Abstract
Purpose:
To describe the concept of aortic elastic deformation (ED) measurement using duplex ultrasonography (DUS) as a tool for detection of high aneurysm sac pressure following endovascular aortic repair (EVAR).
Technique:
High aneurysm sac pressure, with or without proven endoleak, will result in a less compressible aneurysm. Using the dual image function in B-mode of the DUS device and a standardized amount of applied probe pressure, ED can be measured. It is defined as the percentage of deformation of the aneurysm sac on probe pressure application. We hypothesize that less ED of the aneurysm sac can be related with high aneurysm sac pressure and possibly the presence of clinically relevant endoleak. In this note, we describe the technical details of the procedure and report on the applicability and results of ED measurements in the framework of aortic aneurysm and EVAR follow-up in a cohort of 109 patients.
Conclusion:
ED measurement is the first noninvasive pressure-based method in the quest to find a practical and reliable diagnostic tool to exclude high aneurysm sac pressure. In our patient cohort, patients with proven endoleak showed a smaller ED (less compressible), implying the presence of high aneurysm sac pressure. Further research should confirm whether ED measurement using DUS could reliably exclude endoleak after EVAR and further explore its potential for clinical application in EVAR follow-up.
Clinical Impact
For the first time, a simple, fast, and inexpensive diagnostic tool is presented in this study for detecting high sac pressure following EVAR. High sac pressure is typically caused by clinically significant endoleaks, which can have significant consequences. Currently, computed tomography scanning is the most common method used to identify and characterize endoleaks. However, measuring elastic deformation may potentially replace more invasive and expensive modalities, such as the computed tomography in the future.
Introduction
Endovascular aortic repair (EVAR) is increasingly replacing open reconstruction for both elective and emergent repair. The success of EVAR depends on the exclusion of the aneurysm sac from systemic blood pressure. The most common complication after EVAR is endoleak. 1 Device-related endoleak and late onset endoleak are significant risk factors for late aneurysm rupture as well as reintervention. 2 One of the disadvantages of EVAR is the need for regular follow-up. Currently, there are several imaging modalities for endoleak detection, of which contrast-enhanced computed tomography angiography (CTA) is most frequently used. However, CTA is not cost-effective, potentially harmful, and less accurate in classifying endoleak type. 3 Duplex ultrasonography (DUS) is being adopted as a potential replacement for CTA surveillance and is easy, fast, cost-effective, and free of radiation exposure. However, differentiation between types of endoleaks is often difficult. This differentiation is important as most of the type II endoleaks resolve spontaneously and their presence (in the absence of aneurysm sac enlargement) leaves room for a conservative follow-up. On detection of type I and III endoleak however, there is a clinically relevant increased risk of aneurysm sac rupture, which requires the need for timely intervention.
Current guidelines center on aneurysm size, for which DUS screening is well suitable. However, sac pressure analysis would better assess the risk of rupture. 4 A DUS-based method to assess sac pressure combined with diameter measurement and endoleak detection would optimize the clinical relevance of DUS as an EVAR surveillance modality. Because it can be hypothesized that high aneurysm sac pressure will result in a less compressible aneurysm, we introduced the elastic deformation (ED) formula, defined as the percentage of deformation of the aneurysm sac on pressure application, using DUS. Less ED of the aneurysm sac could be an indicator for high aneurysm sac pressure and possibly an indicator for clinically relevant endoleak. In this report, we describe the technical details, applicability, and preliminary results of ED measurements in the framework of aortic aneurysm and EVAR follow-up.
Technique
To determine the ED, the dual image mode of the ultrasound machine was used. A cross-sectional B-mode echo image of the aorta, by applying firm, slow, direct pressure on the ultrasound probe and the same image with increased probe pressure on the patient’s abdomen was generated. An example of the ED measurement is shown in a patient after EVAR without endoleak (Figure 1A) and in a patient after EVAR with proven and clinically significant endoleak (Figure 1B). To standardize the applied pressure, the distance between the skin and the upper edge of the aorta was decreased by 25% in the second image (Figure 2). To subsequently calculate the ED of the aorta, the following formula was used: ED = (the maximal diameter between the anterior and posterior wall of the aorta without extended pressure [AP1] – the diameter between the anterior and posterior wall of the aorta after extended pressure [AP2]) / AP1, expressed as percentage (%) (Figure 2). Duplex ultrasonography was routinely performed by an experienced sonographer using the Siemens ACUSON imaging system (Siemens Healthcare, Erlangen, Germany 2019).
