Abstract
Purpose:
To assess the feasibility and safety of intravascular lithotripsy (IVL) for enabling transfemoral abdominal (EVAR), thoracic (TEVAR), and thoracoabdominal (BEVAR) endovascular aneurysm repair in patients with narrow and calcified iliac arteries.
Materials and methods:
Consecutive patients treated with IVL for severe calcified and narrowed iliac access before EVAR, TEVAR, or BEVAR between November 2020 and June 2022 were retrospectively evaluated. All anatomical iliac characteristics were acquired by multi-planar reconstruction of preoperative computed tomography angiography (CTA). The hostility of the vascular accesses was classified based on Peripheral Arterial Calcium Scoring System (PACSS) and calcified access severity score (CASS), a new score considering both anatomical (calcium grade and length, minimum lumen diameter [MLD], and tortuosity index) and aortic stent-graft (SG/MLD index) parameters. Primary endpoint was technical success defined as successful aortic endograft delivery and deployment without iliac rupture. Freedom from complications and primary patency were additionally analyzed.
Results:
Twenty-eight iliac axes were treated with IVL (8 bilateral) in 20 patients (mean age 74.5±6.7 years) with a mean follow-up of 26.5±6.2 (range 17–36) months. Ten patients underwent EVAR: 3 TEVAR, and 7 BEVAR procedures. In 14 patients (70%), aneurysm disease was associated with symptomatic aorto-iliac occlusive disease (AIOD), with Rutherford class III to IV. The PACSS was grade IV in 89% of the cases and the CASS (mean 14±2) was grade III to IV in all cases. The stent-graft (SG) outer diameter (5.60±1.65 mm) was significantly larger by 50% than MLD (3.96±1.20 mm), with an SG/MLD index of 1.50±0.51 (p<0.001). Technical success was 100%. No dissection, rupture, or distal embolization occurred. One (3.4%) bail-out stenting was necessary as endoconduit after IVL treatment. One month CTA showed that postoperative luminal gain increased by 93% (p<0.001). An improvement of 2 Rutherford classes occurred in all AIOD patients with a primary patency of 100% at last follow-up.
Conclusions:
This study shows the safety and feasibility of IVL as a valuable option to treat narrow and calcified iliac arteries to facilitate endograft delivery. Further studies will be useful to confirm these results.
Clinical Impact
In this article, the use of intravascular iliac artery lithotripsy to facilitate aortic endograft delivery is explored. The presence of iliac severe calcifications still represents a contraindication for aortic endovascular repair. Intravascular lithotripsy increases the feasibility and safety of endovascular aortic procedures, facilitating endograft delivery and reducing the risk of iliac rupture and/or dissections by improving vessel compliance and luminal gain. This novel vessel preparation could be an alternative to “paving and cracking” and/or iliac conduits. This study describes a new score to classify the severity of iliac calcifications, considering anatomical parameters and the profile of aortic endografts delivery system.
Keywords
Introduction
Narrowed and severely calcified iliac axes represent one of the main contraindications to endovascular aortic repair. The vessel compliance in diffused calcified iliac arteries is very poor and this rigidity represents a major issue for the access of large-bore devices. Large aortic endograft can lead to a variety of vascular complications, such as rupture, dissections, and/or occlusions.
Current available options to overcome this issue are the creation of an open surgical conduit or the “paving and cracking” technique, where covered stents are placed in the iliac arteries, followed by aggressive dilatation to generate a larger iliac lumen. Nonetheless, these are burdened by a high rate of perioperative and postoperative complications.1,2
Intravascular lithotripsy (IVL) has recently emerged as an alternative treatment option for calcified lesions. Sonic pressure waves crack superficial and deep calcium, thereby safely expanding the luminal gain and changing vessel compliance. This technology can improve the trackability of the aortic stent-graft (SG) delivery system in severe diseased access vessels. Its safety and effectiveness has already been shown in coronary and peripheral interventions3,4 while scarce data are available regarding its use in the iliac district. 5
Recent studies have reported successful use of IVL during transcatheter aortic valve replacement (TAVR) 6 and in a few cases of endovascular aneurysm repair (EVAR) and thoracic endovascular aneurysm repair (TEVAR).7 –9
This retrospective study evaluates the safety and feasibility of IVL in facilitating endovascular aortic procedures in a large patient cohort.
Materials and Methods
Study Design
Data of consecutive patients treated with IVL to facilitate EVAR, TEVAR, and branched endovascular aneurysm repair (B-EVAR) in 2 European institutions between November 2020 and June 2022 were prospectively entered into a dedicated database and retrospectively evaluated.
All patients provided written informed consent for the inclusion of their data in the database. The study complied with the principles of the Declaration of Helsinki and was performed in accordance with the rules of the ethical review board of our institutions.
Demographics, comorbidities, and clinical data were collected.
All patients received a preoperative and a postoperative (within 30 days) high-resolution computed tomography angiography (CTA). Every iliac artery measurement was acquired by multi-planar reconstruction and center lumen line with dedicated medical software (EndoSize software; Therenva; Rennes, France, and Acquarius Intuition Viewer, TeraRecon Inc, Foster City, CA, USA). The gray windowing was manually adjusted to reduce the blooming artifact of calcifications.
The presence of associated aorto-iliac occlusive disease (AIOD) was defined as significant iliac stenosis and/or occlusion reported by CTA and confirmed by duplex ultrasound scan (DUS); in this subgroup, the severity of symptoms was evaluated with Rutherford stage.
Patients received a CTA within 30 days from the procedure and duplex-scan at 1, 6, and 12 months after the surgery and yearly thereafter.
Definitions and Indication for IVL Treatment
The severity of hostile calcified iliac access was classified by Peripheral Arterial Calcium Scoring System (PACSS) 10 and calcified access severity score (CASS), 11 taking into account 5 anatomical iliac parameters, each one with a severity score from 1 to 4 (1=mild, 2=moderate, 3=severe, 4=very severe; Figure 1).
