Abstract
Introduction:
This physician-initiated study provides 5-year (i.e., long-term) treatment durability data from 3 top recruitment sites that participated in the prospective, multicenter, nonrandomized, single-arm VBX FLEX clinical study (ClinicalTrials.gov identifier: NCT02080871). It evaluates the long-term treatment durability of the GORE VIABAHN VBX Balloon Expandable Endoprosthesis (VBX Stent-Graft) in the treatment of subjects with de novo or restenotic aortoiliac lesions.
Materials and Methods:
A total of 59 subjects with 94 treated lesions were enrolled at the 3 participating sites from the original 140 intent-to-treat subjects in the VBX FLEX study. The primary durability endpoint was long-term primary patency. Secondary long-term outcomes included freedom from target lesion revascularization (TLR), freedom from target vessel revascularization (TVR), as well as resting ankle-brachial index (ABI), Rutherford category, EuroQol 5 Dimensions, and Walking Impairment status.
Results:
Fifty-nine subjects participated and twenty-eight (47.5%) were available through the end of the study at 5-year follow-up (the median follow-up time was 6.6 years due to complications resulting from COVID-19 precautions). At 3 and 5 years, the Kaplan-Meier estimates for freedom from all-cause mortality were 94.5% and 81.7%, respectively. The Kaplan-Meier estimates for primary patency at 3 and 5 years were 94.0% and 89.5% (by lesion) and 91.7% and 84.4% (by subject). Primary assisted patency at 3 and 5 years were 93.3% and 93.3%. Kaplan-Meier estimate for freedom from TLR at 5 years was 89.1%. The majority of subjects were asymptomatic (Rutherford category 0) at 3 years (29/59; 72%), and at 5-year follow-up (18/28; 64%). The 5-year mean resting ankle-brachial index was 0.95±0.18, an improvement of 0.15±0.26 from the baseline (p<0.001). Quality of life measures also showed sustained improvement through long-term follow-up.
Conclusion:
The 5-year long-term follow-up data underscore the robustness and durability of the Viabahn Balloon-Expandable Endoprosthesis for treating aortoiliac occlusive disease.
Clinical Impact
Durable improvement after endovascular treatment of iliac occlusive disease is clinically important because many of these patients are claudicants with significant life expectancy. This study is the first to evaluate the long-term outcomes in patients with iliac occlusive disease treated with the Viabahn VBX balloon-expandable endopirostheses. The study reports excellent long-term patency outcomes with prolonged clinical benefit. These durable results are likely to be an important consideration for clinicians undertaking iliac artery revascularization procedures.
Keywords
Introduction
In 2010, peripheral arterial disease (PAD) affected 202 million people worldwide. 1 Five years later, that number had increased to 236 million. 2 A third of PAD patients in the United States suffer from aortoiliac occlusive disease (AIOD), 3 comprising a considerable subset of this growing population.
Endovascular intervention, as opposed to open surgery, has become an increasingly recommended lower risk option for the treatment of patients with AIOD. 4 Growing experience with the less invasive endovascular approach and better device engineering have helped physicians treat more complicated lesions, including the more severe Trans-Atlantic Inter-Society Consensus II (TASC II) C and D level lesions. 5 Historically, TASC II C and D lesions have been recommended for open surgery, but multiple consensus and practice guidelines now generally endorse an endovascular-first approach.6–8 Balloon-expandable covered stents, in particular, have shown promising results for treating these complex lesions. 9
While endovascular repair has been clinically successful, the long-term success of balloon-expandable covered stent placements is less understood. Less clinical attention has been given to long-term durability, as evidenced by the inclusion of only one 60-month study in a recent systematic review of balloon-expandable covered stents used in treating AIOD. 6 While two other long-term studies have included balloon-expandable covered stents among bare metal and self-expanding covered stents, they did not use any GORE VIABAHN Balloon-Expandable Endoprosthesis (VBX Stent-Grafts).10,11 One of those studies provisionally concluded, however, that balloon-expandable stents were preferred for lesions with severe calcification or greater recoil. 11 The aim of this physician-initiated study is to evaluate the long-term treatment durability of the VBX Stent-Graft, a stent-graft made of 316L stainless steel rings and expanded polytetrafluoroethylene graft material coated with a thromboresistant CARMEDA BioActive Surface (CBAS) and capable of expansion to large vessel diameters. Previous studies publishing the 9-month and 3-year data have indicated the VBX Stent-Graft is both safe and effective.3,12 This study, however, is the first to report long-term follow-up data at 5 years on the VBX Stent-Graft.
Materials and Methods
Study Design
This physician-initiated study reports on a subset analysis of the prospective, multicenter, nonrandomized, single-arm VBX FLEX clinical trial (ClinicalTrials.gov identifier: NCT02080871). As a subset analysis, it assesses the long-term durability of the VBX Stent-Graft in treating patients with de novo or restenotic aortoiliac lesions. Follow-up imaging analyses, Rutherford category assessment, ankle-brachial index (ABI) measurement, the Walking Impairment Questionnaire (WIQ), the EuroQol 5 Dimensions health status measurement (EQ-5D), and adverse events were captured in a 60-month follow-up visit. Appropriate institutional review boards and ethics committees approved the study, and it was conducted in accordance with the Declaration of Helsinki. The study device and design, subject enrollment, and study procedures were detailed in both the initial pivotal trial and in the 3-year follow-up study publications.3,12
Eligibility and Enrollment
Initially, 26 sites across the United States and 1 in Auckland, New Zealand enrolled 140 intent-to-treat (ITT) subjects with 134 per protocol (PP) subjects who met the inclusion and exclusion criteria between December 2013 and August 2015. All 3-year data reported here came from the PP population, while all data from the 59 subjects included at long-term follow-up came from the ITT population. More data about the VBX FLEX study enrollment specifics, including eligibility criteria and how they were met, can be found in the 2 previous publications.3,12
Endpoints and Definitions
The primary endpoint of this long-term durability study was primary patency defined as uninterrupted patency (lack of stenosis of >50% or occlusion of the treated segment determined by vascular imaging after the procedure) with no procedures performed on the treated segment.
