Abstract
Purpose:
Ruptured mycotic pararenal aortic aneurysms are rare and serious condition that requires prompt treatment. Open surgery with aortic resection and in-situ or extra-anatomic reconstruction is the standard treatment. The aim of this technical note is to report urgent endovascular treatment using a readily available custom-made device (created for another patient), with a back-table modification using pericardium patch and a new fenestration.
Technique:
In preoperative measurements on centerline-based workstation, aortic diameter in proximal and distal landing zone and target vessel position matched the measurements of graft plan of custom-made device (CMD) besides left renal artery. To address current patient`s anatomy, closure of the nonsuitable fenestration with pericardial patch and creation of new fenestration (1 cm above and 1:15 hours posterior to original fenestration) for the respective target vessel have been performed. Postoperative computed tomography angiography (CTA) scan showed complete exclusion of aneurysm, perfused target vessels, and no endoleak. Under resistance-based antibiotic therapy, the patient was asymptomatic and showed normal infection parameters in blood samples postoperatively.
Conclusion:
In the hands of an experienced endovascular aortic surgeon modification of a custom-made device is a quick and feasible technique in this emergency situation. Long-term follow-up must confirm the durability and reliability of this new technique.
Clinical Impact:
The described technique of modification of a custom-made endograft can provide an alternative endovascular treatment option for urgent complex abdominal aortic pathologies. Compared to the current available treatment modalities, like physician modified endografts, off-the-shelf branched devices, parallel grafts and in-situ fenestration, it can save considerable time and provides reasonable sealing in ruptured cases. The technique offers a valuable add-on to the armamentarium of experienced endovascular physicians.
Keywords
Introduction
Similar to standard infrarenal abdominal aortic aneurysms (AAA), endograft techniques and modifications have emerged as one of the leading treatment options for complex AAA (cAAA) and thoraco-abdominal aneurysms (TAAAs).1,2
Technological advances, alongside the development of an ever-increasing array of devices and techniques, has paved the way for complex aortic solutions, such as chimney EVAR (ChEVAR), branched and fenestrated EVAR (B/FEVAR), physician-modified endografts (PMEGs) and/or in-situ laser FEVAR (ISLF) increasing the number of patients susceptible of undergoing endovascular treatment. 2
Although FEVAR is recommended as the first endovascular solution for the treatment of cAAAs, most devices will be custom-made devices, requiring between 4 to 6 weeks of manufacturing time. 2 This aspect may limit their role in the management of patients with large, symptomatic or ruptured cAAAs. In these patients, PMEGs have emerged as an alternative solution with good mid- and long-term results. Jusczak et al 3 have reported on increased suitability with the use of PMEGs for treatment of urgent cAAAs in comparison to ChEVAR, T-Branch and other off-the-shelf available fenestrated and branched devices. Similarly, Gouveia E Melo et al 4 performed a systematic review on PMEGs, showing technical success rates of more than 96% for the treatment of urgent cAAAs and TAAAs. Despite high technical success rates, 25% of patients undergoing urgent repair presented some kind of major adverse event during the perioperative period. 4 Finally, Tsilimparis et al 5 reported successful aneurysm exclusion and target vessel revascularization with low short-term mortality rates with PMEGs for the treatment of urgent cAAAs and TAAAs. Despite promising results of PMEGs in urgent cases, one of limitations is the time required for the back-table modifications, limiting their implementation to hemodynamically stable patients. 5
Mycotic aneurysms, with Salmonella spp., being one of the leading microorganisms involved, represent an additional challenge, especially in patients with ruptured aneurysms.6,7
Treatment of ruptured mycotic aortic aneurysm is complex. Open repair with in-situ (bovine or autologous venous material) or extra-anatomic reconstruction is a referred treatment option. 8 Recent studies suggest that endovascular treatment can be a safe option in selected patients, especially in complex aortic aneurysm and with the use of resistance-based antibiotic therapy in advance.9 –11
The aim of this study was to report on the use of a readily- available custom-made fenestrated device (designed for another patient) that was subsequently modified by the use of a bovine pericardial patch and an additional surgeon-made fenestration for the treatment of a patient with a ruptured mycotic aneurysm.