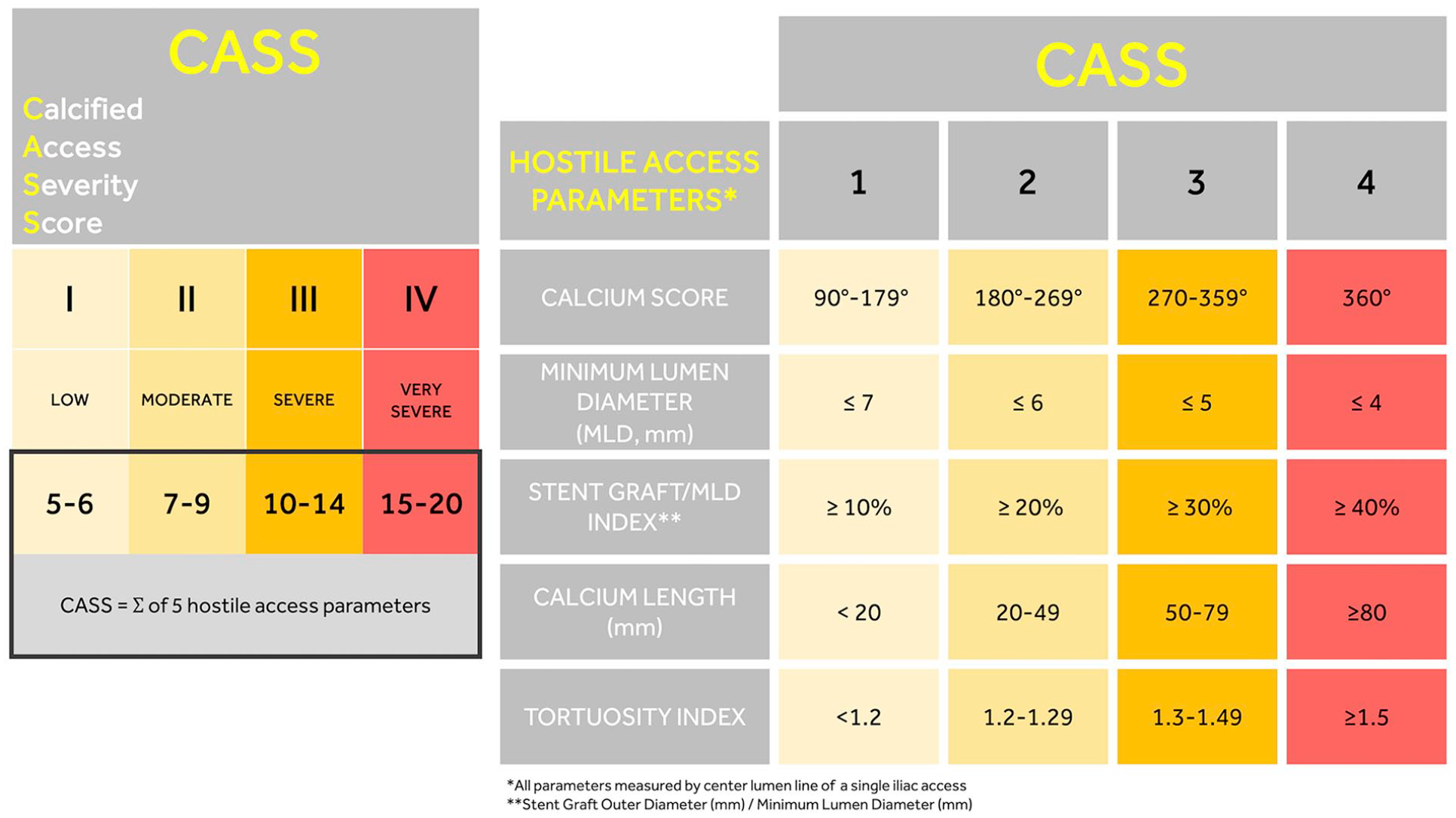
Calcified access severity score (CASS) to assess the severity of hostile access parameters of each iliac axis.
The CASS is calculated as the sum of the 5 severity scores for each iliac axis (Figure 1):
Calcium Grade, classified accordingly to 360° coronary classification 12 and divided into 4 grades (1=90°–179°, 2=180°–269°, 3=270°–359°, 4=360°concentric lesion). The grades 1, 2, 3, and 4 were considered mild, moderate, severe, and very severe, respectively.
Minimum Lumen Diameter, measured as the smallest patent diameter of the vessel excluding calcium thickness. Measurements were obtained using manually adjusted center lumen line and appropriate windowing to reduce the risk of calcium blooming artifacts. An MLD <7 mm, <6 mm, <5 mm, or <4 mm was considered mild, moderate, severe, or very severe, respectively.
Stent-graft/Minimum Lumen Diameter Index, intended as the ratio between the outer diameter of the aortic stent graft delivery system and MLD. The choice of the endograft was planned before the procedure, based on aortic anatomy and iliac vessels characteristics. A decision was taken balancing the available lower profile and the better stent graft fit for patient’s anatomy, considering all features beyond hostile access, such as aortic disease and aortic neck.
An SG/MLD index ≥10%, ≥20%, ≥30%, or ≥40% was considered mild, moderate, severe, or very severe, respectively.
Calcium Length, intended as the sum of all calcified iliac lesions (stenosis >30%) between 2 endpoints, from the aortic bifurcation up to the proximal common femoral artery (CFA). A calcium length <20 mm, 20–49 mm, 50–79 mm, or ≥80 mm, was considered mild, moderate, severe, or very severe, respectively.
Tortuosity Index, considered as the ratio between the center-lumen-line length of the vessel and the linear distance from the aortic bifurcation up to the CFA. 13 A tortuosity index <1.2, 1.2–1.29, 1.3–1.49, or ≥1.5 was considered as a mild, moderate, severe, or very severe score, respectively.
We considered 4 CASS stages divided as I (mild: score 5–6), II (moderate: score 7–9), III (severe: score 10–14), and IV (very severe: score 15–20; Figure 1).
The main indication for IVL was to facilitate aortic endograft delivery through calcified iliac access. Based on the study protocol (Figure 2), we planned to use IVL in case of severe and very severe calcified iliac access with CASS between 10 and 20.
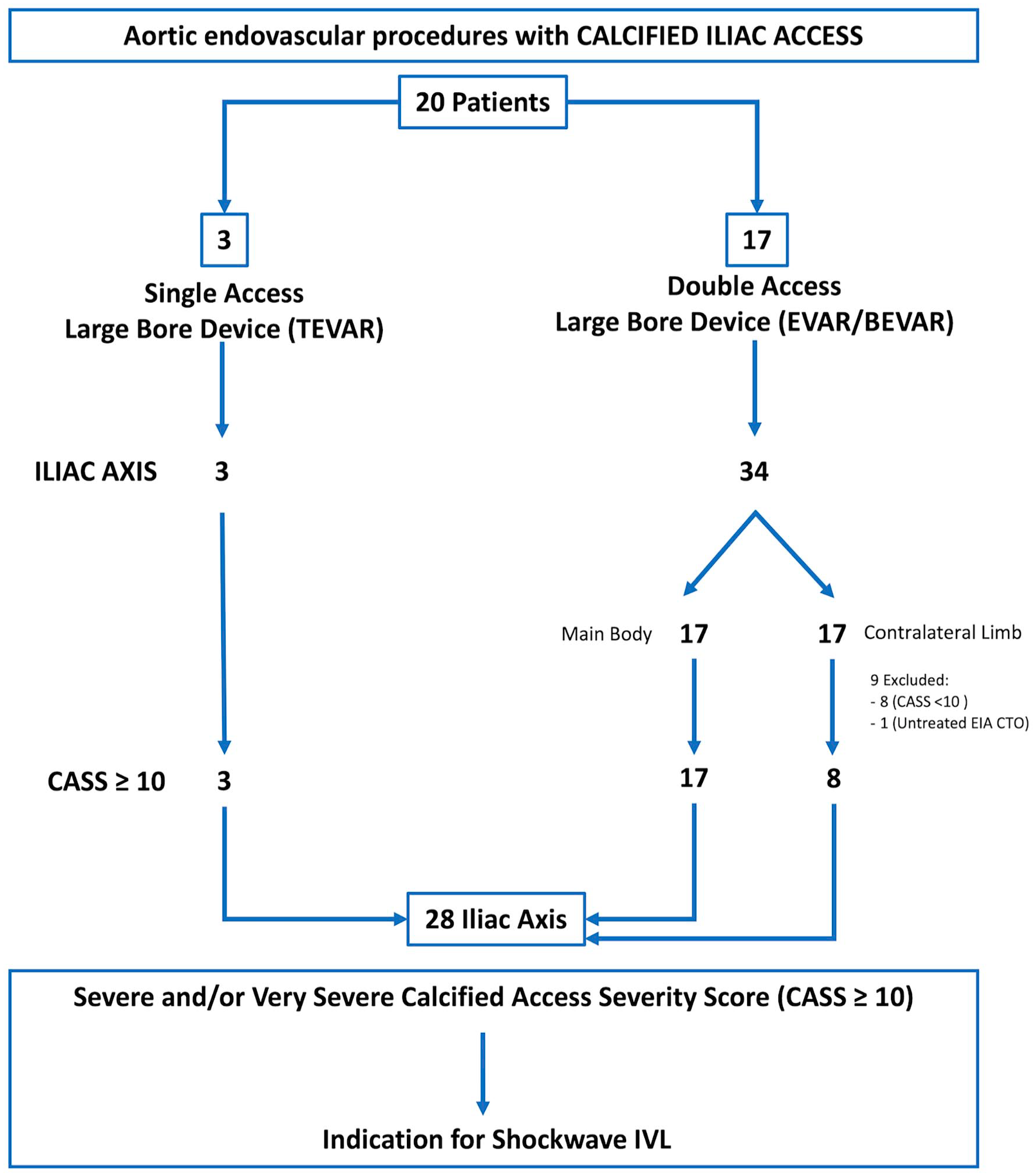
Flow chart of patients treated by intravascular lithotripsy to facilitate the delivery of aortic endografts in case of hostile calcified iliac access. This protocol is based on a calcified access severity score (CASS, Figure 1). Iliac axis with CASS ≥10 was indicated for Shockwave IVL treatment.
