Abstract
Purpose:
To describe the semibranch as new tool to treat patients with thoracoabdominal, para- and juxtarenal aortic pathologies.
Technique:
The technique is demonstrated in 2 patients with aortic pathologies. First, a 76-year-old woman with a type Ia endoleak after endovascular repair of abdominal aneurysm (EVAR). Due to the short mainbody of the EVAR and suprarenal extension of the pathology, a 4-branched device with integrated bifurcation was chosen. To realize a branched device in small diameters of the suprarenal aorta, 2 semibranches, for the superior mesenteric artery (SMA) and the celiac trunc (CT), were planned. The second patient, a 75-year-old man, had a juxtarenal aneurysm. Due to small diameters of the aorta, semibranches for the SMA and the CT were planned. In both cases, a total transfemoral approach was used with a steerable sheath. Placement, cannulation of the semibranch, forwarding of the bridging stentgraft, and sealing was uneventful. All 4 semibranches were successful in sealing the aneurysm.
Conclusion:
The semibranch is another tool in the armamentarium of the endovascular interventionalist, which can expand the range of patients that can be treated. Especially patients with small aortic diameter or short distances between target vessels with the same offspring angle can profit from the semibranch design.
Clinical Impact
The semibranch is a new tool to treat thoracoabdominal as well para- and juxtarenal aortic pathologies. Due to its reduced length, packaging density can be higher and diameter of the stentgraft can be larger. It closes the gap in pathologies where fenestration are not suitable (kinked aortas) and there is not enough space for standard inner branches (small diameters).
Keywords
Introduction
An endovascular approach for patients with thoracoabdominal, para- and juxtarenal aortic pathologies becomes more and more the gold standard, especially for patients unfit for open surgery or prior aortic open repair. At the moment, we have 2 kinds of technologies: fenestrations and branches. Both of them have their advantages and disadvantages. Fenestrations are mostly used in small aortic diameters, as they do not need space to expand. One major disadvantage is that planning and placement must be very accurate. Diameter-reducing technologies facilitate cannulation of the target vessels, but especially in kinked anatomies this can be challenging.
Branches have the advantage of longer sealing zones for the bridging stentgraft, but need space to expand. If outer branches are used, the necessary thrombus free diameter is usually about >28mm. This also depends on the diameter of the main stentgraft. If reduced to 18 mm to achieve outer branches the flow might be reduced, especially if extended with a bifurcated 20mm/22mm device. Inner branches can further reduce the minimal aortic diameter to 22 mm. The reduction of needed aortic diameter has the disadvantage of reduced available space in the device due to flow reduction from up to 4 inner branches. So normally devices with inner branches must be sized larger than those with outer branches. Another pitfall can be, if 2 target vessels are very close and have the same offspring vessel. In this situation, 1 inner branch can compromise the other. Under these conditions, at least one of the inner branches must be changed in orientation and/or height or a twin branch 1 must be used.
The semibranch was designed to overcome these problems. It consists of a shorter inner branch with extended ostium. The smaller amount of material also allows reducing the packaging size of the device.
Technique
The technique is demonstrated in a 76-year-old woman and a 75-year-old man. The first patient had a type Ia endoleak after endovascular repair of abdominal aneurysm (EVAR) with an Anaconda device (Vascutek, Terumo, Inchinnan, UK), due to progression of the disease up to the suprarenal level. The main problem was the short mainbody, with irregular diameters. Sealing with a distal tube configuration was not possible, so extension with a bifurcated graft was necessary. Also using an inverted limb would result in covering at least on renal artery and compromising the second one, if using fenestrations. We decide to use an Artivion (Hechingen, Germany) custom-made device (CMD) with inner branches for the renal arteries and offset them 1 cm higher. As the pathology ended at the level of the highest renal artery, the diameter of the aorta was considerably smaller. Using fenestrations for the superior mesenteric artery (SMA) and the celiac trunk (CT), placement and deployment of the device could be challenging. So, we decided to use a semibranch for the SMA and the CT. The semibranch (Figure 1) consists of a shorter inner branch with a special sinusoid spring and a large ostium to facilitate cannulation also in case of misdeployment (Figure 2).
