Abstract
Purpose:
The treatment of thoracoabdominal aortic aneurysms (TAAAs) using branched endovascular aortic repair (BEVAR) is safe and effective. During deployment, the superior mesenteric artery (SMA) branch can unintentionally open into the celiac trunk (CT) ostium and switched catheterization of the SMA from the CT branch and the CT from the SMA branch can be used as an alternative technique in these cases. This study aimed to investigate the outcome of exchanging the intended target vessels (TVs) for the CT and SMA branches during BEVAR.
Materials and Methods:
A single-center retrospective analysis of patients with TAAAs who underwent BEVAR, using off-the-shelf or custom-made devices (CMDs), with an unintended exchange of TVs for the CT and SMA branches was performed.
Results:
Between 2014 and 2023, 397 patients were treated with BEVAR for TAAA. Eighteen (4.5%) of those patients were treated with an exchange of TVs for the CT and SMA branches. T-branch was used in 9 cases (50%) and the remaining patients were treated with CMDs. Twelve patients were treated electively, 3 were symptomatic and 3 presented with rupture. Of 36 mesenteric TVs in those 18 patients, 34 (94%) were catheterized successfully, including all 18 SMAs and 16 of the 18 CTs. No branch stenosis or occlusion of the switched mesenteric TVs was detected during follow-up. During 30-day follow-up, 3 patients died and during a median follow-up of 3 (interquartile range [IQR]: 1–15) months 3 more patients died. None of the deaths or the 2 unintended reinterventions was induced by the mesenteric TV exchange. The median hospital stay was 14 (IQR: 9–22) days with a median of 4 (IQR: 2–11) days at the intensive care unit.
Conclusion:
The exchange of the mesenteric TVs for the CT and SMA branches during BEVAR with off-the-shelf and CMD endografts is feasible with good TV patency and freedom from TV-related reinterventions. This alternative technique should be considered in selected cases when direct catheterization via the intended branch is deemed more time-consuming or not feasible.
Clinical Impact
This is the first description of using an exchange of target vessels for the celiac trunk and the superior mesenteric artery branches in patients with thoracoabdominal aortic aneurysms undergoing BEVAR, using off-the-shelf or custom-made devices. The high success rate as well as the good clinical results without any branch stenosis or occlusion during follow-up highlight the feasibility of this alternative technique. It could help in challenging cases when catheterization of the intended target vessels is not possible or too time consuming, resulting in higher success rates of BEVAR and better clinical results.
Keywords
Introduction
Branched endovascular aortic repair (BEVAR) is widely accepted as an effective and safe technique for the treatment of thoracoabdominal aortic aneurysms (TAAAs).1–3 High rates of clinical and technical success have been reported with low post-operative morbidity and mortality, particularly in specialized centers.3–5 Alongside, patient-specific custom-made devices (CMDs), standardized off-the-shelf multi-branched endografts are available, which can also be used for urgent cases. 6
Since the introduction of the t-branch, as the first approved off-the-shelf device for BEVAR, its concept to accommodate most anatomic variations has been under close scrutiny. Reported anatomic feasibility rates for the t-branch are considerably based on the relative positions of the visceral arteries and vary in literature.7–9
The superior mesenteric artery (SMA) branch of the Cook Zenith-branched thoracoabdominal devices is usually planned to deploy 1 to 2 cm proximal to the SMA ostium and has mostly a similar clock position as the celiac trunk (CT). Therefore, especially in challenging anatomies, the SMA branch might open into the CT ostium involuntarily (Figure 1) and catheterization from the SMA branch into the CT happens unintended in these cases. In these cases, it can be cumbersome to avoid the CT and connect the SMA branch to the SMA. In addition, this may later even hamper catheterization from the CT branch as the CT ostium may now be obstructed by the SMA branch and its bridging covered stent (BCS).