Measurements of iliac diameters (Table 5) were referred to as the target lesion (TL) that was defined as the iliac lesion with higher SG/MLD index considered for IVL treatment; iliac stenosis larger than 50% were recorded. Reference vessel diameter (RVD) was defined as the media-to-media measurement at the level of TL. In case of bilateral iliac landing zones, the iliac axis with lower CASS was chosen for the aortic main body (larger profile stent graft).
Intravascular Lithotripsy Device and Procedural Protocol
All procedures were performed under local or general anesthesia. Femoral percutaneous access, using the single/double Prostyle or Prostar XL-10F vascular closure device (Abbott Vascular, Redwood City, CA, USA), was performed in most cases. In selected cases, brachial access was performed. Arterial puncture was duplex-assisted, with the advantage of identifying a calcium-free spot to achieve the best puncturing site. In case of CFA severe stenosis/occlusion, an endarterectomy was performed prior to endovascular procedure. Systemic heparinization with 100 UI/kg body weight was administered once gained access to the artery. The aortic endovascular procedures were performed in an AngioSuite (Philips Allura Xper FD 20/20—DS; Philips Healthcare, Eindhoven, The Netherlands) or hybrid room (Siemens Axiom Artis FA; Siemens Medical Solutions, Forchheim, Germany).
The Shockwave Medical Peripheral IVL System (Shockwave Medical, Santa Clara, CA, USA) was performed as already described in calcified TAVR-access. 14
Step-by-Step Procedure
The Shockwave M5 and M5+ IVL catheters contain 5 lithotripsy emitters along the shaft of an integrated 60 mm semicompliant balloon. Through a connected generator, the catheter can deliver a predefined number of pulses (10 cycles of 30 pulses, 2 pulses/second for M5+). Catheter sizing was based on RVD with 10% oversizing, whenever possible. The IVL catheter was introduced into the target vessel over a 0.014” guidewire and positioned across the TL using balloon markers. The integrated balloon, filled with a 50:50 saline/contrast mixture, was inflated to subnominal pressure (4 atm) to provide apposition to the vessel wall and facilitate efficient transmission of sonic pressure waves to disrupt intima and media calcifications.
After IVL treatment, the balloon was inflated to nominal pressure (6 atm) for 10 seconds.
This cycle was repeated multiple times in different locations for each iliac axis, reserving most cycles for the worst lesion.
A predilatation with a 3 mm angioplasty balloon was performed in case of tight iliac stenosis, whereas a postdilatation with nominal diameter was performed in case of high-grade residual stenosis and/or suboptimal IVL sizing. In case of delivery systems larger than 20 French (Fr), a single passage of a large-bore sheath with the same profile was performed per protocol before unboxing the graft and introducing it. 15
After IVL treatment, the 0.014 wire was replaced with a stiff wire and the procedure continued with the introduction of bigger sheaths. From this point, the standard procedure for EVAR, TEVAR, and BEVAR took place. In case of unfeasible iliac crossing, a bail-out endoconduit by using balloon expandable covered stents was performed to allow the large-bore device delivery. The Shockwave M5+ peripheral catheters have been available since January 2022 and this has been the workhorse IVL catheter following its launch (availability of 8 mm catheter, 135 mm shaft, quicker use with 2 cycles/s, and lower profile).
All available aortic abdominal and thoracic endograft were considered, including standard- and low-profile delivery system.
Selected endografts were Cordis Incraft (Cardinal Health, Dublin, OH, USA), Gore Excluder and Conformable Excluder (W.L. Gore & Associates, Flagstaff, AZ, USA), AFX Endovascular AAA Systems (The Endologix, Inc. Irvine, CA, USA), Relay NBS (Terumo Aortic, Sunrise, FL, USA), Valiant Navion (Medtronic, Minneapolis, MN, USA), branched custom-made thoraco-abdominal aortic aneurysm (TAAA) SGs (Cook Medical, Bloomington, IN, USA), and Ankura AAA Stent Graft (Lifetech Scientific, Shenzen, China).
Single antiplatelet therapy was administered lifelong in patients receiving aortic endovascular treatment. Dual antiplatelet therapy with aspirin (100 mg/d) and clopidogrel (75 mg/d) were routinely prescribed for at least 2 months in patients with iliac or visceral stenting.
Endpoints
The primary endpoint of the study was the technical success established with the use of Shockwave IVL to facilitate a successful aortic endograft delivery and deployment without iliac rupture.
Secondary endpoints were freedom from bail-out maneuvers and procedural complications, such as flow-limiting dissections requiring stenting, distal embolizations, and access-site complications.
Early and midterm results were assessed, evaluating primary patency, clinical success, reintervention rate, and mortality.
Primary patency was defined as a target lesion without a severe restenosis/occlusion and without target lesion reintervention (TLR) during follow-up. Restenosis was defined as stenotic vessel area >70% at CT scan or >2.0 peak systolic velocity ratio at DUS calculated as the peak systolic flow velocity in the lesion divided by the peak systolic velocity in the reference vessel.
The clinical success was defined as the procedural success without major complications before discharge; the improvement of one or more Rutherford classes compared with the index procedure was considered for AIOD subgroup only.
Statistical Analysis
Continuous variables are presented as the mean±standard deviation (range) and categorical variables as frequencies and percentages. Comparison between different subgroups were performed using the Wilcoxon-Mann-Whitney test. The threshold of statistical significance was p≤0.05. All analyses were conducted using R language for statistical computing software v.3.6.1 (R Foundation for Statistical Computing, Vienna, Austria, 2020, https://www.R-project.org).
Results
Patient Population
Twenty-eight severely calcified iliac axis (8 bilateral) in 20 patients were selected for IVL use to facilitate the delivery of the aortic stent grafts (Figure 2). Ten patients underwent EVAR (6 LP EVAR), 3 TEVAR, and 7 BEVAR, and 14 (70%) were associated to AIOD (Table 1, Figures 3 and 4). Baseline characteristics are presented in Table 2.
Intravascular Lithotripsy Indications and Procedures.
