Abstract
Introduction:
Data on target vessel (TV)-related outcomes in patients managed with branched thoracic endovascular aortic repair (BTEVAR) are limited. This study aimed to present the TV-related outcomes of BTEVAR in patients managed for aortic arch pathologies at 30 days and during follow-up.
Methods:
A retrospective analysis of consecutive patients, managed between September 1, 2011, and June 30, 2022, with custom-made aortic arch endografts (Cook Medical, Bloomington, IN, USA), presenting at least one branch configuration, were eligible. Primary outcomes were technical success, TV-related patency, and reinterventions at 30 days.
Results:
In total, 255 TVs were revascularized using branches: 107 innominate arteries (IAs), 108 left common carotid arteries (LCCAs), and 40 left subclavian arteries (LSAs). Covered stents were used as bridging stents of which 10.2% were balloon expandable. Relining, using bare-metal stents (BMS), was performed in 14.0% of IAs, 35.2% of LCCAs, and 22.5% of LSAs. Technical success on case basis was 99.2%; no failure was related to unsuccessful TV bridging. At 30 day follow-up, no TV occlusion was detected. In 5.6% of cases, a type Ic or III endoleak, attributed to TVs, was recorded. Two patients needed early branch-related reintervention. The mean follow-up was 18.3±9.2 months. Freedom from TV instability was 94.6% (standard error [SE] 2.5%] at 12 months. No TV stenosis or occlusion was detected up to 48 months of follow-up. Freedom from TV-related reinterventions was 95.4% [SE 2.4%] at 12 months.
Conclusion:
TV stenosis or occlusion in BTEVAR cases is rare and TV-related reinterventions and instability events are mainly attributed to type Ic and III endoleak formation.
Clinical Impact
Previous studies focusing on target vessel (TV) outcomes after endovascular aortic arch repair are limited. In this study, including 255 TVs revascularized using branched arch devices, bridging was performed with covered stents, of which 90% were self-expanding. Relining was at the discretion of the operator and was 14% for the innominate, 35.2% for the left common carotid and 22.5% for the left subclavian artery branches. No 30-day occlusion was detected. The freedom from TV instability was almost 95% at 12 months. TV instability and reintervention were mainly attributed to endoleaks type Ic and IIIc.
Introduction
Branched thoracic endovascular aortic repair (BTEVAR), when anatomically feasible, represents a valuable alternative in patients with pathologies involving the aortic arch. 1 Previous studies showed encouraging outcomes with low early mortality and acceptable follow-up survival rates.2,3 According to the published literature, almost 30% of patients may need an unscheduled secondary intervention during the early follow-up, related to vascular access complications, endoleaks, target vessel (TV)-stenosis, or occlusion. 4 In the same analysis, the reported TV patency rates have been high within the initial year and freedom from TV instability was exceeding 90%. 4
Previous experience of complex endovascular repair in the thoracoabdominal and visceral aorta has shown that fenestrated and branched endografts had acceptable TV-related adverse event rates, while analyses regarding the type of bridging covered stents (BCS) are for the moment inconclusive, providing similar patency rates for both balloon-expandable covered stents (BECS) and self-expanding covered stents (SECS).5–7 These findings, confirming the safety and feasibility of fenestrated and branched aortic repair, in terms of TV outcomes of the visceral aorta, do not allow conclusions for aortic arch cases as supra-aortic trunks (SATs) present significant anatomic differences. Currently, data on TV-related outcomes of the SAT after BTEVAR are quite limited.
The aim of this study was to present the TV-related outcomes of BTEVAR in patients managed for aortic pathologies involving the aortic arch at 30 days, and during the available follow-up, in a single center with >10 years experience in BTEVAR.
Materials and Methods
Study Design
The STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) statement was followed. 8 A retrospective analysis of consecutive patients, managed between September 1, 2011, and June 30, 2022, with custom-made aortic arch endografts and presenting at least one branch configuration for SAT bridging were considered eligible. Urgent cases were not excluded. This study complied with the Declaration of Helsinki and was considered exempt from the need for ethical approval, given its retrospective nature and the use of anonymized data in accordance with local laws. All endovascular aortic interventions were performed in a dedicated hybrid operating room with a fixed imaging system by 4 experienced vascular surgeons. Since January 1, 2015, fluoroscopic fusion guidance was also used to perform complex aortic endovascular repairs, including BTEVAR (Vessel Navigator, Phillips Healthcare, Best, Netherlands). A part of the data presented in this analysis has been previously published, as retrospective cohort studies or after participation in multicenter studies.2–4,9,10
Patient Population
The indications for treatment of the different aortic arch pathologies followed the available guidelines at the time of repair. 1 Patients were discussed in a multidisciplinary aortic board meeting and a decision for endovascular management was made. 2 According to the anatomic restrictions provided by the specific manufacturer (Cook Medical, Bloomington, IN, USA), a proximal landing zone longer than 30 mm and a proximal diameter ≤38 mm, and a distal landing zone ≥30 mm (for single-stage procedures) are considered adequate for endovascular management using custom-made devices (CMDs). 2
Patients with a connective tissue disorder initially had an open repair to provide an adequate proximal landing zone. 9 Patients treated with FTEVAR (including endografts only with fenestrations and/or scallops), surgeon modified grafts, including in situ modified endografts, and the chimney technique were excluded from this analysis.
Technical Characteristics
Only CMDs based on the Zenith platform (Cook Medical) were used. 2 In case of patients needing urgent management, as in symptomatic or ruptured lesions, interventions were performed using available custom-made endografts designed either for the specific patient or from another with compatible anatomy. 10
Technical details on endograft’s deployment have been previously published.2,11 Branched endografts were constructed with 1, 2, or 3 internal funnels according to the number of TV intended to be revascularized and depending on the presence of SAT debranching (eg, left subclavian artery [LSA] transposition or bypass). Landing was mostly in the ascending aorta in zone 0. 2 Depending on the number and anatomy of intended TVs, retrograde access was used from the right common carotid artery (RCCA), right subclavian artery (RSA), the left common carotid artery (LCCA), or left brachial artery (LBA). 2 When available, left carotid-subclavian bypass was used for BCS advancement to the LCCA. 2 Other than left carotid-subclavian bypass, and depending on patient’s anatomy, LSA transposition, right carotid-subclavian bypass, right to left axillary bypass, and LCCA-RCCA bypass were performed. Upper access was used in all cases.