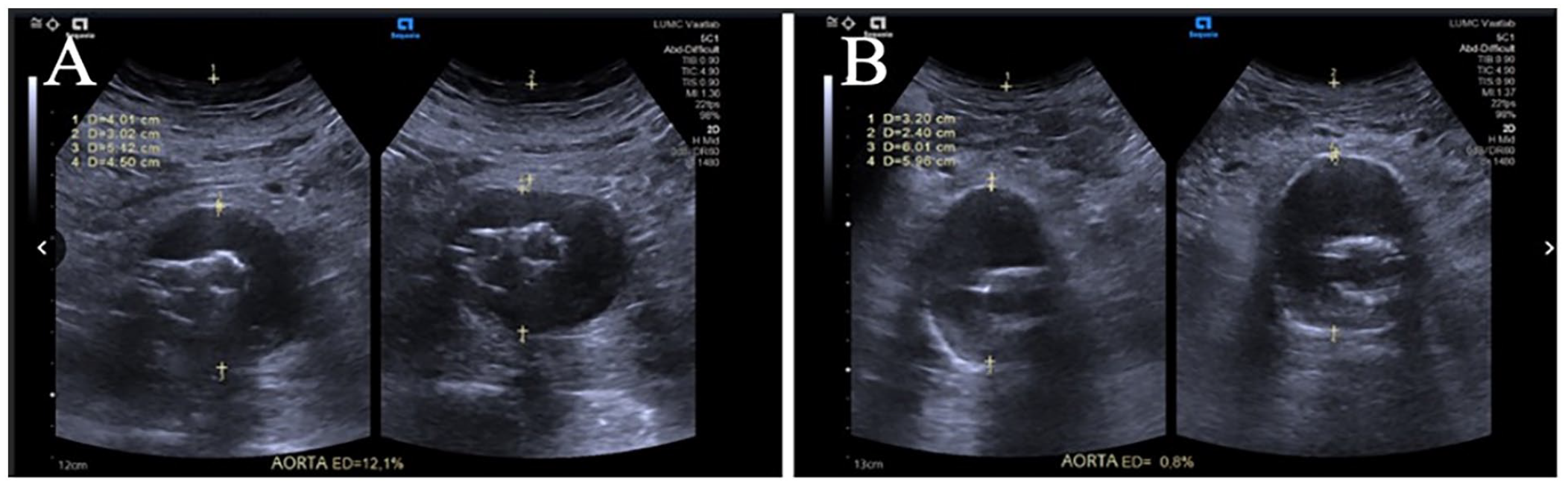
Color duplex ultrasound images of elastic deformation (ED) measurement. (A) Post endovascular aortic repair (EVAR) image in a patient after EVAR without endoleak (ED%=12), (B) Post EVAR image in a patient after EVAR with a proven and clinically significant endoleak (ED%=0.8).
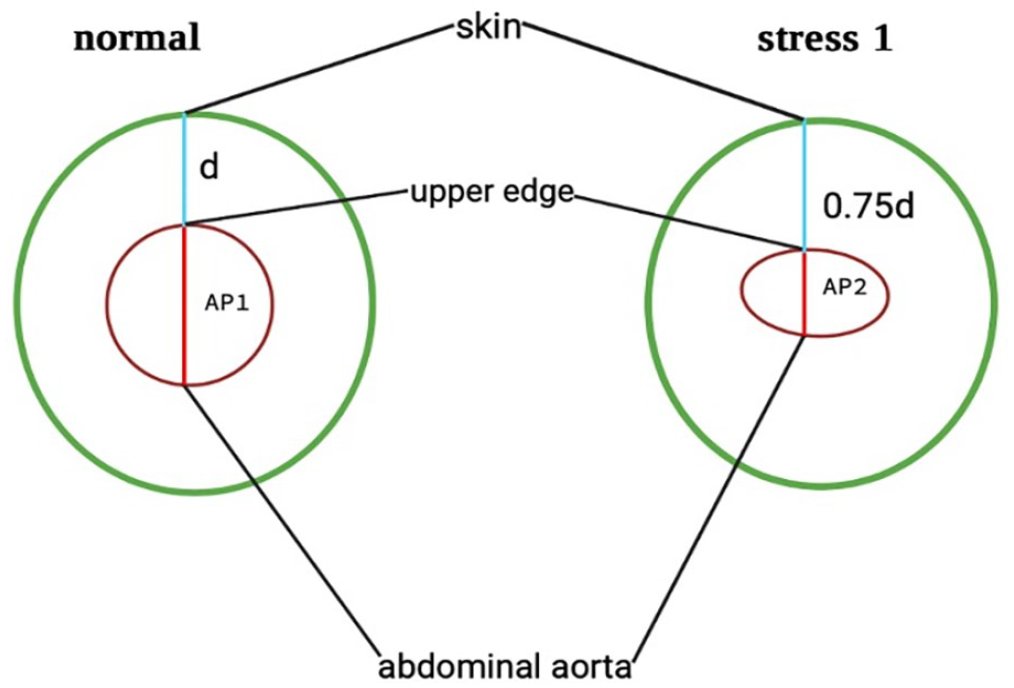
Schematic overview of elastic deformation (ED) measurement with normal distance (d) from skin to upper edge (left) and 75% of this distance (0.75d; right). AP1 = maximal diameter between the anterior and posterior wall of the aorta without extended pressure; AP2 = diameter between the anterior and posterior wall of the aorta after extended pressure.
