Abstract
Introduction:
Rotational atherectomy has shown promise as an adjunctive therapy to percutaneous transluminal angioplasty (PTA) and stenting for the treatment of peripheral arterial disease (PAD). However, published data regarding the safety of these devices are limited. The Food and Drug Administration Manufacturer and User Facility Device Experience (MAUDE) database collects reports of adverse event for medical devices. We present 3 years of MAUDE adverse events data for the Jetstream Atherectomy System (Boston Scientific) for the treatment of PAD.
Materials and Methods:
We searched MAUDE from January 1, 2019 to December 31, 2021. Duplicate reports and those with insufficient information were excluded, leaving a total of 500 reports for analysis. Adverse events were categorized as either patient complication, device malfunction, or both. Adverse events were classified using the Cardiovascular and Interventional Radiological Society of Europe’s (CIRSE) classification system for adverse events.
Results:
The most common patient complications were embolism (22; 4.4%), dissection (17; 3.4%), vessel perforation (12; 2.4%), and device fracture in the patient (6; 1.2%). The most common modes of device failure were entrapment of the device on the guidewire (134; 27%), loss of blade rotation (116; 23%), loss of aspiration (99; 20%), and mechanical damage (57; 11%). As per the CIRSE adverse events classification, most events had no post-procedural sequelae (475; 95%), followed by those requiring prolonged observation (14; 2.8%), and post-procedural therapy without long-term sequelae (10; 2.0%). One hundred six devices (21%) were returned for manufacturer analysis.
Conclusion:
We highlight important adverse events encountered in real-world practice with the Jetstream Atherectomy System. This analysis provides further understanding of the safety profile and modes of failure of Jetstream, and could help guide improvements in product design and manufacturer-user training. There is greater need for root-cause analysis that can aided by returning devices to the manufacturer.
Clinical Impact
We highlight important adverse events encountered in real-world practice with the Jetstream Atherectomy System. The MAUDE database is useful for capturing and characterizing modes of device failure/malfunction not typically described in conventional clinical studies. This may provide valuable information to help guide improvements in product design and manufacturer-user training. This information could also potentially be useful in helping establish manufacturer and product liability in the setting of medicolegal claims. We hope that by contributing to the growing understanding of the safety profile of the Jetstream Atherectomy System, our study may help physicians and patients come to more informed decisions regarding treatment options for PAD.
Keywords
Introduction
Endovascular treatments are now considered first-line therapy for peripheral arterial disease (PAD). The most common treatment is conventional percutaneous transluminal angioplasty (PTA). However, long-term patency after PTA is low in heavily-calcified lesions, even when treated with high balloon pressures, due to severely decreased wall compliance. 1 Endovascular atherectomy has emerged as a contemporary adjunctive technique to debulk calcified plaque, thereby increasing vessel compliance prior to angioplasty, with the goal of increasing long-term patency and avoiding the need for bailout stenting.
Various types of atherectomy devices exist, namely, rotational, directional, orbital, and laser. Rotational atherectomy (RA) uses front-cutting blades to debulk plaque; directional atherectomy uses side-cutting blades to both debulk and capture plaque for removal; orbital atherectomy uses an eccentrically-placed diamond-coated crown that orbits the vessel wall 360° to circumferentially remove plaque; laser atherectomy employs lasers to vaporize plaque. Rotational atherectomy offers the benefit of being relatively fast and easy to use. 2 Among rotational devices, the Jetstream Atherectomy System (Boston Scientific [BSc]) offers the potential advantages of active plaque removal with continuous aspiration. In addition, this particular device has some evidence to support its use as an adjunctive endovascular therapy for the treatment of PAD, with a wide array of reported indications.3–7 However, published data regarding its safety profile are limited. The Food and Drug Administration (FDA) Manufacturer and User Facility Device Experience (MAUDE) database collects reports of adverse events for various medical devices used in the United States. The objective of our study is to query the MAUDE database and identify the most commonly reported adverse events for the Jetstream RA system for the treatment of PAD. We present 3 years of MAUDE data.