Regarding BCS selection, innominate arteries (IAs) were mostly revascularized using patient-specific bridging limbs (Cook Medical), whereas balloon-expandable, self-expanding, or a combination of covered stents were used as bridging stent grafts for the LCCA and/or LSA. 2 Among BECS, Advanta V12 (Atrium Medical Corporation, Merrimack, NH, USA), BeGraft Peripheral (Innomed, Bentley, Hechingen, DE), VBX (Gore & Associates, Flagstaff, AZ, USA), and Lifestream (Bard Medical, GE, USA) were used, depending on operator’s preference. Among SECS, Fluency (Bard Medical), Covera (Bard Medical), Viabahn (Gore & Associates), and WRAPSODY Cell-impermeable Endoprosthesis (Merit Medical Systems, Inc., South Jordan, UT, USA) were applied, with stent choice relying on operator’s discretion. Relining using self-expanding bare metal stents was also at the discretion of the operator and performed with Protégé Everflex (Medtronic, Minneapolis, MN, USA) or Wallstent (Boston Scientific, Marlborough, MA, USA), according to the findings of the intraoperative imaging. Bridging stent oversizing was decided according to the diameter of the TV landing zone; for BECS, the diameter of the stent was equal to TV diameter at the level of the distal landing, whereas for SECS, an oversizing of at least 1 mm according to TV diameter was chosen. For the IA, landing aimed to cover the complete length of the IA, whereas for the LSA the anatomic limit was in most cases the left vertebral artery (LVA). In cases in which the IA could not provide a safe distal landing zone due to length, kinking, or dissection, an RCCA to RSA bypass was used, providing a distal landing zone in the RCCA. 12
Postoperative Surveillance
Follow-up included computed tomography angiogram (CTA) before discharge, at 12 months, and yearly thereafter. Survival and TV-related adverse events, endoleaks type Ic or III, stenosis or occlusion, instability, and reinterventions were documented during surveillance.
Data Collection
Data were introduced in a local database, including baseline and in-hospital information, and underwent pseudonymization to permit further assessment and retrospective introduction of follow-up information. Patients’ data included demographics (age, sex), comorbidities; coronary artery disease (CAD), myocardial infarction (MI), chronic heart failure (CHF), coronary-aortic bypass grafting (CABG), percutaneous coronary angioplasty (PTCA), hypertension, dyslipidemia, ever or active tobacco use, chronic obstructive pulmonary disease (COPD), diabetes mellitus (DM), chronic renal disease (CRD), previous stroke, and peripheral arterial disease. The American Society of Anesthesiologists (ASA) score was also recorded. Any previous aortic valve repair or replacement using metallic valve and any previous open ascending or aortic arch replacement were recorded.
Any underlying pathology, including degenerative or post-dissection aortic arch aneurysms, penetrating aortic ulcers (PAUs), intramural hematomas (IMH), or thoracic or thoracoabdominal aneurysms, with inadequate landing below the LSA, was recorded. The type of aortic dissection (type A or B) was documented, including primary or residual type A aortic dissection. 1 Proximal extension of the disease according to Ishimaru zones was recorded, in addition to aortic arch type: I, II, or III, and presence of bovine aortic arch or aberrant subclavian artery. 1 The presence of stenosis exceeding 50% and dissections extending into the TVs were also noted.
Technical success and any previous or concomitant debranching was noted. Bridging stents characteristics, including SECS or BECS, as well as the use of relining were recorded and analyzed for all 3 SATs. Any 30 day death and major strokes were recorded. TV-related adverse events and reinterventions at 30 days and follow-up were analyzed.
Definitions
Technical success was defined as the successful introduction and deployment of the device at the intended landing zone, with completion of the procedure with TV preservation, absence of surgical conversion or mortality, or endograft or bridging stent occlusion or stenosis. 2 The presence of endoleaks type I or III at final angiography was not considered a criterion for technical failure as in many cases of complex endovascular repair these endoleaks present rather a self-limited behavior within the initial few weeks. 13 Postoperative strokes were classified according to Rankin scale to minor (Rankin score 1–3) and major (Rankin score 4–7). 13 For definitions, due to lack of reporting standards on the endovascular repair of pathologies involving the aortic arch, the reporting standards of the Society of Vascular Surgery for the endovascular aortic repair of aneurysms involving the renal-mesenteric arteries were used, when feasible. 13
Specifically for comorbidities, CAD was defined as any anamnesis of MI and/or electrocardiogram alterations and/or stable defect on dipyridamole-thallium scan and/or stable angina, as confirmed by the preoperative cardiologic evaluation. 13 Myocardial infarction was considered as any confirmed anamnesis (regardless the time of evolvement) of coronary artery syndrome with typical clinical symptoms and/or electrocardiographic changes and/or troponin elevation. Chronic heart failure was any clinical syndrome caused by structural and/or functional cardiac alterations, corroborated by high natriuretic peptide levels and/or pulmonary or systemic congestion, as defined in the preoperative cardiologic evaluation. Coronary-aortic bypass grafting and percutaneous coronary intervention (PCI) were considered as comorbidities when the patient had undergone a previous bypass or angioplasty of the coronary arteries, with or without stenting, and regardless the vessels needing intervention. Hypertension was indicated in any patient with a known history of systolic pressure above 140 mm Hg and/or diastolic pressure above 90 mm Hg, as well as in any patient receiving medication. Similarly, for dyslipidemia and DM, a known disorder of the lipidemic profile or statin treatment was considered as an anamnesis of dyslipidemia, whereas any patient under antidiabetic medication, including insulin or under surveillance for increased glucose values, was considered as diabetic. Chronic obstructive pulmonary disease was any confirmed chronic pulmonary disease characterized by progressive airflow limitation or the use of dedicated medication or need for oxygen therapy. Chronic renal disease was defined as a glomerular filtration rate (GFR) <60 ml/min/1.73 m2. Stroke was the history of any neurological deficit attributed to cerebral infarction as the result of focal injury of the central nervous system, whereas peripheral arterial disease was any anamnesis of previous peripheral open or endovascular intervention, present intermittent claudication, or critical limb ischemia.
Patients were preoperatively evaluated by a cardiologist, using electrocardiogram and transthoracic echocardiography. According to their medical history and findings, further evaluation, including functional tests, was decided. For patients with a history of pulmonary disease (eg, COPD). a preoperative spirometry in addition to thoracic x-ray was performed. The preoperative workup outcomes were evaluated by an anesthesiologist before the procedure.