ED measurements were performed in 109 patients between August 2020 and November 2021. The study was approved by our local institutional review board. Patients were divided into 3 groups: patients with an untreated abdominal aneurysm≥4 cm (AAA group), patients after EVAR with endoleak (leak group), and patients after EVAR without endoleak (no leak group). Endoleak was diagnosed by postoperative CTA. Table 1 presents the outcome of ED in the 3 different groups. Patients in the “no endoleak group” had a significantly increased ED compared with the “leak group” and “AAA group.” The diagnostic accuracy of endoleak exclusion was high (area under the receiver operating characteristic [ROC] curve = 0.91 [95% confidence interval = 0.85–0.97]) with a sensitivity of 0.64 and a specificity of 1.0. The optimal cutoff using ROC-curve analysis was determined at 9.5% ED. A higher ED in the “no leak group” is associated with absence of endoleak on contrast-enhanced computed tomography (Spearman ρ=–0.368; p<0.001). Based on ROC-curve analyses, a cutoff value of 9.5% was chosen, rendering a calculated sensitivity of 64% and specificity of 100% (area under the curve=0.91; p=0.03). This indicates the assurance of the absence of endoleak in patients in whom the ED (%) determined by DUS is higher than 9.5%.
Elastic Deformation in the 3 Different Groups (n=109).
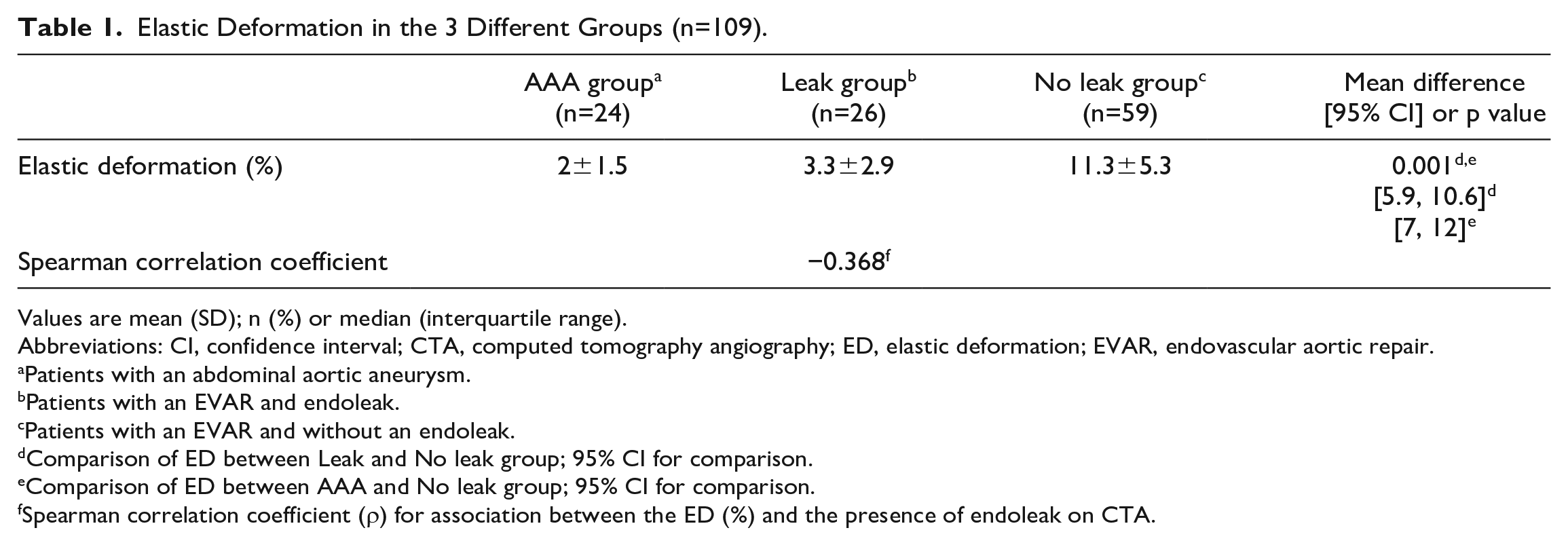
Values are mean (SD); n (%) or median (interquartile range).