Materials and Methods
Study Device
The Jetstream RA system consists of a single-use catheter and control pod and a reusable power console. Various catheter sizes are available, including 2 catheters with expandable cutters (Jetstream XC; 2.1/3.0 and 2.4/3.4 mm) and 2 with nonexpendable cutters (Jetstream SC; 1.6 or 1.85 mm). All catheters are compatible with a 7-French sheath and manufacturer-specified 0.014 inch wires. Each catheter consists of a front-cutting tip that rotates at 70 000 to 73 000 RPM. The catheters are designed for differential cutting whereby abnormal fibrous and calcified tissues are preferentially cut, whereas more compliant normal tissue is theoretically spared. In addition, pumps for saline infusion and aspiration are designed to reduce friction and overheating, and allow for continuous aspiration of thrombus and debris, thereby potentially decreasing the risk of dissection and distal emboli.
MAUDE Database
The MAUDE database collects reports of adverse events for medical devices. It accepts entries from mandatory reporters (device manufacturers, user facilities, and importers), and voluntary reporters (healthcare providers, consumers, and patients). The database is updated monthly, and searches within the database are limited to the past 10 years of data. Individual reports contain information on the device type, event date, whether the device was returned to the manufacturer, and narrative comments by the user and/or manufacturer. Adverse events are categorized into device problems, patient problems, or both. The FDA and manufacturers regularly monitor the database to identify and address device safety issues.
Data Collection
Three independent reviewers (AM, FA, and AA) queried MAUDE from January 1, 2019 through December 31, 2021. Our initial search yielded 515 reports. Fifteen of these reports were excluded as they were either duplicates or contained insufficient information. This left a total of 500 reports for analysis. Adverse events were categorized as patient complications, device malfunction or both. Adverse events were classified using the Cardiovascular and Interventional Radiological Society of Europe’s (CIRSE) classification system for adverse events. 8
Results
A total of 500 reports were included in our analysis (Table 1). The target arteries for RA were: superficial femoral artery (SFA)=300 (60.0%), popliteal=57 (11.4%), infrapopliteal=25 (5.0%), common femoral artery (CFA)=2 (0.4%), iliac=1 (0.2%), profunda femoris=1 (0.2%), and unspecified=148 (29.6%). Of these, 24 (4.8%) targeted multiple arteries. Three hundred twenty-seven (65.6%) of reports specified exclusively above-knee targets, 18 (3.6%) specified exclusively below-knee targets, and 7 (1.4%) cases had both above-knee and below-knee vessel targets. Provided indications for RA were: chronic total occlusion=30 (6.0%), in-stent stenosis=24 (4.8%), claudication=4 (0.8%), critical limb ischemia=3 (0.6%), and unspecified=439 (88.0%).