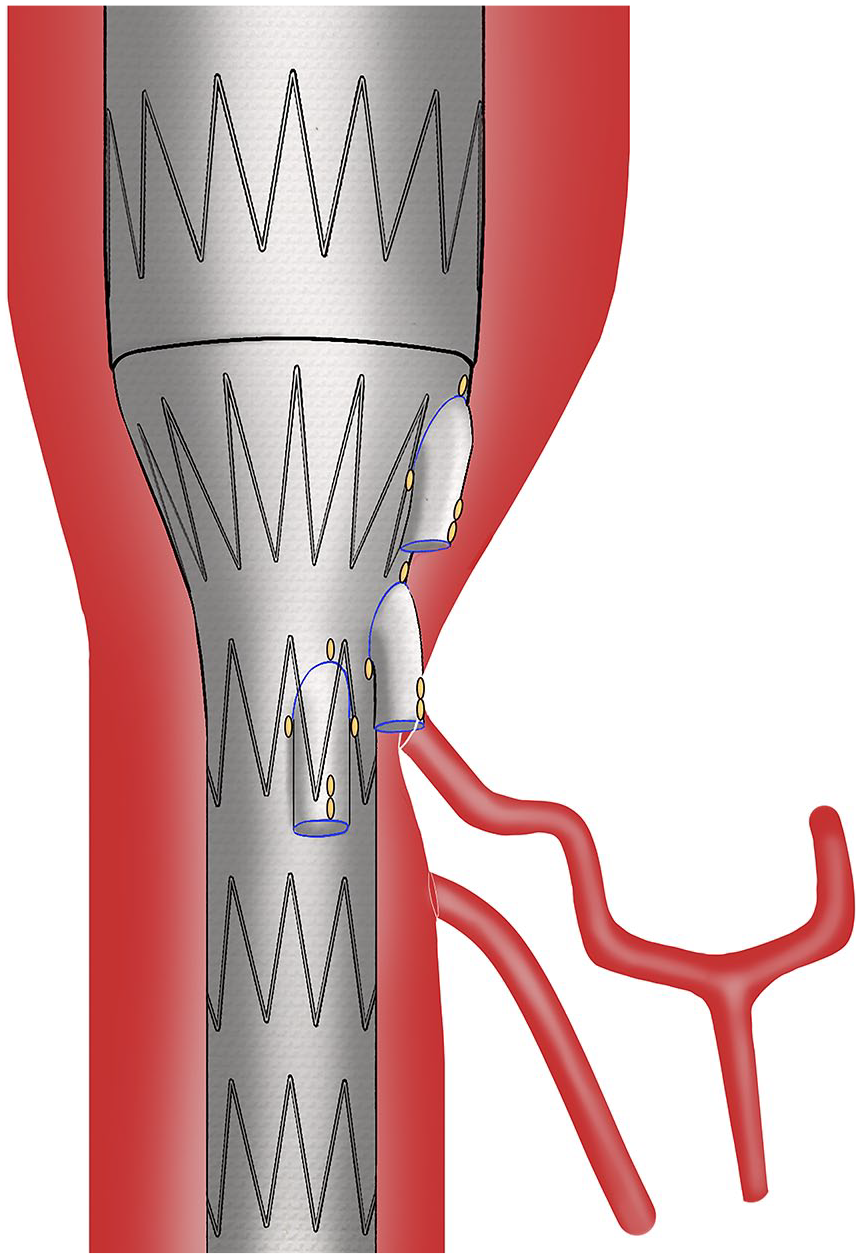
Deployed branched endograft with the superior mesenteric artery branch blocking the ostium of the celiac trunk making it the easier target for catheterization from the superior mesenteric artery branch.
The aim of this study was therefore to investigate the feasibility and durability of an exchange of the mesenteric TVs for the CT and SMA branches during BEVAR with t-branch and CMDs.
Materials and Methods
A single-center retrospective analysis of patients with TAAAs who underwent BEVAR with an unintended exchange of TVs for the CT and SMA branches between January 2014 and March 2023 was performed. Ethical review and approval were waived for this study due to its retrospective design. All patients were pre-operatively assessed for the following parameters: age, sex, American Society of Anesthesiologists classification, renal function, body mass index, chronic obstructive pulmonary disease, coronary artery disease, diabetes mellitus, dyslipidemia, hypertension, peripheral arterial disease, smoking status, and previous stroke. The TAAAs were categorized according to the modified Crawford classification. 10 In addition, maximum aortic diameter, previous aortic interventions, and clinical status of the aneurysm (asymptomatic, symptomatic, and ruptured) were documented.