Technique
A 70-year-old female patient, with known chronic obstructive pulmonary disease Gold III and coronary heart disease, was admitted to our emergency department with a ruptured pararenal AAA. The patient had been admitted to an external hospital 10-day prior to referral due to sepsis with positive blood cultures for Salmonella enteritis and had undergone treatment with intravenous Sultamicillin (broad-spectrum antibiotic). A computed tomography angiography (CTA) was subsequently performed due to the development of acute upper back pain, revealing a ruptured pararenal aortic aneurysm (Figure 1) and an immediate hospital transfer was performed. Considering the concomitant salmonella sepsis, the pararenal aneurysm was categorized as mycotic. The patient presented hemodynamically stable with permissive hypotension (heart rate of 93 bpm, blood pressure 100/60 mm Hg, and a hemoglobin level of 7.5 g/L.).
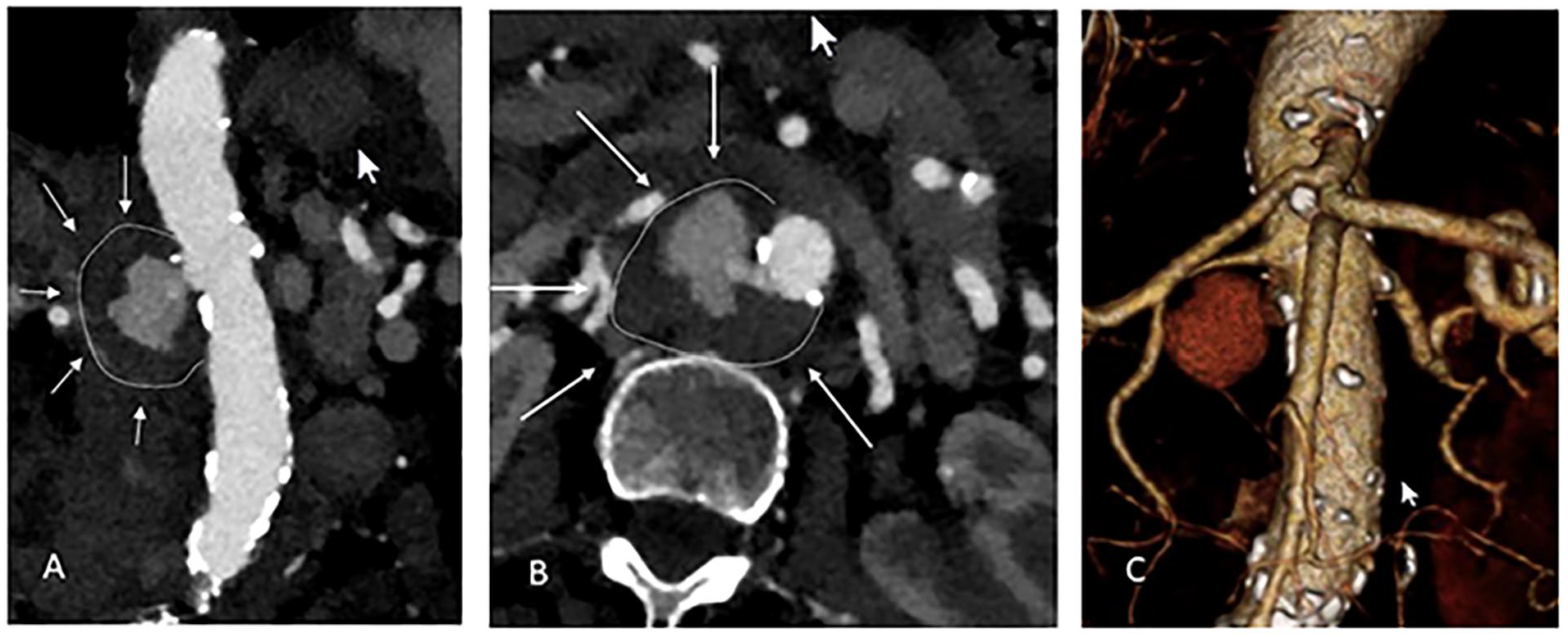
Admission computed tomography angiography (CTA) scan with a ruptured pararenal aortic aneurysm: (A) Coronal axis, (B) Axial axis, and (C) 3-dimensional reconstruction.
Given its ruptured nature, the expected infected aortic situs with vulnerable and damaged tissue in the reno-visceral segment, and serious prior comorbidities, the patient was considered to be high-risk for open surgical repair and an endovascular approach was chosen as the treatment of choice.