Reported Target Artery and Indication for Rotational Atherectomy (RA).
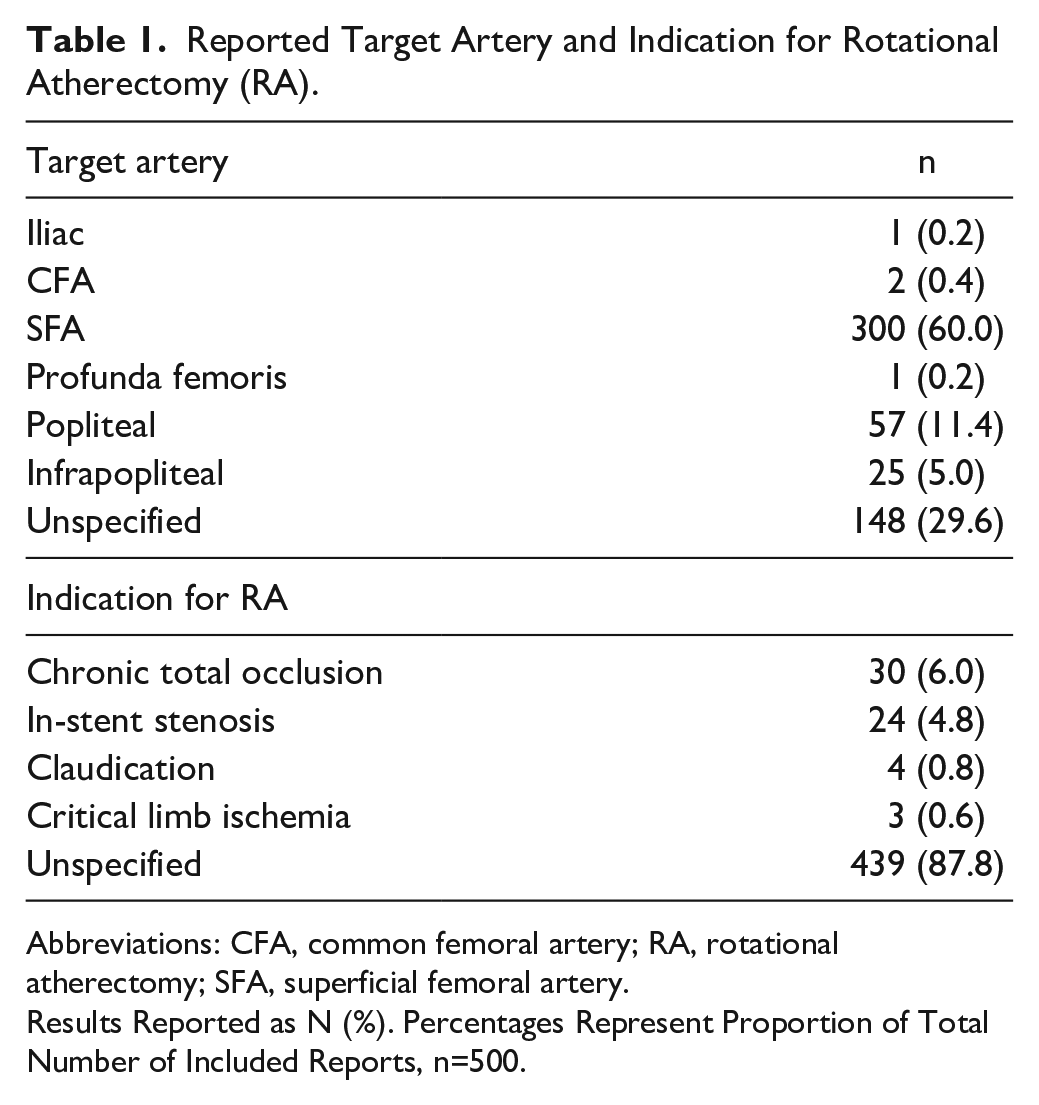
Abbreviations: CFA, common femoral artery; RA, rotational atherectomy; SFA, superficial femoral artery.
Results Reported as N (%). Percentages Represent Proportion of Total Number of Included Reports, n=500.
Patient Complications
Of the 500 reports, 53 (10.6%) reported patient complications. Among all reports, the most common complications were embolism (22; 4.4%), dissection (17; 3.4%), vessel perforation (12; 2.4%), and device fracture in the patient (7; 1.4%) (Table 2). Among the 327 above-knee cases, the most common complications were embolism (20; 6.1%), dissection (14; 4.3%), vessel perforation (10; 3.1%), and device fracture in the patient (4; 1.2%) (Table 3). Among the 17 below-knee cases, the most common complications were embolism (1; 5.9%), dissection (1; 5.9%), and device fracture in the patient (1; 5.9%). In the 7 combined above-knee and below-knee cases, the only reported patient complication was arteriovenous (AV) fistula (1; 14.3%). Most emboli were treated endovascularly during the index procedure, with 4 cases (18%) treated with balloon angioplasty, 3 (14%) with stenting, and 8 (36%) with catheter thrombectomy/aspiration. Three cases of embolism (14%) required surgery, 1 case (4.5%) did not require any therapy, and 3 cases (14%) were not specified. The majority of dissections were also treated endovascularly with either balloon angioplasty or stenting (4 and 6 cases [24% and 35%], respectively). One case (5.9%) required surgery, 3 cases (18%) required no additional intervention, and 3 cases (18%) did not specify the type of treatment. In the setting of vessel perforation, 6 cases (50%) were treated endovascularly during the index procedure with balloon tamponade or stenting (3 cases each), and 6 cases (50%) required surgery. Among the reports of device fracture in the patient, 2 cases (29%) involved the catheter tip, while 5 (71%) involved the guidewire. In 4 cases (57%), the device fragment was abandoned and left in the patient. In 1 case (14%), a snare was used to retrieve the fragment. In another case, a stent was used to trap the device fragment against the vessel wall. In 1 case (14%), the device fragment was retrieved surgically.
