Abstract
Purpose:
Renal artery aneurysm (RAA) is a rare disease. This study proposed and evaluated a new classification for RAA to assist in surgical decision-making.
Materials and Methods:
Single-center data of 105 patients with RAAs from the vascular department of vascular surgery were collected retrospectively. A new classification scheme was proposed. Type I aneurysms arise from the main trunk, accessory branch, or first-order branches away from any bifurcation. Type II aneurysms arise from the first bifurcation with narrow necks (defined as dome-to-neck ratio >2) or from intralobular branches. Type III aneurysms with a wide neck arise from the first bifurcation and affect 2 or more branches that cannot be sacrificed without significant infarction of the kidney.
Results:
There was 50 (47.62%) type I, 33 (31.43%) type II, and 22 (20.95%) type III aneurysms. The classification assigned endovascular repair as first-line treatment (for type I or II), while open techniques were conducted if anatomically suitable (for type III). A kappa level of 0.752 was achieved by the classification compared with a level of 0.579 from the classic Rundback classification. Technical primary success was achieved in 100% and 96.05%, and symptoms were completely resolved in 100% and 84.85%, while hypertension was relieved in 84.21% and 72.92% of patients receiving open surgery or endovascular repair, respectively. No significant difference was observed for perioperative or long-term complications among the 3 classification types.
Conclusion:
The new classification proved to be a convenient and effective method for facilitating choice of intervention for RAAs.
Clinical Impact
This study proposed and evaluated a new classification scheme for renal artery aneurysms, which proved to be a convenient and effective method for facilitating surgical decision-making. Coil embolization was the first-line treatment if suitable, while aneurysm resection and reconstruction with vein graft were conducted for some complex lesions. The safety and efficacy of both open and endovascular methods were validated.
Introduction
Renal artery aneurysm (RAA) is a rare disease that affects approximately 0.1% of the general population. 1 Most RAAs are found incidentally during imaging and remain asymptomatic, while some may present with pain, hydronephrosis, or hematuria. 2 Roughly, two thirds of RAA patients have concurrent hypertension and renal artery stenosis may coexist.1,3 Despite its slow-progressive nature, RAA could be present with a non-gestational mortality rate of 10%. 4
Open and endovascular therapies are both commonly used to treat RAAs.5,6 In 2000, Rundback et al introduced a classification system for RAAs. 7 Saccular aneurysms arising from the main renal artery or proximal large segmental branch are classified as type I, fusiform aneurysms at the same location are classified as type II, and intralobular aneurysms or those on accessory arteries are considered type III. Endovascular therapy was suggested for type I or III, while open surgery was suitable for type II.7,8 However, the advancements in vascular surgery techniques and devices have broadened the treatment options for aneurysms. Treatment strategies vary across different institutions and are usually not consistent with the Rundback classification, especially for aneurysms on the first bifurcation of the renal artery.9 –13
This study retrospectively analyzed 105 patients with RAAs treated at our center during a period of 14 years. Based on our experience with hilum aneurysms, 14 we proposed a new classification system for RAAs, which emphasizes the anatomic features of aneurysms, including the location and morphology of the aneurysm sac as well as the parent-branch vessel relationships. The classification showed better performance in guiding surgical choices compared with Rundback classification. Perioperative and long-term outcomes were analyzed to validate the safety and efficacy of all treatment techniques.
Methods
Patients
Patients diagnosed with RAAs who met the criteria for intervention and were admitted to the vascular surgery department between 2009 and 2023 were retrospectively selected. Renal artery aneurysms were diagnosed using computed tomography angiography (CTA) or magnetic resonance angiography (MRA) and defined as >1.5 times the diameter of the disease-free proximal adjacent arterial segment. Indications for intervention included aneurysm diameter greater than 2 cm, symptomatic RAAs (rupture, hematuria, refractory hypertension not controlled by ≥3 anti-hypertensive agents of different classes, and flank/abdominal/chest pain excluded of other reasons), or aneurysms in women of childbearing age. All participating subjects provided written informed consent, and the study was approved by the institutional review board (protocol number: S-K1965).
Demographic, comorbidity, and other clinical data were collected retrospectively from medical records (Table 1). Perioperative and long-term follow-up, including laboratory results, imaging data, and survival conditions were also recorded. Hypertension was defined as arterial systolic blood pressure (SBP) and diastolic blood pressure (DBP) levels of 3 or more clinical measurements above 140/90 mm Hg or taking anti-hypertensive drugs on admission. Blood pressure response was defined as cured, improved, or failed as previously described. 15 Glomerular filtration rate (GFR) was acquired by isotope renograms. Patients with chronic kidney disease were graded according to the National Kidney Foundation Kidney Disease Outcomes Quality Initiative classification. Estimated GFR (eGFR) was calculated with the Cockcroft-Gault equation, and impaired renal function was defined as a 30% reduction in eGFR.
Baseline Characteristics of Patients and Aneurysms (n=105).
