Abstract
Purpose:
Angio-Seal (Terumo Medical Corporations, Somerset, New Jersey) device is indicated for femoral arteriotomy closure. Real-world published data on complications are limited. We present 1 year of safety events involving Angio-Seal from the US Food and Drug Administration’s post-market surveillance database of Manufacturer and User Facility Device Experience (MAUDE). Steps for managing frequent device-related problems are discussed.
Materials and Methods:
Angio-Seal MAUDE data from November 2019 to December 2020 was classified according to (1) mode of device failure, (2) complication, (3) treatment, and (4) Cardiovascular and Interventional Radiological Society of Europe (CIRSE) adverse event classification system.
Results:
There were 715 safety events, involving Angio-Seal VIP (93.1%), Evolution (5.7%), STS Plus (1.1%), and sizes 6F (62.5%) and 8F (37.5%). Failure mode involved unrecognized use of a damaged device (43.4%), failed deployment (20.1%), failed arterial advancement (6.3%), detachment of device component (4.9%), failed retraction (3.6%), operator error (1.1%), and indeterminate (20.6%). Of total, 44.8% of events were associated with patient harm. Complications involved minor blood loss (34.1%), hematoma (5.6%), significant blood loss (1.4%), and pseudoaneurysm (1.4%). Of total, 43.3% of cases required manual compression (MC), whereas 8.8% required more advanced intervention. Interventions included surgical repair (49.2%), thrombin injection (9.5%), balloon tamponade (6.3%), covered stent (4.8%), and unspecified (30.2%). Majority of safety events were CIRSE grade 1 (92.0%), followed by grades 2 (3.1%), 3 (4.6%), and 6 (deaths, 0.3%). Minority of devices were returned for manufacturer analysis (27.8%).
Conclusions:
The majority of safety events were associated with minor blood loss or local hematoma and could be addressed with MC alone. Most events were attributed to damaged device; however, very few devices were returned to manufacturer for analysis. This should be encouraged to allow for root cause analysis in order to improve safety profile of devices. System-level strategies for addressing barriers to under-reporting of safety events may also be considered.
Clinical Impact
Our study highlights important safety events encountered in real-world practice with Angio-Seal closure device. The MAUDE database captures real-world device malfunctions not typically appreciated in conventional clinical trials. Our study provides valuable insight for clinician-users on anticipating and managing the most common device malfunctions. Additionally, our data provide feedback for manufactures to optimize product design and direct manufacturer user training to improve safety. Finally, we hope that the study promotes system-level strategies that foster reporting of safety events and undertaking of root cause analysis.
Introduction
More than 7 million percutaneous arterial procedures are performed annually worldwide, and the common femoral artery is the most frequent access site. 1 Although manual compression (MC) remains the gold-standard for achieving homeostasis, it may at time be time-consuming, personnel intensive, require prolonged bed rest, and less comfortable for patients. 1 Moreover, MC may not achieve adequate hemostasis in patient demographics with obesity, anticoagulation, or antiplatelet use. 1
Vascular closure devices (VCDs) were first introduced in the early 1990s. Vascular closure devices have the advantage of minimizing time to hemostasis, resource utilization (alleviates staff of providing MC), bed rest, as well as time to ambulation and discharge. 1 The disadvantages include risk of device malfunction, groin infection, limb ischemia, localized thrombosis or distal embolization, as well as device cost and inherent learning curve for operation. Multiple factors influence outcomes of VCDs, including patient characteristics (obesity, anticoagulation status), intervention modality, device features, operator experience, and anatomic challenges (vessel size, degree of calcification). 1 With the rapid expansion of VCDs, the estimate market size has surpassed 1 billion dollars in 2021. 2 As vascular percutaneous interventions grow, VCDs continue to become increasingly more valuable.