Patient-Related Complications Reported in the MAUDE Database.

Abbreviation: TPA, tissue plasminogen activator.
Results Reported as N (%). Percentages Represent Proportion of Total Number of Included Reports, n=500.
Most Common Patient Complications and Modes of Device Failure by Vessel Location.

Results Reported as N (%). Percentages Represent Proportion of Total Number of Included Reports, n=500.
Device Complications
Of the 500 reports, 470 reported device malfunctions (94.0%). The most common modes of device failure were entrapment of the device on the guidewire (134; 26.8%), loss of blade rotation (116; 23.2%), loss of aspiration (99; 19.8%), and mechanical damage (57; 11.4%) (Table 4). Among the 327 above-knee cases, the most common device complications were loss of blade rotation (85; 26.0%), entrapment of the device on the guidewire (80; 24.5%), loss of aspiration (67; 20.5%), and mechanical damage (52; 16.0%) (Table 3). Among the 17 below-knee cases, the most common complications were mechanical damage (8; 47.1%), loss of blade rotation (4; 23.5%), entrapment of the device on the guidewire (4; 23.5%), and loss of aspiration (2; 11.8%). Among the 7 combined above-knee and below-knee cases, the most common device complications were mechanical damage (2; 28.6%), loss of blade rotation (1; 14.3%), entrapment of the device on the guidewire (1; 14.3%), and loss of aspiration (1; 14.3%). Among the reports of entrapment, 59 cases (44%) used a wire not specified as compatible by the device manufacturer, 25 (21%) used a manufacturer-specified compatible wire, and 16 (12%) were unspecified. Among the 116 reports of loss of blade rotation, 28 devices (24%) were returned to the manufacturer for analysis. Of these, 16 (57%) demonstrated visual damage to the catheter in the form of kinks, buckles, or fractures. The remaining 12 returned devices (43%) did not demonstrate any visible damage. Of note, in 15 (54%) of the returned devices, the pinion gear within the catheter had slipped off the driveshaft resulting in a loss of blade rotation. This was seen in catheters both with and without visible external damage. In 5 of the returned devices (18%), manufacturer testing demonstrated normal blade rotation despite user reports of loss of rotation. Of the 99 cases of loss of aspiration, 25 devices (25%) were returned to the manufacturer for analysis. Of these, 7 (28%) demonstrated kinks in the device resulting in air leaks from the aspiration line. Seven other cases (28%) revealed no damage but reported a heavy clot burden within the aspiration line. The remaining 11 cases (44%) cited no device damage and did not comment on the burden of clot within the aspiration line. The most commonly damaged device component was the catheter tip (52 devices; 10%), followed by the guidewire (18 devices; 3.6%). Device-related events resulted in 243 additional Jetstream devices being used during the index procedure.
Modes of Device Failure Reported in the MAUDE Database.