A dedicated 3D workstation (TeraRecon. San Mateo, Calif) was used for case planning. In this emergency situation, the use of T-branch (Cook Medical, Brisbane, Australia) was deemed inappropriate because of the increased risk of paraplegia associated with hemodynamic instability in the event of rupture and greater coverage of healthy aorta. 12 Therefore, we decided to perform a physician modified 4 × fenestrated endograft (Cook Medical, Brisbane, Australia). A custom-made 4 × fenestrated endograft was available that had been developed for another patient who had refused treatment (Figure 2). This endograft suited the patient’s anatomy at every level with the exception of the left renal artery fenestration. Considering the increased time required for creation of a 4 × fenestrated PMEG, the decision was made to use the available 4 × fenestrated custom-made device (CMD), creating a new left renal fenestration and performing closure of the original fenestration with a pericardium patch.
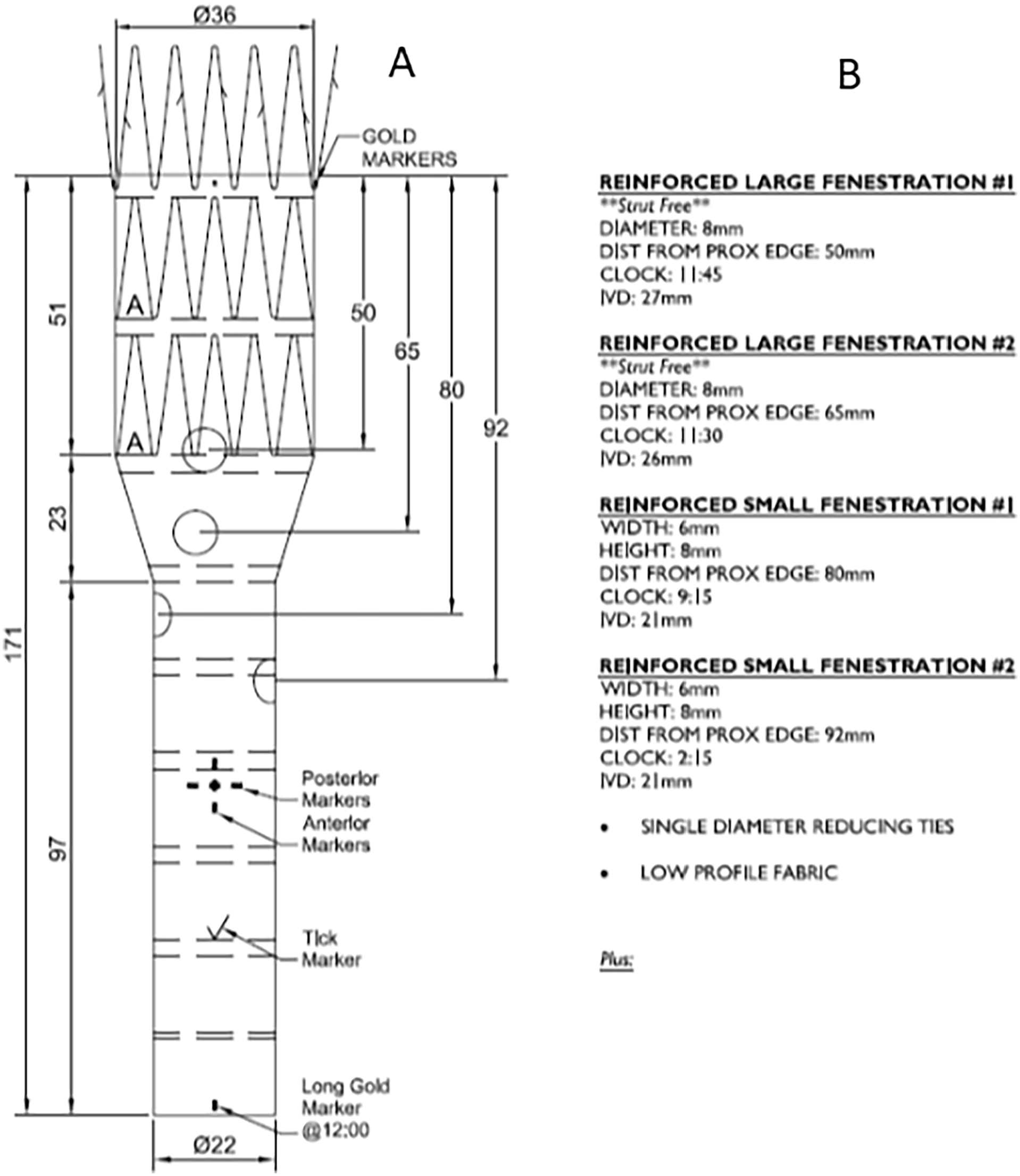
Graft plan of used custom-made device (CMD): (A) Length of graft and distances of fenestrations and (B) Size and clock position of fenestrations. IVD, In-vitro diagnostics.