Angio-Seal (Terumo Interventional Systems, Somerset, New Jersey) is a VCD indicated for diagnostic and therapeutic interventions. There are 3 versions of the device: STS plus, VIP, and Evolution: introduced in 1999, 2005 and 2012, respectively. STS plus is a suture-mediated closure system, whereas VIP and Evolution utilize a bioabsorbable closure mechanism. 1 Evolution is the newest version with a larger plug for faster closure time and a more user-friendly design. 1 Greater than 75% of Angio-Seal devices used worldwide are VIP. 1 The Angio-Seal VIP and Evolution systems are composed of the following components: an insertion sheath, arteriotomy locator (modified dilator), guidewire, absorbable collagen sponge, and absorbable polymer anchor connected by an absorbable suture (Figure 1). The device operates as a passive approximator that deploys a collagen plug over the arteriotomy site with the aid of an intraluminal anchor. 1 After intraluminal placement is confirmed, an anchor abuts the wall and a collagen plug is deployed over the arteriotomy. The plug expands when it enters the subcutaneous tissue and accelerates the clotting cascade. The anchor and plug are resorbed over time. Angio-Seal has been approved to close arteriotomy sites up to 8F; although in practice, it is used to close larger-caliber arteriotomies ranging from 9 to 12F with good efficacy. 3
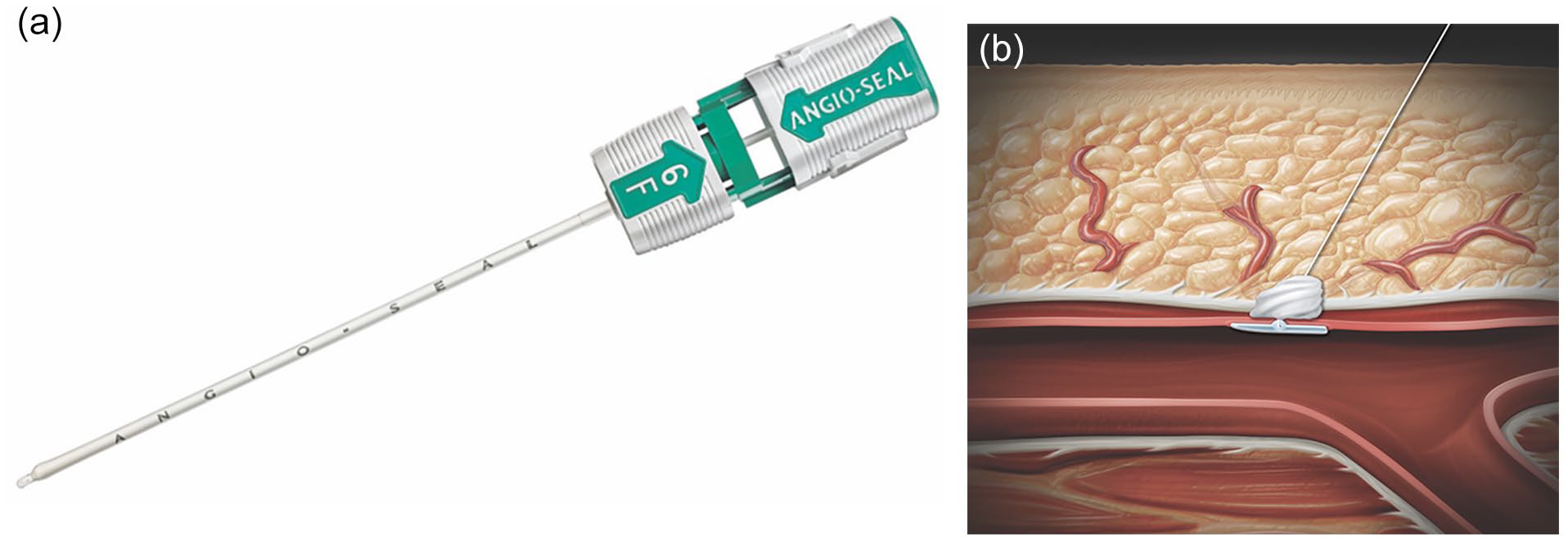
(A) Angio-Seal VIP 6F. (B) Graphic demonstrating mechanism of hemostasis.
Clinical trials of Angio-Seal suggest low complication rates.4–9 Yet, real-world experience may reveal new safety issues not previously appreciated in clinical trials. 10 Over the last decade, numerous devices have been recalled following US Food and Drug Administration (FDA) approval due to safety concerns identified in clinical practice.11,12 This highlights the need for post-market surveillance, which may reveal issues not typically assessed for in clinical trials. Herein, we present real-world safety events associated with Angio-Seal using the FDA’s Manufacturer and User Facility Device Experience (MAUDE) database. 13 We also propose strategies to mitigate and manage complications associated with Angio-Seal use.