Primary, primary assisted (blood flow maintained with or without reintervention in the treated segments), and secondary patency rates (the presence of blood flow with or without reintervention in the treated segments) were analyzed through the 5-year long-term follow-up. Target lesion revascularization (TLR) was defined as revascularization within the treated segments by means of a percutaneous vascular intervention, surgical bypass, thrombolysis, or other invasive means. Target vessel revascularization (TVR) was defined as revascularization of the treated vessel at time of index procedure by means of a percutaneous vascular intervention, surgical bypass, thrombolysis, or other invasive means. Changes in Rutherford category, resting ABI, EQ-5D, and WIQ were assessed from the initial baseline through all follow-ups.
Major adverse events (MAEs) included device- or procedure-related events causing TLR occurring within 5 years of the index procedure and major amputation of the treated limb(s) within 5 years of the index procedure.
Follow-up
This physician-initiated, long-term follow-up study was undertaken to provide 5-year durability data for VBX Stent-Graft from 3 of the initial study’s highest enrolling recruitment sites (Auckland City Hospital, Wellmont Holston Valley Medical Center, and Greenville Hospital System). The 5-year long-term follow-up refers to the intended 60-month follow-up. COVID-19 caused significant delays in the data capture and widened the data capture window.
At the 60-month follow-up evaluation, an ABI measurement and Rutherford category assessment were taken, the WIQ and EQ-5D were completed by the subject, and data regarding any emergent treatments or MAEs—including event circumstances and outcome—were collected. It was confirmed that the subject was continuing on antiplatelet/anticoagulation therapy, and an iliac artery duplex ultrasound was taken. No core laboratory services were used for the follow-up evaluation data.
Statistical Analysis
Kaplan-Meier estimates were calculated with censoring at the latest available follow-up, except in cases where the follow-up took place after the 5-year mark. Such cases are included, however, in the associated tables. A t test was conducted to evaluate the difference in mean ABI in each study limb at long-term follow-up versus pre-procedure, with statistical significance at p<0.05. All other data were summarized using descriptive statistics (frequencies and percentages or appropriate measures of mean and spread). Statistical analysis was performed using R (version) 4.1.1 (R Foundation for Statistical Computing, Vienna, Austria) and SAS v9.4 (SAS Institute, Cary, North Carolina).
Results
Enrollment and Long-Term Follow-up
Fifty-nine subjects from the VBX FLEX study’s ITT population (n=140) were enrolled from 3 sites, and 28 agreed to participate in the 5-year long-term follow-up. Of the 31 subjects who did not participate, 12 subjects had died (of other causes), 3 were alive but could not be contacted; the remainder of the subjects elected not to continue with the study. We report and compare here on the 3-year PP population (n=134) to stay consistent with the study’s previous publications.3,12 However, 3 subjects of the 59 enrolled for 5-year follow-up did not meet the PP criteria. Therefore, all 59 subjects were a part of the ITT cohort, but not all 5-year subjects were PP subjects. Moreover, 5-year long-term follow-up refers to the intended 60-month follow-up. Due to complications with COVID-19 restrictions, the median follow-up was 6.6 years, ranging from 5.7 to 7.4 years. To help manage this extended follow-up and report statistics accurately, Kaplan-Meier estimates were calculated with a strict 5-year cutoff.