To assess the fit of the graft to the patient`s anatomy, the proximal and distal landing zone diameters were calculated, which were 27 and 18 mm, respectively (Figure 3). The custom-made device presented a 30% proximal oversizing (27 mm native aorta vs. 36 mm of graft) and an 18% distal oversizing (18 mm vs. 22 mm). We considered proximal oversizing as moderate, nevertheless relevant risk of infolding remains. The clock position and distance between target vessels are shown in Figure 4. All parameters were compared to the CMD graft plan. Only the fenestration for the left renal artery would have a misalignment that would not allow catheterization of the target vessel (1 cm caudal and 1:15 hours misalignment).
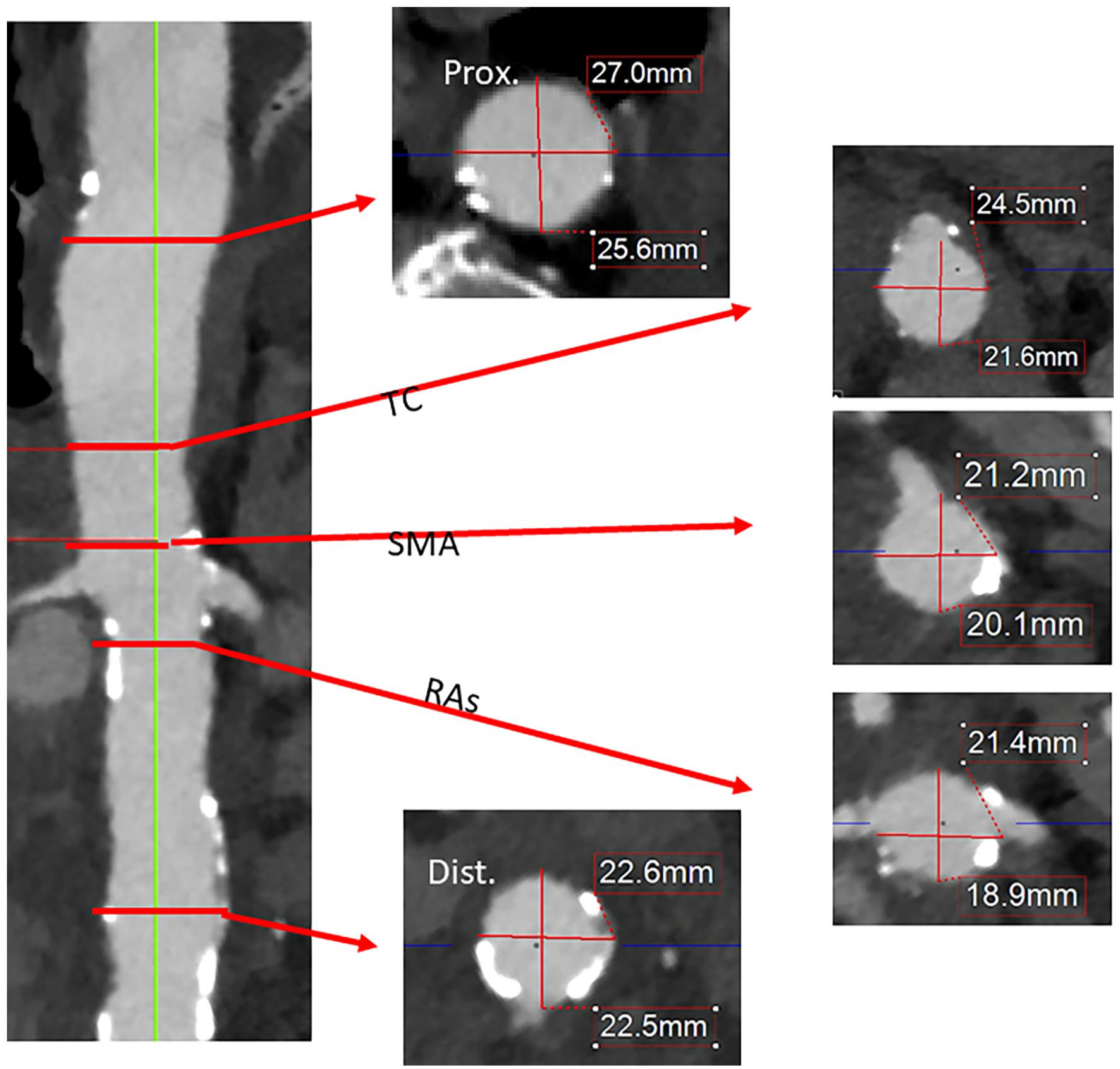
Aortic diameters according to proximal/distal landing zone and reno-visceral level. TC, Truncus coeliacus; SMA, Superior mesenteric artery; RAs, Renal arteries.