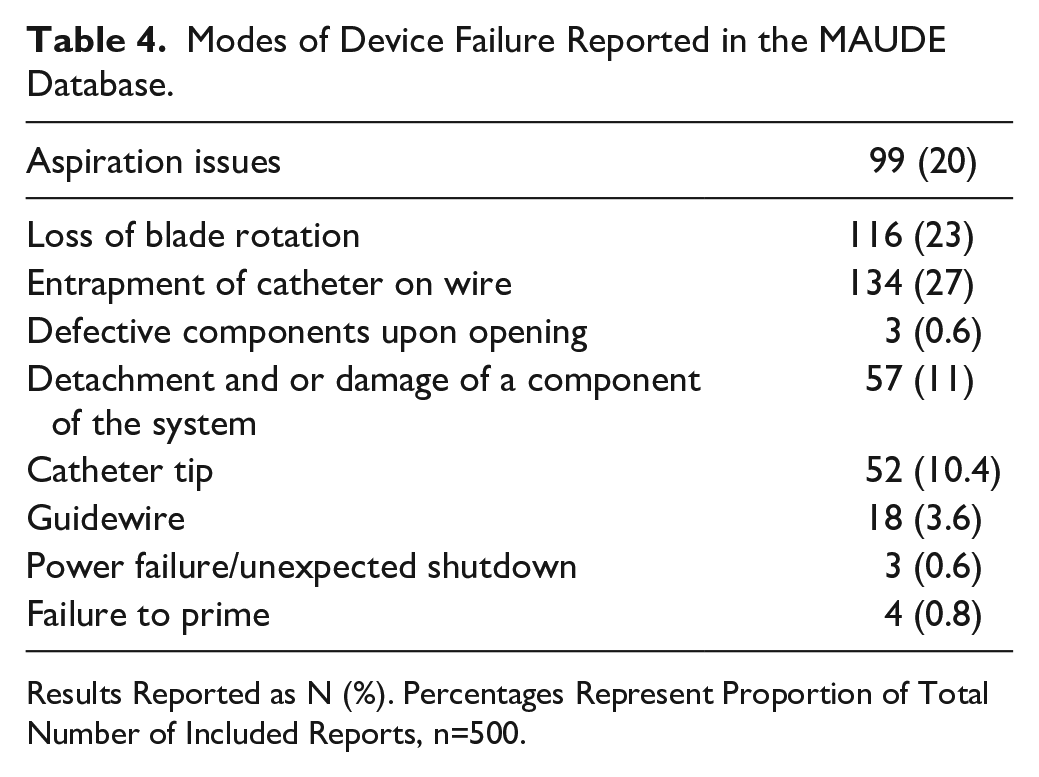
Results Reported as N (%). Percentages Represent Proportion of Total Number of Included Reports, n=500.
There were 24 reports of a device malfunction with related patient harm. This represents 45.2% (24/53) of all patient complications in this study and 5.2% (24/470) of all device malfunctions. For example, 5 of 12 reported cases (42%) of vessel perforation were reported as due to entrapment of the Jetstream catheter on the guidewire, resulting in difficult removal from the patient and vascular damage. Of these 5 cases, 2 patients (40%) required surgical repair of the vessel. In the case of distal embolism, 4 of the 22 reported cases (18.2%) were caused by loss of catheter aspiration, all of which required further treatment with catheter embolectomy.
As per the CIRSE adverse events classification, most events had no post-procedural sequelae (class 1; 475; 95%), for example, in the case of minor mechanical damage to the catheter (Table 5). This was followed by events requiring prolonged observation but no post-procedural sequelae (class 2; 14; 2.8%), such as a case of non-flow-limiting arterial dissection requiring no treatment. Ten events (2.0%) required additional post-procedure therapy or prolonged hospital stay without post-procedural sequelae (class 3), for example, a case of vessel perforation successfully treated with stenting. There were no events resulting in permanent post-procedural sequelae (class 4–6).
Adverse Event Severity as per the CIRSE and SIR Classifications, Respectively.