Demographics and Baseline Characteristics
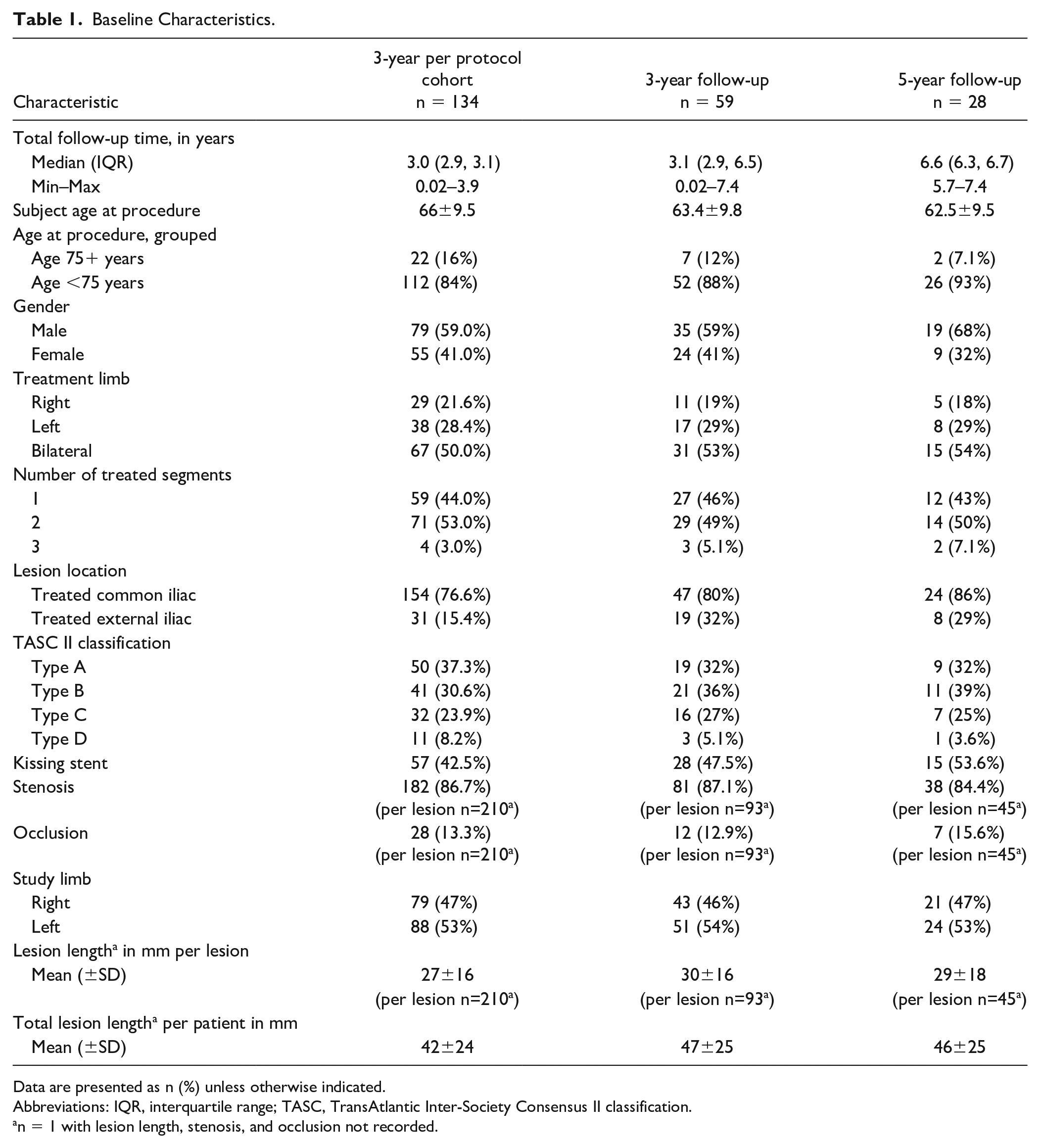
The baseline characteristics of the 28 subjects who completed 5-year follow-up were comparable to the 59 subjects enrolled and to the 134 per protocol cohort of original subjects enrolled in the VBX FLEX 3-year study (Table 1). The mean age at procedure was 63.4±9.8 years old and 62.5±9.5 years old for subjects in the 3-year and the 5-year cohorts, respectively, compared with 66±9.5 years old for the original VBX FLEX study. The proportion of males was higher in the 5-year group, compared with the 3-year groups (68% vs 59%).
Baseline Characteristics.
Data are presented as n (%) unless otherwise indicated.
Abbreviations: IQR, interquartile range; TASC, TransAtlantic Inter-Society Consensus II classification.
n = 1 with lesion length, stenosis, and occlusion not recorded.
Lesion Characteristics
The baseline lesion characteristics of participants who completed 5-year follow-up were also similar to those in the 3-year study and to the per protocol cohort (Table 1). A similar percentage of subjects had bilateral lesions (per protocol cohort, 50.0%; 3 years, 53%; 5 years, 54%). There was a higher proportion of subjects with kissing stents in the 5-year cohort compared with the per protocol cohort (53.6%–42.5%), which may have contributed to the longer lesion length observed in this group. The mean total lesion length per patient for the 3-year and 5-year cohorts was 47±25 mm and 46±25 mm, respectively, compared with the PP cohort’s 42±24 mm. In all groups, the majority of treated lesions were in the common iliac artery. The TASC II distribution followed a similar pattern across the groups. In the 5-year group, the majority were TASC II A/B lesions (71%, 20/28) with TASC II C (25%, 7/28) and TASC II D (3.6%, 1/28). Across the 59 subjects included in the study, there were 94 treated lesions and there were 46 included at 5 years (Table 1). The distribution of lesions across the right and left limbs was similar across groups. The mean lesion length was 27±16 mm for the PP cohort. In the 3-year and 5-year follow-up, it was slightly longer at 30±16 mm and 29±18 mm, respectively. At 5-year follow-up, 96.4% remained on antiplatelet therapy (46.5% dual).
Primary outcomes
Primary, primary assisted, and secondary patency
Data on patency at 5-year follow-up are presented in Table 2. Kaplan-Meier estimates for primary patency by lesion were 94.0% and 89.5% at 3 and 5 years (Figure 1A). Primary patency by subject at 3 and 5 years were 91.7% and 84.4% (Figure 1B). The Kaplan-Meier estimates for primary assisted patency by subject at 3 and 5 years were 93.3% and 93.3%; primary assisted patency by lesion at 3 and 5 years and were 96.1% and 96.1%. Figure 2 shows a Kaplan-Meier plot of primary patency by subject to 1 year, comparing the 5-year subjects (n=59) to the per protocol cohort (n=134), as well as the 78 subjects not included in the 5-year follow-up. The 1-year estimate of primary patency was 98.2% for the 5-year group, compared with 94.4% for the PP cohort.
Study Outcomes at 5-Year Follow-up.