All procedures were carried out in a hybrid operating room with a fixed imaging system. All patients were under systemic heparinization to achieve a targeted activated clotting time of 250 to 350 seconds with patients under general anesthesia. All BEVAR devices used in this study were either t-branch or Cook Zenith thoracoabdominal CMDs (Cook Medical, Bloomington, IN, USA). In all cases, the SMA was connected via the CT branch and the CT was connected with the SMA branch (Figure 2). A variety of BCSs was used based on operator’s choice. Both retrograde transfemoral access with steerable sheaths and antegrade trans-brachial access were used in this cohort.11,12 For elective Crawford type I, II, III, and V TAAAs, multi-staged endovascular repair was used to minimize the risk for spinal cord ischemia with a primary thoracic endovascular aortic repair (TEVAR) stage as well as the use of perfusion branches and limbs during the BEVAR stage. 13
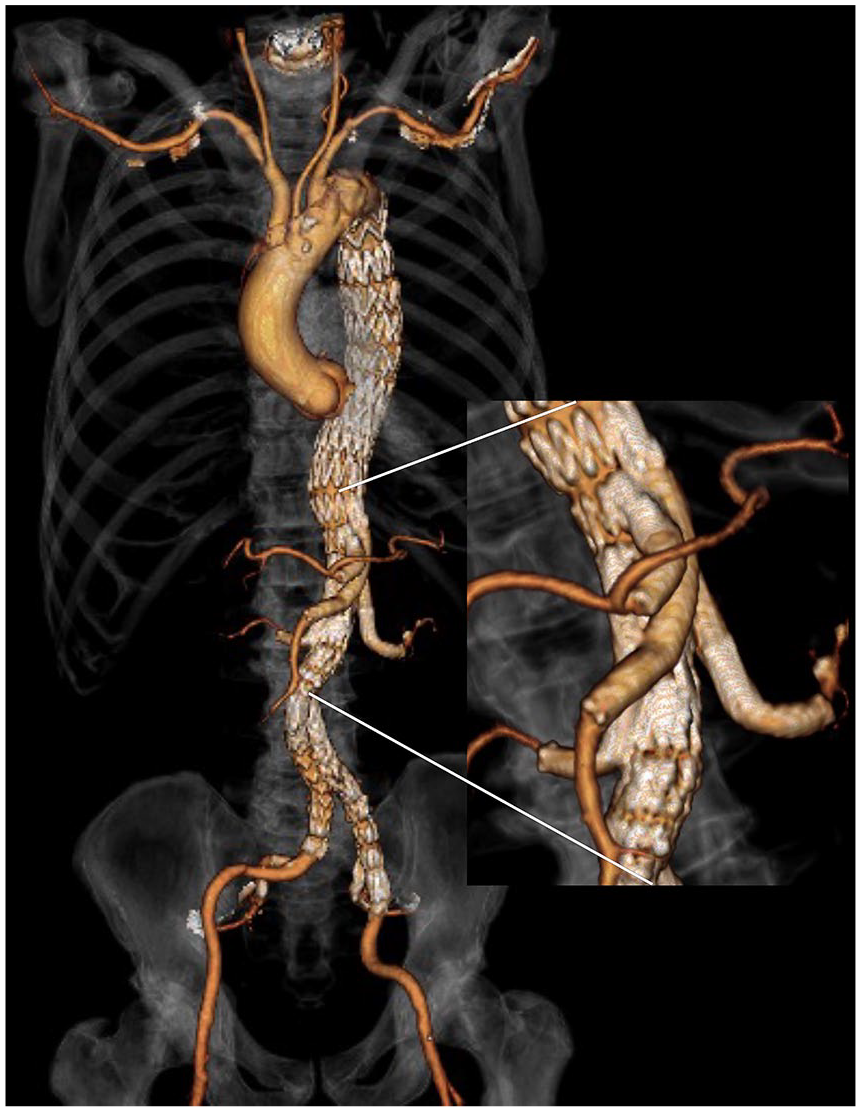
Volume rendering reconstruction of computed tomography angiogram depicting the exchange of the celiac trunk and superior mesenteric artery branch after the elective repair of a Crawford type II thoracoabdominal aneurysm with a t-branch.