Outcomes
Primary outcomes were 30 day TV-related outcomes, including patency and reinterventions. The TV-related outcomes during follow-up were assessed as secondary outcomes.
Statistical Analysis
Continuous data were reported as mean±SD. Categorical data were expressed as absolute numbers and percentages. Independent 2-sample t tests were used for normally distributed continuous variables, and the Mann-Whitney U test for non-normally distributed continuous and ordinal variables. Chi-square test was used for categorical data comparison. Kaplan-Meier estimates were performed to assess follow-up outcomes. No correction for multiple hypothesis testing was applied. Statistical analysis was performed by SPSS 22.0 for Windows software (IBM Corp, Armonk, NY, USA).
Results
Patient Cohort
In total, 116 patients were managed with BTEVAR, whereas 10 patients were managed with scallop and branched configuration. Seventeen patients (14.7%) were managed using 3-branch arch devices, with 2 antegrade and 1 retrograde branch for the LSA; 4 patients were managed with single branch device; and 95 patients were managed with double-branch devices (85 with branches targeting the IA and LCCA). The mean age for the total cohort was 70.8±4.8 years and 60.3% were men. Among comorbidities, the most common was hypertension, reported in 90.5% of patients. Comorbidities are presented in Table 1.
Comorbidities Distribution in Patients Treated With Branched Thoracic Endovascular Aortic Repair.
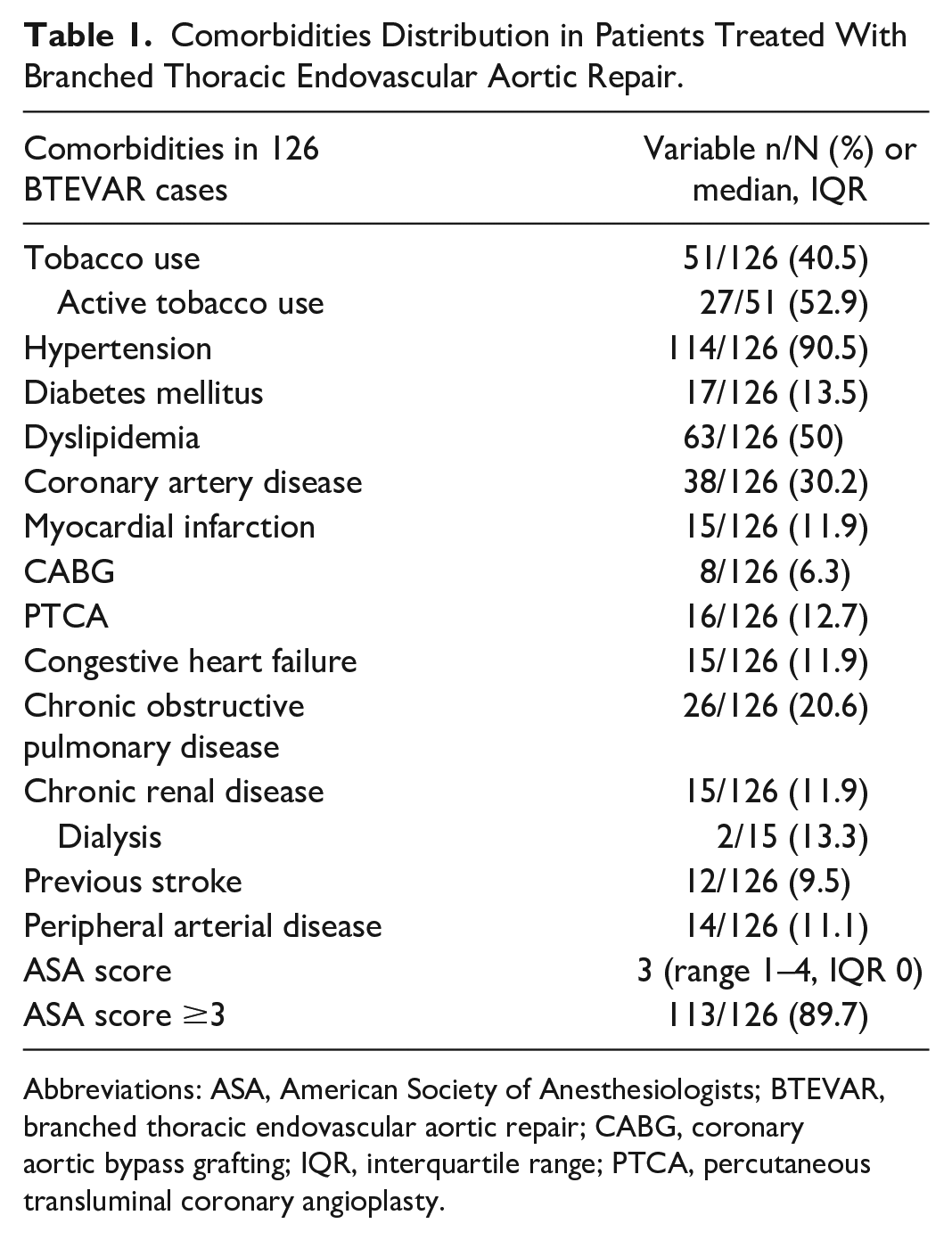
Abbreviations: ASA, American Society of Anesthesiologists; BTEVAR, branched thoracic endovascular aortic repair; CABG, coronary aortic bypass grafting; IQR, interquartile range; PTCA, percutaneous transluminal coronary angioplasty.
Seven (5.6%) patients presented a bovine arch configuration, whereas aortic arch type distribution was 16.7% type I, 59.5% type II, and 23.8% type III. Sixty (47.6%) patients had an aortic dissection and 23.3% (14/60) had a type B aortic dissection. The mean aortic diameter was 63.5±5.8 mm. Among patients, 45 (38.8%) had a history of ascending aortic repair and 26 (20.6%) of previous aortic arch repair. The lesions’ characteristics are given in Table 2.
Aortic Lesions’ Classification, According to the Indication for Repair, and Proximal Extension of the Disease, According to Ishimaru’s Zones.