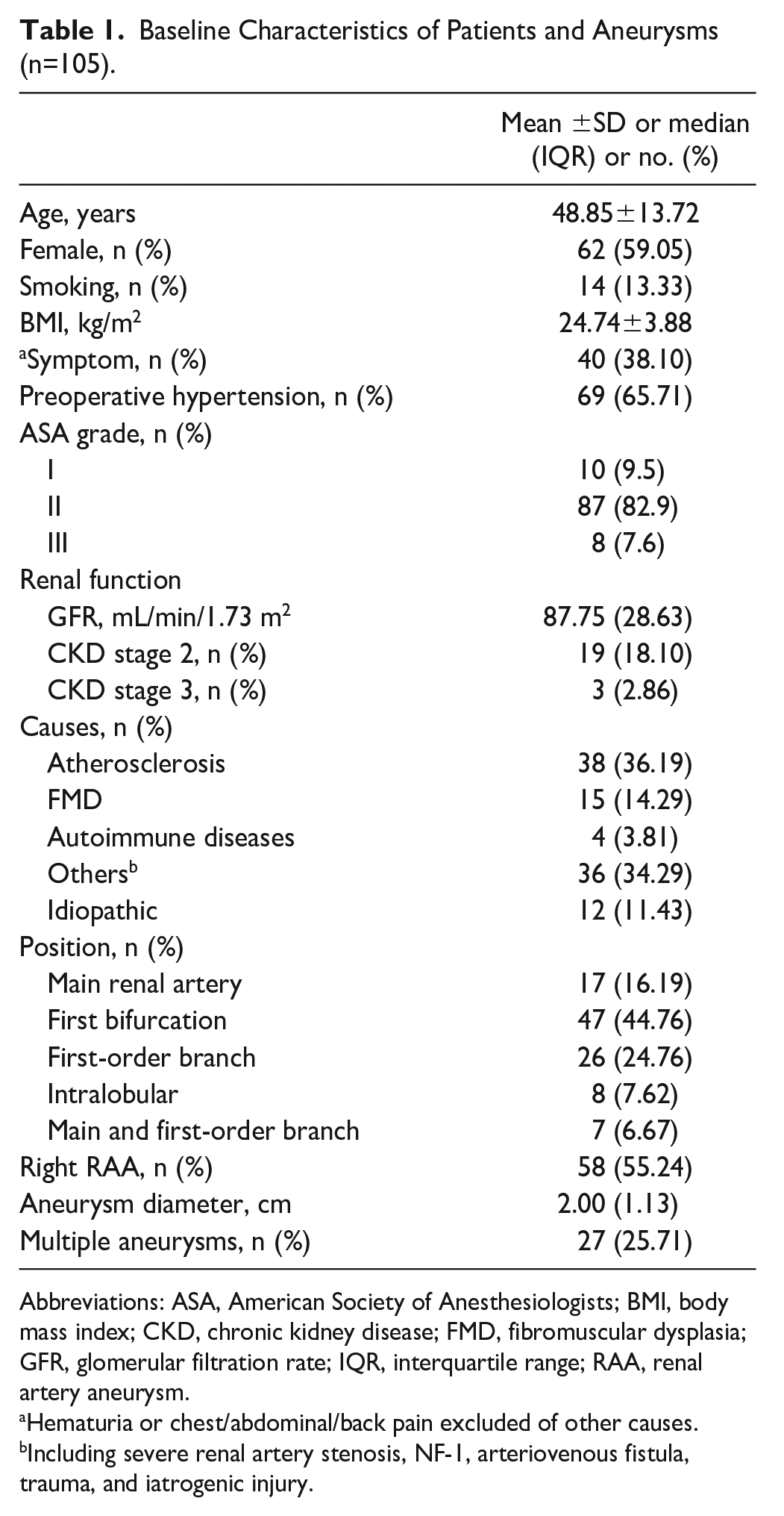
Abbreviations: ASA, American Society of Anesthesiologists; BMI, body mass index; CKD, chronic kidney disease; FMD, fibromuscular dysplasia; GFR, glomerular filtration rate; IQR, interquartile range; RAA, renal artery aneurysm.
Hematuria or chest/abdominal/back pain excluded of other causes.
Including severe renal artery stenosis, NF-1, arteriovenous fistula, trauma, and iatrogenic injury.
Classification System
The 3D model of each RAA was reconstructed from pre-operative imaging. Based on the location and morphology of the aneurysm, we proposed a new classification system as portrayed in Figure 1:
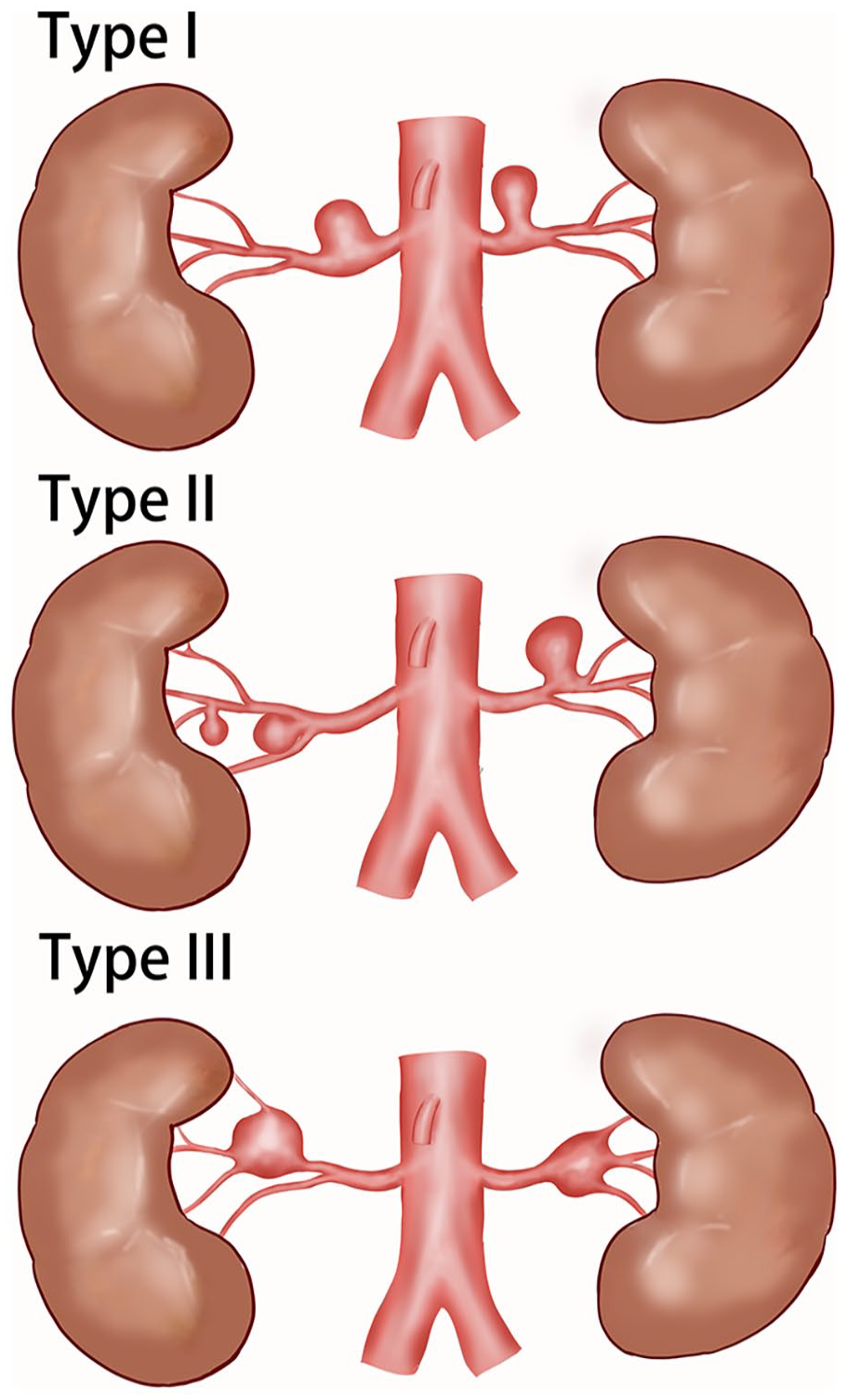
The Peking Union Medical College Hospital (PUMCH) classification. A schematic drawing of the new classification. Type I aneurysms arise from the main trunk, accessory branch, or first-order branches away from any bifurcation. Type II aneurysms arise from the first bifurcation with narrow necks (defined as dome-to-neck ratio >2) or from Intralobular branches. Type III aneurysms have wide-neck arise from the first bifurcation that affects 2 or more branches that cannot be sacrificed without significant infarction of the kidney.
Procedure