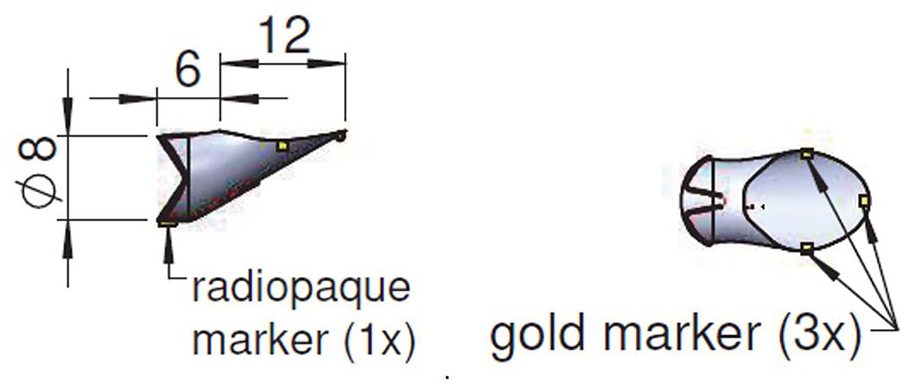
Semibranch design. The inner branch consists of a shorter inner branch with sinusoid spring, available in 6 and 8 mm diameter. The length of the branch is 6mm and of the ostium 12mm. The proximal ring marker is on the top of the sinusoid spring and the 3 ton markers are the same as in the standard inner branch.
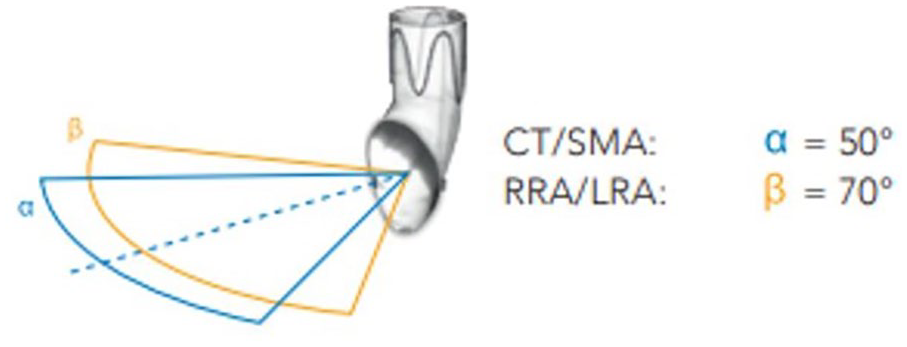
The larger ostium enhances the range of cannulation also in case of misdeployment. CT/SMA, celiac trunk\superior mesenteric artery; RRA, right renal artery; LRA, left renal artery.
The technical drawing of the CMD is shown in Figure 3. Due to a relevant kink in the descending thoracic aorta the proximal part was shortened and a double proximal spring was used.