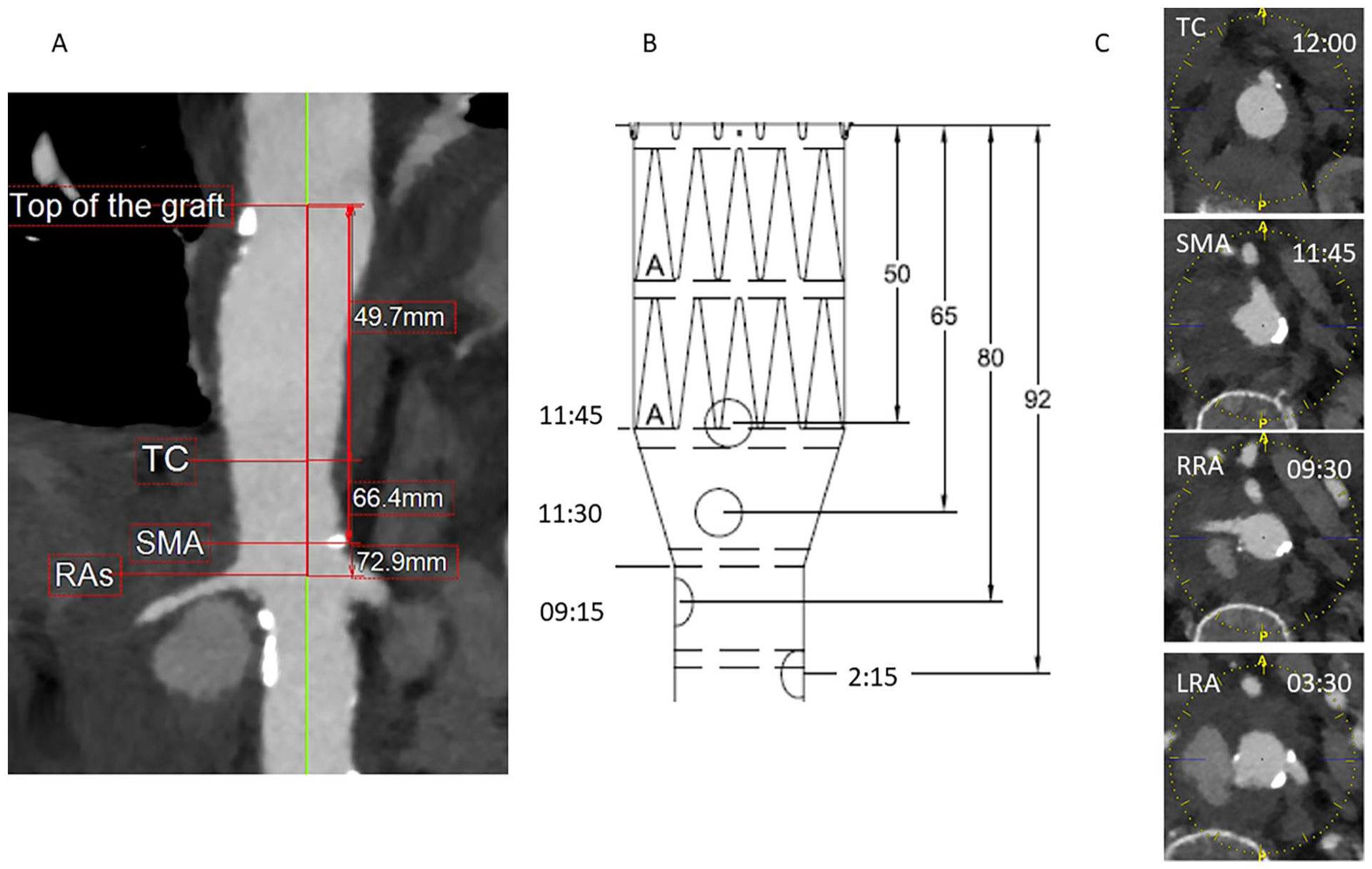
(A) Distances of target vessels to top of the graft, (B) Graft plan with distances to top of the graft and clock positions, and (C) Clock positions of target vessels in current patient. TC, Truncus coeliacus; SMA, Superior mesenteric artery; RRA, Rigth renal artery; LRA, Left renal artery.