Endovascular interventions included primary coiling, stent-assisted coiling (SAC), and simple stent implantation. The main SAC techniques employed were “jailing” or “coil-stent” as reported earlier. 16 Open surgery consisted of aneurysmoplasty, resection and primary anastomosis, tailoring, and resection and graft reconstruction with either saphenous vein or prosthetic graft.2,5,13,17 For some complex lesions, ex vivo reconstruction with renal auto-transplantation under cold perfusion was performed. 15 Examples were illustrated in Figure 2, and detailed operation types were listed in Supplemental Table S1.
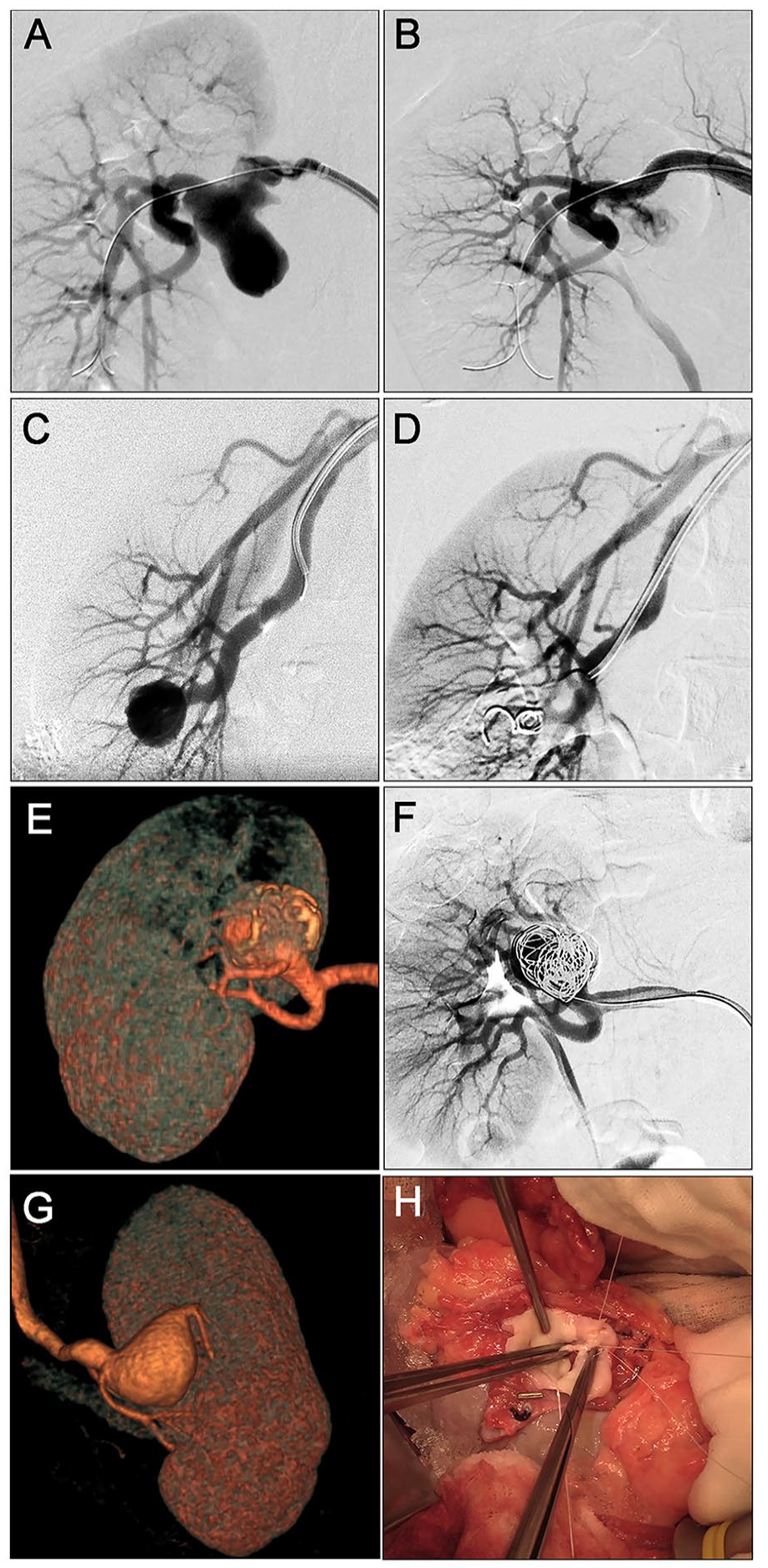
Examples of renal artery aneurysm (RAA) repair. (A-B) Simple stenting for type I aneurysm. (A) Preoperative angiography. (B) Angiography performed at the end of the procedure. (C-D) Coil embolization for type II aneurysm. (C) Preoperative angiography. (D) Angiography performed at the end of the procedure. (E-F) Coil embolization for type IIIa aneurysm. (E) Preoperative computer tomography angiography (CTA). (F) Angiography performed at the end of the procedure. (G-H) Aneurysm resection and saphenous vein graft autogenous reconstruction for type IIIb aneurysm. (G) Preoperative CTA. (H) Suturing of the renal artery during aneurysm resection.