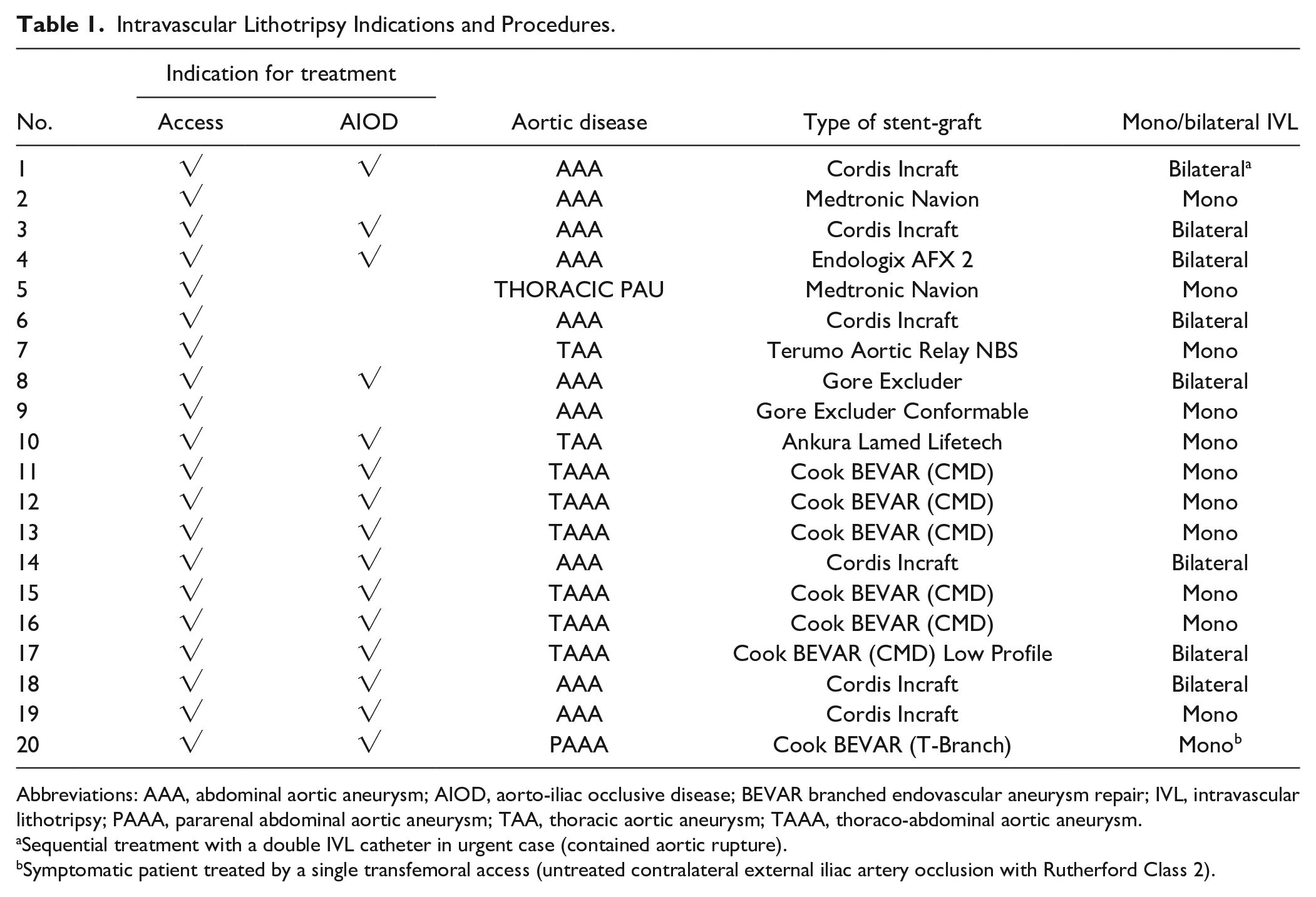
Abbreviations: AAA, abdominal aortic aneurysm; AIOD, aorto-iliac occlusive disease; BEVAR branched endovascular aneurysm repair; IVL, intravascular lithotripsy; PAAA, pararenal abdominal aortic aneurysm; TAA, thoracic aortic aneurysm; TAAA, thoraco-abdominal aortic aneurysm.
Sequential treatment with a double IVL catheter in urgent case (contained aortic rupture).
Symptomatic patient treated by a single transfemoral access (untreated contralateral external iliac artery occlusion with Rutherford Class 2).
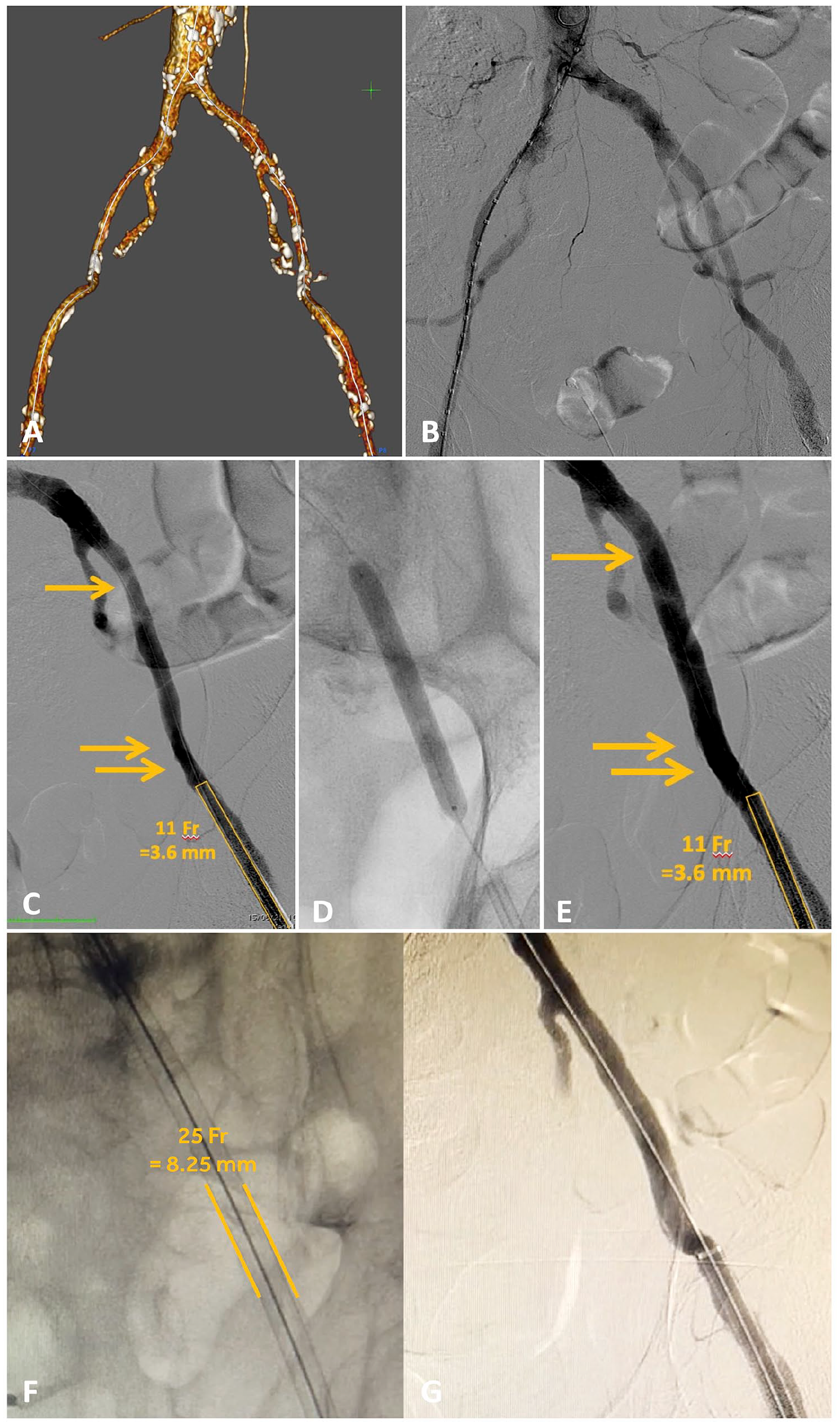
“ACCESS” case. A and B: Preoperative computed tomography angiography (A), and angiogram (B) of narrowed and calcific iliac axes in case of aortic arch disease (thoracic aortic endograft with 25 French [Fr] outer diameter delivery system). C to E: Pre-intravascular lithotripsy (IVL) angiogram (C), vessel preparation by IVL treatment (D), and post-IVL angiogram (E). F: Delivery of a thoracic aortic endograft with 25 Fr outer diameter. G: Completion angiogram showing no signs of iliac ruptures and/or dissections.