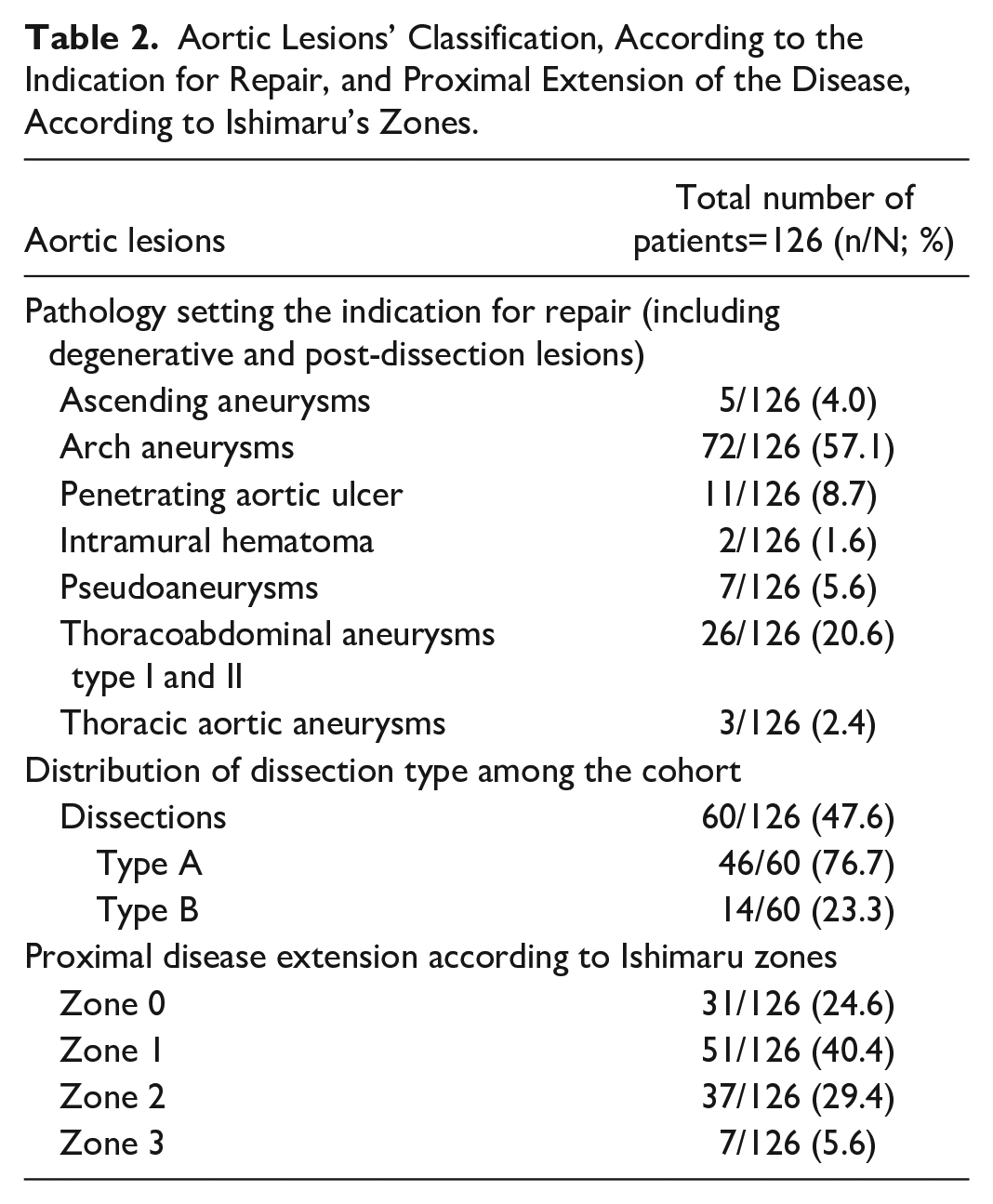
Target Vessel Findings
In total, 265 TVs were addressed in 126 patients, 255 using branches. Among them, 107 were IAs, 108 were LCCAs, and 40 LSAs. Among the bridged SAT, TV stenosis exceeding 50% was detected in 0.9% of the IAs, 0% of the LCCA, and 0% of the LSAs while 15.8% of the IAs were dissected, 6.5% of the LCCAs and 2.5% of the LSAs. Ninety-eight (77.8%) patients needed preoperative debranching; 6 of them underwent more than 1 revascularization procedures. Eighty-four (85.7%) LCCA-LSA bypasses and 5 (5.1%) LSA transpositions were performed. A right carotid-subclavian bypass was performed in 8 cases (in 5, the right bypass was combined with a LCCA-LSA bypass), 5 patients needed a right to left axillary bypass, and 2 needed an LCCA-RCCA bypass (in 1, it was combined with an LCCA-LSA bypass). Upper access was used in all cases.
Regarding the distribution of bridging stents, the IAs were bridged in 91 cases (85%) using a patient-specific bridging stent-graft; in 14 (13.1%) cases using an SECS while in 2 cases (1.9%), with landing within the RCCA, BECS were used. Similarly, for the LCCA and LSA, 17 (15.7%) and 7 (17.5%) cases, respectively, were revascularized using BECS (16.2%). The remaining LCCAs and LSAs were revascularized using SECS (Table 3).
Bridging Stent Distribution Among Target Vessels.