Follow-up
Technical primary success was defined as the successful introduction and deployment of the device without surgical conversion or any complication. Hypertension relief was defined as SBP/DBP below 140/90 mm Hg at 3 or more clinical measurements without the reduced number of anti-hypertensive drugs. Transient postoperative complications included back/abdominal pain, fever, and discomfort or nausea before discharge. Color Doppler, CTA, or angiography was applied to evaluate arterial patency, which was defined as blood flow within the target renal artery or bypass and perfusion in the respective renal parenchyma. Laboratory tests, including creatinine (µmol/L), white blood cell count (WBC, 109/L), hemoglobin (HGB, g/L), platelet (PLT, 109/L), and
Statistical Analysis
Statistical analysis was performed using SPSS 26.0 software (IBM Corp., Armonk, NY). Continuous variables were analyzed for normality and summarized with median and interquartile range (IQR) or mean ± standard deviation. Wilcoxon, Kruskal-Wallis, and Friedman tests were employed for cross-group or within-group comparisons. Categorical variables were presented with frequencies and compared using the χ 2 or Fisher’s exact test as appropriate. A p-value of <0.05 was considered significant.
Results
Patient Demographics
As shown in Table 1, the mean age of 105 patients was 48.85±13.72 years, and 62 (59.05%) were female. Symptomatic patients constituted 38.10% and hypertension was present in 65.71% of all subjects. The majority of the patients were American Society of Anesthesiologists (ASA) grade II (87, 82.9%). The median GFR was 87.75 mL/min/1.73m2, and 22 patients had stage 2 or 3 chronic kidney disease. Considering the etiologies, atherosclerosis (38, 36.19%), fibromuscular dysplasia (FMD; 15, 14.29%), and autoimmune diseases (4, 3.81%) were the most recognizable causes.
Aneurysm Anatomy
Although 27 (25.71%) patients had ipsilateral or bilateral multiple aneurysms, only one side of the renal arteries was repaired for each patient. Most of the aneurysms were located at the first bifurcation (47, 44.76%), followed by 26 (24.76%) at the first-order branch, 17 (16.19%) at the main renal artery, and 8 (7.62%) at intralobular branches. All 7 (6.67%) patients with 1-side multiple aneurysms had them located at the main renal artery, first-order branch, or both. In addition, 58 (55.24%) were right side and the median diameter was 2.00 (IQR=1.13) cm.
Comparison of Treatment Options Between the Rundback and New Classification
Among 105 patients, 102 agreed to undergo surgical operation. Table 2 showed the results of kappa value between the Rundback or new classification planning and RAA repair. Only 58.97% (23/39) of Rundback type II fusiform aneurysms received open surgery as proposed, and the kappa level was 0.579 for the classification. Regarding the new classification, 91.36% (74/81) of type I and II aneurysms underwent endovascular repair. And 90.48% (19/21) received open surgery for type III. The kappa level for the 3 types achieved 0.752.
Results of Kappa Value Between Classification Planning and Renal Artery Aneurysm (RAA) Repair.