Materials and Methods
MAUDE Database
The MAUDE database stores post-market adverse events involving medical devices.13,14 The database is updated monthly by reports from manufacturers, device users, health care professionals, and patients. Reports contain information about the device and narrative description of the safety event. The database is reviewed routinely by the FDA and manufacturers to address device-related safety events. The FDA issues safety alerts or recalls if a device is found to be defective. Although the database does not have utility in establishing definitive adverse event rates as it relies on clinician reporting, it can provide insight into the mechanism of device associated failure. There are a number of studies that have explored the MAUDE database for obtaining real-world safety data on various devices.15–22
Data Collection
The MAUDE database was searched on February 2, 2022 using search terms “Angio-Seal,” “Angioseal,” and “Angio Seal.” Data from November 2019 to December 2020 were independently evaluated by 2 radiology resident reviewers. After excluding duplicate (68) and incomplete entries (82), 715 reports were included in the final analysis. Reports were classified according to (1) mode of device failure, (2) type of complication, (3) treatment modality used to manage the complication, and (4) level of associated patient impact using the Society of Interventional Radiology (SIR) and Cardiovascular and Interventional Radiological Society of Europe (CIRSE) adverse event classification systems (Tables 1 and 2).23,24
Society of Interventional Radiology’s Adverse Event Classification. 23

Cardiovascular and Interventional Radiological Society of Europe Adverse Event Classification. 24
Results
There were a total of 715 reported safety events. These involved Angio-Seal VIP (666; 93.1%), Evolution (41; 5.7%), STS Plus (8; 1.1%), with sizes 6F (447; 62.5%) and 8F (268; 37.5%). These ratios are reflective of the model’s prevalence in the market during the study period (2019-2020), where VIP was by far the most commonly used model, followed by Evolution and a minority included STS Plus. A minority of devices were returned to the manufacturer for root cause analysis (199; 27.8%).
Among reports, the most common procedures involved coronary intervention (238; 33.3%), coronary angiogram (62; 8.7%), peripheral intervention (67; 9.0%), peripheral angiogram (58; 8.1%), neurointervention (28; 3.9%), and coiling procedure (55; 7.7%). Of note, 2 of the entries involved brachial artery closure (off-label use for this device), whereas the remainder involved the common femoral artery.
The mode of failure involved damaged device, defined by unrecognized use of a damaged device (310; 43.4%), failed deployment (144; 20.1%), failed arterial advancement (45; 6.3%), detachment of device component during use (35; 4.9%), failed retraction (26; 3.6%), operator error (8; 1.1%), and the remainder were indeterminate (147; 20.6%) (Table 3).
Mode of Device Failure Reported in the MAUDE Database Related to Angio-Seal.
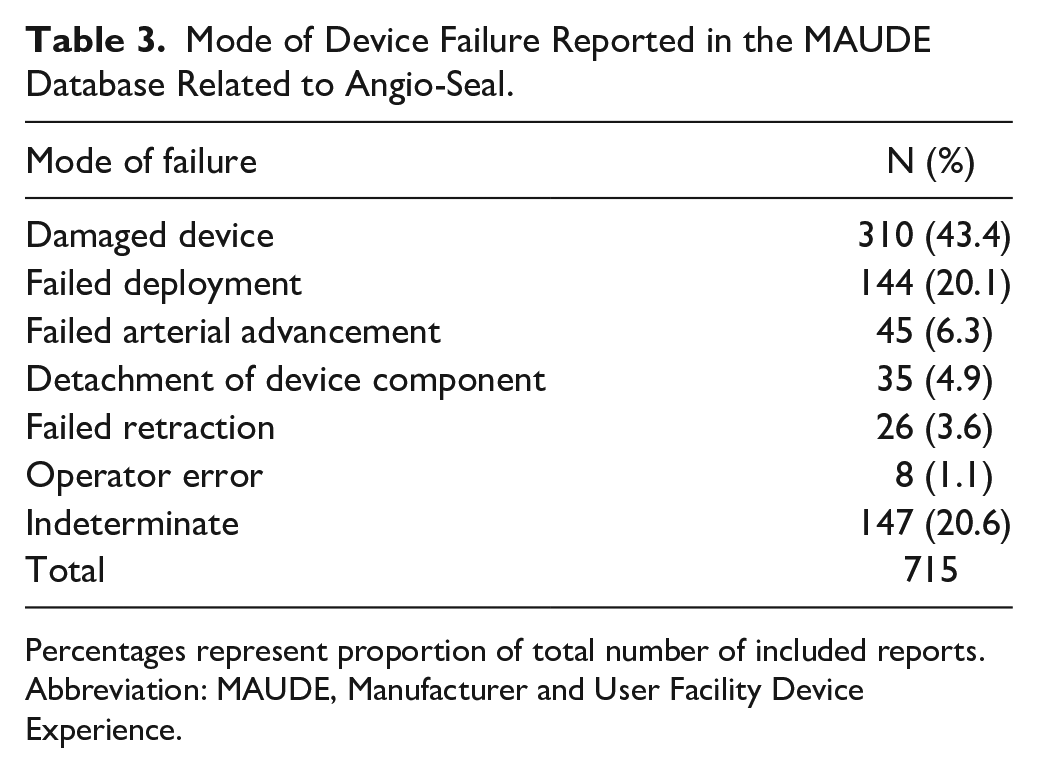
Percentages represent proportion of total number of included reports.
