Abstract
Purpose:
The cauda equina syndrome (CES) is a rare condition affecting less than 1 in 100,000 patients annually. Diagnosing CES is challenging because of its rare incidence, potentially subtle presentation, and various underlying etiologies. Vascular causes, such as inferior vena cava (IVC) thrombosis, are uncommon but should be considered, since timely recognition and treatment of deep vein thrombosis (DVT) as a cause of CES can avoid irreversible neurological damage.
Case report:
A 30-year-old male presented with partial CES caused by nerve root compression due to venous congestion from an extensive iliocaval DVT. He completely recovered after thrombolysis and stenting of the IVC. His iliocaval tract remained patent until the last date of follow-up at 1 year without signs of post-thrombotic syndrome. Broad molecular, infectious, and hematological laboratory tests did not reveal any underlying disease for the thrombotic event, particularly no hereditary or acquired thrombophilia.
Conclusion:
Timely recognition of venous thrombosis as a cause of CES is essential. This is the first case report of CES caused by an extensive iliocaval DVT successfully treated with thrombolysis and venous stenting with good resolution of DVT and CES.
Clinical Impact
This case-report describes a patient with cauda equina syndrome resulting from an extensive iliocaval deep vein thrombosis due to an underlying stenosis of the inferior vena cava. Thrombolysis and venous stenting succesfully restored venous patency and thereby relieved symptoms and signs of cauda equina syndrome, in addition to (long-term) therapeutic dose anticoagulation. It is important to timely recognize deep vein thrombosis as a cause of cauda equina syndrome and to consider endovenous treatment in a specialized center.
Keywords
Introduction
The cauda equina syndrome (CES) is a rare condition affecting less than 1 in 100,000 patients annually. 1 Symptoms and signs of CES, such as sciatica, loss of urinary function, and altered saddle sensation can be challenging to recognize, because of the occasionally subtle presentation. 2 There are various etiologies of CES such as disk herniation, tumors, infection, spinal stenosis, and inflammatory disease. 3 A timely diagnosis and adequate treatment of the underlying cause is important to avoid severe irreversible neurological damage. 4
Although uncommon, vascular causes of CES have been described and should not be overlooked. 3 Epidural vein engorgement has been revealed by magnetic resonance imaging (MRI) in patients with deep vein thrombosis (DVT) or compression of the IVC, causing a radicular syndrome by spinal cord or nerve root compression. 5 So far, three cases of CES caused by IVC thrombosis or obstruction have been reported: one IVC thrombosis was treated with anticoagulants only, 6 another IVC thrombosis with thrombectomy and thrombolysis, 7 and a case of IVC atresia was treated by open surgical nerve root decompression. 8 Treatment of CES with a venous stent has only been reported once for a patient with left common iliac vein compression. 9
Deep venous stenting is increasingly performed for occlusion or atresia of the IVC in patients with symptoms and signs of the post-thrombotic syndrome (PTS), 10 but has never been described in the context of treating CES. We describe a case of CES resulting from an extensive iliocaval DVT due to an underlying stenosis of the IVC, successfully treated with thrombolysis and stenting. Informed consent was obtained from the individual described in this case-report.
Case Report
Presentation
A 30-year-old male initially presented at a local hospital with progressive complaints of back pain and swollen legs for 4 days. He had no relevant medical history besides a pyloromyotomy and orchidopexy as a child. He was able to walk, but his legs felt heavy and numb. Defecation was absent for 2 days and urinary miction was difficult, but still possible. He had no cardiac or respiratory complaints. His body mass index was 26.3 kg/m2, he was a current smoker for 10 years, and did not use any medication.
Examination
Both legs of the patient were red and swollen, with intact motor functions. He had an altered saddle sensation with numbness around the anus, a lowered sphincter tension, and a lowered anal reflex. Plantar reflexes of the feet were indifferent on both sides. The femoral, popliteal, tibial posterior, and dorsalis pedis pulses were all palpable bilaterally. His clinical presentation was suggestive for an incomplete CES. 11
Computed tomography angiography (CTa) showed an extensive thrombus in the IVC and left and right common and external iliac veins, as shown in Figure 1. Collaterals at the level of the renal veins were indicative of a chronic stenosis of the IVC. Coincidental findings were a mild splenomegaly and a small abnormality around the pyelum of the right kidney. Direct nerve root compression due to venous congestion was revealed on MRI with decreased venous outflow of the nerve roots itself, shown in Figure 2. Cerebrospinal fluid was missing around the nerve roots of L4-L5, possibly because of epidural lipomatosis combined with mild bulging of the lumbar disk and congestion of the venous plexus leading to enlarged epidural veins.