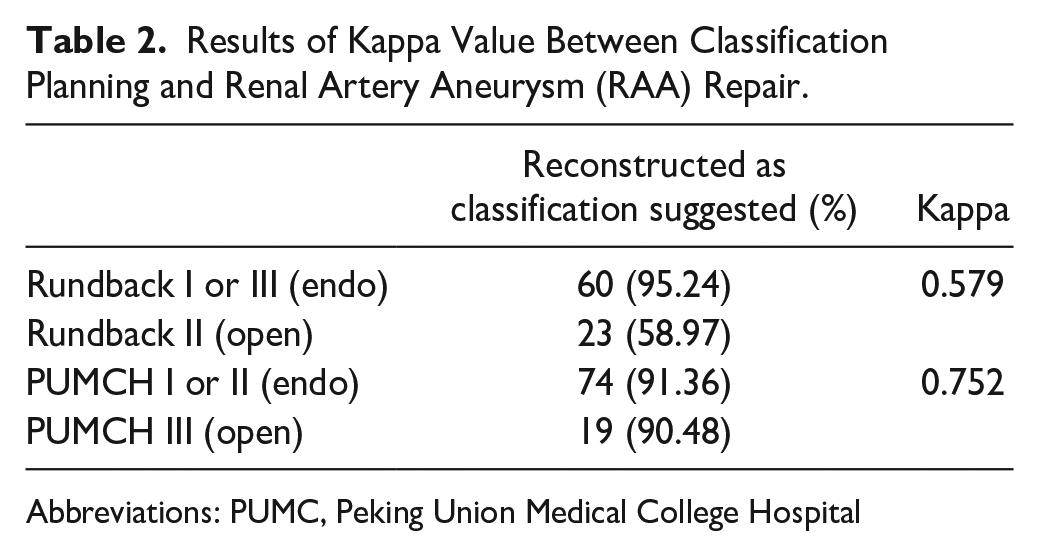
Abbreviations: PUMC, Peking Union Medical College Hospital
Short-Term and Long-Term Results
Table 3 summarized the 30-day and long-term results of 102 patients receiving operation comparing open and endovascular repair. Technical primary success was achieved in all 26 patients undergoing open surgery and 96.05% (73/76) of those receiving endovascular repair. Two underwent angiography and gave up operation due to the high risk of renal loss. One patient had intraoperative renal artery spasm recurrently and the guidewires could not be introduced. Symptoms were completely relieved in 100% (5/5) and 84.85% (28/33), and hypertension relief was observed in 84.21% (16/19) and 72.92% (35/48) of patients in each group, respectively. Significant differences in transient postoperative complication, blood transfusion, and hospital stay were found between the 2 groups. Only 1 patient receiving primary coil embolization had impaired renal function (not shown in table).
Thirty-Day and Long-Term Results After Reconstruction of Renal Artery Aneurysms (RAAs) in 102 Patients.
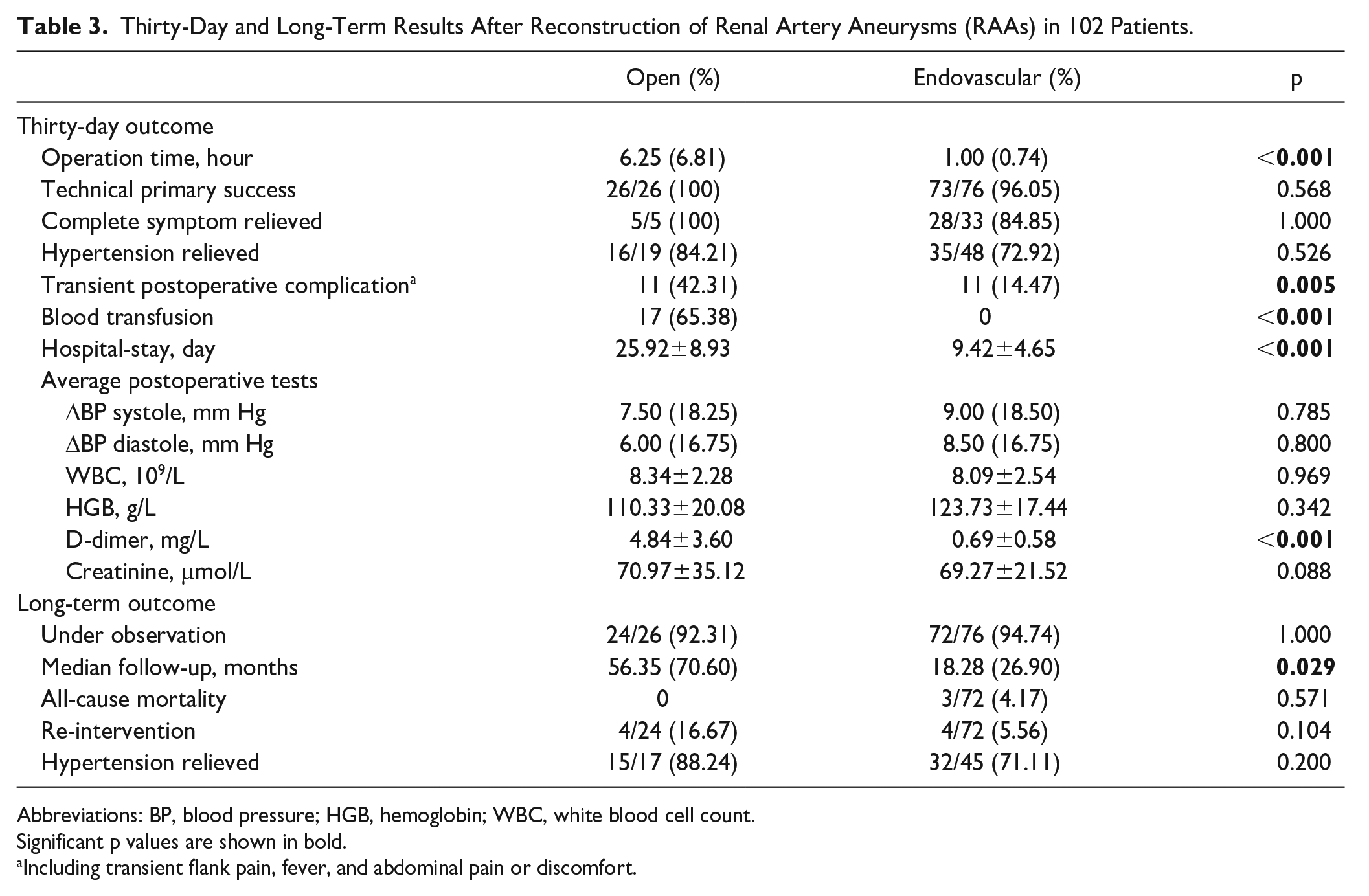
Abbreviations: BP, blood pressure; HGB, hemoglobin; WBC, white blood cell count.
Significant p values are shown in bold.
Including transient flank pain, fever, and abdominal pain or discomfort.