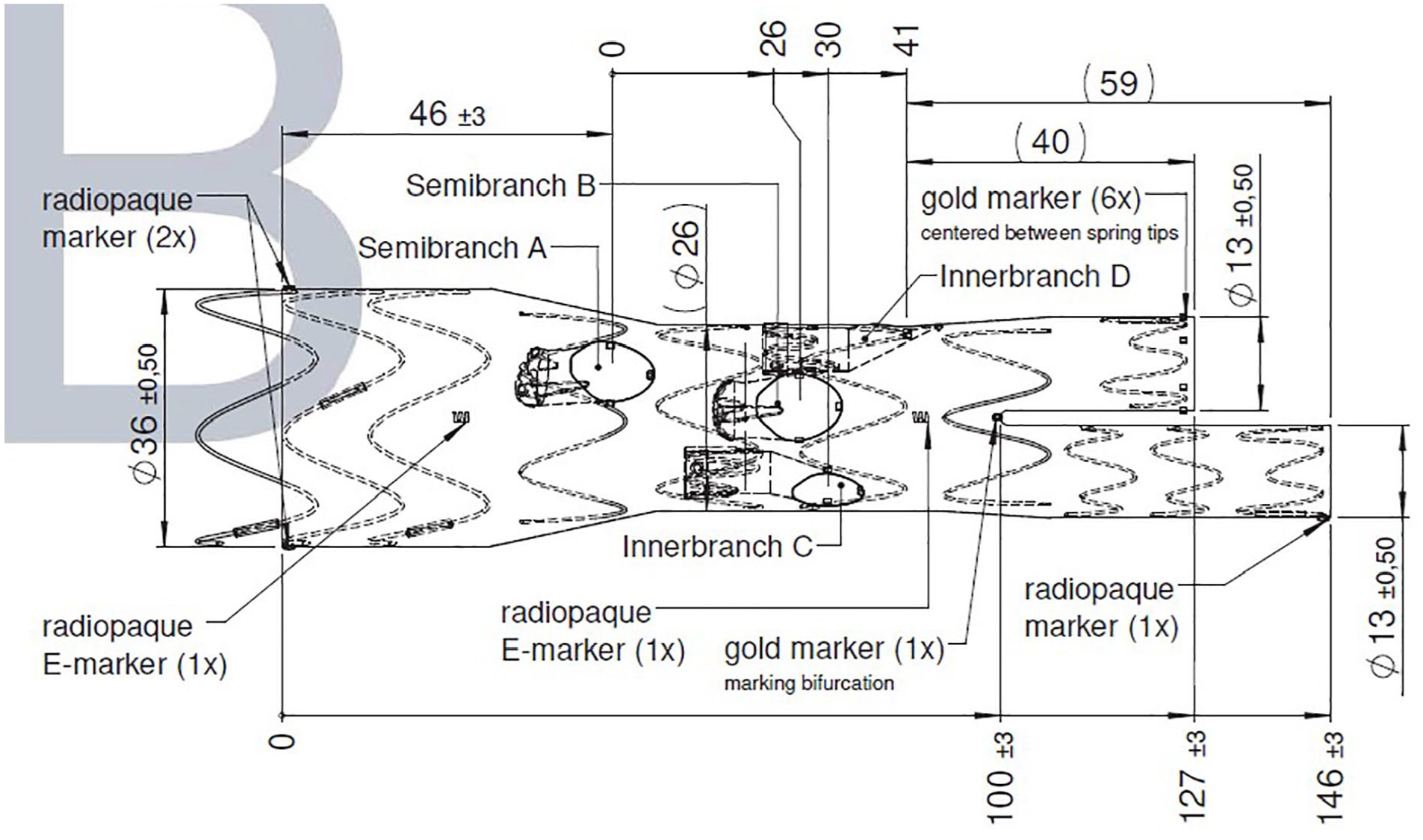
Technical drawing.
Our standard approach for branch cannulation is transfemoral, using an 8.5F Destino Reach steerable sheath (Oscor, Palm Harbor, FL, USA). As bridging stentgrafts 6mm Viabahn VBX (Gore W. L. Gore & Associates, Newark, DE, USA) over a Rosen wire (COOK Medical, Bloomington, IN, USA) were used and flared inside the semibranch with an 8 × 20mm balloon (Admiral Extreme, Medtronic, Minneapolis, MN, USA). Control angiography showed no endoleak and good flow (Figures 4A–F).