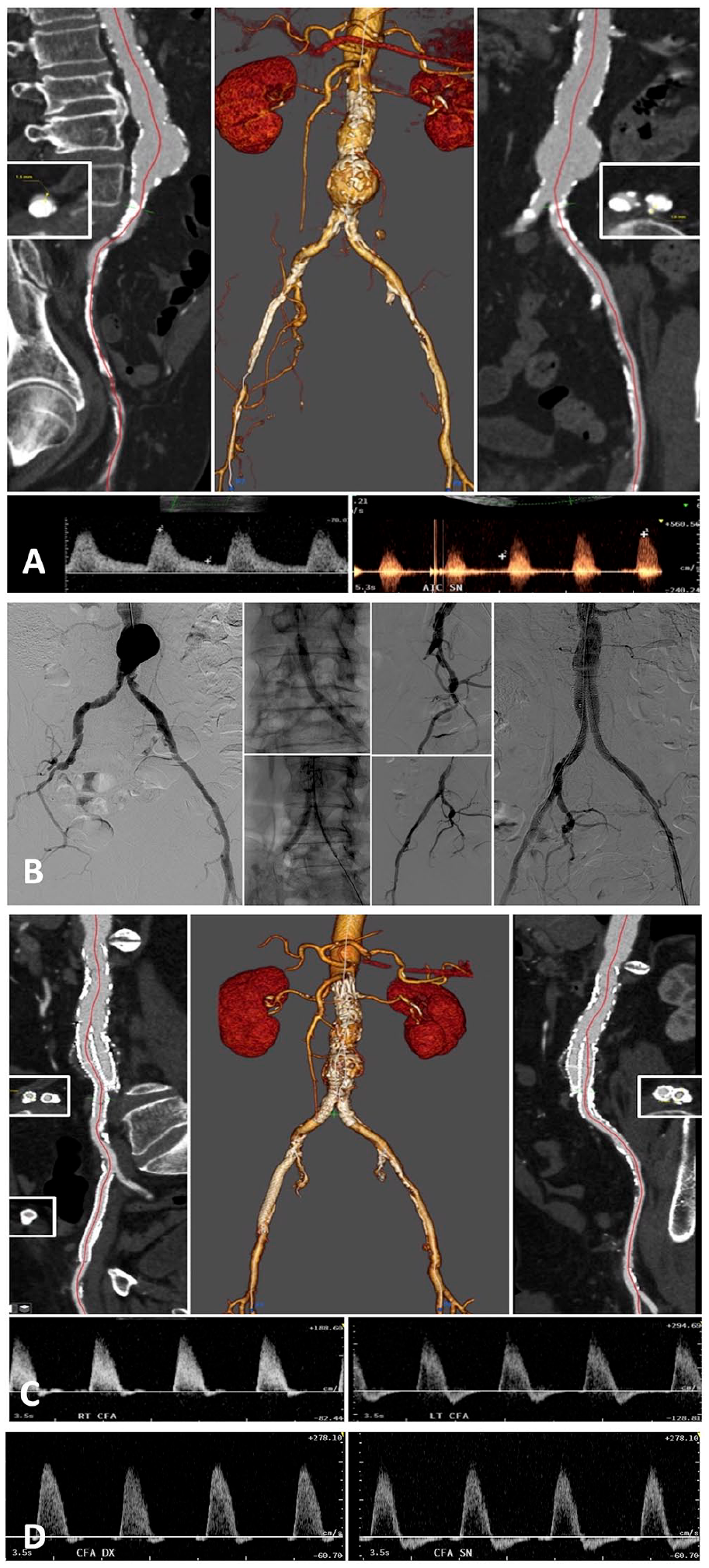
“Aorto-iliac occlusive disease (AIOD)” case. A: Preoperative computed tomography angiography and duplex-scan. B: Procedure. Preoperative angiogram, vessel preparation by IVL treatment from brachial access, EVAR, iliac kissing covered stenting in place of iliac limbs, right external iliac artery recanalization-IVL-stenting, and completion angiogram. Procedure performed in local anesthesia by using 2 percutaneous access (left brachial and left common femoral). C: Postoperative computed tomography angiography and duplex-scan control on right and left common femoral artery. D: Two years’ duplex-scan control showing stable bi-3 phasic waveforms at the same locations.
Patients Characteristics.
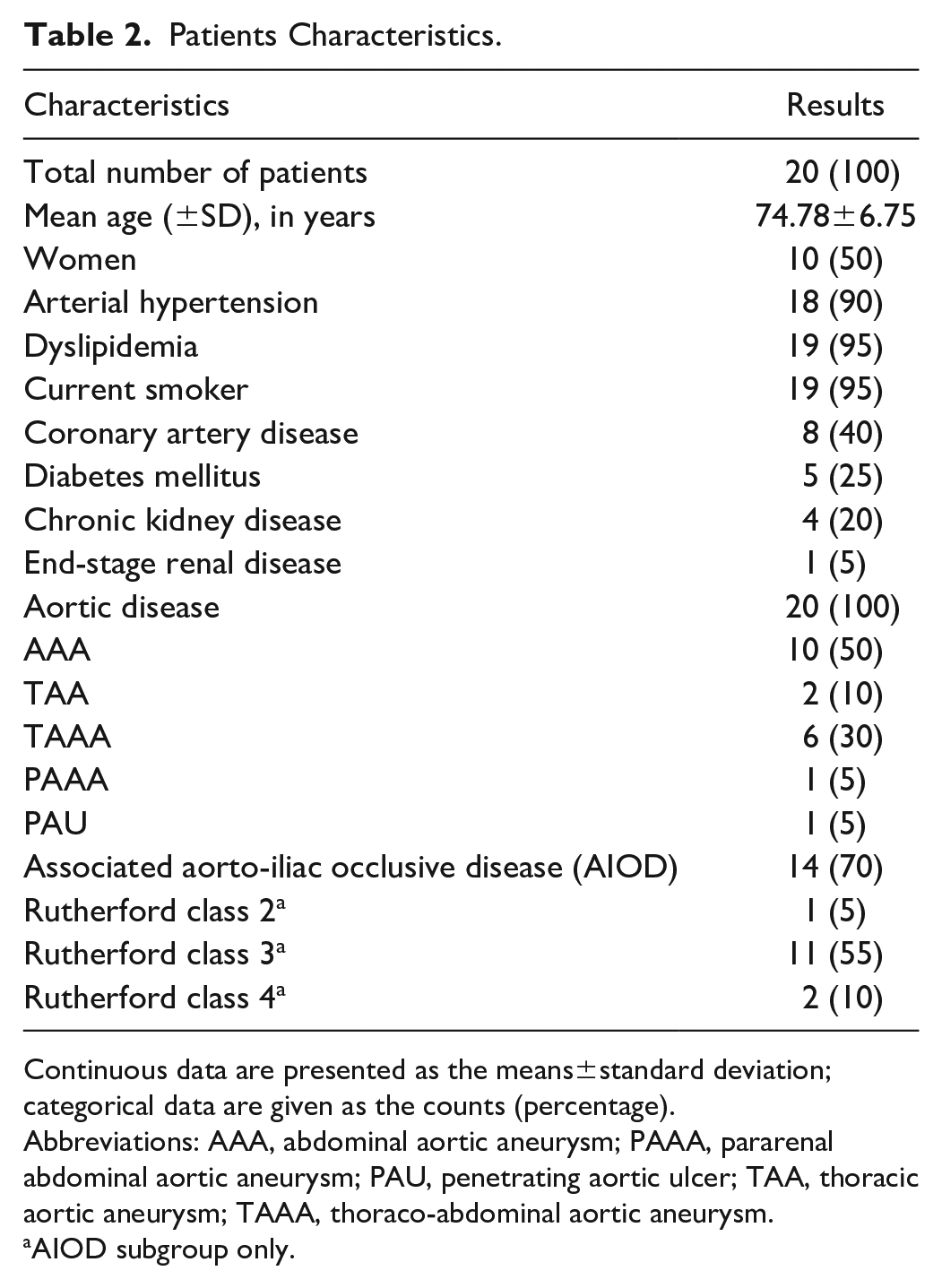
Continuous data are presented as the means±standard deviation; categorical data are given as the counts (percentage).