In total, 24 of 26 (92.31%) and 72 of 76 (94.74%) patients in each group received at least 1 long-term examination, with a median follow-up of 56.35 and 18.28 months. The median follow-up for the whole group is 22.88 (IQR=49.93) months. Figure 3 showed the Kaplan-Meier curves for survival, intervention, and patency. No significant difference was observed for long-term outcomes. Three patients died of non-RAA-related reasons (pancreatic cancer, aortic dissection, and stroke). Four and 3 patients in each group had occluded renal arteries or stent/graft upon imaging examination, of which only 1 underwent subsequential nephrectomy. Re-intervention happened in 4 of 24 (16.67%) and 4 of 72 (5.56%) for each group. One patient had undergone tailoring technique and received coiling 9 months later due to an unsatisfactory resolve of an aneurysm. Ex vivo repairs and auto-transplantation had been carried out in the other 3 patients, of which 2 received balloon angioplasty and stent implantation for anastomotic stenosis, whereas 1 accepted nephrectomy due to renal failure. Three of the 4 patients receiving endovascular repair underwent coiling for a second time. One patient received balloon dilatation due to in-stent stenosis. Regarding patients with hypertension, 88.0% and 71.11% in each group had relief at long-term follow-up. In addition, there was no significant difference among the 3 Peking Union Medical College Hospital (PUMCH) classification types for perioperative or long-term results (Table 4).
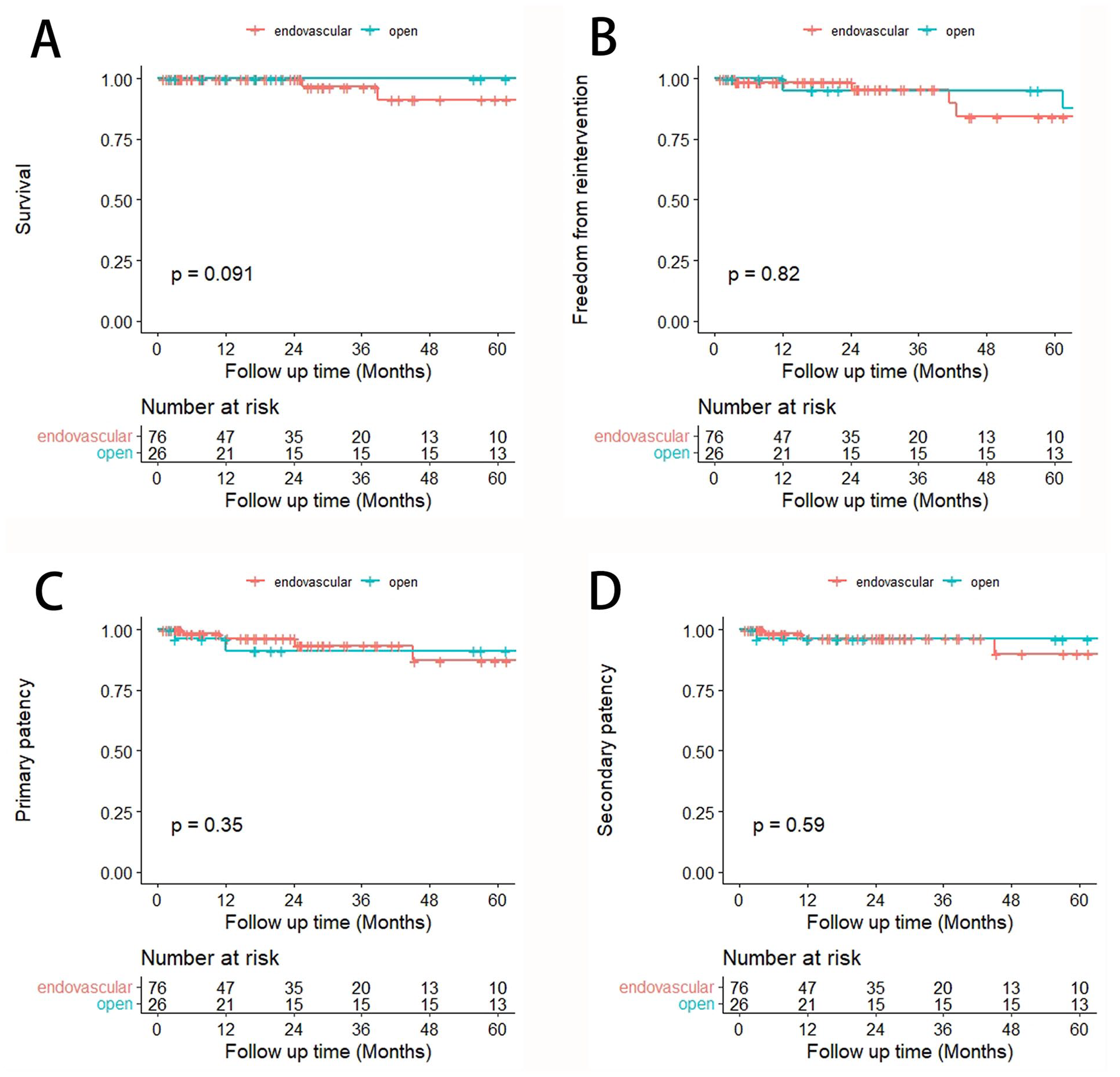
The Kaplan-Meier curves for survival, intervention, and patency between open and endovascular repair. Comparison between patients receiving open and endovascular repair was shown with Kaplan-Meier curves for (A) survival, (B) freedom from re-intervention, (C) primary patency, and (D) secondary patency.
Thirty-Day and Long-Term Results After Reconstruction of Different Renal Artery Aneurysm (RAA) Subtypes.