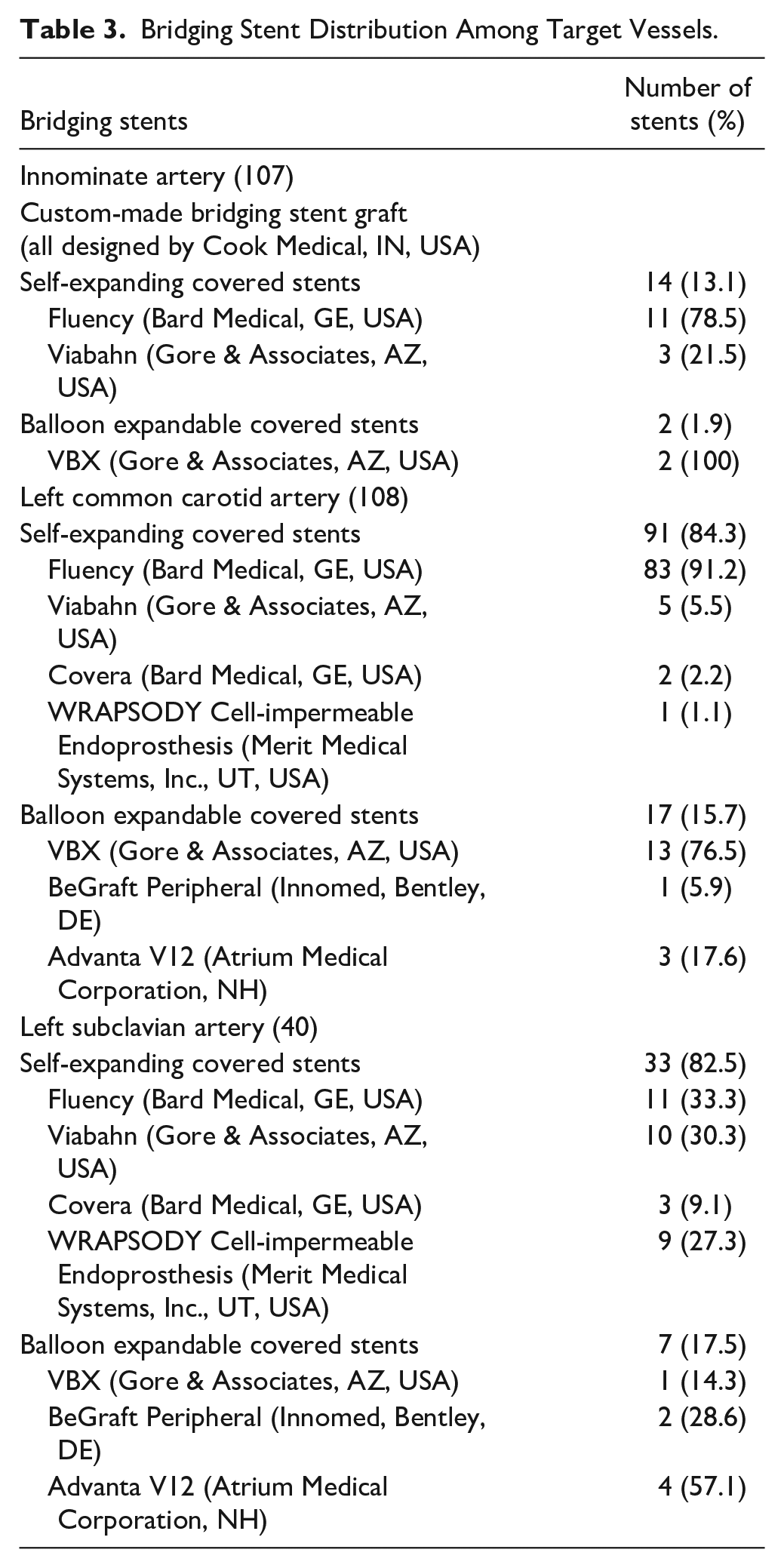
As stated in the “Methods” section, the application of relining was at the discretion of the operator and relied on the intraoperative imaging findings. Relining using self-expanding bare-metal stents (BMS) was performed in 14.0% of IAs, 35.2% of LCCAs, and 22.5% of LSAs. In 11.2% of the IAs, 12% of the LCCAs, and 25% of the LSAs, an extension using an additional covered stent was needed. In 83 cases of double-branch devices, the LSA was revascularized in a staged manner and intraoperatively occluded using vascular plugs (93.7%), whereas in the remaining cases, coil embolization was performed for the occlusion of the LSA.
Early Outcomes
Technical success was 99.2% among patients. One patient presented signs of hemodynamic instability and needed urgent conversion for cardiac tamponade drainage. All TVs were successfully bridged. Endoleaks type Ic and III were detected on final angiography in 4 cases (3.1%). None of them were considered significant and all patients were managed conservatively until the predischarge CTA.
The total 30 day mortality rate was 10.3%, while strokes were recorded in 14 cases, leading to a 11.0% rate (major 7.1%). None of these events was related to TV occlusion or stenosis. Among 126 patients, 124 underwent a predischarge CTA. No TV occlusion was recorded. One patient presented a bridging stent LCCA stenosis and was treated by relining with BMS. Furthermore, TV-related (type Ic and IIIc) endoleaks were detected in 7 (5.6%) patients in the predischarge CTA. Among them, 1 patient was managed with reintervention due to endoleak type Ic. An extension of the LCCA bridging stent was performed before discharge. For the remaining 5 cases, a conservative management was decided until reevaluation while 1 patient died due to cardiac complications (he presented with a type IIIc endoleak between the arch component and the IA bridging stent).
Follow-up Outcomes
The mean follow-up was 18.3±9.2 months. The loss to follow-up was 32%. The estimated survival at 12 months was 82.7% (standard error [SE] 3.8%) and 79.4% (SE 4.3%) at 24 months. Two deaths were aneurysm-related during follow-up: 1 related to abdominal aortic aneurysm rupture (6 months) and 1 related to graft infection (3 months).
Nine events (per case analysis) of TV instability were recorded; none of them led to patient’s mortality. The estimated freedom from TV instability was 94.6% (SE 2.5%) at 12 months and 90.4% (SE 3.7%) at 24 months (Figure 1). The freedom from TV-related endoleaks was 92.8% (SE 3.4%) at 24 months (Figure 2). Five events of TV-related endoleaks were recorded; 4 type Ic (2 related to IA branch and 2 to the LCCA branch) and 1 type IIIc (related to IA branch). Among them, the patient with the type IIIc died in-hospital due to cardiac complications, before having further evaluation or surgical treatment. For the type Ic endoleaks, 4 were managed with stent extension, whereas 1, related to the IA branch, was managed with coil embolization of the false lumen. Regarding the time of evolvement, as depicted also in Figure 2, 2 events were diagnosed within 30 days, 1 at 6 months, and 2 at 24 months.