Abbreviations: AAA, abdominal aortic aneurysm; PAAA, pararenal abdominal aortic aneurysm; PAU, penetrating aortic ulcer; TAA, thoracic aortic aneurysm; TAAA, thoraco-abdominal aortic aneurysm.
AIOD subgroup only.
Preoperative Characteristics
A total of 28 iliac axis were classified as PACSS IV in 89% of the cases, and as CASS III (10–14) and IV (15–20) in 53% and 47% of the cases, respectively; the CASS grade was significantly higher in AIOD subgroup (15 vs 13, p<0.001; Figure 5A).
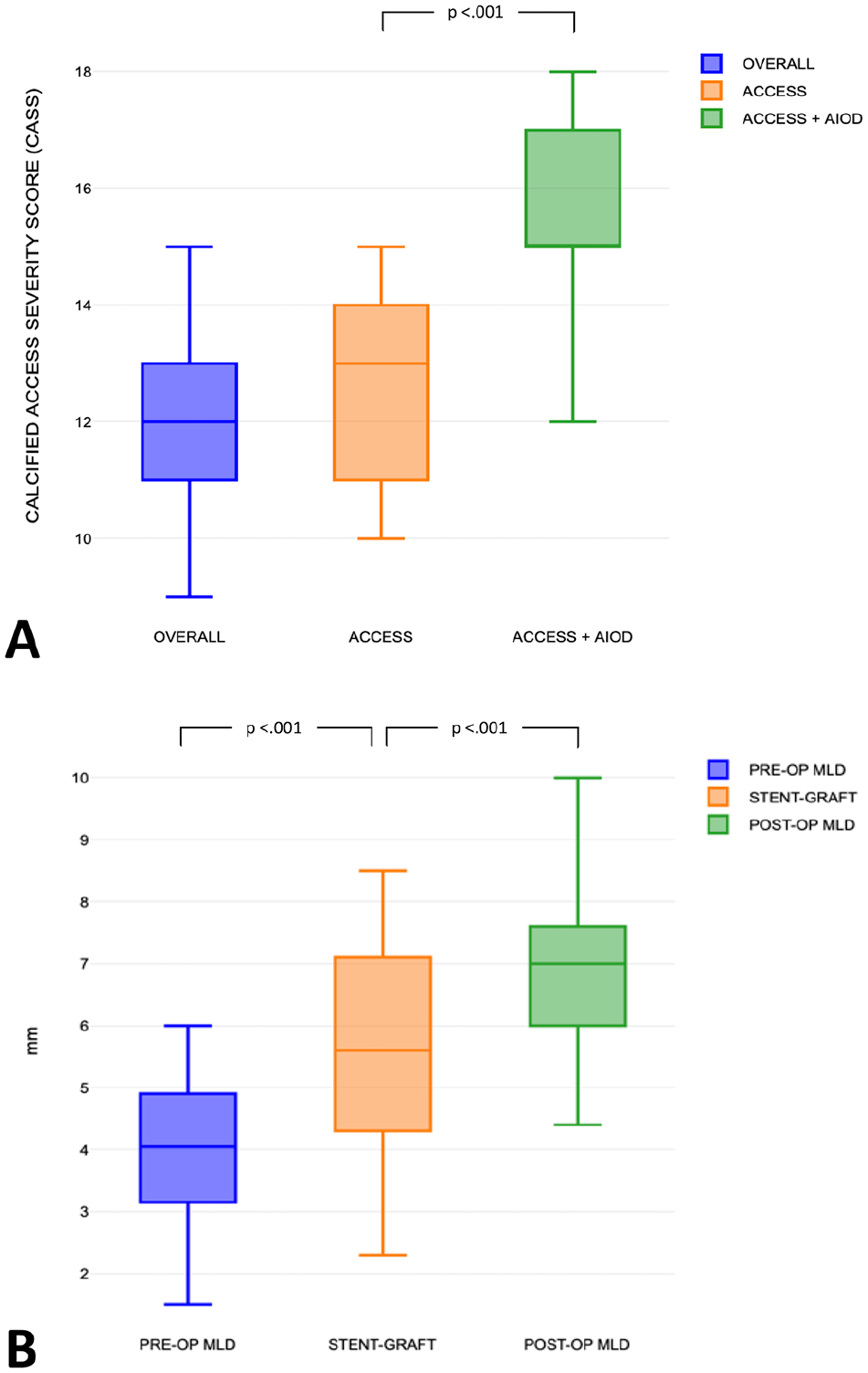
Preoperative and postoperative results. A: Overall, calcified access severity score (CASS) and compared in 2 subgroups, ACCESS and ACCESS+AIOD (aorto-iliac occlusive disease). B: Comparison in millimeters (mm) between preoperative/postoperative minimum lumen diameter and stent-graft outer profile.
The mean calcium grade was 3.46±0.50, with calcifications larger than 180°, 270°, or concentric in 35.7%, 17.8%, and 46% of the target lesions, respectively.
Preoperative mean MLD was 3.96±1.20, with an SG/MLD index of 1.50±0.51 (SG larger than MLD by 50%, p<0.001). The target lesion was localized in external and common iliac artery in 78.5% and 21.5%, respectively, with a mean RVD of 8.20±1.85.
The SG/MLD index was significantly higher in women than in men, 1.64±0.56 vs 1.32±0.43, respectively (p<0.001). Significant gender-related difference was not encountered for MLD (3.55±1.47 mm in female vs 4.23±0.75 mm in male).
Mean calcium total length was 88.71±34.59 mm (considering 4.29±1.46 iliac lesions) with a rate of iliac calcified lesions of 50%±16% for each iliac axis length. The iliac tortuosity index was considered moderate in 35% of the cases. All preoperative characteristics are summarized in Table 3. Two patients had complete occlusion of the external iliac artery and one was not treated because of a mild claudication (BEVAR procedure performed with a single femoral access for a symptomatic PAAA).
Preoperative/Postoperative Iliac Axis and Aortic Stent-Graft Characteristics.
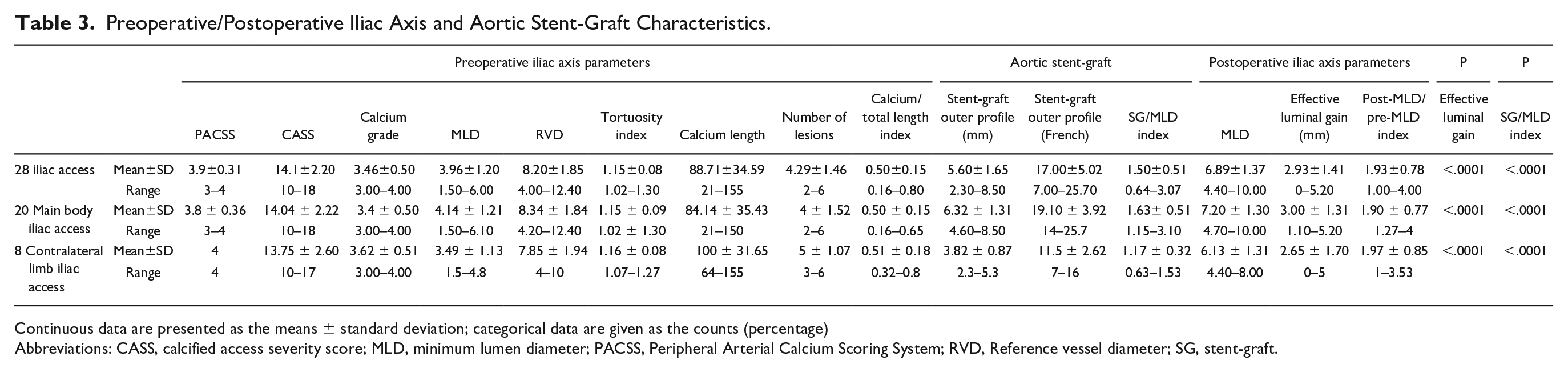
Continuous data are presented as the means ± standard deviation; categorical data are given as the counts (percentage)
Abbreviations: CASS, calcified access severity score; MLD, minimum lumen diameter; PACSS, Peripheral Arterial Calcium Scoring System; RVD, Reference vessel diameter; SG, stent-graft.
