Abstract
Purpose:
To describe the X-over reversed iliac extension technique in a patient with severe peripheral arterial disease (PAD) scheduled for inner branched endovascular aortic repair (iBEVAR).
Technique:
A multimorbid 62-year-old male patient was planned for iBEVAR due to a 58 mm suprarenal aortic aneurysm. The patient had a previous right femoropopliteal bypass and stenting of the left iliac axis. At admission, he presented with recent onset severe left limb claudication, which was attributed to left iliac stent occlusion. To avoid the postoperative compression of the right common femoral artery (CFA) and preserve the patency of the bypass, a single left CFA access, followed by left iliac artery recanalization, was decided. The right iliac axis was catheterized with a Lunderquist wire using X-over access from the left CFA. An iliac extension (ZISL, 24–59, Cook Medical, Bloomington, USA) was reversed and resheathed on back-table and implanted in the right common iliac artery using the X-over technique. The left CFA access was used to complete the remaining steps of the procedure. The predischarge computed tomography angiography confirmed bilateral iliac artery and femoropopliteal bypass patency.
Conclusion:
The X-over reversed iliac extension technique may be applied in selected PAD patients, when undergoing complex endovascular aortic repair.
Clinical Impact
As the number of patients with peripheral arterial disease (PAD) is expected to increase the upcoming decades, out of the box solutions may be needed to assist complex endovascular aortic management. The X over technique, which consist of the contralateral advancement of an on-table reversed iliac limb, was successfully applied in a patient with severe PAD and numerous previous peripheral interventions, who was managed with branched endovascular aortic repair . The X Over technique may provide an additional alternative in well-selected patients with demanding vascular access undergoing complex endovascular aortic procedures.
Keywords
Introduction
Hostile iliac access may affect any decision-making and operative planning of endovascular aortic procedures and in many cases, it may obligate out-of-the-box solutions. 1 Alternative access options and physician-modified endograft (PMEG) have been used in patients with anatomic restrictions for a standard endovascular approach, especially when urgent repair is indicated.1–3 Aortic endografts and iliac components, including iliac extensions and branch devices, have been used out of the instructions for use and modified in situ or on table to adapt to patient’s anatomy and achieve aneurysm sac exclusion.1–3,4
Herein, we report the X-over reversed iliac extension technique, which was used in a patient with severe peripheral arterial disease (PAD) and a suprarenal abdominal aortic aneurysm (sAAA) managed with inner branched endovascular aortic repair (iBEVAR).
Technique
A 62-year-old male patient with an anamnesis of PAD, with a previous right above the knee femoropopliteal (common femoral artery [CFA]-P1 popliteal artery) saphenous vein bypass (Figure 1A) and a left iliac axis stenting, coronary artery disease, with previous percutaneous coronary artery stenting and aorto-coronary bypass grafting, active smoking, dyslipidemia, diabetes type II and hypertension was scheduled for elective treatment of an asymptomatic 58mm sAAA. The preoperative computed tomography angiography (CTA) confirmed in addition the presence of a dorsal penetrating aortic ulcer located at aortic zone 6 (Figure 1B), a 15mm right hypogastric artery aneurysm and a left hypogastric artery occlusion. The patient was under best medical treatment with aspirin 100mg and rosuvastatin/ezetimibe 20/10mg, daily. Considering his medical history and aortic and iliac anatomy, an endovascular management was suggested.