Abbreviation: MAUDE, Manufacturer and User Facility Device Experience.
The most common damaged device incidents involved (1) bypass tube found detached from the carrier tip (87 cases), (2) deformed tip of the Angio-Seal (54), (3) resistance when advancing the locator into the sheath (54), (4) missing Angio-Seal component (16), and (5) bent insertion sheath (12). Among the failed retraction category, most commonly (1) the hemostatic component was found protruding at the skin (37), (2) the collagen plug was found in the extravascular space (4), or (3) embolized intravascularly (6).
Of the 715 device complications, a total of 320 events (44.8%) were associated with patient harm (Table 4). Complications involved minor blood loss defined as <500 cc (244; 34.1%), hematoma (40; 5.6%), significant blood loss defined as >500 cc (10; 1.4%), pseudoaneurysm (10; 1.4%), thrombus (6; 0.8%), pulseless extremity (4; 0.6%), vascular dissection (3; 0.4%), emboli (2; 0.3%), and infection (1; 0.1%).
Patient-Related Complications Reported in the MAUDE Database Related to Angio-Seal Use.
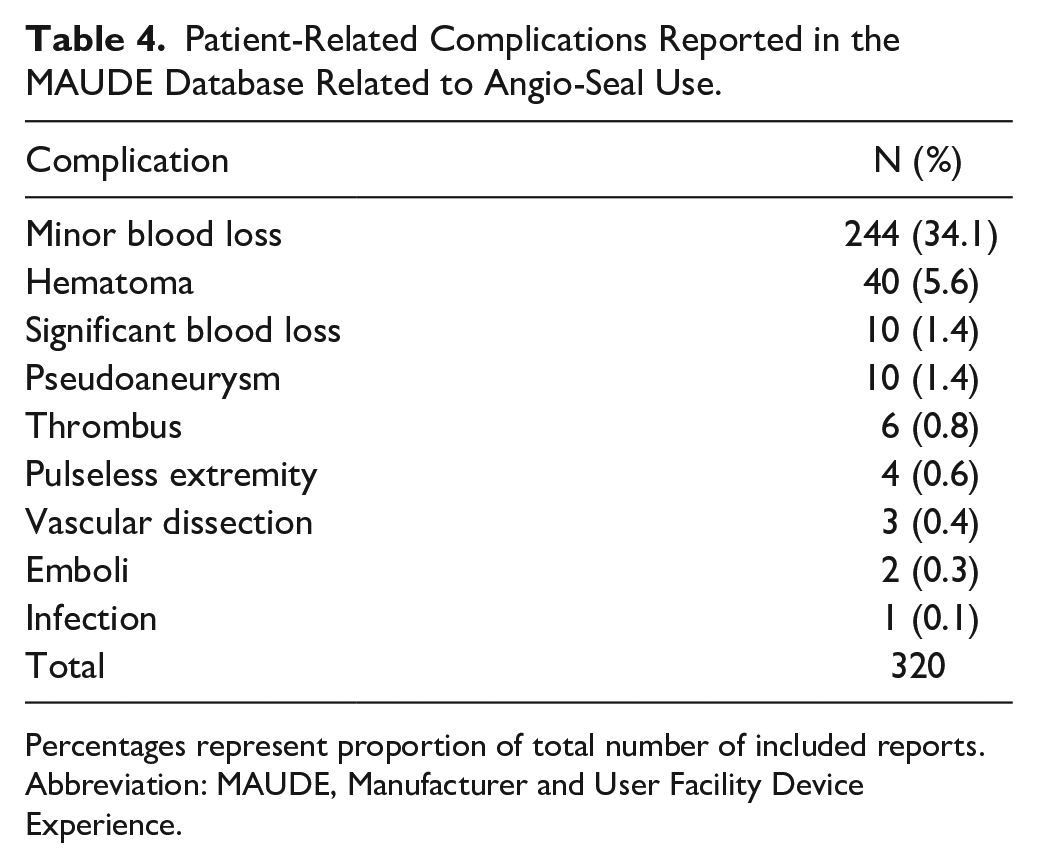
Percentages represent proportion of total number of included reports.