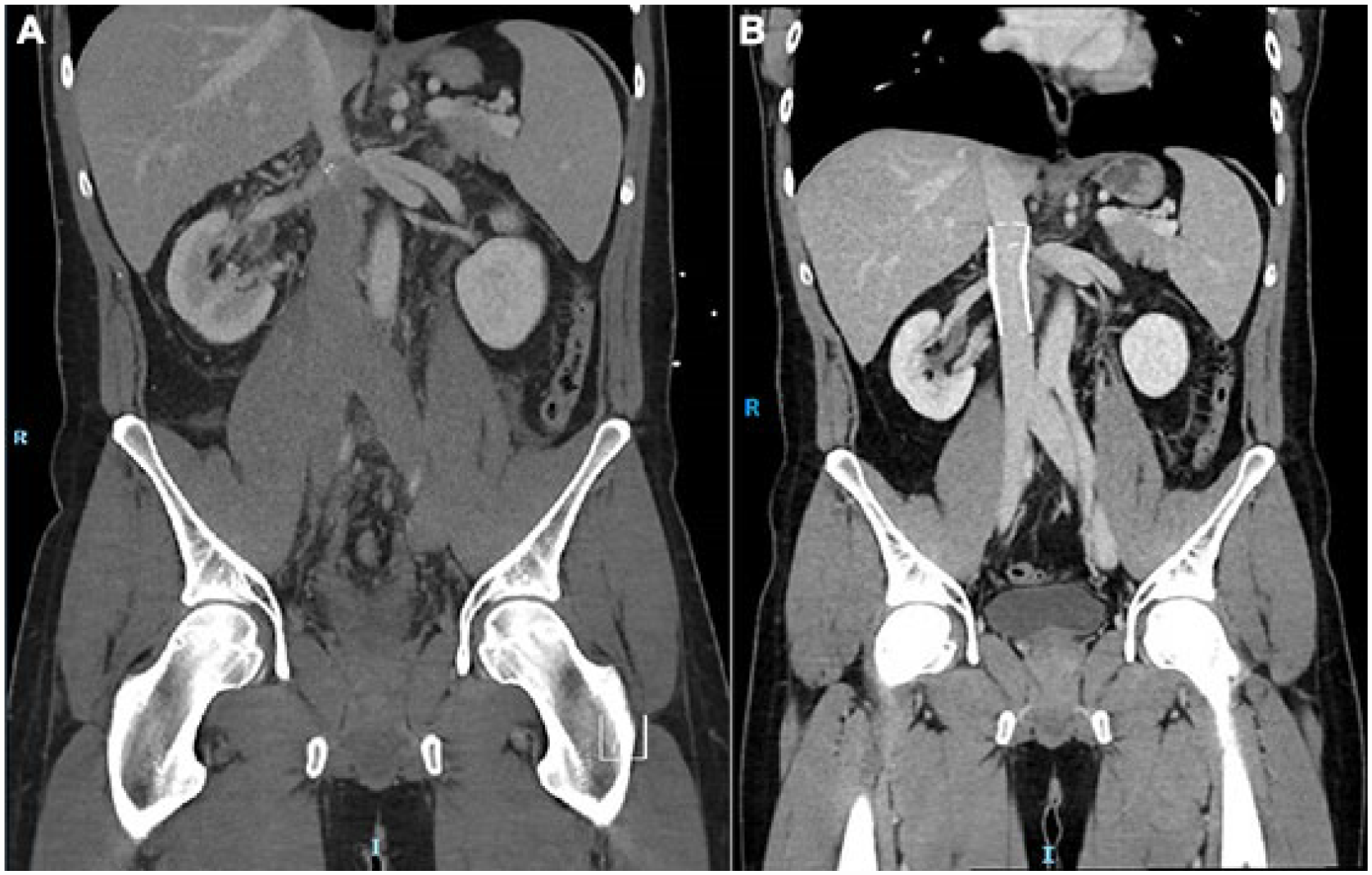
Computed tomography angiography showing extensive iliocaval thrombosis at the emergency (A), and patent inferior vena cava 3 months after deep venous stenting (B).