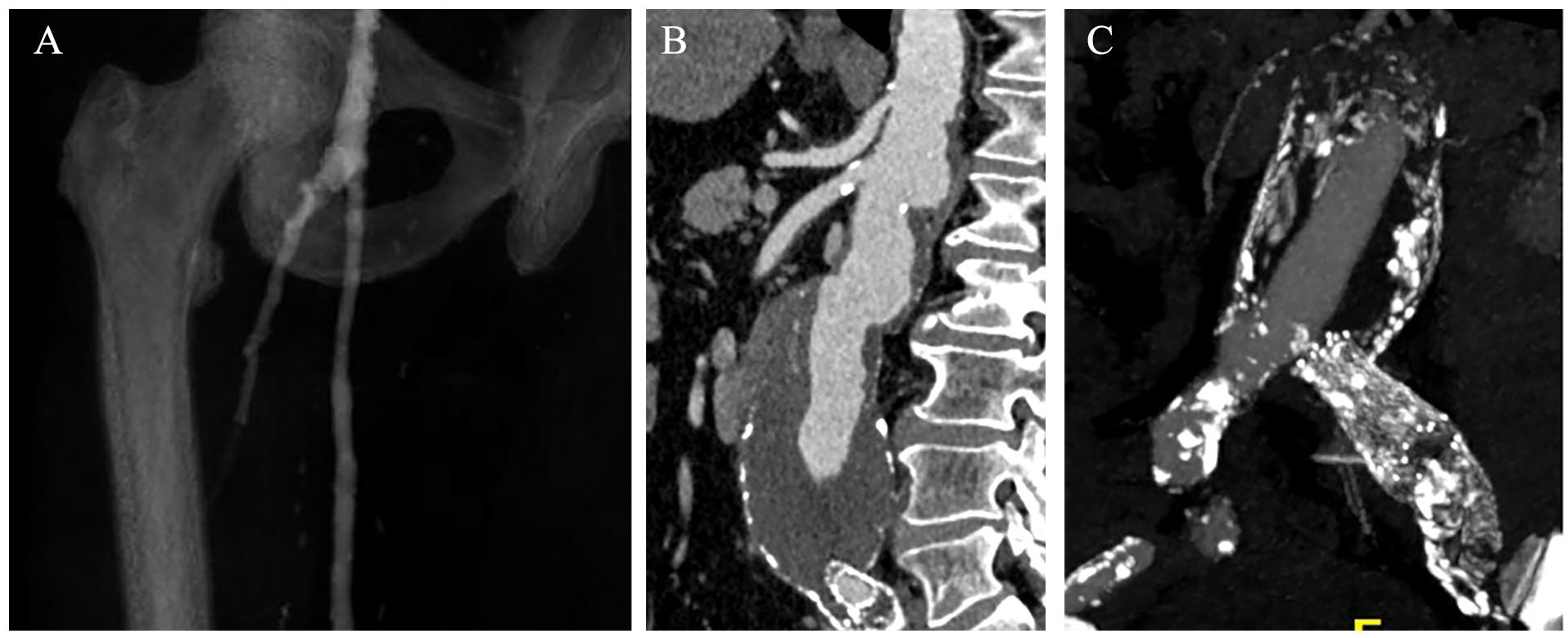
The patient had a history of a right femoropopliteal bypass with saphenous vein graft, which was patent (A), while he had undergone a left iliac angioplasty and stenting 5 years ago. The computed tomography angiography confirmed the presence of a suprarenal abdominal aortic aneurysm of 58mm (B). A total left iliac in-stent thrombotic occlusion was diagnosed with computed tomography angiography and confirmed intra-operatively (C).
However, at admission for the scheduled procedure, he reported a recent onset severe claudication of the left lower limb. An urgent CTA confirmed the previously described PAD findings, in addition to the presence of a left iliac stent occlusion, due to thrombotic material (Figure 1C).