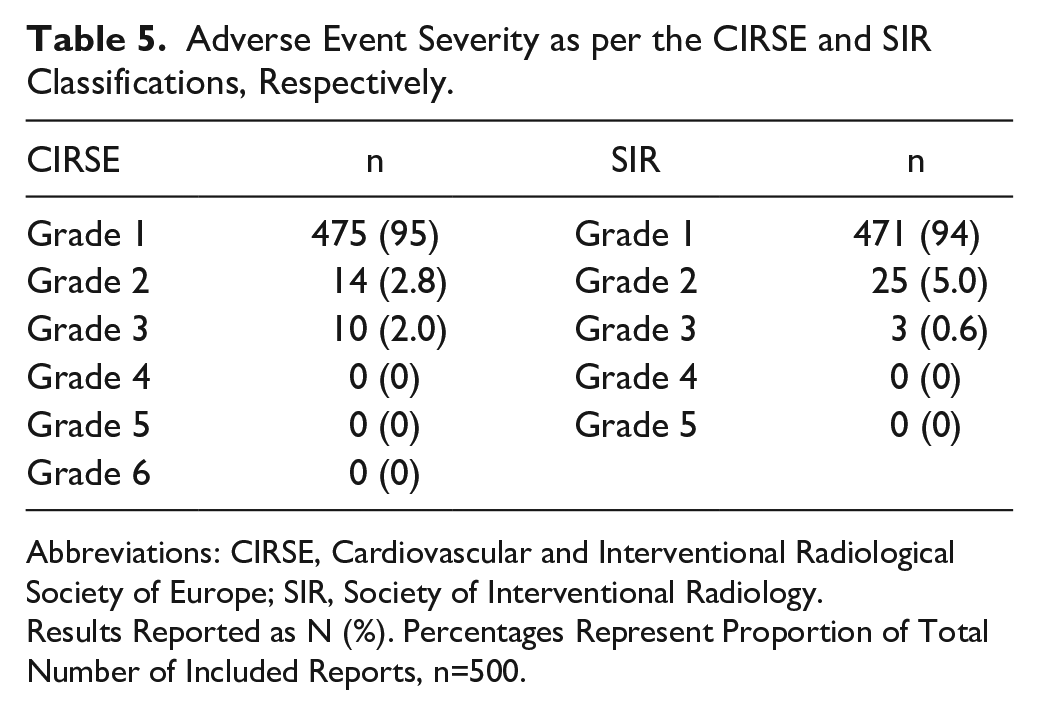
Abbreviations: CIRSE, Cardiovascular and Interventional Radiological Society of Europe; SIR, Society of Interventional Radiology.
Results Reported as N (%). Percentages Represent Proportion of Total Number of Included Reports, n=500.
Among all reports, 106 (21.2%) devices were returned for manufacturer analysis. The root cause of device failure was identified in 81 (76.4%) of the returned devices.
Discussion
We present the most common patient complications and modes of device failure for the Jetstream RA system reported in the MAUDE database over a 3-year period. The following are key findings from our study:
The most common patient complications were embolism and vessel dissection/perforation.
The most common modes of device failure were entrapment of the Jetstream catheter on the guidewire, and loss of blade rotation and/or aspiration.
The majority of reports overall were of device malfunction without report of patient harm.
A significant proportion of patient complications had an associated device malfunction (45.2%).
Most adverse events had no immediate post-procedural sequelae.
A minority (21.2%) of reported devices were returned to the manufacturer for analysis. Of these, the root cause of device failure was identified in the majority (76.4%) of devices.
Prior studies of the safety and effectiveness of Jetstream have focused on patient-related complications.2–7 Adverse event rates in the RA arm of these studies are shown in Table 6. Similar to our findings, distal embolism and vessel dissection/perforation were among the most common patient complications in these studies. With regards to device malfunction, a recent study of MAUDE database reports for different RA devices for treatment of coronary artery disease demonstrated similar results to ours, with device entrapment and mechanical damage representing the most commonly reported modes of failure. 9 However, to our knowledge, our study is the first to report modes of device failure in this device in the setting of PAD; there are currently no other studies of Jetstream or other atherectomy devices for use in PAD using MAUDE data. Modes of failure are an important area of continued study since device malfunctions can be associated with patient complications and likely increased procedure time and cost.
Reported Complications in Prior Rotational Atherectomy Studies.