Primary Endopoints and Procedural Characteristics
Technical success was 100% with a correct deployment of aortic stent grafts as planned in absence of iliac rupture. Types and profile of aortic SGs and procedural characteristics are described in Tables 1, 2, and 4 respectively. The mean SG outer profile was 17±5 Fr overall, with 19±3.9 Fr and 11.5±2.6 Fr of of the main body and contralateral limb, respectively. Intravascular lithotripsy was successfully delivered in all 28 iliac axis focusing on target lesion to facilitate a smooth and safe crossing of the aortic stent grafts. The mean delivered IVL pulses were 136.1±45.1 per iliac axis, considering a single IVL catheter in all cases but one. In 2 patients (10%) a percutaneous brachial access was used for IVL and iliac stenting, avoiding ipsilateral femoral approach (Figure 4).
A 7 mm IVL catheter was used in most of the cases (82%). In one case (3.4%), a bail-out stenting (see further, “Secondary Endpoints”) was required.
Five (17.8%) balloon-expandable covered stents (Viabahn VBX, Gore) were implanted in place of iliac limbs at the level of common iliac artery to reduce the profile and increase the radial force. Four external iliac artery (14.2%) stenting (bare metal self-expanding stents but one) were performed in AIOD cases (2 for dissections occurred during wire crossing prior to IVL treatment, 1 for long chronic total occlusion, and 1 for severe stenosis).
One procedure (case 1) was performed in urgent setting for a contained ruptured AAA with double IVL catheter used sequentially in both iliac axis to save procedural time.
Secondary Endpoints and Postoperative Characteristics
The mean follow-up was 26.5±6.2 (range 17–36) months. Clinical success was 100% without any major complication before discharge. No IVL-related flow-limiting dissections and/or distal embolizations occurred. One iliac axis (3.4%) required a bail-out stenting (concentric lesion at the origin of external iliac artery treated by suboptimal IVL sizing) with a balloon-expandable covered stent (Viabahn VBX, Gore) successfully used as endoconduit to allow the aortic endograft delivery. Two patients (10%) with AIOD required a planned femoral endarterectomy.
The postoperative MLD (6.89±1.37 mm) significantly increased by 93% (p<0.001) with an effective luminal gain of 2.93±1.41 (Table 3). The CTAs and duplex scan controls showed no signs of iliac (target lesions, iliac limbs, or different iliac locations) dissection, occlusion, restenosis, and/or recoils postoperatively and up to last follow-up.
No perioperative complications occurred except for a bleeding of a percutaneous femoral access that was converted to open surgery at the end of the procedure. No other complications, reinterventions, or deaths were reported during hospitalization or follow-up.
In the AIOD subgroup, all treated patients were discharged with a clinical improvement of 2 Rutherford classes, and no clinically driven revascularization occurred during follow-up. No type I or III endoleaks occurred, with a stable/shrinked aneurysm sac in all cases. Secondary endpoints and clinical outcomes are summarized in Table 4.
Procedural Characteristics and Clinical Outcomes.
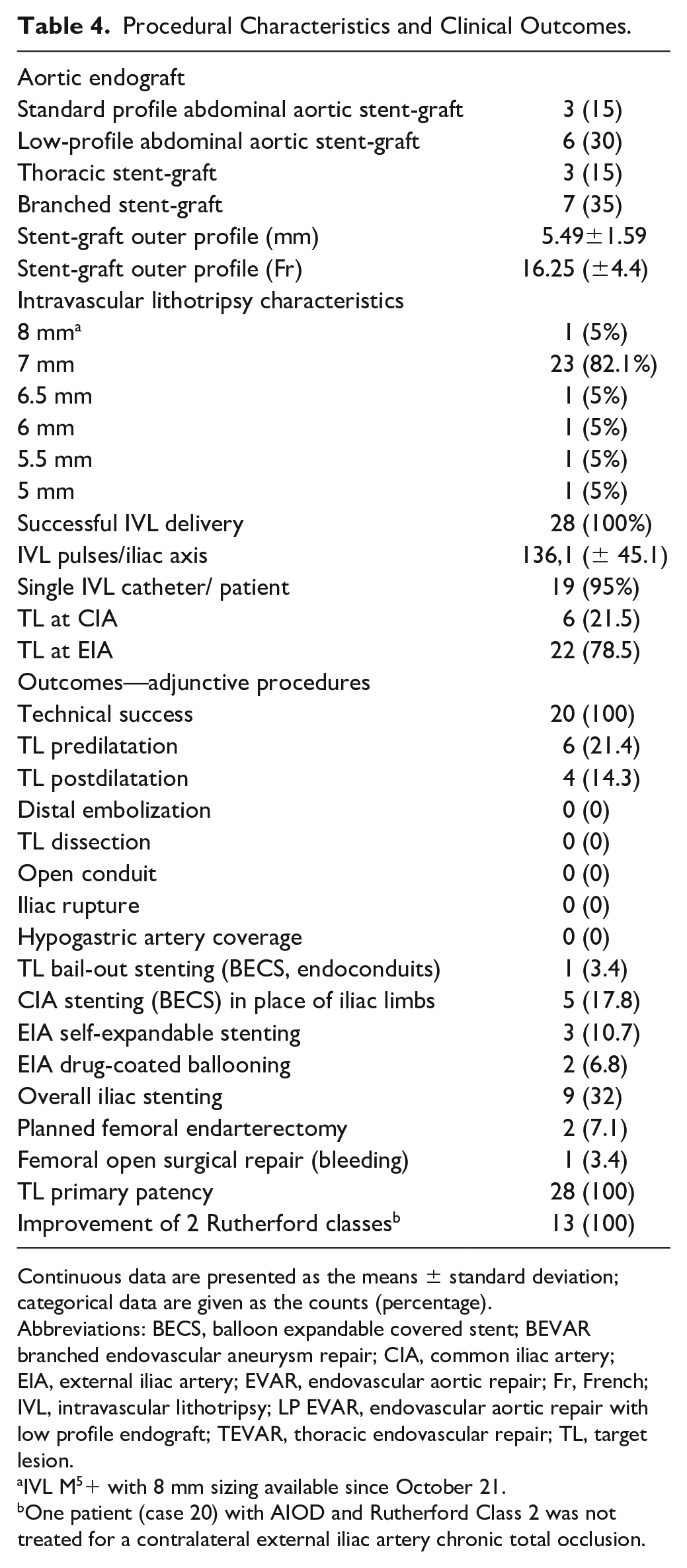
Continuous data are presented as the means ± standard deviation; categorical data are given as the counts (percentage).
Abbreviations: BECS, balloon expandable covered stent; BEVAR branched endovascular aneurysm repair; CIA, common iliac artery; EIA, external iliac artery; EVAR, endovascular aortic repair; Fr, French; IVL, intravascular lithotripsy; LP EVAR, endovascular aortic repair with low profile endograft; TEVAR, thoracic endovascular repair; TL, target lesion.
IVL M5+ with 8 mm sizing available since October 21.