Abbreviation: MAUDE, Manufacturer and User Facility Device Experience.
Manual compression was used in 43.3% of cases (310) to achieve hemostasis upon Angio-Seal failure. More advanced intervention was required in 8.8% of cases (63) to manage associated complications (Table 5). Interventions included surgical repair (31; 49.2%), thrombin injection (6; 9.5%), balloon tamponade (4; 6.3%), covered stent (3; 4.8%), and the remainder were unspecified (19; 30.2%).
Interventions to Manage Complications Associated With Angio-Seal Use Reported in the MAUDE Database.
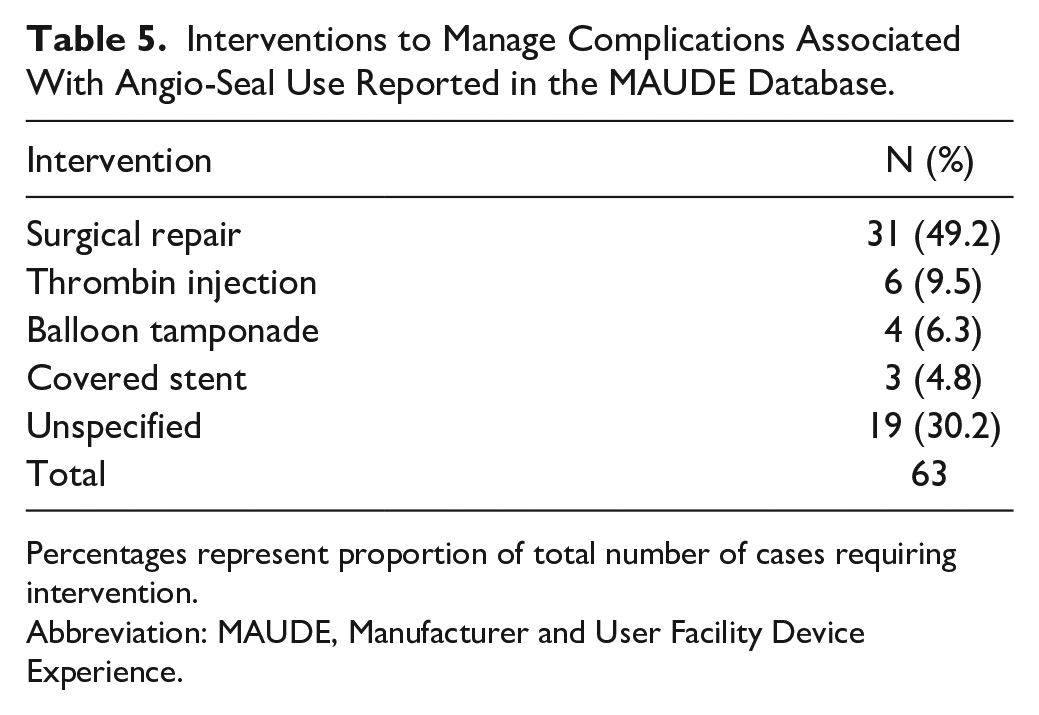
Percentages represent proportion of total number of cases requiring intervention.
Abbreviation: MAUDE, Manufacturer and User Facility Device Experience.