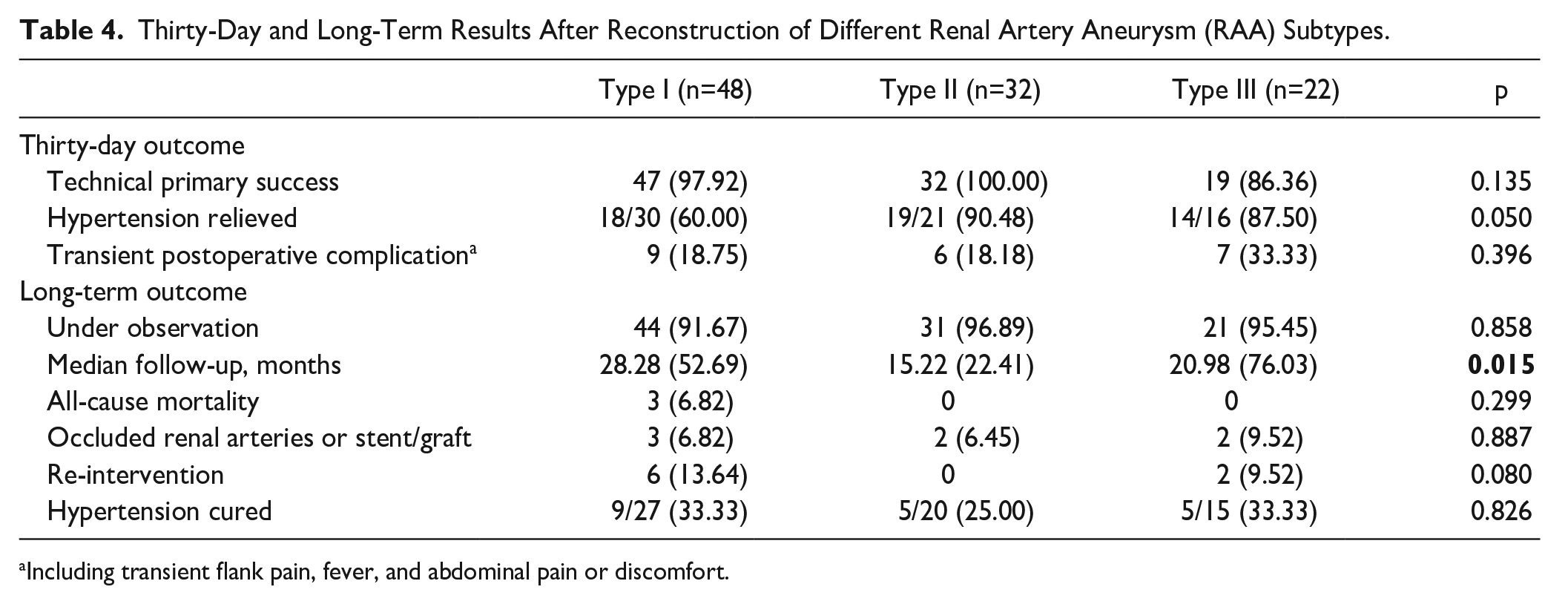
Including transient flank pain, fever, and abdominal pain or discomfort.
For laboratory results, as listed in Table S2, a significant drop in HGB, rise of WBC, and neutrophils (NEUT) were found immediately after the operation, yet short-term and long-term re-examination of all parameters changed to normal values. Creatinine level remained normal both immediately after the operation (68.00±11.74 μmol/L) and at follow-up examination (74.88±15.80 μmol/L within 1 year and 71.88±14.82 μmol/L after 1 year).
Discussion
Since it was first reported, 18 identification and intervention of RAA have gained increasing attention due to the development of both imaging and vascular surgery techniques. Symptoms relief, prevention of rupture, as well as preservation of renal function are crucial targets of surgery. A large part of patients at our center were referred from the outpatient clinic, where symptomatic RAA was usually the main complaint. Hence, surgical intervention was more radically conducted regarding aneurysm size. Besides atherosclerosis and FMD, vessel wall degeneration induced by autoimmune disorders or rare genetic diseases, such as neurofibromatosis type 1 (NF1) could also result in RAA. Pseudoaneurysms from trauma or iatrogenic injury were occasionally encountered.
Recently, endovascular approaches have become the predominant choice for RAA in several institutions,19 –21 yet open surgery is still performed for complex lesions.13,22,23 High-volume studies and meta-analyses found little difference in short-term or long-term mortality and major morbidity between the 2 choices.9,24,25 Most RAA patients are young and have few comorbidities, making them well-tolerated under general anesthesia. Since the kidneys are essential organs with little collateral circulation, the choice of treatments is based more on the anatomic features of RAAs as well as branch protection and renal function preservation.
Previous literature has summarized some experience in selecting treatment strategies for RAA. Coil embolization, as a minimally invasive endovascular procedure with good effectiveness and safety,21,26 could be easily performed in narrow-necked saccular aneurysms and assisted stent broadened its use in wide-necked side-wall aneurysms. 8 Aneurysms affecting distal secondary or tertiary order branches could be repaired with hypothermic ex vivo approaches. 13 For those who are unsuitable for endovascular therapy or at young ages, aneurysm resection and reconstruction with autologous or prosthetic graft are conducted. Rundback et al provided a rough guide for management, but the location of the aneurysm was not emphasized in this classification, and the specific definition of aneurysm morphology (fusiform/saccular) has not been precisely clarified. 7 As calculated by Henke P. K. and colleagues and is consistent with our data, 5 the majority of RAA resides on the first bifurcation of the renal artery, which could involve more than one segmental branch and influence blood supply.