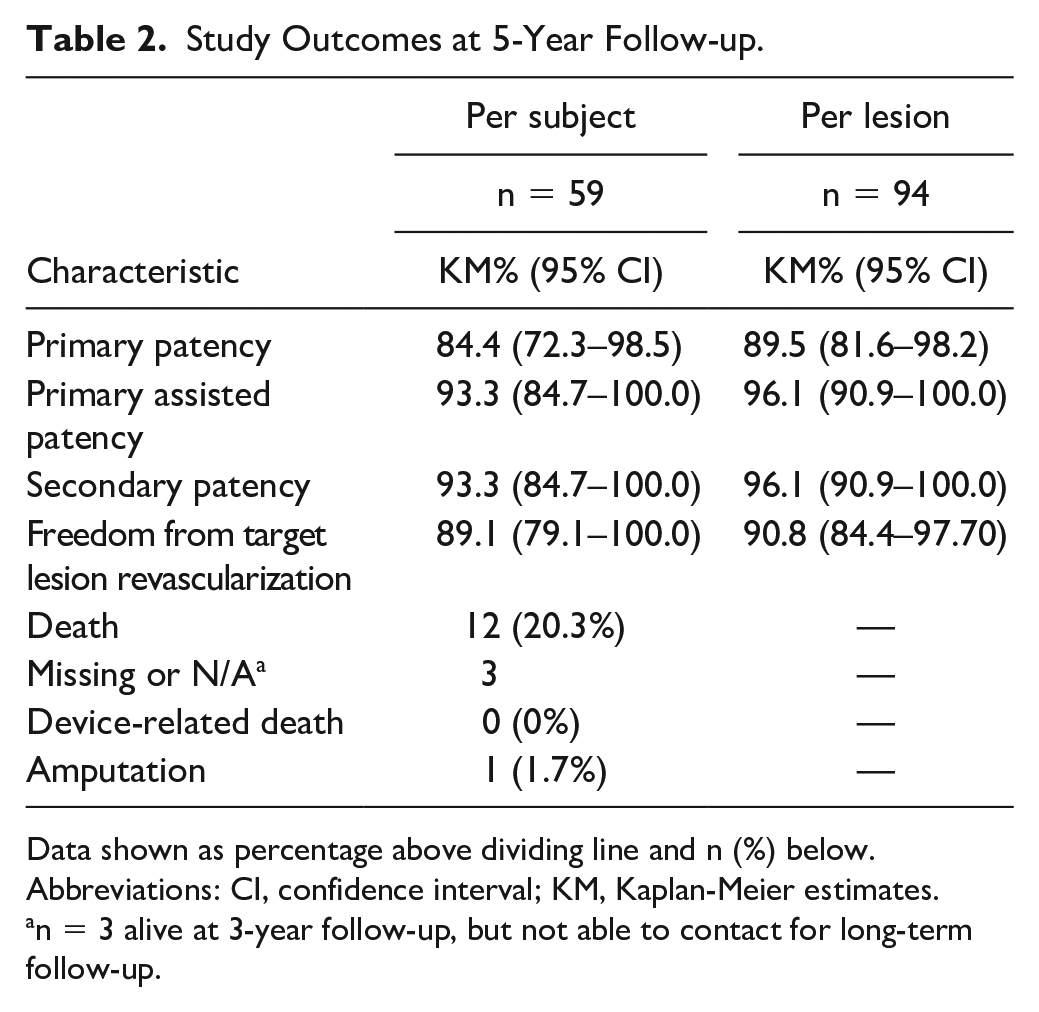
Data shown as percentage above dividing line and n (%) below.
Abbreviations: CI, confidence interval; KM, Kaplan-Meier estimates.
n = 3 alive at 3-year follow-up, but not able to contact for long-term follow-up.
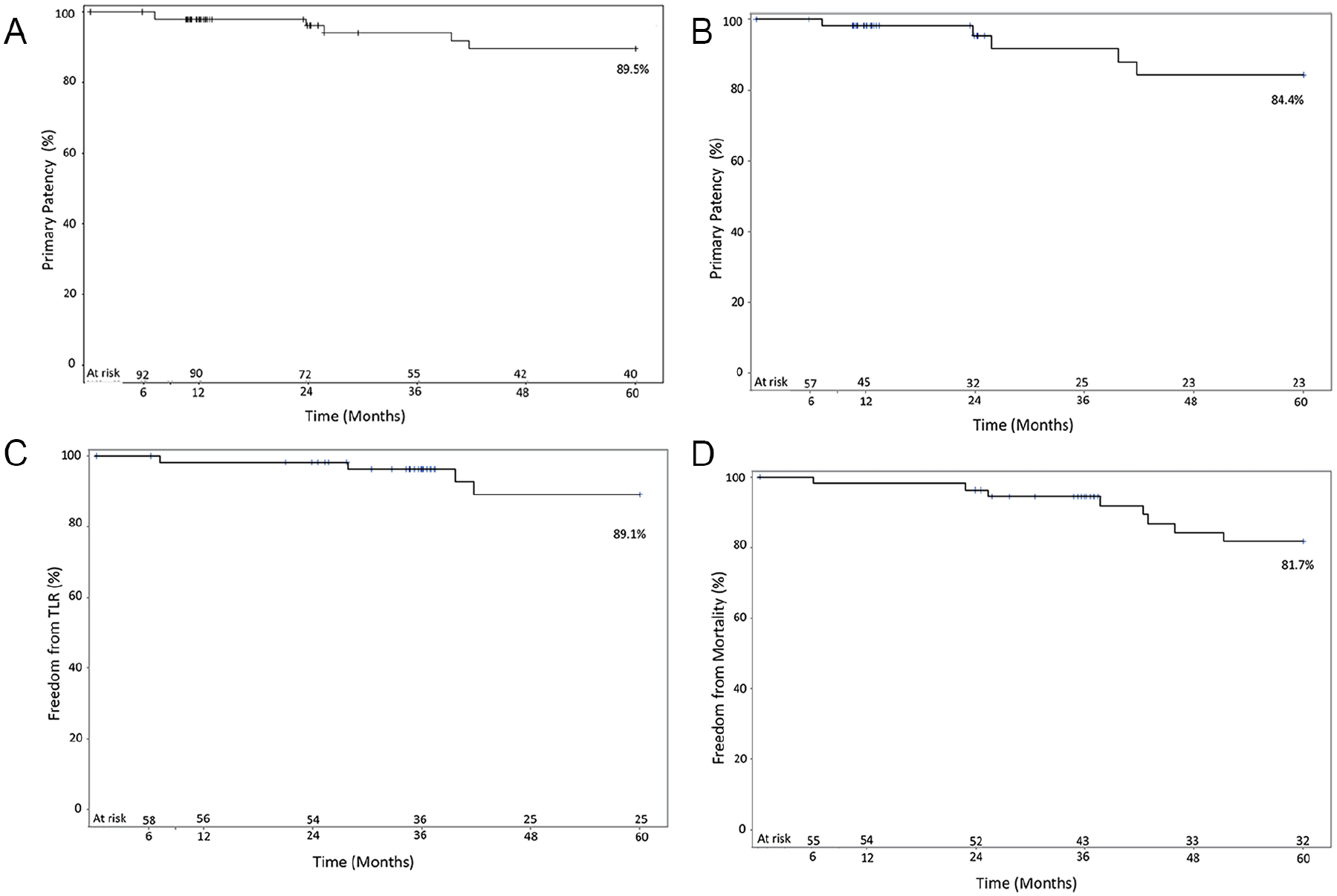
(A) Kaplan-Meier plot of primary patency, by lesion. (B) Kaplan-Meier plot of primary patency, by subject. (C) Kaplan-Meier plot of freedom from TLR, by subject. (D) Kaplan-Meier plot of freedom from all-cause mortality. TLR, target lesion revascularization.