One patient (case 20) with AIOD and Rutherford Class 2 was not treated for a contralateral external iliac artery chronic total occlusion.
Discussion
The main findings are as follows: (1) Shockwave IVL clearly facilitated the advancement through calcified iliac arteries of large-bore devices by significantly increasing vessel compliance, (2) no periprocedural ruptures occurred, confirming an excellent safety profile, (3) acute luminal gain and freedom from residual stenosis were significantly improved after IVL treatment.
Hostile and calcified iliac arteries are critical preoperative and intraoperative issues creating severe difficulties in achieving adequate access for endograft delivery. In the coronary and/or peripheral arteries, different calcium scores are currently used without clear indication for the iliac district in combination with aortic procedures.10,12,16–18
In this study, we introduced a new score (CASS) 11 considering anatomical vessels’ characteristics and device profile at the same time. The broad range of aortic endograft profiles, from 14 Fr up to 26 Fr, has been considered a key parameter in correlation to concentric calcified lesions; therefore, the SG/MLD index together with the severity of iliac calcium and tortuosity were combined to assess CASS and indicate the use of IVL as vessel preparation to facilitate aortic stent graft delivery.
In our opinion, the SG/MLD index is essential to give the right indication for IVL in large-bore access and to guarantee the ideal aortic SG to the patients. Some drawbacks should be considered: low-profile endografts are available for EVAR only, different IFUs are present for the aortic neck, and absence of frictions during rotations should be necessary during FB-EVAR deployments.
Iliac characteristics should be taken into account to correctly stratify the surgical risk in FB-EVAR. Endograft twisting and difficult target vessels cannulation is higher in hostile iliac access, procedures are technically more demanding (need of intraoperative adjunctive manouvers), and late mortality is increased. 19 Hertault et al 2 described 8 distinctive strategies to overcome access issues, including the use of preloaded renal catheters in the endograft delivery system or graft modification (branches instead of fenestrations) to allow an easier cannulation in case of misalignment. This approach is confirmed by our series with all branched endografts in case of TAAA.
In our study, all patients had one or both hostile iliac access with a mean MLD <4 mm associated to very severe (arc calcifications larger than 180°, 270°, or concentric in 35.7%, 17.8%, and 46% of the target lesions, respectively) and diffuse calcifications (mean extension of 50% compared with the iliac axis).
Despite using low-profile stent grafts whenever possible, the aortic stent graft outer profile was almost 50% larger than the MLD of the iliac vessels. Nevertheless, all the devices were safely delivered and no major complications occurred, showing how cracking calcium and achieving a better vessel compliance by using IVL plays a key role for this indication.
Currently available alternative techniques in such cases consist of open or endovascular procedures. The creation of an open iliac conduit is associated with higher surgical complications and mortality, and its use decreased 3-fold in the past 5 years. 1 Endovascular approaches can vary between balloon angioplasty and more complex techniques such as “paving and cracking.”
A retrospective study by Asciutto et al, 20 comparing endoconduits and open iliac conduits, showed a 57.9% complication rate of the endovascular treatment during the first 30 days after surgery.
The extension of the endoconduit from common to external iliac artery invariably covers the hypogastric artery, leading to a higher risk of pelvic ischemia, limb occlusion, and/or spinal cord ischemia. 21 Moreover, the placement of covered stent does not always guarantee the endograft delivery in case of calcific and concentric lesions. In our experience, no hypogastric artery coverage, nor rupture or open conversion occurred, demonstrating an excellent safety profile.
The use of IVL in large-bore access initially appeared in case reports describing its use to facilitate EVAR 7 and TEVAR. 8 Large registries exist and were published, analyzing the added value in TAVR or in peripheral endovascular treatments.
A recent multicenter registry evaluated the use of IVL in 108 patients to facilitate TAVR in complex iliac access with a technical success of 100%. An IVL-associated vascular complication occurred in 7.4%. The majority of dissections occurred in the group where a predilatation was performed (21.1% vs 3.4%, p<0.001). 22 In our study, we used predilatation with a 3 mm balloon, just to facilitate IVL catheter advancement through a tight calcified stenosis, if necessary.
Palmerini et al reported outcomes of 3 different access routes for 1707 TAVRs among which 518 were transfemoral: 183 patients were treated with lithotripsy and 335 with PTA alone prior to TAVR.
Despite assuming IVL has made possible transfemoral access to TAVR patients who were previously deemed unsuitable, they reported how this approach was used in case of low hostile access, considering calcifications and tortuosities of iliacs and femoral accesses. 23
Differently from TAVR, endovascular aortic procedures are feasible just from femoral and/or iliac access without alternative approaches, such as thoracic and/or axillary; for this reason, finding the better and safer option to overcome a severely calcified access route is extremely important, and IVL could represent a new adjunctive treatment for these cases.
From the iliac subgroup of Disrupt PAD III observational registry, Armstrong et al reported safe and effective results of 17 IVL cases to optimize delivery of large-bore devices. They described only procedural outcomes among which was a stenting rate of 41.2% (3.5% in our study). 24
Recently, a retrospective, single-center analysis with 9 patients, including 4 TAVR, 1 EVAR, 1 TEVAR, and 3 FEVAR was published, reporting perioperative outcomes of IVL in hostile access. Technical success was 100% despite that more than half the cases required adjunctive maneuvers. 25
As precise quantification of the anatomic effect of IVL was difficult to assess, future studies could consider a deeper analysis of acute luminal gain and its effect on vessels’ hemodynamic and changing compliance. From this point of view, the duplex ultrasound could be extremely useful as adjunctive completion assessment, showing the waveform improvement (multiphasic in place of monophasic) and the absence of significant stenosis/dissections. 26
Randomized trials and/or studies comparing IVL and other currently used techniques have never been investigated. A new level of evidence could be useful to determine the worthiness of IVL use and its role in heavily calcified lesions treatment.
Study Limitations
This study is limited by a small sample size and its retrospective design. Calcific lesions were analyzed for many characteristics, such as length, tortuosity index, and calcium score but no standardized calcification grading system is available. For this reason, we proposed a new CASS.
A new device (IVL M5+) with the same number of cycles has been used for the last group of enrolled patients; the availability of 8 mm sizing and the lower profile (6 Fr up to 7 mm) represents an improvement in terms of wider feasibility for ideal IVL sizing and access profile.
A larger Shockwave IVL catheter (L6, diameter of 8–12 mm) very recently has been released in United States and could be very effective in case of calcific lesions at the level of common iliac arteries.
While analysis from the initial experience is auspicious, further prospective studies with a larger sample of patients and/or matched control groups are mandatory to assess clinical effectiveness and long-term effects of the IVL. In this context, a multicenter prospective pilot study (SHOCK-ACCESS, ClinicalTrials.gov, number NCT0580641) has been recently approved to evaluate the use of Shockwave M5+ IVL catheter in hostile and calcified access during aortic endovascular procedures. 27
Conclusions
In this study, Shockwave IVL was safely and successfully applied to enable transfemoral EVAR, TEVAR, and BEVAR in patients with hostile calcified iliac arteries. The IVL represents a valid option to increase the feasibility of endovascular aortic procedures, facilitating endograft delivery and reducing the risk of iliac rupture and/or dissections by changing vessel compliance and optimizing luminal gain. Moreover, it represents a new tool not only to expand indications and improve immediate results, but also to get better clinical outcomes in patients with associated AIOD, confirmed by our good midterm results. Its ease of use and excellent safety profile gives this innovative technology a central role in the treatment of severely calcified access arteries. Ongoing prospective multicenter study will be useful to confirm our promising results.
Footnotes
Authors’ Note
Presented as talk at CX Symposium, London, 2023.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Stefano Fazzini and Michel Joseph Bosiers have a consulting agreement with Shockwave Medical.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