According to SIR adverse event classification, the majority of events were associated with mild patient harm (658; 92%), followed by moderate (22; 3.1%), severe (15; 2.1%), life-threatening (18; 2.5%), and death (2; 0.3%). According to the CIRSE adverse event classification, the majority of safety events were associated with class 1 (658; 92.9%), followed by class 2 (22; 3.1%), class 3 (33; 4.6%), and class 6 (2; 0.3%) level of patient impact (Table 6). Case description of events which involve patient death or life-threatening outcomes are summarized in Table 7.
Safety Event Associated Level of Patient Impact as Per the CIRSE and SIR Classifications.
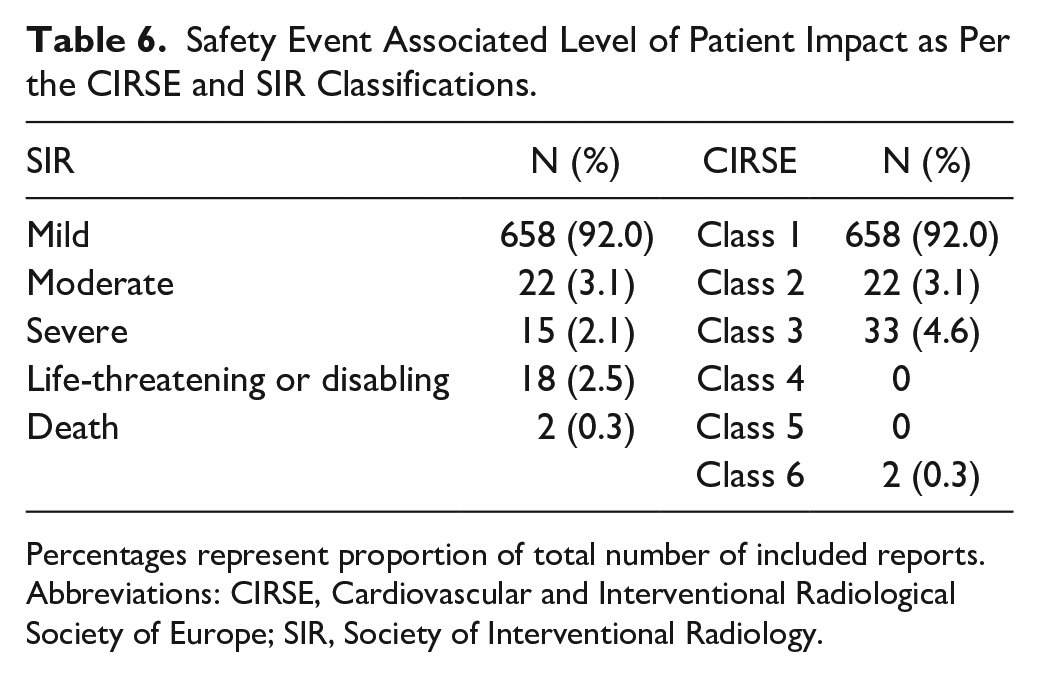
Percentages represent proportion of total number of included reports.
Abbreviations: CIRSE, Cardiovascular and Interventional Radiological Society of Europe; SIR, Society of Interventional Radiology.
Description of Cases Which Involved Patient Death or Life-Threatening/Disabling Events.

Abbreviation: CT, computed tomography; MC, manual compression.
Discussion
The literature has shown that although complications of Angio-Seal are extremely rare, they do occur and it is important for clinicians to be aware of them. 1 These include failure of device deployment, access-site hematoma, infected hematoma, pseudoaneurysm, arterial laceration, oozing of blood up to 1 hour post-deployment, and new onset pain at the groin.9,25–30 In addition, vascular occlusion, loss of distal pulse, retroperitoneal bleed, and dissection have all been described, although they remain exceedingly rare.28–30
The most frequent complications in our study involved minor blood loss as defined by <500 cc (34.1%), puncture site hematoma (5.6%), and significant blood loss as defined by >500 cc (1.4%). This is similar to findings of a large multicenter trial by Wong et al 9 in which common complications were access-site rebleeding or hematoma formation; however, they had no reports of major access-site–related adverse events. The most common cause of safety events in our data was the unrecognized use of a damaged device (43.4%), which points to the simple yet crucial step of device inspection prior to use. Notably, it is possible that some cases of operator error led to a damaged device following use, which was identified and labeled retrospectively as an unrecognized use of a damaged device. The vast majority of events in our study were associated with minor adverse patient effect (92%, grade 1 level of patient impact as per the CIRSE adverse event classification). In addition, a minority of devices were returned to the manufacturer for root cause analysis (27.8%). Similarly, Khalid and Ahmad 31 found that across a wide review of the MAUDE database, only a minority of devices were actually returned to the manufacturer. As a result, the input of the device manufacturer is lacking in many reports and a root cause analysis could not be conducted. Hence, return of device to manufacturer should be encouraged.