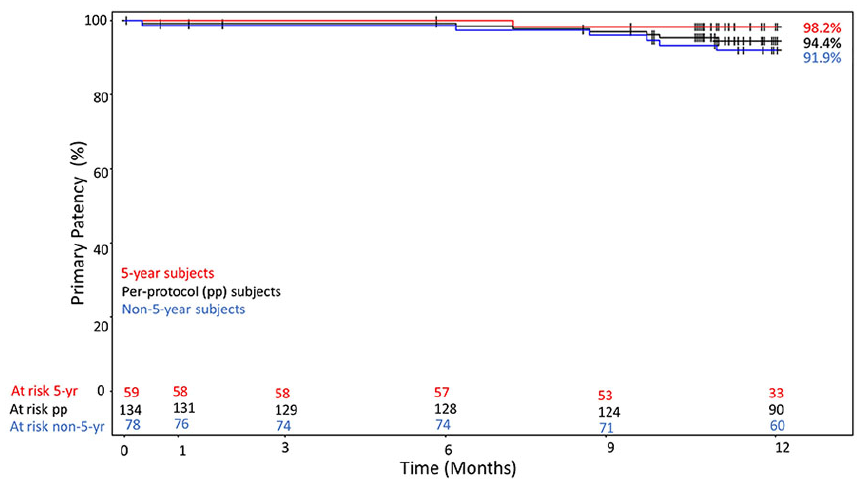
Kaplan-Meier plot of primary patency at 12 months.
The Kaplan-Meier estimates for freedom from TLR were 96.3% at 3 years and 89.1% at 5 years and are shown in Figure 1C. One of the target limb TLR events was reported before the 3-year follow-up. Kaplan-Meier estimates for freedom from TVR were 96.3% at 3 years and 89.1% at 5 years. There were 6 MAEs (target limb TLR or amputation) observed in the 5-year group. One amputation was reported at the 5-year follow-up, which did not occur in the stented segment and the subject had a patent stent. The Kaplan-Meier analysis for freedom from all-cause mortality at 5 years is shown in Figure 1D. At 3 and 5 years, the Kaplan-Meier estimates for freedom from all-cause mortality were 94.5% and 81.7%, respectively. While 12 subjects (21%) died during the study, none of the deaths appeared related to the study device (cause of death for 6 subjects was cardiovascular, 2 pneumonia, 2 cancer, 1 respiratory arrest, 1 unknown).
Change in Rutherford clinical category and ABI
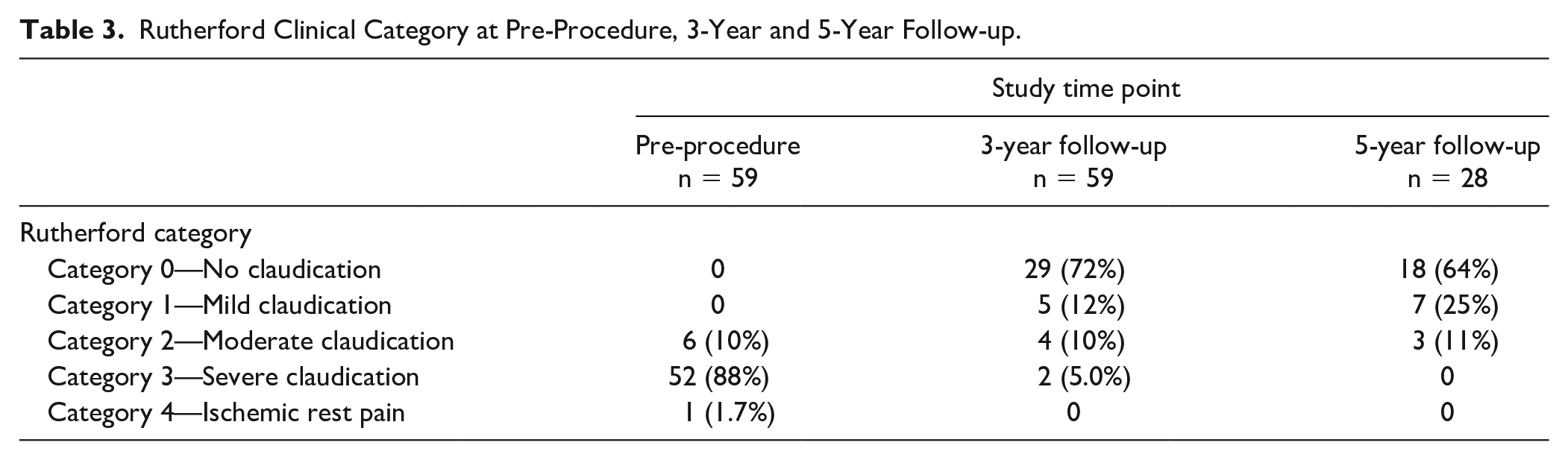
Table 3 shows the change in Rutherford category over follow-up. The majority (52/59; 88%) of subjects had severe claudication (Rutherford category 3) at baseline. At the 3-year and 5-year follow-ups, the majority of subjects had no claudication. At 3 years, 1 subject had a 4-point reduction in Rutherford category (from category 4—ischemic rest pain to category 0—no claudication; Table 4). Twenty-five subjects with severe claudication had a 3-point reduction in Rutherford category (to category 0). At 5-year follow-up, 16 subjects had a 3-point reduction in Rutherford category from category 3 (severe claudication) to category 0. At pre-procedure, the mean resting ABI was 0.82±0.23. The 5-year mean resting ABI, however, was 0.95±0.18, which was 0.15±0.26 higher than the pre-procedure baseline (p<0.001).