The operation was performed in a hybrid room with a Siemens Zeego C-arm, utilizing fusion imaging. We partially released the endograft on the back-table, exposing the mainbody about 2 cm above the custom-made fenestration of the left renal artery. With electric cautery, a new fenestration of 6 × 6 mm size (according to target vessel size of 6 mm) was created 10 mm above the original fenestration at 3:30 o’clock (1 stent-row above and 1:15 hours posterior to the original fenestration). The new fenestration was reinforced with an Amplatz Goose Snare and fixated with a running suture using a 4.0 polyfilament thread. Afterwards, the original fenestration was closed with a pericardium patch (Synovis Life Technologies), using a 5.0 polyfilament running suture. Finally, the distal part of the endograft was shortened (4 cm) between the stent-rows to adjust it to the desired length and preplanned landing zone (Figure 5). Afterwards, the main-body was reloaded onto the delivery device with the aid of a Mersilene band and 2.0 polyfilament sutures. In total modification took 25 minutes.

(A) Creation of surgeon modified fenestration with electric cautery and reinforcement with Amplatz Goose Snare, (B) Closure of original fenestration with pericardial patch, and (C and D) Shortening of stentgraft with electric cautery for adjustment of distal landing zone.
Parallel to the modification, the anesthetic team prepared the patient for surgery. Subsequently, bilateral sonographic-guided percutaneous transfemoral accesses with 2 ProGlide systems (Abbott) was performed, followed by administration of 5000 IE of heparin, with a target of activated clotting time (ACT) >250 s. A double-curved Lunderquist guidewire (Cook Medical) was placed in the ascending aorta, followed by dilatation of the right femoral access with 16- and 18-Fr dilatator sheaths. The modified fenestrated endograft was introduced via the right femoral access and partially released under serial angiography. All fenestrations were aligned corrected to their respective target vessels. A 20-Fr sheath was then introduced through the left femoral access into the endograft. Target vessels were cannulated using a 6 × 55 or 7 × 55 Fr Flexor sheath (Cook Medical), a hydrophilic Terumo guidewire (Terumo Europe), and a 5 F Berenstein (Merit Medical Systems) and UF catheter (Cordis) from bottom to top. After correct cannulation, angiography was performed to confirm adequate guidewire and catheter positioning, followed by replacement of the hydrophilic guidewire with a stiff Rosen guidewire. Finally, all target vessels were secured by insertion of 6 × 45-Fr or 7 × 45-Fr sheaths, and bridging stents (7 mm BeGraft Peripheral for the CT and SMA in 8-mm fenestrations, 6 mm BeGraft Peripheral for both renal arteries in 6 × 8-mm fenestrations) were inserted into their respective target vessel.
After cannulation, the endograft was fully deployed and subsequently ballooned with a compliant Coda balloon (Cook Medical), to ensure correct endograft-aortic wall apposition before delivery of the bridging stents. Finally, the bridging stents (BeGraft Peripheral/Bentley InnoMed, Hechingen) were deployed from highest to lowest target vessel. After deployment, the bridging stents were flared using a short (20 mm) balloon with a nominal diameter 2 mm greater than the bridging stent used. Finally, the selective angiography of the reno-visceral vessels confirmed adequate placement and distal perfusion.
Final angiogram confirmed an adequate exclusion of aneurysm without the presence of endoleaks (Figure 6). All catheters and sheaths were removed and access vessels were successfully closed with Proglide percutaneous closure systems.