The majority of the complications identified in our study were similar to the prior literature. However, our study revealed some additional rare complications. These included a case of collagen caught intravascularly such that the device could not be removed, requiring surgical removal. Another case showed failure to deploy Angio-Seal due to a knot in the device suture. Although no deaths were reported in the literature to our knowledge, our data revealed 2 patient deaths reported within the Angio-Seal MAUDE data. Further analysis of these 2 cases involved retroperitoneal bleeding as a cause; however, although both reports described injury to the inferior epigastric artery found at the time of attempted rescue surgery, it remains unclear if this was related to the initial access, arterial puncture failure, or secondary to anticoagulation status. If in fact there was an inferior epigastric injury, it seems unlikely this would be related to Angio-Seal deployment or failure.
Intra-arterial dislocation of vascular plug causing arterial occlusion has been described in a number of studies.9,25,32 Atherosclerosis of the femoral vessels has been shown to contribute to vessel occlusion by entrapping the Angio-Seal plug in the stenotic area. 32 Some authors suggest that Angio-Seal use should be contraindicated in cases of heavy atherosclerotic disease or vessel diameters less than 5 mm. 32 The polymer anchor has also been reported to embolize and cause complete occlusion or stenosis of the femoral artery, requiring surgical retrieval and definitive arterial repair.33,34 According to the manufacturer, the product anchor is meant to be 95% absorbed by 42 days and 100% by 60 to 90 days. However, vascular obstruction has been reported up to 3 months after placement. 34
Delayed complications of Angio-Seal are reported in the MAUDE database. For example, on post-operative day 10, the Angio-Seal anchor was found to have embolized and caused vascular occlusion, requiring surgical retrieval. In another instance, a patient presented following discharge with a retroperitoneal bleed and at the time of surgical intervention, the Angio-Seal anchor was found dislodged in the inguinal ligament. Another case involved a patient who developed a femoral hematoma and hypotension 60 hours post-cardiac intervention. A covered stent was used to seal the hematoma, although no clear cause for the hematoma was identified. Notably, the author’s experience has also seen cases of delayed hematoma whereby the anchor was found dislodged from the artery several days following device deployment. A limitation to consider is the lack of data regarding the puncture zone (vessel calcification, diameter, tortuosity) as well as puncture type (fluoroscopic or ultrasound guided vs no image guidance), which inherently affects the rates of delayed complications.
Late vascular complications of Angio-Seal have also been reported in the literature as case reports. For example, in a patient with persistent pain at access site 3 weeks following intervention, intraluminal collagen causing eccentric stenosis was found. 34 In another case, patient presented 3 weeks post-procedure with leg claudication and was found to have intra-arterial collagen. 34 Follow-up Dopplers did not show any absorption after 2 weeks, at which point percutaneous extraction of the collagen plug was performed, although incompletely as part of the collagen had begun to dissolve. 34 In a similar case, angiogram 3 months post-intervention showed persistent subtotal arterial occlusion secondary to intraluminal collagen plug, subsequently treated with atherectomy and balloon angioplasty. 34
Potential mechanisms of Angio-Seal–related arterial occlusion include the following: (1) anchor-induced dissection flap with a superimposed thrombus from dragging of the anchor along the inner arterial wall during deployment; (2) thrombosis induced by the intraluminal anchor or a portion of the collagen plug that prolapses into the lumen perhaps secondary to dynamic flexion forces; (3) atherosclerotic plaque rupture from device-induced trauma; (4) delayed detachment of the footplate from the collagen plug and its subsequent embolization; and (5) intimal hyperplasia at the closure site as a delayed cause of stenosis. Taken together, the data suggest that in patients with symptoms of arterial insufficiency following the use of Angio-Seal in both the acute and delayed stages, local arterial stenosis at the closure site should be considered in the differential diagnosis.