Rutherford Clinical Category at Pre-Procedure, 3-Year and 5-Year Follow-up.
Rutherford Clinical Category at 3- and 5-Year Follow-up.
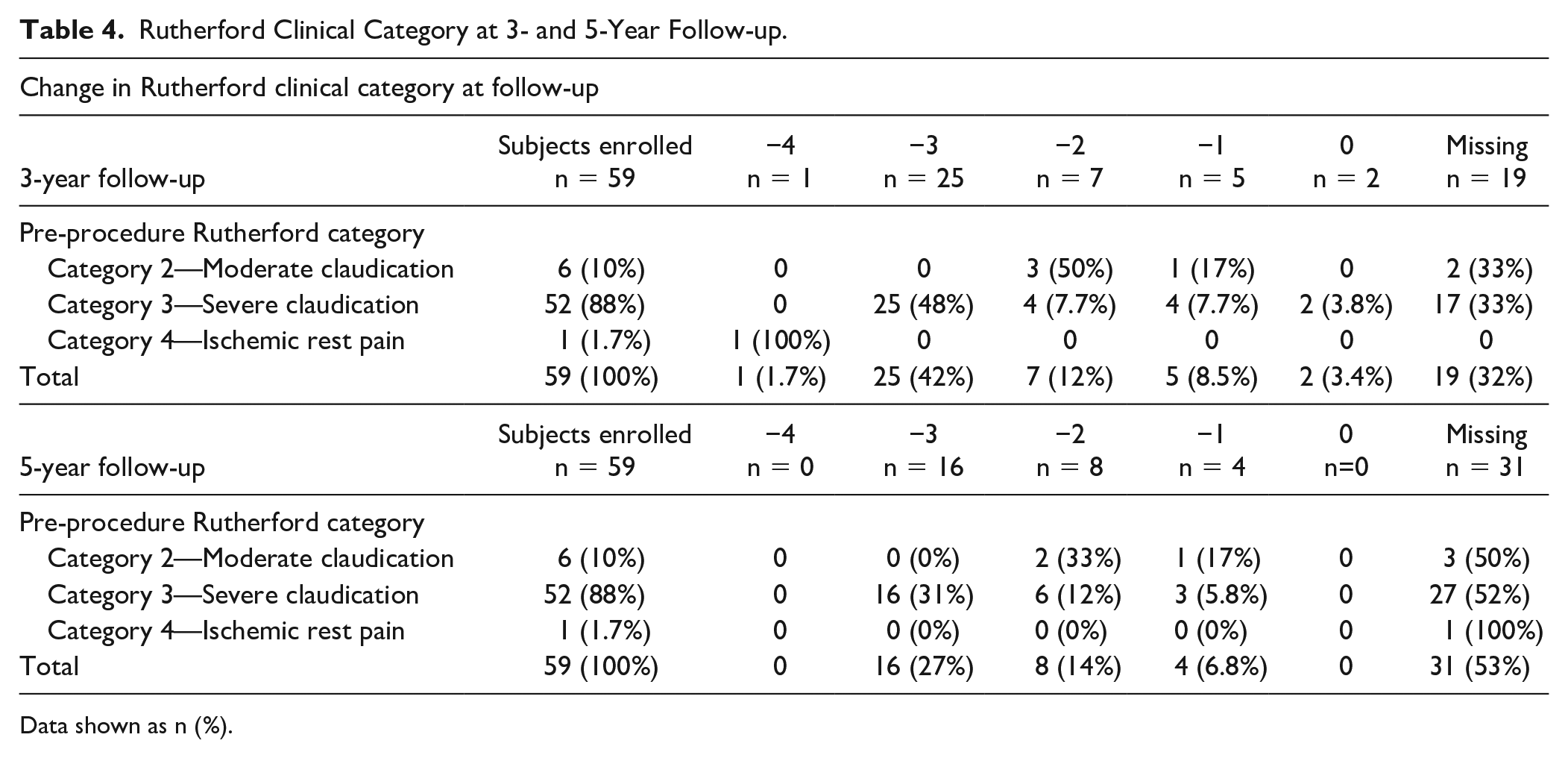
Data shown as n (%).
Change in quality of life
Quality of life saw consistent changes from baseline to 5-year follow-up, and WIQ measures for walking distance, walking speed, and stair climbing were all improved, too (Figure 3). Subjects were also surveyed using the EuroQol-5D (EQ-5D) standardized questionnaire on health-related quality of life (Table 5). Overall, the results of the EQ-5D showed that quality of life was maintained or improved for the majority of subjects.
Quality of Life Measures (EQ-5D) at 5-Year Follow-up.