Technical success was considered as the successful catheterization and BCS placement in all intended TVs. Technical aspects like stent-graft design, number of TVs, operation time, fluoroscopy time, dose area product, volume of contrast agent, type of arterial access, use of prophylactic cerebrospinal fluid drainage, planned staged aneurysm repair, as well as type and number of BCS were analyzed.
Intra-procedural and post-procedural complications, such as bleeding, access-related complications, myocardial infarction, pulmonary complications, renal failure, reinterventions, and spinal cord ischemia were evaluated. Target vessel (TV)-related outcomes were evaluated according to the Society for Vascular Surgery reporting standards for endovascular aortic repair of aneurysms involving the renal-mesenteric arteries. Target vessel instability was defined as a composite end point used to define any death or rupture related to side branch complications (eg, endoleak, rupture) or any secondary intervention indicated to treat a branch-related complication, including endoleak, disconnection, kink, stenosis, occlusion, or rupture. 14 Follow-up was carried out including computed tomography angiography examinations at 1 and 12 months, followed by yearly examinations. The causes for reinterventions and mortality were analyzed.
All data were stored in Microsoft Excel (Microsoft; Redmond, WA, USA). For the statistical analyses, including Kaplan-Meier analyses for patency, freedom from reinterventions and survival rates SPSS 27.0 for Windows (SPSS, Chicago, IL, USA) were used.
Results
Patient Demographics and Clinical Details
Between 2014 and 2023, 397 patients were treated with thoracoabdominal BEVAR at our center, 218 with CMDs, and 179 with t-branch. Eighteen (4.5%) of those patients were treated with an exchange of TVs for the CT and SMA branches during BEVAR. T-branch was used in 9 cases (50%) and the remaining patients were treated with branched CMDs.
Demographic data and aneurysm characteristics of the 18 patients are shown in Table 1. Of the 8 patients with previous TEVAR, 2 were treated with arch-branched devices. For all symptomatic and ruptured aneurysms, t-branch was used. Sixteen of the endografts had a 4-branch design; 1 had inner branches. In the 2 remaining cases, a 5-branch and 7-branch designs were utilized.
Demographic Data and Aneurysm Characteristics.

Values are median (IQR) or number (percentage).
Abbreviations: ASA, American Society of Anesthesiologists; COPD, chronic obstructive pulmonary disease; eGFR, estimated glomerular filtration rate; EVAR, endovascular aortic repair; TEVAR, thoracic endovascular aortic repair.
Procedural Details
Procedural findings are depicted in Table 2. Of 36 mesenteric TVs, 34 (94%) were successfully catheterized. Of the remaining 40 branches (39 renal arteries and 1 large-segment artery), 37 (93%) were treated successfully. Full percutaneous access was used in 11 cases (61%), of which 2 had access-related complications and needed surgical revision. The remaining 7 cases had a cutdown for femoral access as well as brachial access. For one of the patients, an open surgical iliac conduit was used due to insufficient iliac access.
Procedural Findings.