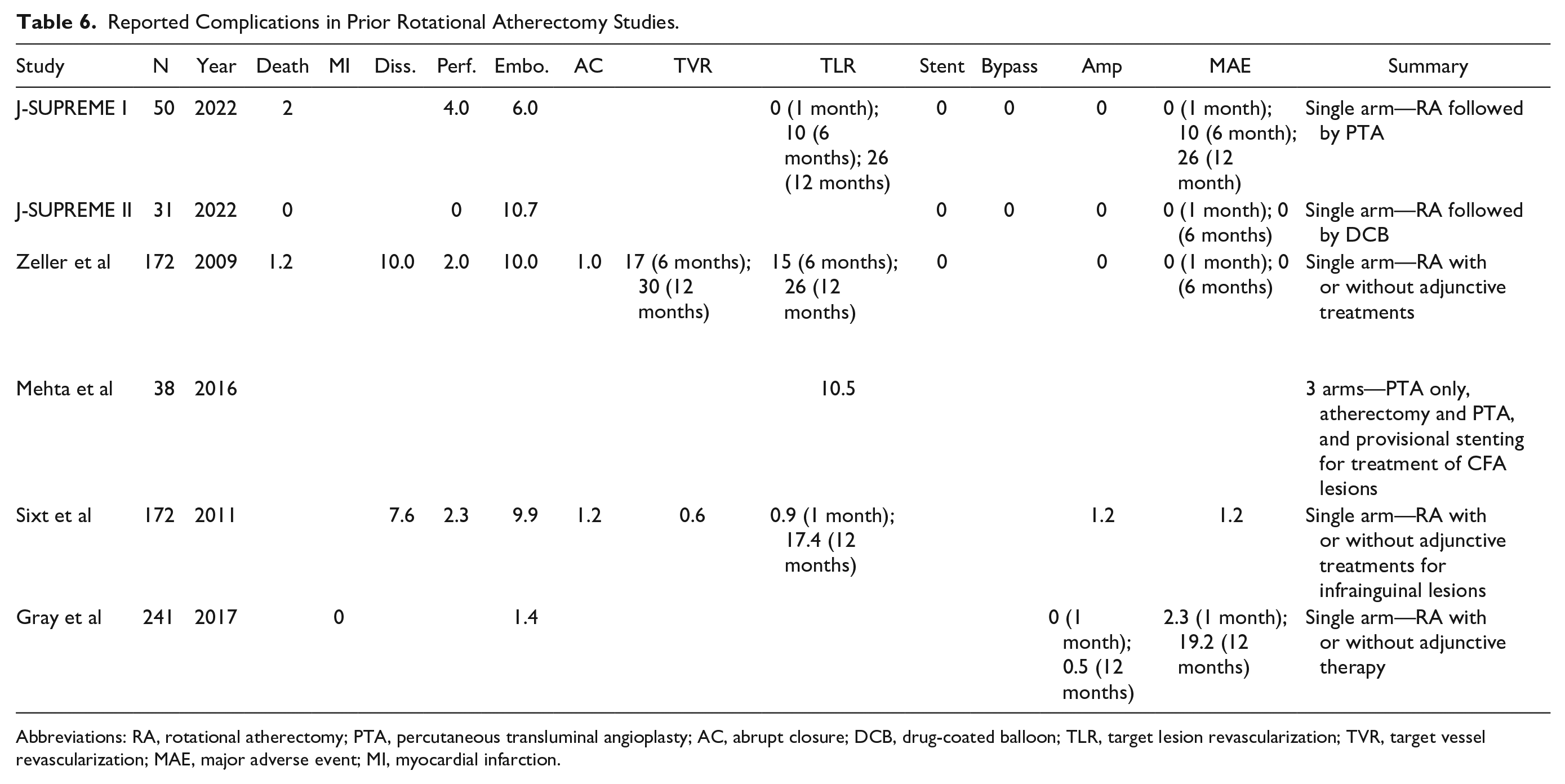
Abbreviations: RA, rotational atherectomy; PTA, percutaneous transluminal angioplasty; AC, abrupt closure; DCB, drug-coated balloon; TLR, target lesion revascularization; TVR, target vessel revascularization; MAE, major adverse event; MI, myocardial infarction.
Complications by Location of Use
When comparing reported device complications for above-knee versus below-knee treatments, mechanical damage was more frequently reported in below-knee procedures compared with above-knee procedures (47.1% versus 16.0%) and was the only complication that showed a clinically significant difference. In contrast, loss of device aspiration was more frequently reported in above-knee procedures (20.5% versus 11.8%), although not clinically significant. With regards to patient complications, vessel perforation was reported in 10 (3.1%) above-knee cases but was not reported in any below-knee case. Analysis of combined above- and below-knee treatments is limited as the reporters in these cases did not specify in which vessel the device or patient complication occurred. It should also be noted that the number of above-knee treatments far outweighed the number of below-knee treatments (327 versus 17).