In the past 14 years, our center has gained experience in treating RAAs and classifying rare diseases.14,27 –30 The new classification considers both aneurysm location and morphology in surgical planning. Regardless of their shape, type I aneurysms could mostly be managed with advanced endovascular techniques regardless of their shape. For type II RAAs, saccular aneurysms on the first bifurcation are also treated with endovascular approaches, especially coil embolization. The definition of saccular could be found in literature concerning intracranial aneurysm, and a dome-to-neck ratio >2 has been generally accepted. 31 For aneurysms where only intralobular branches were affected, an endovascular approach was preferred if sacrificing the branch led to acceptable renal function loss. Generally, branches supplying <50% of renal mesenchyme on CTA were considered worth trying endovascular therapy. For type III RAAs, the percentage of renal perfusion of affected segmental branches should be assessed. Open surgery was the first consideration for aneurysms where most or all first-order branches originated from the aneurysm. In most clinical scenarios, only 2 or 3 branches join at the first bifurcation making it easy to recognize the blood supply range of these branches following their distribution into the kidney. Importantly, the patients’ opinions should be respected under these circumstances.
Using this classification for quick surgical decisions helps patients avoid radiation exposure from multiple angiographies. In addition, timely selection of antiplatelet or antithrombotic treatment for patients planned for either endovascular or open repair reduces postoperative complications. The kappa analysis demonstrated that compared with the Rundback classification, this classification scheme had good accuracy. Among the 7 patients for type I or II choosing open surgery instead, 1 patient was an 11-year-old girl and ex vivo repair was selected to ensure durable patency,32,33 whereas 5 received operations received operation more than 5 years ago. One patient has combined renal artery stenosis and aneurysms on both the main and first-branch artery, and open surgery was selected for better exposure. The 2 patients for type III who did not receive open repair underwent angiography and gave up endovascular repair due to high occlusion risk.
Several special types of aneurysms should be noted. Two saccular pseudoaneurysms, as well as 1 saccular aneurysm combined with arteriovenous fistula included in this study, were all embolized following previous research. 19 Multiple aneurysms were separately viewed using our classification system, and treatment strategies took all of them into account. Interestingly, all multiple aneurysms encountered by us were on the main or first-order branch, hence, coil embolization was routinely performed. However, open surgery should still be considered if more complicated branches were involved, as it has proven reliability.3,5 No ruptured or hemodynamically unstable RAA was present in our study.
Since the new classification mainly dictated the choice of open or endovascular operation, we retrospectively analyzed and compared their results at our center. Overall, technical primary success was high for both techniques with explicit relief of symptoms and hypertension. Consistent with previous findings,25,34 endovascular repair had the advantage of fewer postoperative complications, little need for blood transfusion, and less hospital stay. Back/abdominal pain arose from renal microinfarction, and fever may be due to thrombus or necrosis absorption or infection. Although patients in the open surgery group had higher levels of
This study is limited by its retrospective design. Our RAA management mainly involves stenting or coil embolization for endovascular repair and aneurysm resection and saphenous vein graft for open surgery. Other institutions have proven the safety and efficacy of alternative techniques. A prospective study would be necessary to further validate and refine the classification in the future and to explore its role in risk stratification. More evidence is needed to set the optimal diameter threshold for RAA. Experience with rare aneurysm types also requires accumulation.
Conclusions
Surgical approaches for RAA should be tailored individually to the location and morphology of each aneurysm. We have proposed a convenient and accurate classification system based on CTA images that facilitate the choice of intervention between endovascular or open techniques, revealing value in guiding treatment selection with good short-term and long-term outcomes.
Supplemental Material
sj-docx-1-jet-10.1177_15266028241229014 – Supplemental material for An Anatomic Classification Scheme for Surgical Planning of Renal Artery Aneurysms
Supplemental material, sj-docx-1-jet-10.1177_15266028241229014 for An Anatomic Classification Scheme for Surgical Planning of Renal Artery Aneurysms by Fangda Li, Siting Li, Zenghan Cao, Rong Zeng, Xiaolong Liu, Changwei Liu, Bao Liu, Yuexin Chen, Wei Ye, Lei Wang, Leng Ni and Yuehong Zheng in Journal of Endovascular Therapy
Supplemental Material
sj-docx-2-jet-10.1177_15266028241229014 – Supplemental material for An Anatomic Classification Scheme for Surgical Planning of Renal Artery Aneurysms
Supplemental material, sj-docx-2-jet-10.1177_15266028241229014 for An Anatomic Classification Scheme for Surgical Planning of Renal Artery Aneurysms by Fangda Li, Siting Li, Zenghan Cao, Rong Zeng, Xiaolong Liu, Changwei Liu, Bao Liu, Yuexin Chen, Wei Ye, Lei Wang, Leng Ni and Yuehong Zheng in Journal of Endovascular Therapy
Footnotes
Acknowledgements
The authors thanked Dr Zhili Liu, Jiang Shao, Wei Ye, and Xiaojun Song for their expertise in clinical assessment and operation performance. They appreciated the help from Dr Yi Wang, Jiong Zhou, Xue Bai, and Yonghong Tian for data collection.
Author Contributions
F.L. contributes to the study design. S.L. contributes to the data analysis. F.L., S.L., Y.Z., and L.W. contribute to the manuscript writing and revision. F.L., S.L., and Z.C. contribute to electronic image analysis. Z.C. contributes to the graph drawing. R.Z. and Y.Z. contribute to guidance of classification system establishment. R.Z., X.L., C.L., B.L., Y.C., W.Y., L.N., and Y.Z. contribute to performing operation and clinical data collection. The manuscript has been read and approved by all the authors, the requirements for authorship have been met, and each author believes that the manuscript represents honest work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Natural Science Foundation of China (grant numbers: 51890894, 82070492, and 82100519), the CAMS Innovation fund for Medical Science (CIFMS, grant no. 2021-I2M-C&T-A-006 and 2021-I2M-1-016), and the National High-Level Hospital Clinical Research Funding (grant no. 2022-PUMCH-B-100 and 2022-PUMCH-C-062). The funders had no role in study design, data collection, analysis, decision to publish, or preparation of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