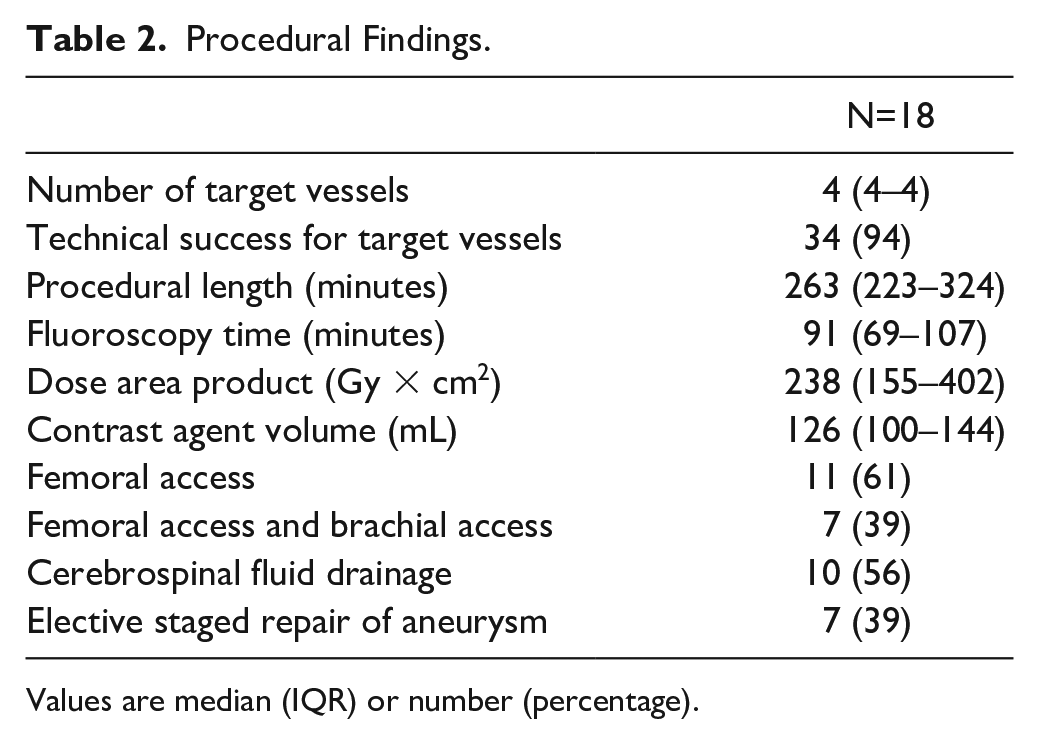
Values are median (IQR) or number (percentage).
Catheterization of the SMA via the CT branch was successful in all patients. However, in 2 cases, subsequent catheterization of the CT via the SMA branch was not successful. One SMA branch of an elective 7-branch CMD was compressed against the vessel wall in combination with a severe stenosis of the CT. The other case was also an elective t-branch with an unfavorable angle of the SMA branch to the mesenteric TVs. In both cases, the SMA branch was occluded using an Amplatzer plug (Abbott, Chicago, IL, USA). 9 The inability to connect the CT did not result in any malperfusion or clinically relevant endoleak.
Regarding the bridging stents, self-expandable covered stent (SECS) as well as balloon-expandable covered stent (BECS) were used. For the CT, SECSs (Viabahn, W.L. Gore and Associates, Flagstaff, AZ, USA; Fluency, Becton Dickinson, Franklin Lakes, NJ, USA) were utilized in 10, BECS (Advanta V12, Getinge, Göteborg, Sweden; Viabahn VBX, W.L. Gore and Associates, Flagstaff, AZ, USA) in 5, and a combination of both in 1 case. The SMA was treated with SECS in 10 patients, with BECS in 4, and a combination of BECS and SECS in 4 patients (Table 3). Post-operatively, 9 patients received single-antiplatelet therapy (SAPT) with aspirin, 6 had dual-antiplatelet therapy (DAPT, aspirin and clopidogrel) and 3 patients received only rivaroxaban due to their atrial fibrillation. Antithrombotic therapy had no significant influence on the outcome in this study.
Bridging Covered Stents Used for the Celiac Trunk and Superior Mesenteric Artery.