Treatment Plan
As the maximum visceral aortic diameter was 27mm, a custom-made four inner branch device (Figure 2) for the revascularization of the celiac trunk, superior mesenteric artery and renal arteries was decided. Distal landing would be performed into the right common iliac (CIA) and left external iliac artery (EIA). The diameter of the right CIA landing zone was 21mm, and a 24mm stent-graft diameter was planned to achieve sealing. Considering the complex iliac access, previous multiple peripheral interventions and need to preserve the patency of the right femoropopliteal bypass, a single large left CFA access was chosen, in addition to a back-table modification of the right iliac limb, which would be reversed and resheathed, for X-over placement. A left iliac axis revascularization would follow to manage the newly diagnosed thrombosis. The diameter of the right hypogastric artery aneurysm set no indication for intervention at that moment. 5
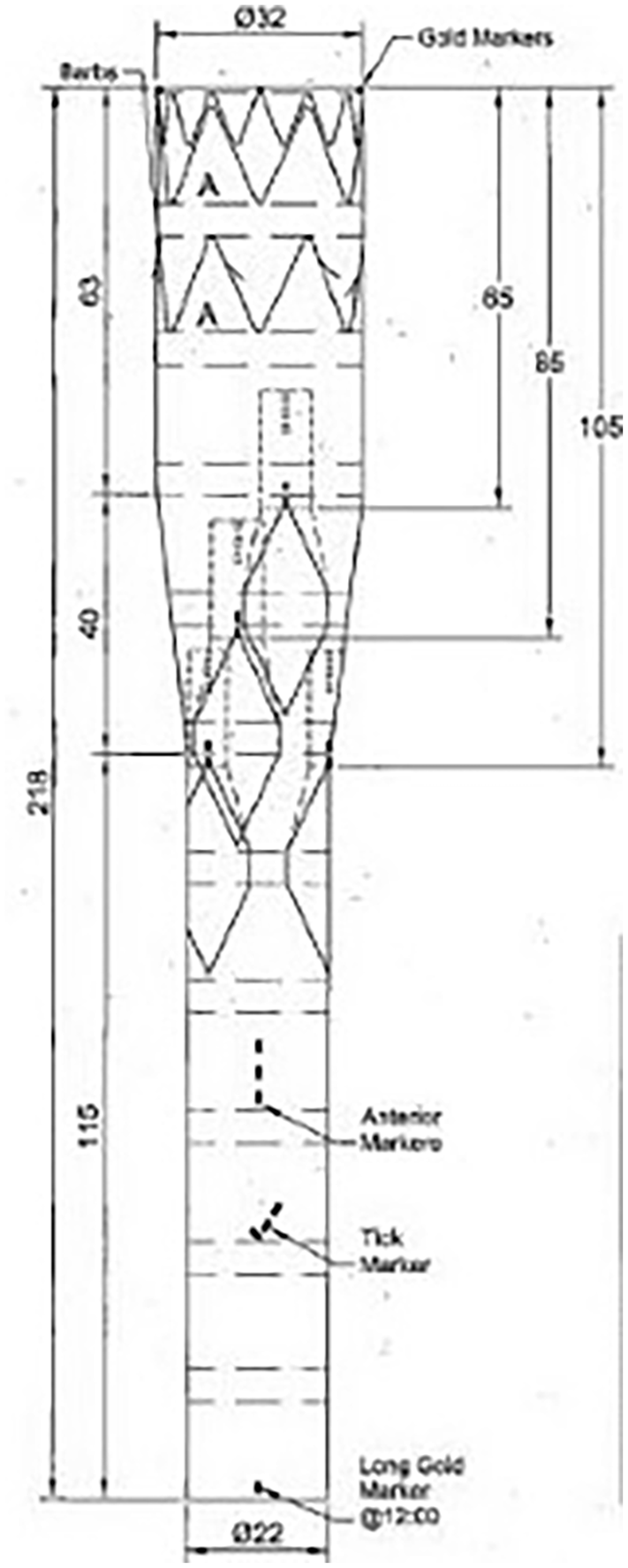
Both the relatively narrow visceral aorta (27mm) in addition to execute the procedure, using single left access led to the selection of a custom-made inner branch device for proximal aneurysm exclusion. The device relied on the Zenith Platform (Cook Medical, Bloomington, IN, USA).
Procedure
The patient was in supine position under general anesthesia. A cerebrospinal fluid drainage was in place. A cut-down of the left common and profunda femoral artery was performed. Back table, an iliac extension (ZISL, 24–59mm, 14Fr introducer sheath, Cook Medical, Bloomington, IN, USA) was deployed, reversed, and re-loaded into its sheath. Systemic heparinization at 100 IU/kg with a target-activated clotting time between 250 and 350 sec was obtained. The right iliac axis was catheterized, after crossing the occluded left external and common iliac artery, using the X-over technique. An extra-stiff Lunderquist wire was advanced and placed in the right superficial femoral artery. The reversed iliac extension was then advanced over the wire (Figure 3A) and deployed in anterograde fashion above the right iliac bifurcation (Figure 3B). After deployment, a bare-back 4F universal flush catheter was directly advanced (without sheath; Figure 3B) from the right side to perform visceral aortic angiography and further, CT-fusion mask readjustment.