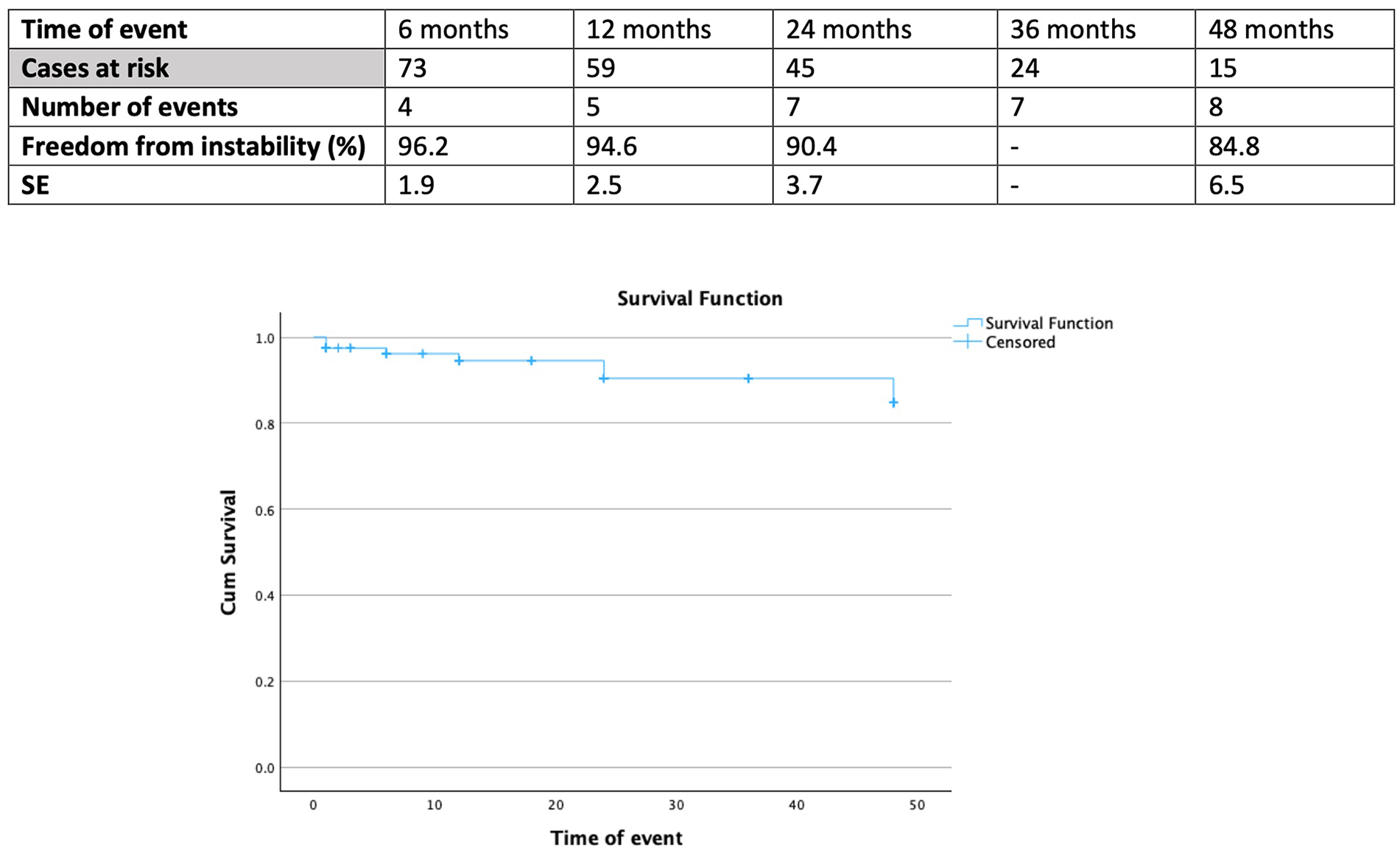
Follow-up freedom from target vessel instability in 126 patients managed with branched thoracic endovascular aortic repair for aortic pathologies involving the arch. SE, standard error.
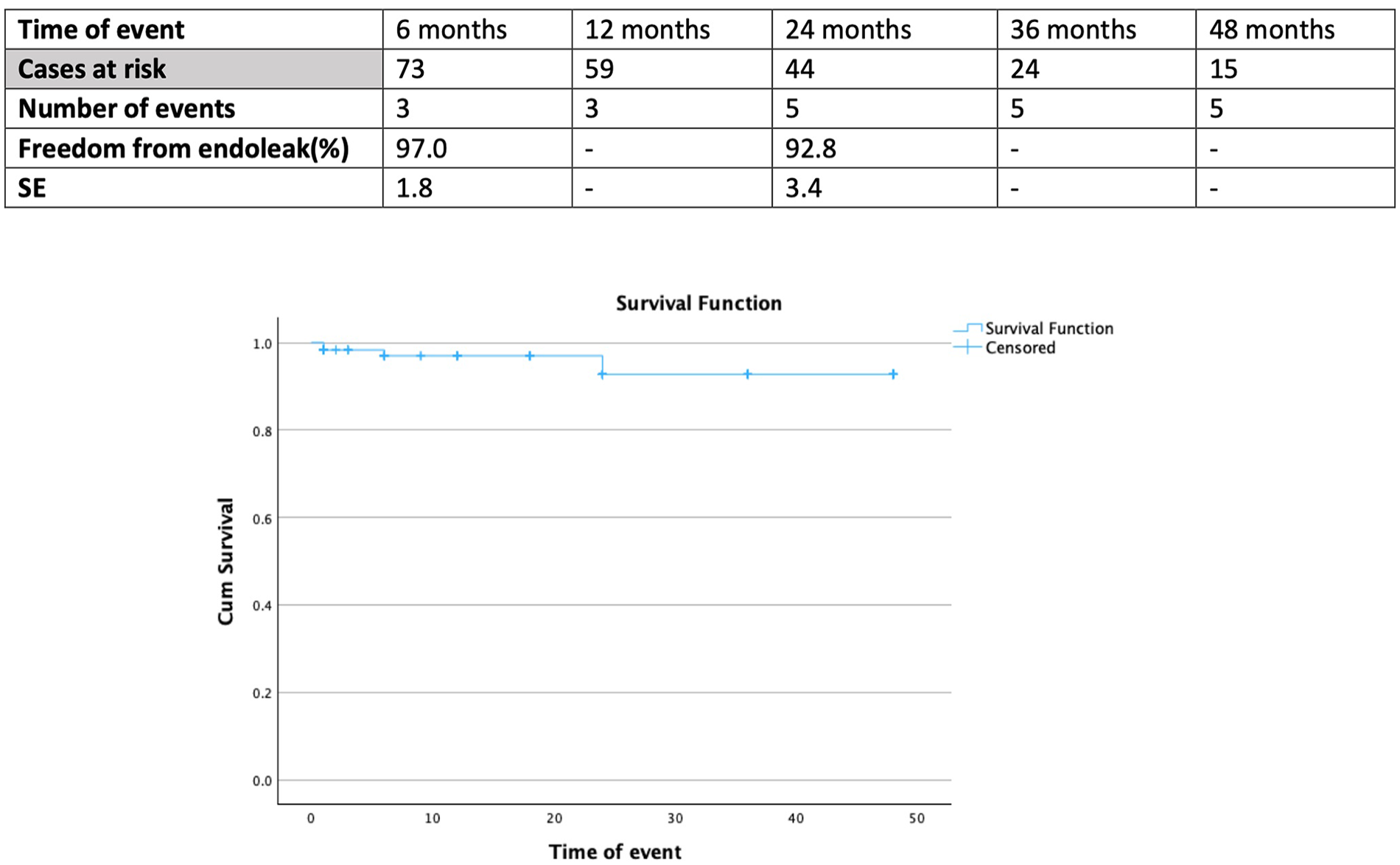
Follow-up freedom from target vessel–related endoleak in 126 patients managed with branched thoracic endovascular aortic repair for aortic pathologies involving the arch. SE, standard error.