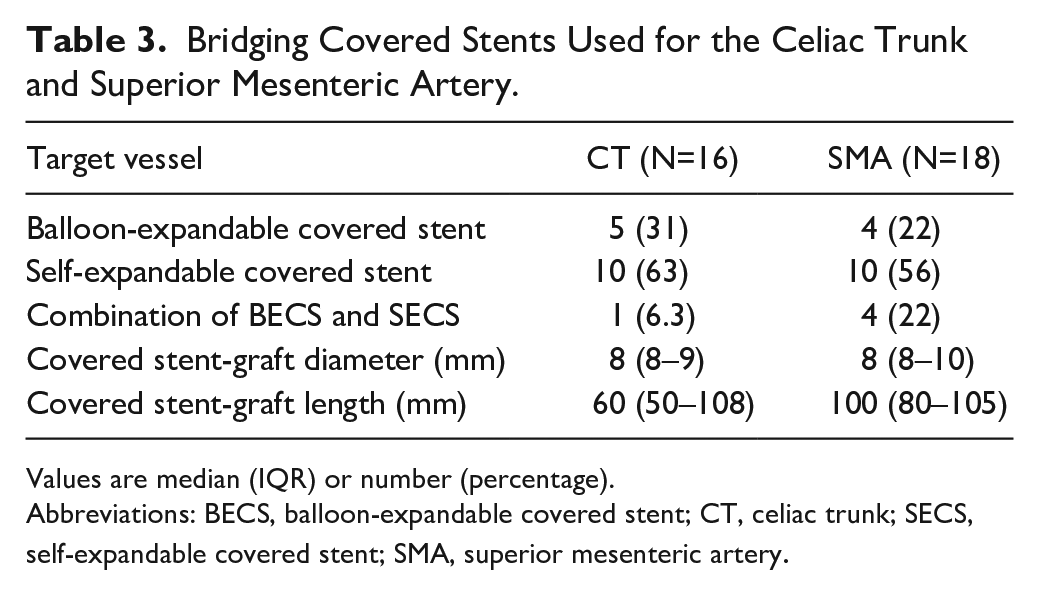
Values are median (IQR) or number (percentage).
Abbreviations: BECS, balloon-expandable covered stent; CT, celiac trunk; SECS, self-expandable covered stent; SMA, superior mesenteric artery.
Complications included 3 patients with post-operative acute kidney injury and 1 case of spinal cord ischemia. No case of mesenteric ischemia was detected. The median hospital stay was 14 (interquartile range [IQR]: 9–22) days with a median of 4 (IQR: 2–11) days at the intensive care unit.
Follow-up
During the 30-day follow-up, 3 patients died, 1 after rupture, 1 symptomatic, and 1 elective TAAA case. One death was attributed to heart failure, 1 to intracranial bleeding, and 1 to sepsis. After 3 months, 1 patient succumbed to multi-organ failure, followed by 2 deaths after 14 and 16 months due to sepsis and heart failure, respectively. This resulted in a total of 6 patients who died during follow-up. Kaplan-Meier analyses of patency, freedom from reinterventions, and survival were 100%, 75%, and 73% at 1 year, respectively. No stenosis or occlusions of the TVs were detected during follow-up. In 4 cases, planned completion procedures as part of a staged aneurysm repair were performed. Two reinterventions were reported, which were TEVARs due to a late type 1a and late type 3 endoleak. None of the reinterventions was related to mesenteric TV instability. The median follow-up was 3 (IQR: 1–15) months.
Discussion
This retrospective analysis of 18 TAAA cases, who underwent BEVAR with an exchange of TVs for the CT and SMA branches with off-the-shelf and CMD endografts, reports the feasibility and clinical results of this alternative technique. In literature, only 1 case report of a redistribution of branches to the supra-aortic TVs in an emergency repair with a triple-branched TEVAR was found. 15 For the thoracoabdominal aorta, no case report or case series was found.
Despite rigorous planning even CMDs might not deploy as intended, especially in challenging anatomies. In our experience, similar clock positions and short distances between the CT and SMA can result in the unintended opening of the SMA branch into the CT ostium as well as higher deployment of branched endografts.
Since the introduction of the t-branch in 2012, endovascular techniques have further evolved. 1 Increased experience with BEVAR, the application of steerable sheaths and other alternative techniques have broadened the indications for complex aortic endovascular treatment, which in some cases must also be performed in urgent settings and outside of the instructions for use. Hongku et al 16 reported that only 18% of patients in their small emergency BEVAR series complied with the anatomical instructions for use of the t-branch. 17 Higher deployment of the t-branch was used in some of the reported cases to facilitate catheterization of all TVs.