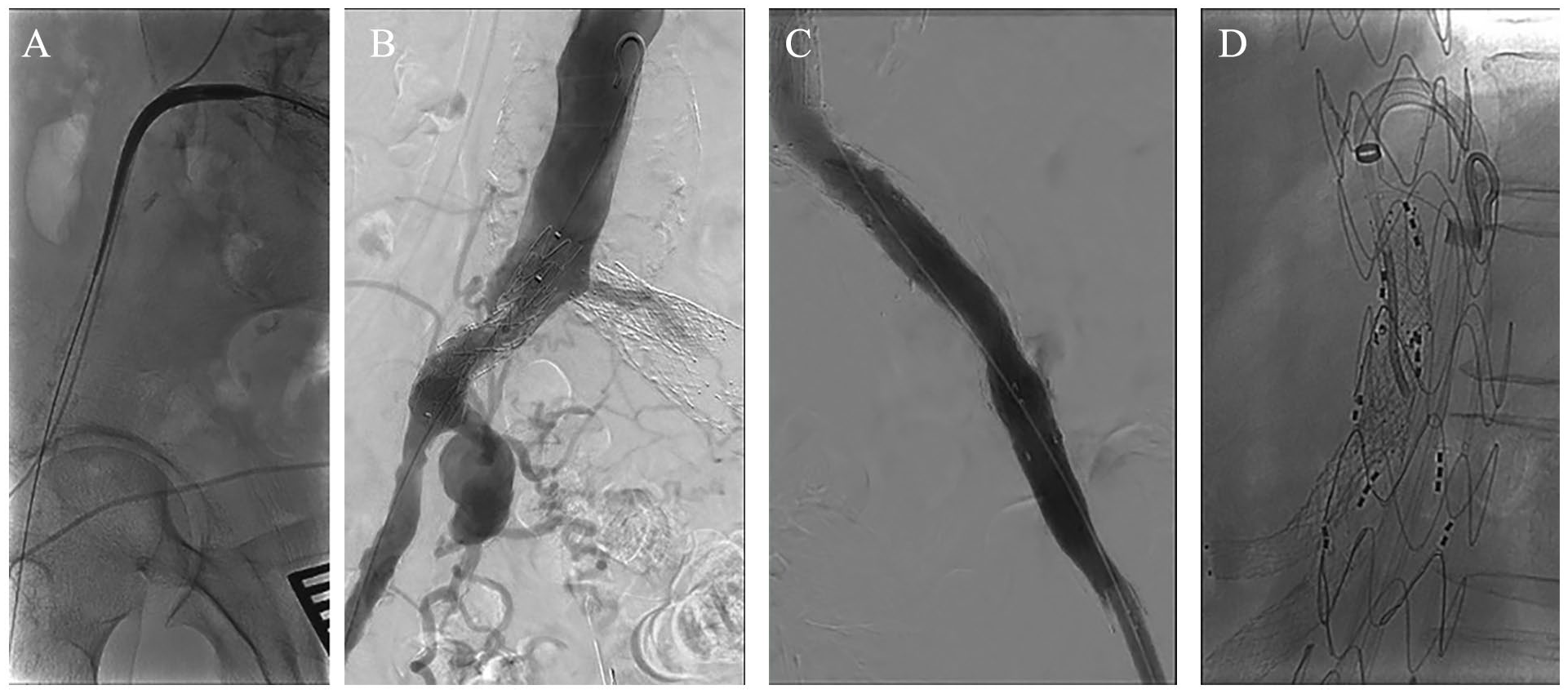
After trough-and-through wire creation, the reversed iliac extension was then advanced (A) and deployed in anterograde fashion above the iliac bifurcation. After deployment the wire was advanced into the aorta and replaced by a bare-back 4F universal flush Catheter (B) for visceral aortic angiography. Through the left femoral access, the iBEVAR, bifurcated device and left iliac extension were advanced and deployed while to restore full patency of the left iliac axis, an over-the-wire thrombectomy was performed (C). Subsequently, using a 10Fr steerable sheath (D), all target vessels were catheterized and bridged.