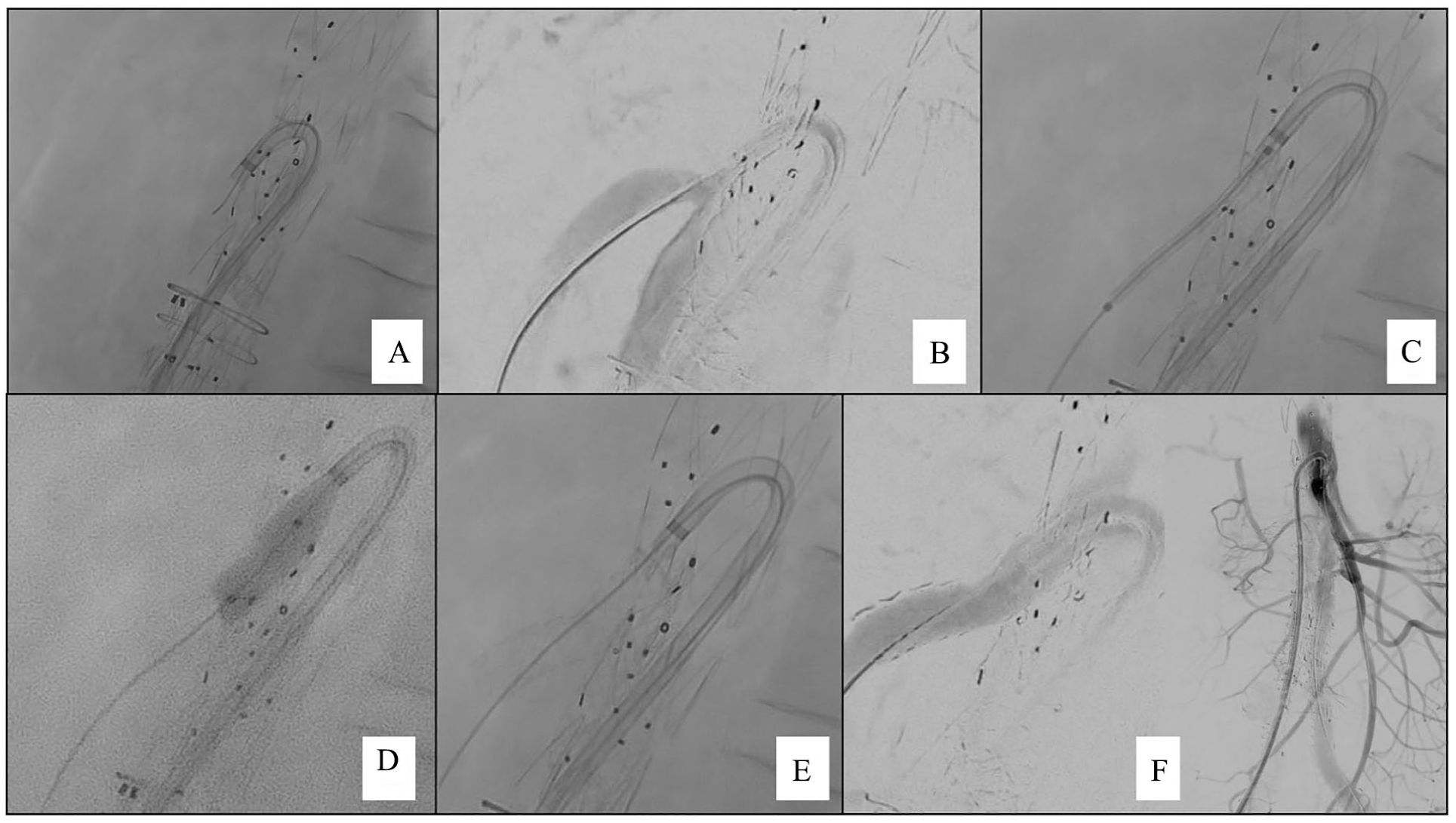
Intraoperative imaging. (A) Cannulation of the SMA semibranch via a transfemoral approach using a steerable sheath. (B) Angiography, showing the exact placement of the semibranch slightly above the target vessel. (C) Placement of the bridging stentgraft. (D) Flaring of the bridging stentgraft inside the semibranch and outside of the target vessel. (E) Fluoroscopy of the final result. (F) Final angiography showing a good perfusion of the SMA without endoleak. SMA, superior mesenteric artery.
