yamume.tshomba@unicatt.it): Unit of Vascular Surgery, Fondazione Policlinico Universitario A. Gemelli IRCCS, Università Cattolica del Sacro Cuore, Rome, Italy.
69. Brant W. Ullery (Email: brant.ullery@providence.org): Providence Heart and Vascular Institute, Portland, OR, USA.
70. Çağdaş Ünlü (Email: Cagdas.Unlu@nwz.nl): Department of Vascular Surgery, Noordwest Ziekenhuisgroep, Alkmaar, The Netherlands.
71. Gilbert Upchurch (Email: gib.upchurch@surgery.ufl.edu): Division of Vascular Surgery and Endovascular Therapy, University of Florida College of Medicine, Gainesville, FL, USA.
72. Gian Franco Veraldi (Email: gianfranco.veraldi@aovr.veneto.it): Department of vascular surgery, University Hospital and Trust of Verona, University of Verona-School of Medicine, Verona, Italy.
73. Hence Verhagen (Email: h.verhagen@erasmusmc.nl): Department of Vascular Surgery, Erasmus MC University Medical Centre, Rotterdam, The Netherlands.
74. Eric Verhoeven (Email: eric.verhoeven@klinikum-nuernberg.de): Department of Vascular and Endovascular Surgery, General Hospital Nuremberg, Paracelsus Medical University, Nuremberg, Germany.
75. Fabio Verzini (Email: fabio.verzini@unito.it): Division of Vascular Surgery, Department of Surgical Sciences, University of Torino, A. O. U. Città della Salute e della Scienza, Molinette Hospital, Turin, Italy.
76. Anders Wanhainen (Email: Anders. Wanhainen@surgsci.uu.se): Department of Surgical Sciences, Vascular Surgery, Uppsala University, Uppsala, Sweden.
77. Alexander Zimmermann (Email: Alexander. Zimmermann@usz.ch): Department of Vascular Surgery, University Hospital Zurich, University of Zurich, Zurich, Switzerland.
Prior Presentation
This work has been presented on the Vaatdagen (National vascular surgery conference in the Netherlands) April 2024 where it won the prize for best oral presentation.
Author Contributions
All authors contributed to the design of the study and formulation of the statements. All authors contributed to the critical analysis of data collected. Maria-Annette Kooijman wrote the manuscript and performed the statistical analysis with input from all authors. All authors contributed to the critical revision of the manuscript. All authors read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Luca Bertoglio is consultant for Cook, Artivion and WL Gore. Isabelle Van Herzeele is supported by a Senior Clinical Fellowship of the Fund for Scientific Research, Flanders, Belgium; is involved in education and training duties for Medtronic International Trading Sarl, Tolochenaz, Switzerland. Ross Milner is Consultant for Cydar, CyndRx, Endoron, Endospan, Medtronic, Shockwave, Silk Road, and WL Gore. Jean-Paul P.M. de Vries and Richte C.L. Schuurmann are cofounders of Endovascular Diagnostics B.V., which holds patent rights to the VIA software used in this study. The other authors state that there is no financial/personal interest or belief that could their influence objectivity.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this project was provided by the Jaap Schouten Foundation (JSF). The JSF had no involvement in the study design, collection analysis and interpretation of data. The JSF was not involved in the decision to submit the manuscript for publication.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