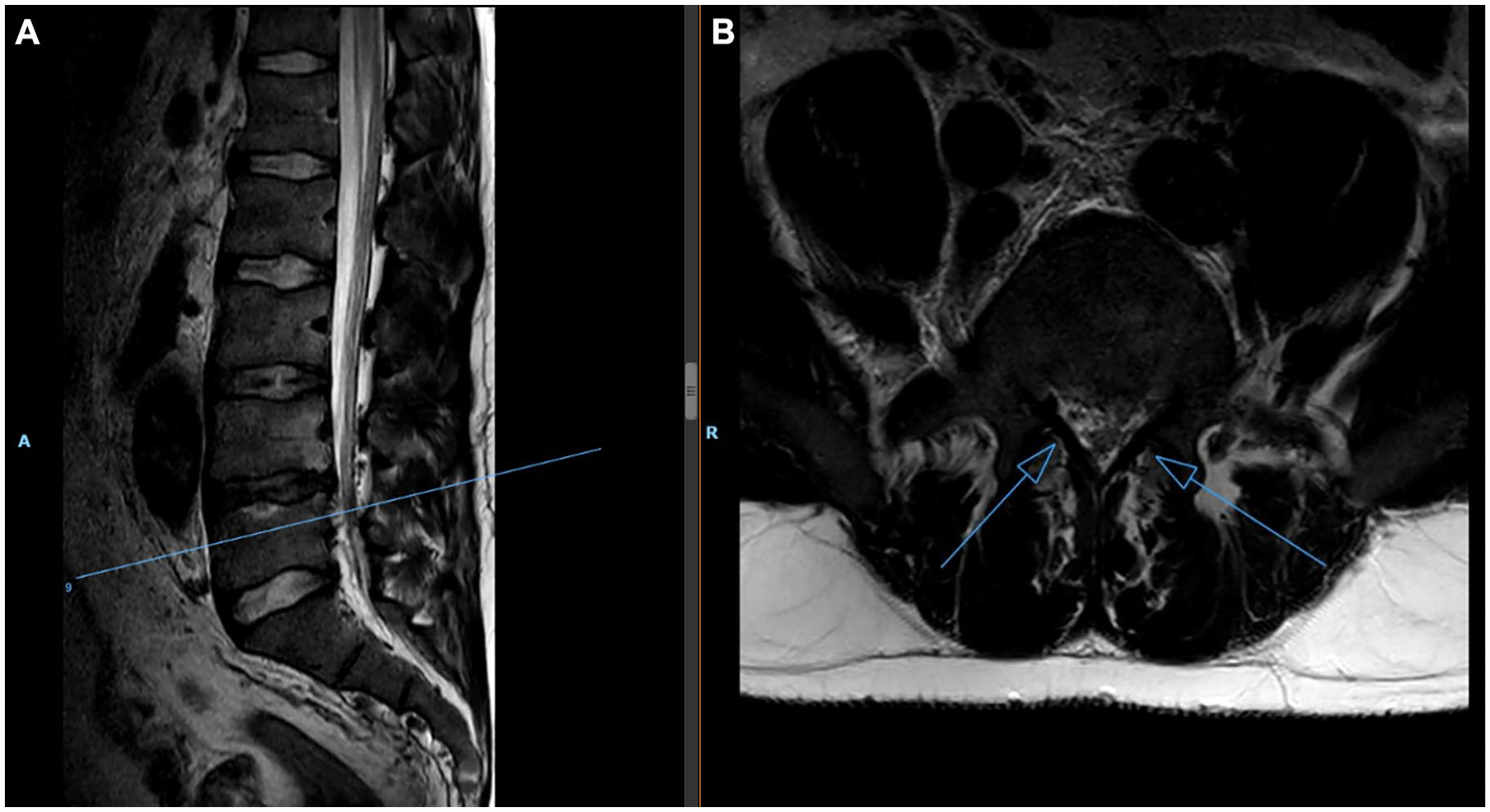
Magnetic resonance imaging during presentation at the emergency room. Sagittal imaging of the spine shown on the image (A). Venous congestion at the level of L4-L5 indicated by open arrow on axial imaging (B).
Medical specialists of the department of vascular surgery, neurology, neurosurgery, hematology, and (interventional) radiology were involved. Based on the clinical findings and findings on CTa and MRI, the most likely cause of CES was the iliocaval DVT. The patient started therapeutic anticoagulation with intravenous unfractionated heparin and was referred to our tertiary hospital for thrombolysis and potentially venous stenting of the IVC.