The estimated freedom from TV stenosis or occlusion was 93.0% (SE 6.1%) at 48 months (Supplemental Figure S1). Except the patient with the in-hospital LCCA managed with relining, another patient was diagnosed with LSA stenosis, without clinical consequences, managed with recanalization and stenting using a balloon-expandable and a self-expanding bare metal stent at 48 months of follow-up. An additional event was recorded at 96 months of follow-up in a patient presenting with an anastomotic stenosis of an LCCA-LSA bypass managed with stenting using 2 balloon expandable stents: 1 covered and 1 bare metal.
Freedom from TV-related reinterventions was 95.4% (SE 2.4%) at 12 months and 91.1% (SE 3.7%) at 24 months (Figure 3). All reinterventions were endovascular, except for 1 case needing carotid endarterectomy in a patient with aortic dissection extension to the LCCA. The remaining reinterventions are as described previously: 3 were related to TV stenosis/occlusion and 4 were related to type Ic endoleaks (Supplemental Table S1).
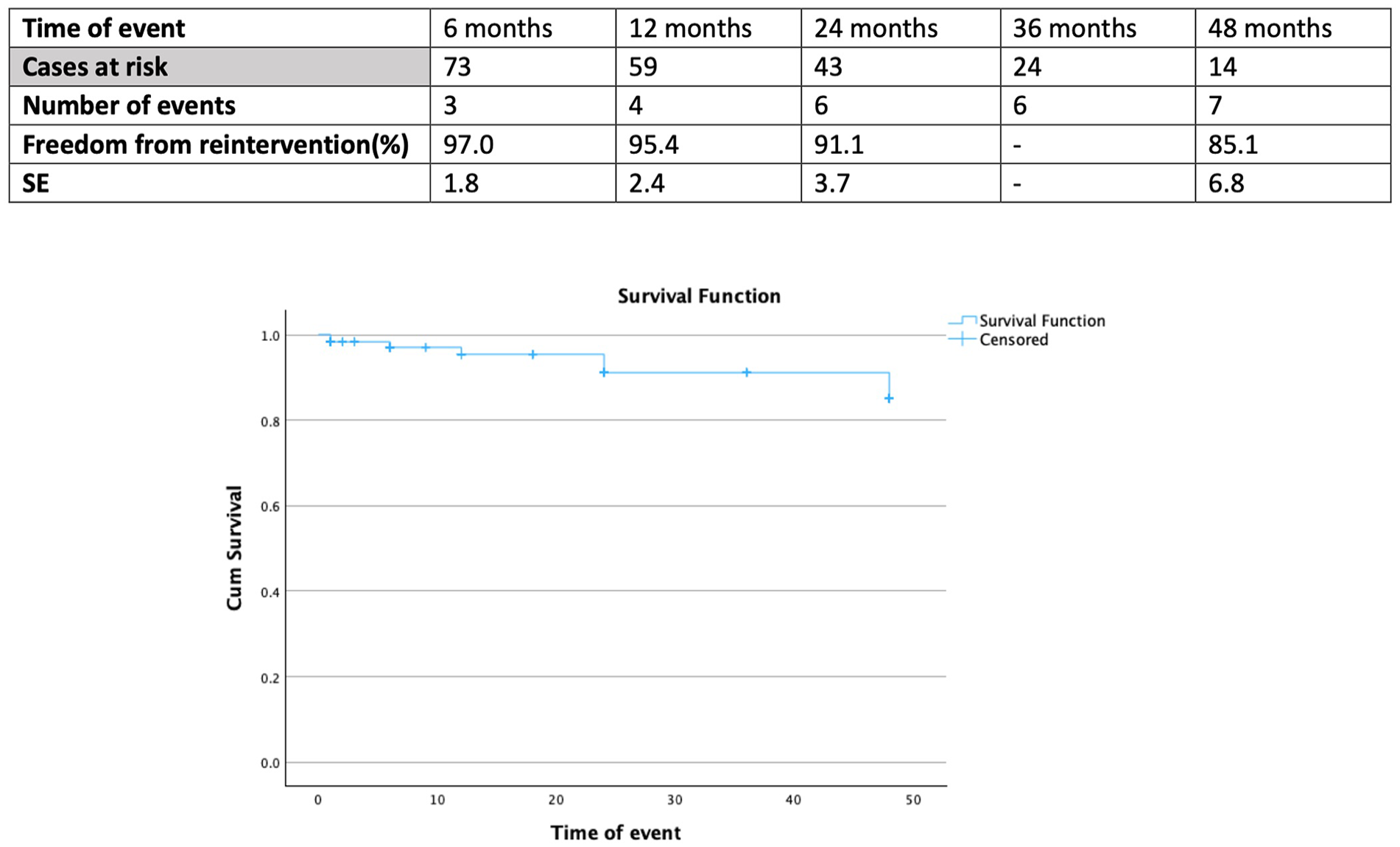
Follow-up freedom from target vessel–related reintervention in 126 patients managed with branched thoracic endovascular aortic repair for aortic pathologies involving the arch. SE, standard error.
Discussion
Branched thoracic endovascular aortic repair was related to low mortality and acceptable cerebrovascular adverse event rates compared with previously reported outcomes from our own experience as well as in patients needing extensive aortic repair.2,3,14,15 Furthermore, TV-related outcomes and adverse events have been extensively investigated in patients managed with complex endovascular repair of the thoracoabdominal aorta and showed high freedom from occlusion.6,16 However, almost 25% of patients needed a secondary TV-related intervention. 16 Studies reporting on TV-related adverse events in BTEVAR are limited. 4 However, high patency rates are reported. 4 This analysis showed that BTEVAR is safe regarding TV outcomes, with 100% technical success and no early occlusion, whereas re-interventions were mainly attributed to TV-related endoleaks. However, none of the TV-related adverse events was associated with mortality.