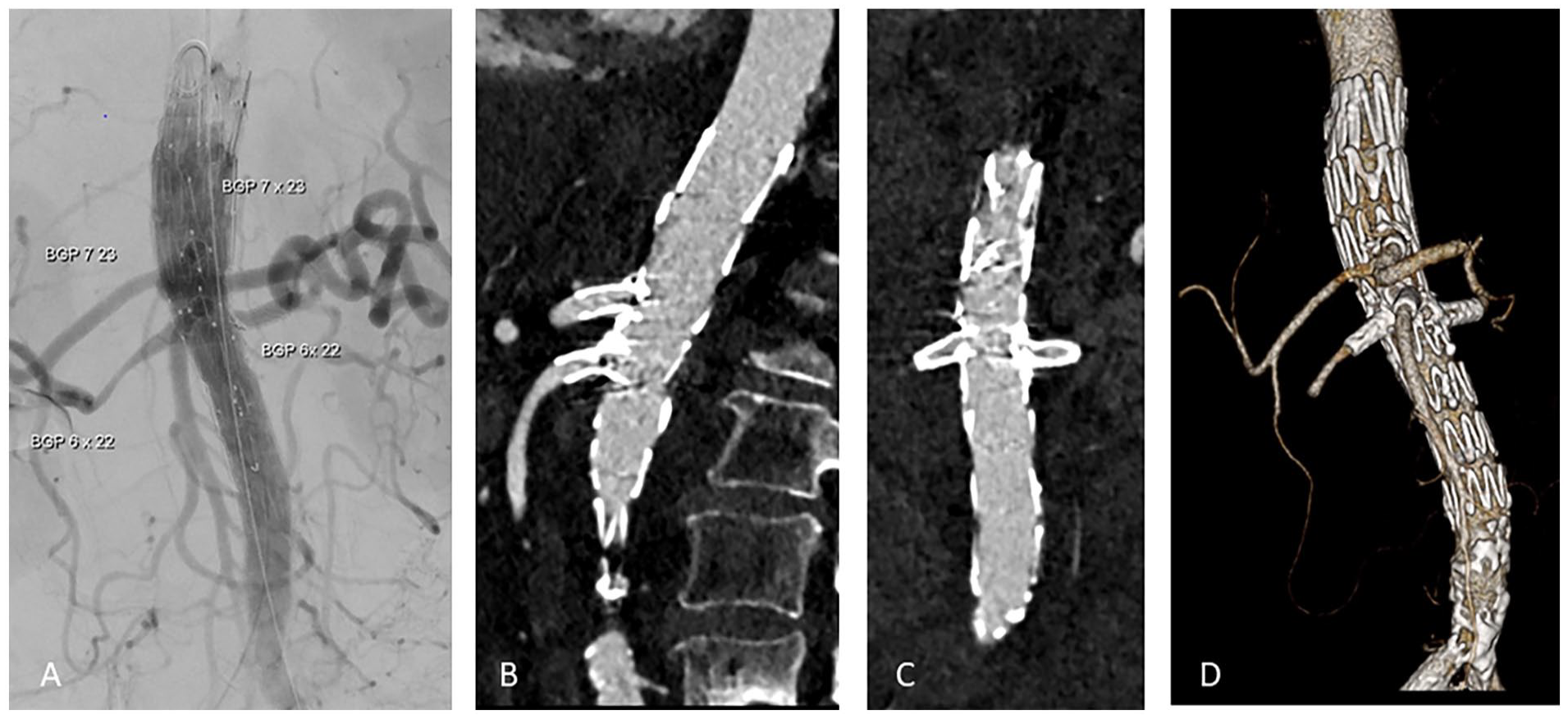
(A) Final angiogram with excluded aneurysm and perfused target vessels, (B) Sagital axis of postoperative computed tomography angiography (CTA) scan with perfused computed tomography (CT) and SMA, (C) Coronal axis with perfused renal arteries, and (D) 3-dimensional reconstruction. SMA, Superior mesenteric artery; BGP, BeGraft Peripheral.
The initial postoperative period was uneventful, and the patient was transferred from the intensive care unit (ICU) to the regular ward. A postoperative CTA was performed on the third postoperative day, revealing successfully excluded aneurysm with patent bridging stents and target vessels without evidence of any endoleaks, stenosis, or relevant kinking (Figure 6). However, the scan raised suspicion of a potential perforation in the upper gastrointestinal tract. A diagnostic laparotomy confirmed a duodenal perforation caused by an ulceration. No aorto-enteric fistula was found, and local excision and direct suture were performed. Cholecystectomy was also performed due to salmonella enteritis.
After multidisciplinary consultation with infection disease specialists intravenous treatment with Ceftriaxone (third-generation cephalosporin) was administered until discharge. To minimize the risk of infection of prosthetic material used, long-term antibiotic treatment was indicated. After good response on I.V. therapy with normal infection parameters and afebrile patient, an oral therapy with Co-Amoxicillin for additional 3 months was recommended (total antibiotic treatment period of 4.5 month). The remaining postoperative course was uneventful with negative blood cultures and normal infection parameters. The patient remained afebrile without any abdominal pain. 30 days postoperatively patient was discharged to rehabilitation clinic.
Finally, at 3-month follow-up in our outpatient clinic, the patient presented in a good general condition without fever, no loss of weight, or any signs of infection.
Discussion
Treatment of ruptured mycotic aortic aneurysm is complex. Open repair with in-situ (bonvine or autologous venous material) or extra-anatomic reconstruction is a referred treatment option. 8 Recent studies suggest that endovascular treatment can be a safe option in selected patients, especially in complex aortic aneurysm.9 –11
There are no long-term data about use of endografts in mycotic aneurysm. But in case of controlled infection with resistance-based antibiotic therapy and without evidence of aorto-enteric fistula, it might be a bailout option in selected emergency cases.