The 75-year-old man had a juxtarenal aneurysm with narrow (24mm) aortic diameter in the suprarenal part. A standard inner branch device would be feasible, but with reduced diameter, alternatively a fenestrated graft would be necessary. Due to the experience with the first case, we planned an Artivion (Hechingen, Germany) CMD with inner branches for the renal arteries and semibranches for the SMA and CT.
Approach and technique was the same as described above. In this case, cannulation and bridging of the semibranches was uneventful as well (Figure 5). In Figure 6, the SMA semibranch is depicted with the flared bridging stentgraft inside the semibranch achieving seal in the sinousoid spring to the beginning of the ostium.
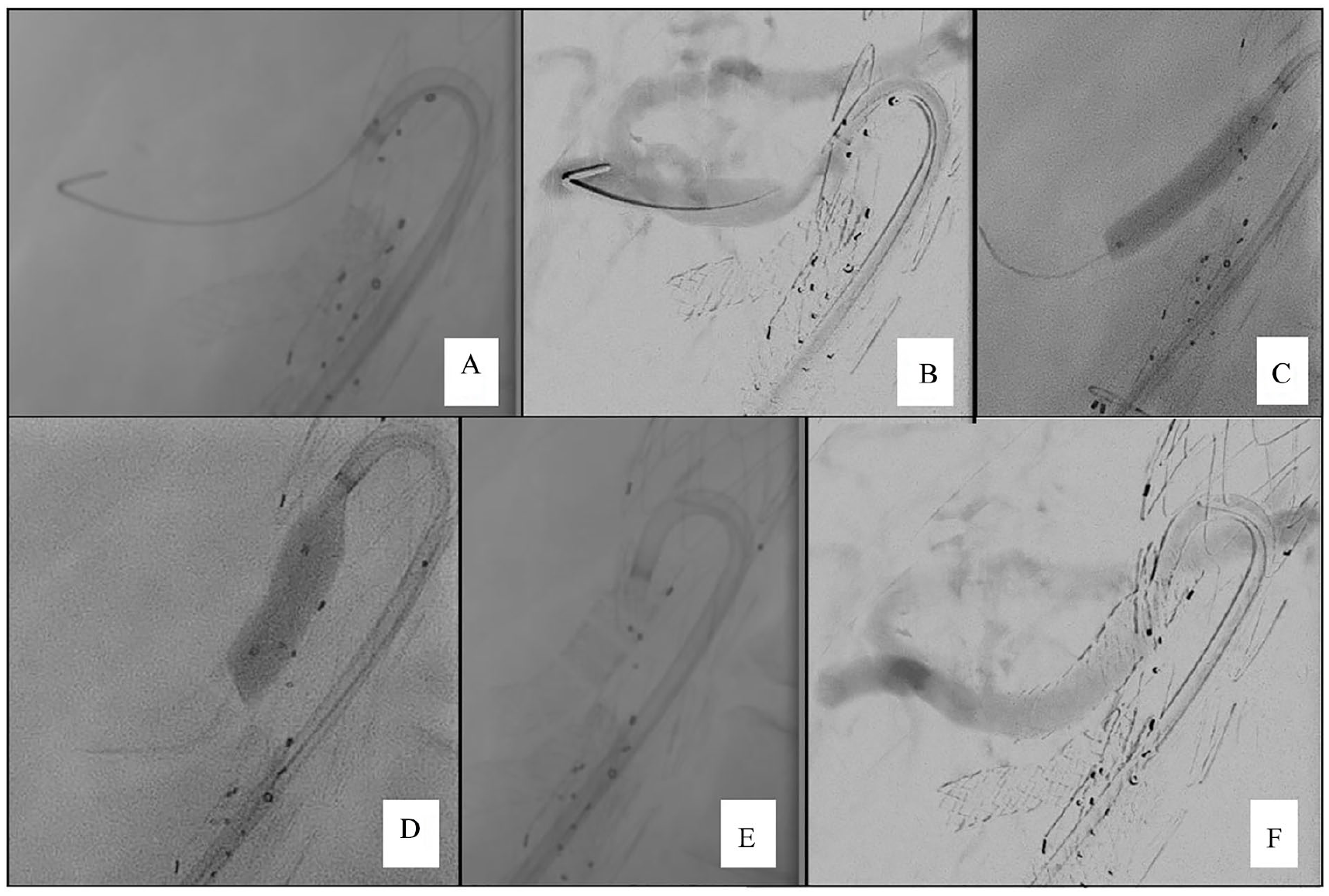
Intraoperative imaging. (A) Cannulation of the CT semibranch via a transfemoral approach using a steerable sheath. (B) Angiography, showing the exact placement of the semibranch slightly above the target vessel. (C) Deployment of the bridging stentgraft. (D) Flaring of the bridging stentgraft inside the semibranch and outside of the target vessel. (E) Fluoroscopy of the final result. (F) Final angiography showing a good perfusion of the SMA without endoleak. CT, celiac trunk; SMA, superior mesenteric artery.
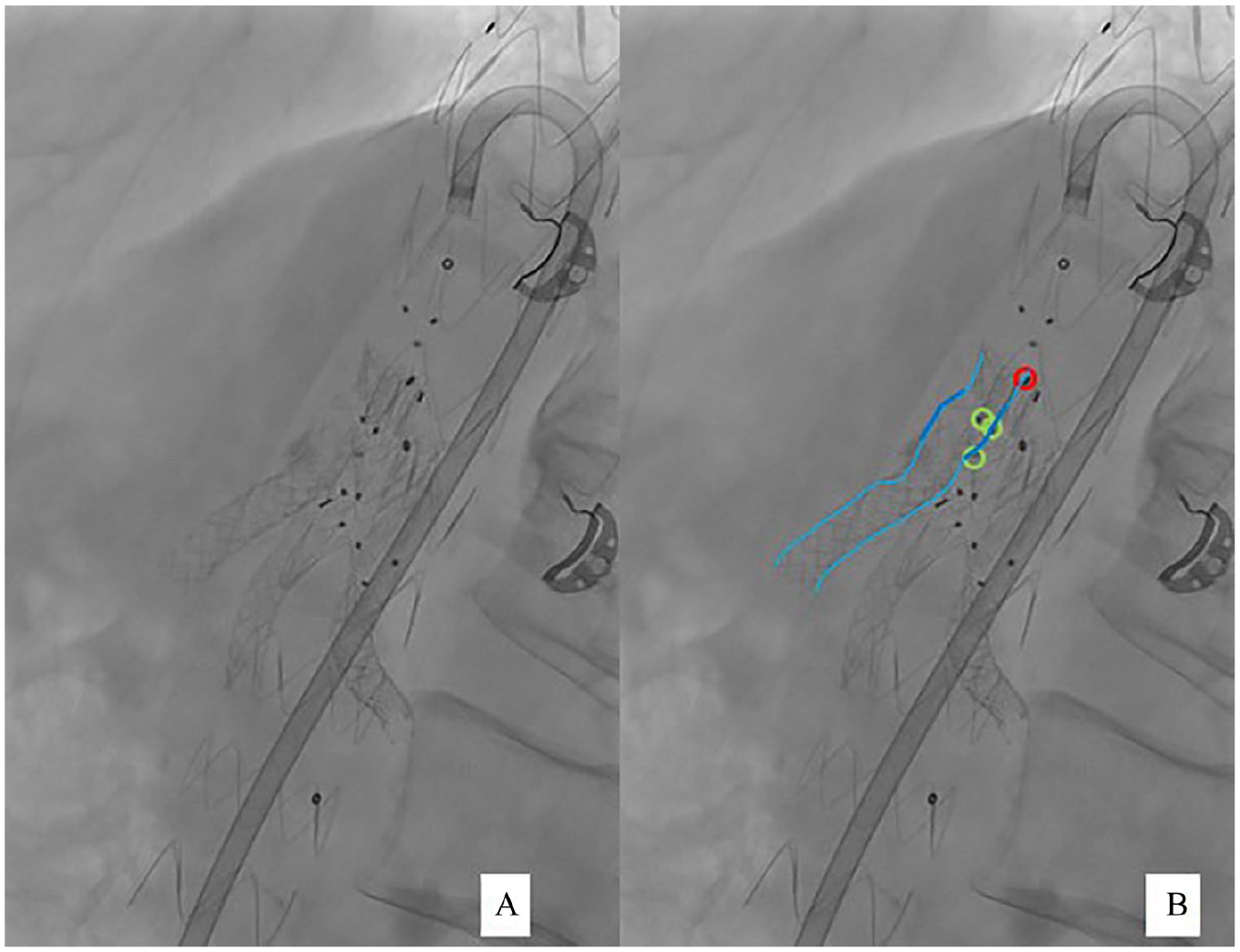
(A) Original version. (B) Marked version. Red circle: proximal ring marker of the semibranch; green circles: distal ton markers of the semibranch; light blue line: contours of the bridging stentgraft; dark blue line: flared part of the bridging stentgraft.