In addition, TV instability is a term used to describe the composite outcome of TV adverse events and define durability of branches in visceral aortic repair. 17 This definition seems applicable also in BTEVAR cases to summarize TV-related outcomes. In this analysis, the estimated freedom from TV instability was 90.4% at 24 months of follow-up and mainly related to endoleaks than occlusions. Previous studies, reporting on fenestrated and branched aortic arch repair, provided similarly encouraging findings in terms of freedom from TV instability.4,18 In patients managed with triple-branch devices, the estimated freedom from TV instability was 91% at 12 months; a rate conforming to the findings of this analysis. 4
As previously reported, TV patency was high at 12 months in patients managed with BTEVAR, suggesting that TV instability in SATs mainly relates to the presence of type Ic and IIIc endoleaks. 4 In this analysis, TV-related endoleaks, including type Ic and III, were estimated at 7% within 24 months. The 30 day outcomes also signified a rate at 5% of type Ic and IIIc endoleaks. However, among cases with early endoleaks detected in the predischarge CTA, only 1 patient with a type Ic of the LCCA required an early secondary intervention at 30 days. Initial identification of endoleaks in predischarge CTAs may not always trigger immediate reintervention as many of these patients may benefit from a “wait and see” approach until the first follow-up evaluation. At least according to our experience, as also reported in the literature, early endoleak detection in complex endovascular procedures should not be interpreted as failure as these endoleaks tend to disappear within a few weeks. 13 In our cohort, 5 out of 7 endoleaks were self-limited.
On the contrary, TV occlusion seems to be a rather sporadic phenomenon in this analysis and also in previous studies.3,4 With the exception of 1 LCCA stenosis detected and managed before patient’s discharge from the hospital, a second TV stenosis was diagnosed in 1 case at 48 month follow-up and had no clinical impact. Anatomic factors, including large TV diameters and adequately long landing zones may contribute to this low stenosis and occlusion rate. Both in CMD and off-the-shelf arch devices, TV patency seems similarly high.19,20 Further investigation, including SAT anatomy and bridging stent characteristics, would add valuable information.
In addition, TV reintervention rates were low in this analysis; less than 10% within the 24-month follow-up and comparable to the current literature. 4 All reinterventions were performed with endovascular means, except for a case needing an open repair for a dissection of the LCCA. In this analysis, no late stroke was recorded and all events were attributed to the main procedure. However, despite the low reintervention rate shown in this analysis, follow-up is mandatory for the prevention of major adverse events, including strokes related to TV occlusion or stenosis. 21
The choice of BCS may affect TV outcomes. For the moment, no data exist on the impact of bridging stents’ characteristics on TV adverse events in BTEVAR. However, in the current cohort, the vast majority of the LCCA and LSA were stented using SECS, whereas 85% of the IAs were bridged using a patient-specific bridging stent-graft. The lack of adverse events did not permit further analyses on the role of the specific stent-type. Regarding the potential stent-selection, especially IA bridging, except for the use of patient-specific limbs, large SECS were applied in selected cases, whereas balloon-expandable stents were used only when landing was performed in the RCCA. For the LCCA and LSA stenting, a variety of stent types were used, depending on patient anatomy, surgeon’s preference, and availability of materials.
On the potential role of relining on aortic arch TVs, at least to our knowledge no data are available. In this cohort, relining was applied depending on the intraoperative imaging and surgeons’ opinion. This led to a quite high rate, especially for LCCA branches, where almost 1 in 3 arteries was relined with a self-expanding bare metal stent. The need to reassure carotid patency, to avoid stroke events in the future, could be a subjective criterion for relining application. As the occlusion rate in this cohort was low, an analysis on the role of relining on TV adverse events was not feasible. A correlation between more aggressive relining and high TV patency in endovascular arch repair cannot be excluded.
Limitations
The retrospective nature of this study is the main limitation of this study. Type II errors cannot be excluded due to the number of cases. In addition, the inclusion of patients with various underlying pathologies and extent of coverage (including single-, double-, or triple-branched devices) could affect the outcomes of this analysis and further studies are needed to report on TV-related adverse events in each subgroup of cases. To our knowledge, this study represents the only available literature focusing on TV adverse events during the early and midterm follow-up in patients managed with BTEVAR. Longer follow-up may provide further information on the durability of the technique. The findings of this analysis should be evaluated under the presence of 32% loss to follow-up, which may affect outcomes. This rate may lead to false estimation of the real need for secondary procedures and underestimation of the mortality rates during follow-up. Anatomic data, including length of landing zones and TV diameter, were not reported and further investigation of this topic may provide additional information. The lack of TV-related adverse events, along with the conformity of bridging stent type, did not permit for the moment further investigation on the potential factors affecting outcomes.
Conclusion
Target vessel stenosis or occlusion in patients managed with BTEVAR is rare at 30 days and during midterm follow-up. TV-related reinterventions and instability are mainly attributed to type Ic and IIIc endoleak formation, which affect almost 9% of patients at the initial 24 months of follow-up.
Supplemental Material
sj-docx-1-jet-10.1177_15266028241231905 – Supplemental material for Target Vessel–Related Outcomes in Patients Managed With Branch Thoracic Aortic Endovascular Repair
Supplemental material, sj-docx-1-jet-10.1177_15266028241231905 for Target Vessel–Related Outcomes in Patients Managed With Branch Thoracic Aortic Endovascular Repair by Petroula Nana, Giuseppe Panuccio, Fiona Rohlffs, Konstantinos Spanos, Jose I. Torrealba and Tilo Kölbel in Journal of Endovascular Therapy
Supplemental Material
sj-docx-2-jet-10.1177_15266028241231905 – Supplemental material for Target Vessel–Related Outcomes in Patients Managed With Branch Thoracic Aortic Endovascular Repair
Supplemental material, sj-docx-2-jet-10.1177_15266028241231905 for Target Vessel–Related Outcomes in Patients Managed With Branch Thoracic Aortic Endovascular Repair by Petroula Nana, Giuseppe Panuccio, Fiona Rohlffs, Konstantinos Spanos, Jose I. Torrealba and Tilo Kölbel in Journal of Endovascular Therapy
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.K. is a consultant and proctor for, and has intellectual property with, Cook Medical, receiving royalties, speaking fees, and research, travel, and educational grants. All authors have completed the International Committee of Medical Journal Editors (ICMJE) uniform disclosure form and declare no support from any organization for the submitted work, no financial relationships with any organizations that might have an interest in the submitted work, and no other relationships or activities that could appear to have influenced the submitted work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