Treatment
Open surgical decompression was discussed with the neurosurgeon but was not indicated because of the partial manifestation of CES and deemed also too high risk with the concomitant use of anticoagulation. The medical team decided to treat the patient with an endovenous approach. As per protocol both popliteal veins were punctured for venous access with placement of two 10Fr sheaths. The popliteal, femoral, and common femoral veins were open on venogram. Large amounts of blood clots were removed by thrombosuction from the IVC and both iliac veins with improved venous blood flow. Thrombolysis using alteplase was administered in a split dose through two multisidehole catheters (0.5 mg per hour per catheter) with a split dose of heparin per sheath (5000 IU per 24 hours per sheath). Venous stenting was obviously indicated because of an IVC-stenosis on CTa, however, as the patient was not prepared to undergo sedation and there was no anesthesiologist available, stenting was postponed until the next day.
Clinical symptoms remained similar the next morning. Patency of the femoral and iliac veins was improved on a control-venogram, but the mid IVC remained narrowed on venography, shown in Figure 3, which was also visible with on intravascular ultrasound (IVUS). The anesthesiologist was consulted for peri-procedural sedation. The mid-caval stenosis was stented with a 24 mm x 60 mm sinus-XL stent and extended cranially with a second 24 mm x 60 mm sinus-XL stent, pre- and post-dilated with a 24 mm transluminal angioplasty balloon. Flow substantially improved afterwards. Because of cardiac arrhythmias during the procedure without wire manipulation in the heart, a CTa was performed immediately which excluded pulmonary emboli. The stent was adequately deployed with minimal thrombus proximal to the stent and in the left common iliac vein without limited flow.
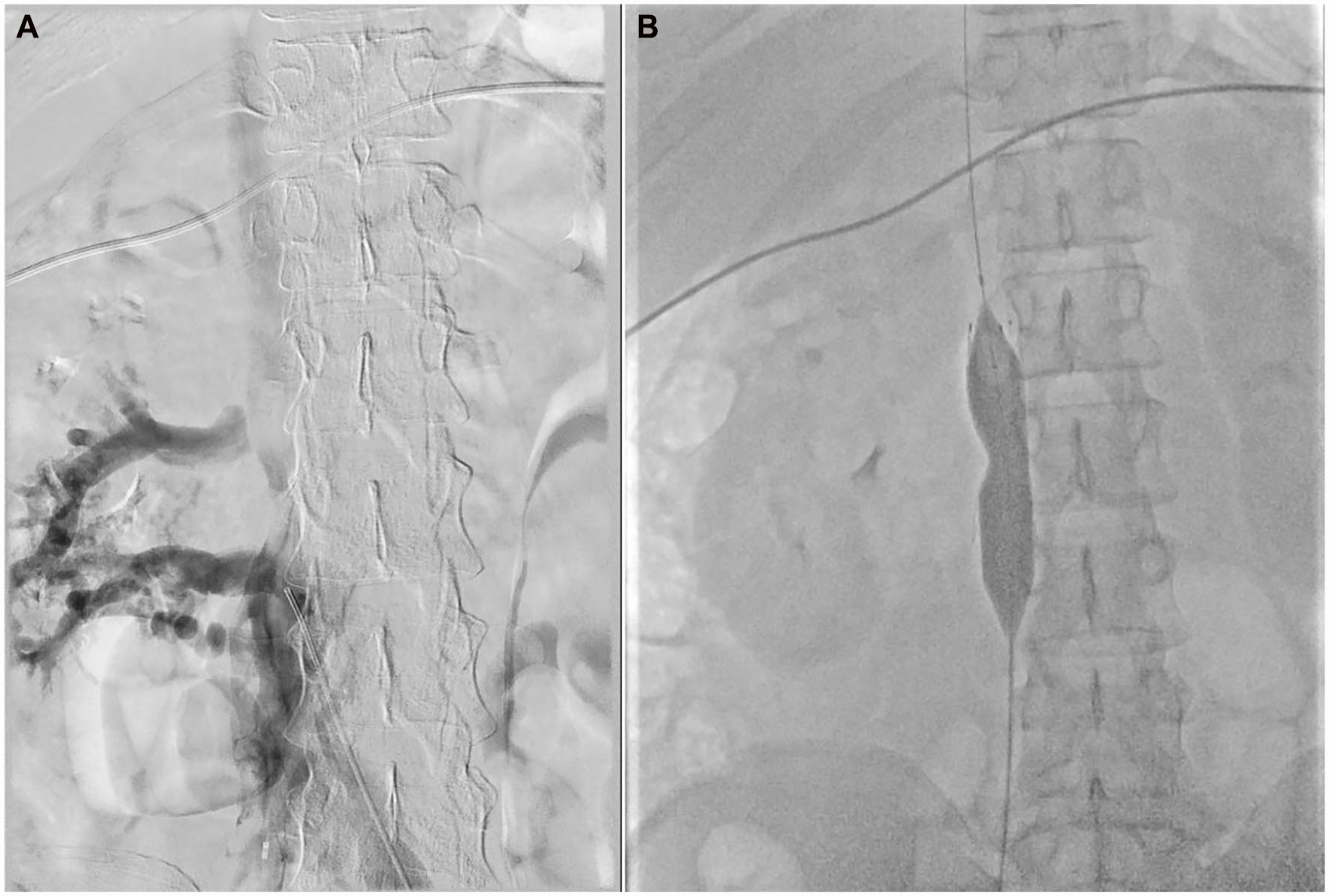
Occlusion of the inferior vena cava on venography (A) and stenosis of the inferior vena cava during percutaneous transluminal angiography and venous stenting (B).