The inner branch endograft, the bifurcated device and the left iliac extension (ZISL 13–77, Cook Medical, Bloomington, IN, USA) were all advanced and deployed through the left CFA access. Molding ballooning was performed at the proximal and distal landing and graft-overlapping zones. To restore and secure patency of the left iliac axis, an over the wire thrombectomy using a Fogarty catheter was performed (Figure 3C). The left distal landing was extended to the EIA using self-expanding covered stents (from proximal to distal: 13/60mm Fluency (BD, Franklin Lakes, NJ, USA), 13/50mm Viabahn (Gore, Flagstaff, AZ, USA), and 10/60mm Fluency (BD, Franklin Lakes, NJ, USA).
Subsequently, a platform consisting of a 14Fr sheath and a 10Fr steerable sheath (through-and-through suture technique, Figure 3D) was advanced through the left CFA access to perform visceral target vessel (TV) catheterization and bridging in standard fashion. Afterwards, the right iliac extension was catheterized from the contralateral gate (up-and-over technique), 6 and the bifurcated device and right reversed iliac extension were bridged using balloon-expandable covered stents (10/59mm and 10/38mm Advanta V12, Atrium Medical, Merrimack, NH), which were post-dilated using a 14mm angioplasty balloon (Armada, Abbott, Lake Forest, IL, USA). The distal 24mm iliac extension was molded using a 20mm Atlas balloon (BD, Franklin Lakes, NJ, USA). The completion angiography confirmed aneurysm’s exclusion, TV and bilateral iliac artery patency without any type I or III endoleak (Figure 4A).
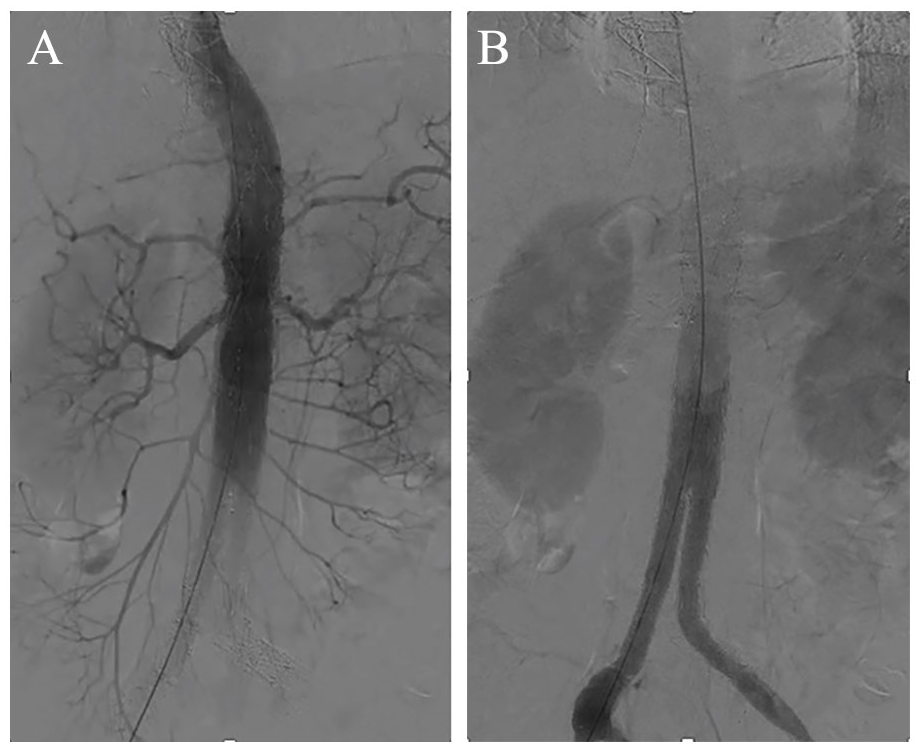
Completion angiography confirmed aneurysm exclusion, with target vessel (A) and iliac axes patency, without endoleak (B).