Especially in ruptured aortic aneurysm cases, time is of the essence to stabilize the patient. As a consequence, any technique that may expedite the procedure should be utilized. Catheterizing the SMA via the branch intended for the CT and vice versa can save time in selected cases, as it can be time-consuming or even impossible to attach the TVs as planned to their respective branches. This experience has led us to mostly accept the unintended switch of the mesenteric TVs in case the SMA branch opens into the CT ostium in more narrow anatomies. After deployment of the branched endograft, the bifurcated component and the ipsilateral iliac limb we usually start with the most proximal branch.
If catheterization of the CT via the CT branch is not feasible, we continue with the SMA branch. However, in emergent cases, we tend to initially focus on the SMA. As we depicted in Figure 1, in some cases, the SMA branch can block the ostium of the CT or it is positioned closely to the CT ostium, making it the easier target from the SMA branch. This reassignment can create a configuration of the CT and SMA BCS similar to the crossed limbs in the infrarenal aorta (Figure 2). 18 In these cases, we prefer to use SECS instead of BECS due to their higher flexibility. When compression of the SECS was suspected, relining with BECS was performed. As described in literature, BCS lengths and configurations were analyzed in this study. Due to the exchange of TVs the visceral branches were longer with a median of 60 mm (CT) and 100 mm (SMA) compared with other BEVAR series reporting lengths between 64 and 77 mm.19,20 The role of the SMA as the most important TV is evident but of course bridges toward the CT must not be burnt without good reason. Of the 18 cases reported in our cohort, catheterization of the CT via the SMA branch was not possible in only 2 cases. However, in both cases, the initially deployed BCS from the SMA branch to the CT did not inhibit the catheterization of the SMA via the CT branch, as described above. Also, patency was not negatively affected by this exchange of both mesenteric TVs during follow-up. Post-operative SAPT or DAPT had no significant influence in this study but since 2021 our protocol changed toward DAPT for this patient group in accordance with a recent Delphi consensus. 21
In comparison with larger series of BEVAR procedures, the technical success, TV patency, and survival rates were comparable with the current study.3,5,9 We believe that the exchange of TVs for the CT and SMA branches is a feasible and effective alternative technique, which should be considered in selected cases.
Limitations
The main limitation of this study is the retrospective nature and the highly selected patient group of only 4.5% of the thoracoabdominal BEVARs performed during the study period. The 2 not connected CTs caused no major complications in this cohort but this might result in serious morbidity or even mortality in other patients. Although we did not encounter difficulties with BCS blocking catheterization of the TVs or crossed BCS compressing each other, this technique might influence long-term TV patency. Also, the longer BCS might influence patency. Their risk should be considered when using this alternative technique. We could not identify specific anatomic risk factors for TV instability. Also, the procedures were performed in a national referral aortic center, therefore, the results may not directly translate to a “real-world” cohort of patients, and larger studies should evaluate the described technique.
Conclusions
The unintended exchange of TVs for the CT and the SMA branches during BEVAR with off-the-shelf and custom-made endografts is feasible with good TV patency and freedom from reinterventions. This alternative technique should be considered in selected cases, when direct catheterization via the intended branch is deemed more time-consuming or not feasible.
Supplemental Material
sj-pdf-1-jet-10.1177_15266028241251985 – Supplemental material for Unintended Exchange of Target Vessels for Celiac Trunk and Superior Mesenteric Artery Branches in Complex Endovascular Aortic Repair
Supplemental material, sj-pdf-1-jet-10.1177_15266028241251985 for Unintended Exchange of Target Vessels for Celiac Trunk and Superior Mesenteric Artery Branches in Complex Endovascular Aortic Repair by Florian K. Enzmann, Alessandro Grandi, Giuseppe Panuccio, José Ignacio Torrealba, Michaela Kluckner, Petroula Nana, Fiona Rohlffs and Tilo Kölbel in Journal of Endovascular Therapy
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