Current treatment of urgent cAAAs consists of endovascular solutions. Treatment options include BEVAR, for example, T-Branch (Cook Medical) or other off-the-shelf solutions, PMEG, ChEVAR, or ISLF.1,2,13
Concerning the use of an off-the-shelf solution, Kölbel et al 14 reported high technical success and early target vessel patency rates with the use of T-Branch (off-the-shelf multi-branched stent-graft) in cAAA and TAAA repair.
Physician-modified endografts have become a progressively used technique, especially in urgent settings due to the immediate availability and adjustability to different aortic anatomies. Manunga et al 15 reported 2021 on technical aspects and options of modifications of Cook devices in emergency and elective treatment of complex aortic aneurysm. Although PMEGs are not treatment of choice, especially in mycotic aneurysms, the technique is a valuable option in treatment of emergency cases and high-risk patients.4,5 These complex modifications should be performed by endovascular surgeons experienced in measuring and using branched and fenestrated endografts.
In the patient herein presented, with a ruptured pararenal aortic aneurysm, aneurysm was also suspected to be mycotic. Open surgical repair was considered as first treatment option. However, due to patient complex situs with infected and vulnerable aortic tissue and patient comorbities, it was considered too high risk.
The use of T-branch (Cook Medical, Brisbane, Australia) was deemed unsuitable due to an increased risk of paraplegia associated with hemodynamic instability in case of rupture and more extensive coverage of healthy aorta. 12
Due to the emergency setting, a PMEG was considered to be the most feasible solution. Given preoperative broad-spectrum antibiotic treatment, increasing long-term outcomes of endovascular repair for mycotic aneurysms, an endovascular solution was selected as the best potential treatment option. Furthermore, Salmonella spp have shown improved outcomes and lower rates of reinfection compared with other organisms.6,10
Although originally considered for a 4 × fenestrated PMEG, due to availability from other CMDs, we were able to use a CMD 4 × FEVAR from another patient, which was considered suitable to the anatomy of the patient, except from the left renal fenestration. Through the creation of an additional fenestration and pericardium patch closure of the original fenestration, modification time was significantly reduced in comparison to a 4 × fenestrated PMEG. Compared to this elegant and rapid solution, a complete 4 × fenestrated PMEG would have required a longer modification time. 5 In addition, a standard endograft has to be completely deployed, the 4 × fenestrations adequately orientated and created, sized, reinforced with circular sutured snare, followed by construction of the diameter reducing ties and reloading of the complete main body. In this case, the PMEG was only partially deployed, leaving all the constraining wires in place, and only one fenestration had to be created, alongside patch closure of the original fenestration. Both mentioned aspects reduced the risk of infection, mal-rotation during repacking, and potential graft defects leading to endoleaks. Finally, it allowed deployment of the graft from back to front with the option of repositioning of fenestrations after partial release. However, these partial modifications are not always possible, as it is necessary to have at least a small bridge between the original and new fenestration to maintain integrity of graft material and still be able to size the fenestration as planned.
For closure of original left renal fenestration, pericardium patch was used due to its good flexibility and hemostatic abilities in terms of reloading to delivery device and sealing of fenestration. Furthermore, it has proved its sealing ability and durability in several other anatomical sites, like in the carotid and femoral arteries, and even the aorta. 16 To avoid disrupture of patch after release of constraining ties size of patch was chosen 2 to 3 mm larger than fenestration. In case of relevant leckage, coiling or stenting and closure of the fenestration with a vascular plug can be performed. In general initial small leckages disappear in follow-up. The use of synthetique material like PTFE or Dacron was considered as inappropriate, due to stitch hole bleeding, stiffness in case of repacking, and missing hemostatic ability with a possible potential for type III endoleak; especially in this ruptured situation. But for sure there is no available evidence.
Conclusion
Physician-modified endografts can be considered for treatment of acute complex aneurysms in high-risk patients. The modification of a readily available custom-made 4 × fenestrated device to accelerate the process is a rapid and feasible alternative technique in emergency situations and may significantly reduce the modification time increasing the effectiveness of the repair.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NT: Travel and institutional research grants from and proctoring for Cook Medical and Bentley.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability
The data used to support the findings of this study are included within the article.
Informed Consent
The patient consented to the publication of his case details and images and written informed consent was obtained.