Abbreviations: CI, confidence interval; CTA, computed tomography angiography; ED, elastic deformation; EVAR, endovascular aortic repair.
Patients with an abdominal aortic aneurysm.
Patients with an EVAR and endoleak.
Patients with an EVAR and without an endoleak.
Comparison of ED between Leak and No leak group; 95% CI for comparison.
Comparison of ED between AAA and No leak group; 95% CI for comparison.
Spearman correlation coefficient (ρ) for association between the ED (%) and the presence of endoleak on CTA.
Discussion
Lifelong EVAR surveillance is still needed for the detection of late complications such as stent graft migration and endoleak associated with subsequent aneurysm-related death or morbidity. Current guidelines of the American College for Vascular Surgery recommend a baseline measurement within 1 month after EVAR for the detection of endoleak or aneurysm sac enlargement using DUS or CTA. 5 Computed tomography is often the primary imaging modality for surveillance, because of its widespread availability and validation in several major trials. 6 The routine use of CTA is a subject of debate because of its radiation load and associated high costs, and also because ultrasonography screening has proven equal to CTA for measuring aneurysm size and detecting sac expansion. 7 Several studies on EVAR surveillance demonstrated promising results using DUS; compared with protocol-based CTA, it may perform superiorly in detecting endoleaks, identifying clinically relevant endoleaks, and has high sensitivity and specificity. 8 A disadvantage is that DUS is largely operator-dependent. This calls for a noninvasive technique assessing sac pressure in addition to aneurysm diameter and the occurrence of endoleak.
Our findings show that using a standardized ED calculation, high aneurysm sac pressure could easily be identified. Elastic deformation is the first noninvasive pressure-based method in the quest to find a reliable diagnostic tool to exclude or diagnose endoleaks. Using ED as a prediction tool for high aneurysm sac pressure, it appeared possible to exclude the presence of clinically relevant endoleaks. This technique has to be evaluated in a longitudinal clinical study to determine whether it could eventually replace CTA in EVAR surveillance and be an additive to normal DUS. This would contribute to the well-needed reduction of lifetime costs of EVAR, the unnecessary repeated radiation load, and less usage of potentially (nephro) toxic contrast agents. Future projects should aim to validate the associated intraobserver and interobserver variabilities of our proposed technique of ED measurement as proving low variability is needed for clinical implementation in the follow-up after EVAR. Another area of research will be the correlation of ED with endoleak type and concurrent relation with aneurysm sac growth which can aid in clinical decision-making.
A potential advantage of the currently described technique is the standardization of DUS measurements. Several DUS measurements have been proposed; from outer to outer wall, outer to inner wall, and inner to inner wall measurements, all with pros and cons. 9 A study by Hall and Duprat 10 demonstrated that aortic pressure assessment (which was assessed invasively) is feasible and may assist in the detection of clinically significant endoleak postoperatively. Our technical note describes a simple to use and standardized technique which can identify high sac pressure noninvasively. The technique has some limitations. The use of DUS is limited by the presence of excess bowel gas and habitus; by creating a standardized method for the measurement of the ED, it is however possible to overcome this. Second, DUS examination could be influenced by a high interobserver variability leading to surveillance discrepancies. This could be reduced when using a dedicated vascular sonography laboratory, as most aortic surgery centers have.
Conclusion
Simple and noninvasive assessment of ED can be used to exclude the presence of high aneurysm sac pressure after EVAR. Our findings suggest that ED of the aneurysm sac could be of additional value to rule out clinically relevant endoleaks in patients after EVAR. Further research designed to confirm these findings and to explore the association between ED and types of endoleaks in more detail, is needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