The sheaths and wires were retrieved and the left CFA was sutured. Moderate manual compression was applied for 15 minutes on the right CFA. No further compression bandage was used. The duration of the procedure was 250 minutes, and the fluoroscopy time was 60 minutes. The patient was extubated and transferred for 48 hours to the intensive care unit. He remained hemodynamically and neurologically stable. A pre-discharge CTA was performed on the third postoperative day, confirming the findings of the completion angiography (Figure 4B).
He was discharged the seventh postoperative day with instructions for smoking cessation and double antiplatelet treatment for 6 months, followed by a lifetime aspirin treatment. The patient was diagnosed with lung cancer 6 months after the procedure and continued his treatment in another health facility near his home. Until cancer diagnosis, the patient reported no symptoms that could be attributed to lower limb ischemia.
Discussion
We demonstrated the feasibility and safety of the X-over reversed iliac extension technique in a severely comorbid PAD patient with complex aortic disease, avoiding the use of bilateral large femoral access, iliac conduits, and compression banding onto the pre-existing femoropopliteal bypass. 7 Access during complex endovascular aortic repair can be challenging, especially in patients with severe atherosclerotic disease.8,9 After multiple peripheral interventions, including open revascularizations, cut down access may not be contraindicated but groins may present excessive scar tissue, adding a potential disadvantage for safe and sufficient use of closure devices, emergent conversion in case of bleeding and wound infection. 10
To decrease the risk of access complications and bypass occlusion, we decided to puncture the right CFA and use it only to perform angiographic controls through a 4Fr catheter. Moderate manual compression was applied at the end of the procedure for 15 minutes. Both the intraoperative use of large bore sheaths and postoperative compression banding could affect the patency of the femoropopliteal bypass. Upper extremity access could also have been used, with respect to the higher complication and embolization rate, especially in patients with severe PAD. 11 Different techniques have been described to overcome technical difficulties accompanying hostile aortic and iliac anatomies, including the up-and-over technique in patients with previous aortic repair and the use of steerable sheaths and reversed endografts.12–14 The described X-over reversed iliac extension technique may be also an alternative approach in patients with EIA occlusion, needing landing into the contralateral patent hypogastric artery.
Patients with abdominal aortic aneurysm extending to the common iliac arteries may also benefit from the X-over approach, as regular balloon- or self-expandable covered stents are not available in tapered designs and may not be applicable as iliac extensions in large diameter iliac arteries. On the contrary, limb reversing can be completed in a few minutes on back-table, without affecting the operating time. For a smooth X-over advancement of the reversed iliac extension over the aortic bifurcation, which should not present a steep angle, the preoperative anatomic evaluation is mandatory. Special attention should be given in the selection of the appropriate iliac extension length, to avoid deployment of its proximal end into the ipsilateral CIA. The risk of graft infection after ex vivo endograft modifications may be increased, despite that previous studies do not confirm such a direct association. 15 An extensive discussion with the patient on the potential future complications is mandatory.
As the number of PAD patients is suspected to increase the upcoming decades, the number of cases needing complex endovascular repair and presenting hostile iliac access will follow the same tendancy. 10 Current recommendations suggest the cautious use of physician-modified techniques and restrict their application mainly as bailout interventions. 5 Despite that the main published experience reports on modified aortic endografts, iliac components may also undergo surgeon-modifications.2,5 The technical success of these procedures and early uneventful survival depend on cautious patient selection, detailed preoperative planning and adaption to each patient’s anatomic demands.
Conclusion
Patients with previous peripheral interventions due to PAD and hostile femoral and/or iliac access may benefit from on-table endograft modifications and single-access approach. The X-over reversed iliac extension technique is feasible and offers a safe option in selected cases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TK is a consultant and proctor for and has intellectual property with Cook Medical, receiving royalties, speaking fees, and research, travel, and educational grants. All authors declare no support from any organization for the submitted work; no financial relationships with any organizations that might have an interest in the submitted work; no other relationships or activities that could appear to have influenced the submitted work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