Follow-Up
Only saddle sensation slightly improved after thrombectomy (day before venous stenting). All symptoms and signs related to CES completely resolved 1 day after venous stenting. The urinary catheter was removed, and neurological findings normalized. The patient was able to mobilize without pain. Stent patency was confirmed with CTa, shown in Figure 1. The patient was discharged after 4 days without signs or symptoms of CES. He was treated with therapeutic dose low-molecular-weight heparin based on therapeutic anti-Xa levels and was switched to a vitamin-K-antagonist at our outpatient clinic after 2 weeks (INR range 2.0–3.0). The patient was followed up after 6 weeks, 3 months, 6 months, and 1 year. Venous stents and iliofemoral tracts were checked with CTa and duplex ultrasound and have remained patent so far. Symptoms and signs of CES completely resolved until the last date of follow-up and he had no signs of PTS (Villalta score of 2) at the last visit. 12 He even noticed an improved overall fitness level compared with before the thrombotic event, probably caused by opening up the chronic stenosis of the IVC. At the most recent visit, he was switched to a direct oral anticoagulant (apixaban 5 mg, twice daily) because of very instable INR values. An indefinite duration of anticoagulant therapy was discussed with the patient and agreed upon by shared decision making.
Etiology
The hematologist was consulted to screen for thrombotic risk factors and underlying disease or thrombophilia in this young patient with severe presentation of DVT, mild splenomegaly and coincidental finding of a small abnormality around the pyelum of the right kidney. Underlying hematological malignancies, such as malignant lymphoma, were excluded. Infectious diseases were ruled out by serology for the human immunodeficiency virus and various other acute viral infections. Myeloproliferative neoplasms were excluded by molecular diagnostics (JAK2V617F, JAK2 exon 12, CALR, MPL and BCR-ABL all negative), as well as paroxysmal nocturnal hemoglobinuria by flow cytometry. Thrombophilia testing, including the factor V Leiden and prothrombin G20210A mutation, antiphospholipid antibodies including lupus anticoagulant, were all negative and antithrombin, protein C, and protein S levels were normal. No other underlying cause was found for the extensive DVT besides the mid-caval stenosis.
Discussion
To our knowledge this is the first case of CES caused by epidural vein engorgement due to an extensive iliocaval DVT provoked by a chronic stenosis of the IVC. CES completely resolved after thrombectomy, catheter-directed thrombolysis, and stenting of the stenosis in the IVC.
The exact etiology of IVC obstruction is not completely understood. Thrombotic events during early childhood, resulting from pediatric surgery, trauma, malignancy, or neonatal central lines, seem the most likely cause in general, although true congenital absence has also been described. 13 Based on the collaterals seen on CTa in our patient, it was clear that the IVC stenosis did not occur recently. None of the factors described above affected our patient, besides two minor surgical procedures during childhood. However, an IVC obstruction that originates at a young age may remain asymptomatic over time. 14
Stasis of venous blood around spinal nerves leading to venous congestion, intraneural edema, and nerve compression seem the underlying mechanism causing CES in IVC thrombosis or obstruction. 15 Our patient recovered within 1 day after endovenous intervention and was discharged after 4 days. The quick and complete resolution of the DVT and the IVC stenosis may have prevented remaining disabilities or severe progression of neurological complaints.
Conclusion
It is important to timely recognize DVT as a cause of CES and to consider endovenous treatment in a specialized center. Venous stents could restore venous patency and thereby relieve symptoms and signs of CES, in addition to (long-term) therapeutic dose anticoagulation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Prior Presentation
This case-report is accepted as a fast-track presentation (4+2 minutes Q&A), planned at the annual meeting of the European Society for Vascular Surgery in Rome on 20 September 2022.