Discussion
Complex aortic interventions with fenestrated and branched devices are increasingly becoming the standard therapy for thoracoabdominal, para- and juxtarenal aortic pathologies especially in older and unfit patients. More and more companies are getting in the market with dedicated devices and for thoracoabdominal aneurysms there are at least 4 off-the-shelf devices in different markets. In branched stentgrafts, we have outer and inner branches, so the therapist can decide what technology fits best patient’s anatomy. In Crawford type I aneurysms, dissections with small true lumen or in juxta-/pararenal aneurysms the application of branched technologies is limited.
Fenestrations are used best, if the target vessel has a 90° take off, whereas for directional branches a steeper angle is recommended. 2 The semibranch can have an advantage due to reduced fixation length at the inner side of the device, so that also a flatter angle can be managed.
In a recent study, 2 fenestrations showed better results than branches in complex endovascular aortic procedures. In this study, 1004 fenestrations with 100 branches were compared, but only 11 of them were inner branches. The authors state, that a higher stress at the transition zone from the stentgraft to the target vessel due to the downward facing branches may be the reason for lower patency rates. Total diameter of the graft and total diameter of the aorta were not stated in this trial, as well as the percentage of failure of the 11 (of course only 1% of all targeted renal arteries) inner branches. Maybe an inner branch or semibranch, that can help to improve device diameter and due to its design a flattener angle is possible, can overcome this problem, although this is not proven yet. Fenestrations require direct contact with the aortic wall. A gap >5mm is an independent risk factor for target vessel instability. 3 This problem is less significant in branched devices.
A wide range of indications of the semibranch is possible. Due to the shorter design than a standard inner branch, packaging of the device is smaller; as a result, the diameter of the stentgraft can be wider. The diameter of the stentgraft in small aortic diameters is an issue. Wang et al 4 and highlighted by Tenorio and Oderich 5 described their outcomes after postdissection aneurysms and the importance of a wider diameter of the grafts. Other indications can be redo-interventions after failed branched or fenestrated stentgrafts. 6
Conclusion
The semibranch design is an additional tool for treating patients with thoracoabdominal, para- and juxtarenal aortic pathologies. Longer follow-up and larger cohorts are needed to evaluate the technique.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The author received travel and research grants from Artivion, and is a member of the advisory board.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