Moreover, risk of infection has been reported with Angio-Seal in a number of case reports whereas the overall incidence remains extremely rare.35–37 In theory, the collagen plug is foreign material inside the arterial lumen and can act as a nidus for infection. Proposed risk factors associated with infection are obesity, diabetes mellitus, and previous placement of percutaneous closure devices. 36 To this end, patients with high probability of needing further vascular access at the same site in the near future may not be ideal candidates for closure device use. 36 Angio-Seal recommendations are that vessels closed with this product not be accessed again for 6 weeks. 36
Although it is “off-label,” the use of Angio-Seal closure for brachial access has been studied with a reported success rate of 96.9% among 140 patients. 38 Major complications (3.1%) at 30 days consisted of puncture site hematoma, brachial artery occlusion, and pseudoaneurysm. 38 Minor complications included minimal blood loss from the access site and mild pain at the cubital fossa. 38 In another smaller study which included 36 patients, only minor complications including mild paresthesia, pain, and hematoma were observed. 39
Reflecting on the data presented herein, several complications unique to Angio-Seal along with management options are discussed as follows:
Importance of device inspection: This analysis has shown that damaged devices account for nearly half of reported failures. Inspection of the device prior to use is therefore a simple yet crucial step in mitigating potential adverse events.
Collagen deposition into the artery: If this condition is suspected, the diagnosis can be confirmed by duplex ultrasound. Management is controversial; however, options include leaving the collagen plug in situ with anticoagulation, endovascular snaring, or surgical removal. 40 In addition, intraluminal collagen deposition may remain undetected with ischemic symptoms developing several weeks after use. 41 Therefore, all cases of lower limb ischemia within several months after Angio-Seal deployment should be investigated with duplex ultrasound to exclude intravascular collagen deposition as the potential cause.
Ultrasound-guided deployment: Ultrasound-guided Angio-Seal deployment has been described to be useful in 3 scenarios.30,42 It is useful to avoid entrapment of the anchor within posterior wall plaque or a small caliber vessel leading to intraluminal deployment of the collagen plug, to avoid extraluminal deployment in patients in whom pseudoaneurysms develop during the intervention, and to determine whether there is intraluminal deployment of the anchor and collagen plug when there is primary failure in achieving hemostasis. 42
Very thin patients: Collagen may protrude from the skin after compaction has been completed. This is normal occurrence in thin patients and should not be mistaken for erroneous closure, as was the case in a number of adverse event reports in the database. If this occurs, attempt can be made to push the collagen under the skin using the compaction tube or a sterile hemostat. Alternatively, pre-puncture dissection of the subcutaneous space can be done to free the skin edges which can be sutured subsequently over the exposed plug to avoid skin protrusion. Vigorous compaction should be avoided as it may result in anchor fracture. Cutting off the excess collagen should also be avoided, as the suture woven through the collagen may be cut and the integrity of the anchor/collagen could be compromised.
Limitations
General limitations of the MAUDE database are inclusion and selection bias given that it is a voluntary reporting system with contributions from manufactures, users, health care professionals and patients, incomplete and non-standardized reporting of details surrounding safety event, and lack of a rigorous data verification process. Moreover, although the database establishes an associative relationship between the adverse event and subsequent complication, a definitive cause and effect cannot be confirmed. In addition, a minority of devices were returned to the manufacturer, hindering root cause analysis. A degree of bias is also inherent to the process of a manufacturer-led device root cause analysis which should be considered. Furthermore, true incidence rates of safety events are unknown given the unspecified number of Angio-Seal devices used per year. Finally, the database does not reflect long-term complications.
Conclusions
We present safety events encountered in real-world practice with Angio-Seal. The MAUDE database captures real-world device malfunctions not typically appreciated in conventional clinical trials. The database provides valuable feedback for manufacturers to optimize product design and direct manufacturer-user training. Moreover, the data provide valuable insight for clinician-users on managing common device malfunctions to foster patient safety. Ideally, greater detail would be available in safety reports along with return of device to manufacturer in order to facilitate in-depth root cause analysis of safety events. System-level strategies for addressing barriers to under-reporting of safety events include ensuring that clinicians are aware of the processes involved in reporting and simplifying the process. From a policy perspective, aligning legislation to foster a system that mandates reporting of adverse events is central to making MAUDE and similar databases more impactful. Equally important is the reporting of near misses as awareness of the factors that might lead to error is the first key to error reduction. 43
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