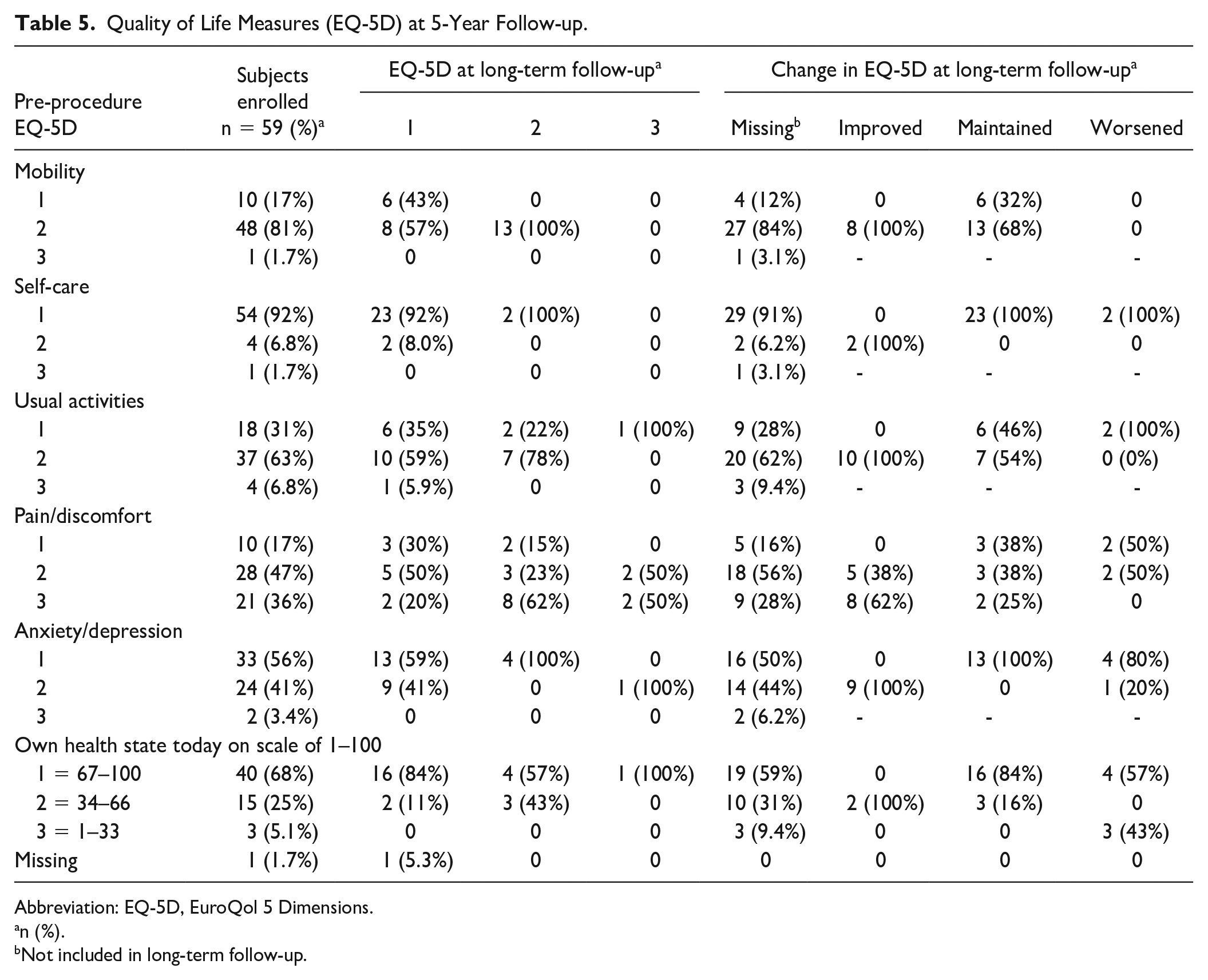
Abbreviation: EQ-5D, EuroQol 5 Dimensions.
n (%).
Not included in long-term follow-up.
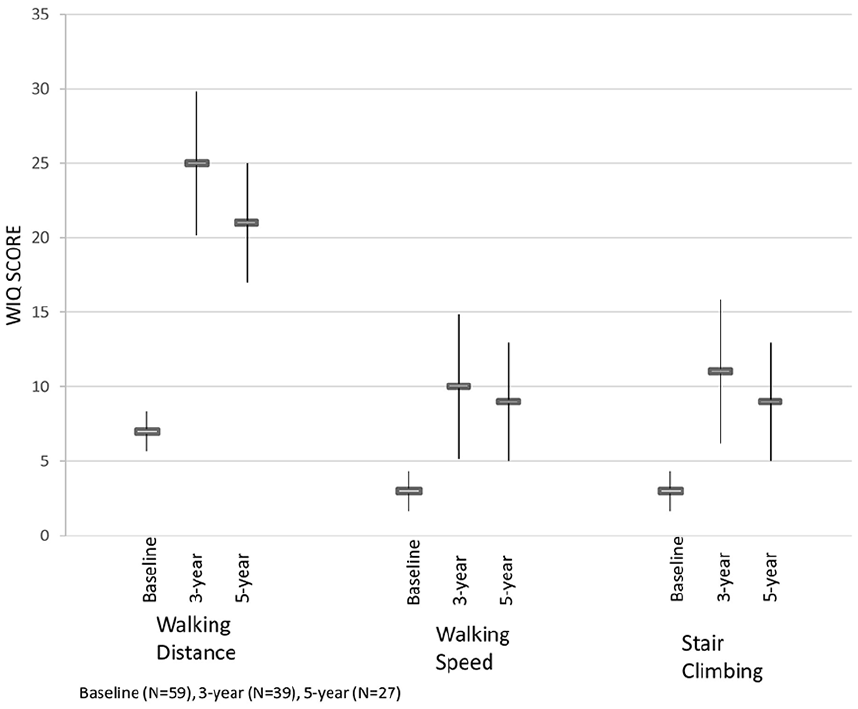
Walking Impairment Questionnaire. WIQ, Walking Impairment Questionnaire.