Devices Returned to the Manufacturer
Only 21.2% (106) of devices were returned to the manufacturer for analysis. However, of these, a root cause of device failure was identified in 76.4% (81). For example, most cases of poor device aspiration were due to either a kinked/ruptured infusion line or a heavy clot burden. In several cases of catheter entrapment on guidewires, manufacturer analysis revealed that a non-compatible wire with a coating was used, resulting in the wire coating peeling off, becoming trapped in the catheter, and impeding wire advancement/withdrawal. In several cases of loss of blade rotation, manufacturer inspection identified an issue with the pinion gear within the catheter slipping off the driveshaft leading to the loss of blade rotation. Information, such as this may inform both users and manufacturers on how to prevent device complications as well as guide future design improvements.
Proposed techniques to minimize complications:
1. Embolism. Use an embolic filter device distal to atherectomy. The filter device must be free-spinning on the wire so that it does not rotate along with the RA device. Note, the Instructions for Use (IFU) of the Jetstream device do not mandate embolic protection; however, its use has been reported 10 and at our institution, it is considered prudent, particularly in higher-risk patients, for example, those with single-vessel run off.
2. Dissection. Prudent case selection and avoidance of highly tortuous vessels.
3. Perforation. Avoid unintentional device oversizing and excessive angulation. Avoid “blades up” position in small caliber vessel.
4. Device fracture. Avoid excessive force on the guidewire and catheter. Ensure appropriate wire positioning distally avoiding side-branches. When going “up and over” the iliac bifurcation, in acute angles, oversizing the sheath to 8Fr can be helpful to allow more freedom of device movement and prevent entrapment/elongation.
5. Entrapment of the catheter on the guidewire. Use guidewires compatible for use with the device as per manufacturer guidelines. Any guidewire with a coating risks being sheared off during rotation and can become embolic. Furthermore, shearing off of the wire coating within the lumen of the catheter can further contribute to entrapment. In the authors’ experience, excessive use of Atherectomy mode without “Rexing” can result in heating of the device that may result in fusion to the guidewire. Liberal use of “Rexing” is optimal.
6. Clogging of the catheter resulting in poor aspiration. Use a short “pecking” motion, whereby the tip of the catheter is repeatedly gently advanced into the lesion and then withdrawn in “Rex” mode. The “Rex” mode allows for the lubricant solution saline/lubricant/ (Rotaglide) vasodilator that can further prevent blocking of the catheter.
Device failures and complications are often hard to address. Many of them, such as perforation, dissection, and embolization are experience related. These complications would decrease with more experience. However, the most common device-related complication was wire entrapment due to incorrect wire choice. This could be easily addressed by education or simple labeling on the packaging. Choosing an inappropriate or incompatible wire could be considered a medical error.
Limitations
Our study has several limitations. First, given that our data do not provide an incidence rate for the adverse events, it is not possible to compare our data with those of previous studies. Next, only a minority of reported devices were returned to the manufacturer for root-cause analysis, limiting our ability to establish and confirm a cause-and-effect relationship between the adverse event and the device. As well, reporting of the vessel treated, treatment indication, and the presence of user error were inconsistent and often not specified. The MAUDE database may not have captured all adverse events since it is a passive surveillance system. For example, cases of intravascular hemolysis and infection following RA have been reported but were not captured in our data.11,12 Moreover, since reporting in MAUDE is voluntary, adverse events are likely under- reported, and reports may be subject to recall bias. Also, our study focuses on a single device. As such our results and experience are limited to the Jetstream Atherectomy System and are not necessarily applicable to the broader category of all RA devices. Finally, the adverse events captured by the MAUDE database occurred intra-procedurally or during the index admission, and long-term complications may not have been captured.
Conclusion
We highlight important adverse events encountered in real-world practice with the Jetstream Atherectomy System. The MAUDE database is useful for capturing and characterizing modes of device failure/malfunction not typically described in conventional clinical studies. This may provide valuable information to help guide improvements in product design and manufacturer-user training. This information could also potentially be useful in helping establish manufacturer and product liability in the setting of medicolegal claims. There is greater need for root-cause analysis that can be aided by encouraging users to return devices to the manufacturer and to provide more detailed and standardized reports. Finally, we hope that by contributing to the growing understanding of the safety profile of the Jetstream Atherectomy System, our study may help physicians and patients come to more informed decisions regarding treatment options for PAD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