Discussion
This physician-initiated durability study’s Kaplan-Meier analyses estimate primary patency maintained in 89.5% of lesions and freedom from TLR maintained in 89.1% of subjects at 5 years. The prior study reported 12-month primary patency Kaplan-Meier estimates of 94.5% by lesion and it reported 3-year freedom from TLR in 91.4% of subjects and 91.2% by lesion. 3 The previously reported mean ABI was 0.93±0.19 at 3 years, whereas at 5 year the mean resting ABI was 0.95±0.18. In short, the 5-year long-term durability outcomes of the VBX Stent-Graft were similar to those recorded in the 3-year follow-up study.
This study’s long-term data further solidify the viability of endovascular interventions for effective treatment in patients with AIOD.5–8 While there is less direct comparative data between balloon-expandable versus self-expanding covered stents than covered versus bare metal stents, balloon-expandable covered stents tend to perform better in areas of calcification, whereas self-expanding covered stents demonstrate better conformability for areas with more tortuosity.3,13,14 For this reason, balloon-expandable covered stents have shown clinical efficacy for treating TASC II C and D lesions. 15 Evidence also suggests encouraging outcomes when the VBX Stent-Graft is used in a kissing stent technique at the aortoiliac bifurcation, or in tortuous external iliac arteries. 16
This study had a majority of intermittent claudicate subjects. Management of subjects is important and should be adhere to the Society for Vascular Surgery Appropriate Use Criteria when possible. 17
Four other balloon-expandable covered stents are currently available, the LifeStream (Bard, Tempe, AZ), JOSTENT (JoMed, Atlanta, GA), BeGraft (Bentley, Hechingen, Germany), and the iCAST/Advanta V12 (Atrium Medical Corp, Hudson, NH). While the iCast/Advanta V12 has long-term clinical data for durability, no data exist on the long-term viability for any of the others. 18 The majority of the 21% of patients who died were of cardiac-related causes and is of a similar rate found in other long-term follow-up iliac occlusive disease studies. 18 The Kaplan-Meier primary patency estimate in the COBEST study using iCast/Advanta V12 was 74.7% and 62.9% in bare metal stents at 5 years, slightly lower than this study’s Kaplan-Meier estimate of 89.5%. 18 These results suggest positive durability outcomes for balloon-expandable covered stents; however, key differences in study design and patient population hinder a direct comparison of results from COBEST and this study. Yet it is worth noting that the mean age at index procedure was 65 years old in the COBEST trial and 62.5 years old in this one, suggesting that many patients could end up living with these stents for years if not decades, making durability a vital performance point.
Previous studies have already shown that the VBX Stent-Graft improves clinical outcomes and that it is safe and effective.3,12 This follow-up study for the VBX FLEX trial, however, presents evidence for the device’s long-term durability as well as its safety and efficacy outcomes. No device-related MAEs were reported and quality of life measures stayed consistently high. After 5-year follow-up, 16 subjects (31%) had a 3-point reduction in Rutherford category from category 3 (severe claudication) to category 0, 18 subjects (64%) had no claudication (category 0), and 7 (25%) had mild (category 1) (see Table 4). Moreover, the high number of kissing stents (15 subjects) included in the 5-year data, as well as the inclusion of subjects with TASC II type C (25% 7/28) and D lesions (3.6% 1/28), further points toward the VBX Stent-Graft’s continuing performance outcomes for complicated occlusions.
Study Limitations
This study’s limitations, including its nonrandomized design, eligibility criteria, and diverse outcome definitions have been discussed in Bismuth et al. 12 The reduced number of sites included for this 5-year study reduced the original sample size. Analysis of subset could not be powered due to the low sample size. As the 3 sites with the highest original enrollment, however, this should introduce little selection bias to the outcomes. Fifteen subjects elected not to continue with the study for unspecified reasons, which further limited the available data. The majority of lesions were TASC II A/B (71%, 20/28) with most subjects being Rutherford category 3 pre-procedure which is a limitation of these results to real-world practice patients and the uniqueness of this long-term follow-up cohort. The baseline patient and lesion demographics are similar, however, between the PP cohort and the 5-year cohort. The 5-year cohort did have a slightly longer mean lesion length than the PP cohort, which may suggest a poorer prognosis, but 1-year patency in the 5-year cohort was slightly better than the PP cohort (Figure 2). Overall, the 5-year cohort is likely to be a reasonable representation of the entire patient group.
As with the initial study cohort, the majority of 5-year group was male (68%) and/or less than 75 years old (93%), though it was a higher percentage of males at 5 years compared with 3 years (68 to 59%). The freedom from TLR was 89.1%, yet the inclusion of more female subjects for a more even distribution could alter that percentage as female subjects are more likely to require revascularization. This consideration, which was more fully explained in the 3-year study, persisted at 5 years. 3
Conclusions
The 5-year long-term follow-up data underscore the robustness and durability of the Viabahn VBX Balloon-Expandable Endoprosthesis in the treatment of AIOD with a range of lesion complexity, including a significant percentage of TASC II C/D lesions, aortic bifurcation involvement, chronic occlusions, and calcific lesions. Long-term data are now available for 2 balloon-expandable covered stents, and while direct comparisons between these 2 device studies are limited by differences in patient population and study design, this study reported 5-year Primary Patency Kaplan-Meier estimates of 89.5% compared with 74.7% for the other device. Both of these studies point toward the safety and efficacy of balloon-expandable covered stents for endovascular treatment.
Footnotes
Acknowledgements
Áine Duffy, PhD, and Scott Dill, PhD (NAMSA, New York, NY), assisted in the preparation of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Andrew Holden is on the Scientific Advisory Board Member and is a Clinical Investigator for Gore Medical. Bruce H. Gray has consulting agreements with W.L. Gore and Associates, Surmodics, and Hart Clinical Consultants. Elleni Takele, Andrew Hill, Rahul Sakhuja, Christopher Metzger, and Alana Cavadino have no declared conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a research grant from W.L. Gore and Associates.
