Abstract
Purpose:
Unintended false-lumen thoracic endovascular aortic repair (TEVAR) is under-reported and often fatal. We present percutaneous endovascular rescue techniques for true-lumen reperfusion with strategies to address the peripheral artery perfusion needs unique to each case.
Case Report:
Two patients deteriorated 24 hours after uncomplicated and complicated acute aortic type-B dissection TEVAR treatment at a tertiary community hospital. Reevaluation of index and repeated imaging studies revealed inadvertent false-lumen TEVAR with severe visceral and peripheral ischemia. Stepwise subtraction angiography was used to confirm continuous true-lumen catheterization from femoral puncture into the thoracic aorta. Retrograde fenestration distal to the misplaced TEVAR from the true- into the false-lumen allowed for snorkeling by distal stent-extension across the dissection membrane. In one case, TEVAR stent graft extension resulted in true-lumen re-expansion and in the other case, bare-metal stent-extension for stabilization of the dissection membrane resulted in true-lumen re-expansion in addition to preservation of visceral and peripheral perfusion via the false-lumen. Despite excellent acute hemodynamic results and initial signs of recovery, both patients eventually died (multiorgan failure; cerebral hemorrhagic infarction).
Conclusion:
Correct indication and prompt recognition of potential inadvertent false-lumen stenting is critical to avoid disastrous sequelae, for example, malperfusion. Endovascular salvage is feasible for restoration of correct perfusion and must be performed in a timely manner.
Clinical Impact
Clinically apparent, complicated type-B dissection necessitates instantaneous treatment. Prerequisite for an endovascular approach is a true-lumen guidewire continuously from access to the ascending aorta. DSA after TEVAR and prompt clinical re-evaluation are to verify effective visceral and peripheral restoration of blood flow. Despite these measures, inadvertent false-lumen TEVAR may occur rarely. Immediate recognition may allow for timely true-lumen re-expansion using percutaneous endovascular salvage techniques e.g. retrograde fenestration from the true- into the false-lumen for distal snorkeling of the misplaced TEVAR across the dissection membrane. However, morbidity and mortality are very high, if erroneous stent graft placement is not identified early.
Keywords
Introduction
Complicated acute type B aortic dissection (TBAD) is a life-threatening disease typically characterized by visceral and/or peripheral ischemia and hypertension, and may lead to aortic rupture. Lower limb malperfusion may occur in up to 40% of complicated TBADs. 1 While thoracic endovascular aortic repair (TEVAR) is commonly used to treat complicated TBAD, unintended false lumen (FL) stent graft implantation is an under-reported, often fatal, yet potentially treatable complication. Here, we present percutaneous endovascular rescue techniques for true-lumen (TL) reperfusion with strategies to address the peripheral artery perfusion needs unique to each of two cases.
Case Presentations
Two consecutive male patients treated with TEVAR for TBAD (case 1, 63 years; case 2, 55 years) presented to the emergency department of a tertiary community hospital suffering from severe acute back and abdominal pain. Both patients showed uncontrollable arterial hypertension. Contrast-enhanced electrocardiogram-gated computed tomography angiography (CTA) depicted an acute aortic dissection originating from the proximal descending thoracic aorta into the common iliac arteries. Thoracoabdominal TL collapse with consecutive visceral malperfusion was only present in case 2. TEVAR was immediately performed in both patients. A single aortography of the aortic arch and descending thoracic aorta was acquired for stent graft implantation. Post-implantation angiography showed apparently patent, normal looking thoracic aortas with regular vessel diameters. However, the full extent of the persistent impairment of visceral perfusion was not immediately recognized in either case. Within 24 hours, both patients showed progressive deterioration with increasing abdominal pain.
Physical examination showed increasing tachycardia and uncontrollable arterial hypertension (systolic blood pressure up to 220 mmHg) in both patients. There were pulse deficits and differences in blood pressure between both upper and lower extremities. Ankle-brachial index was significantly reduced for both of lower extremities. Laboratory analysis revealed progressive elevation of serum lactate, lactate dehydrogenase, and creatine kinase as signs of severe ischemia.
Reevaluation of the index imaging studies and follow-up CTA confirmed erroneous stent graft implantation into the FL with complete compression of the descending thoracic aortic TL (Figure 1).
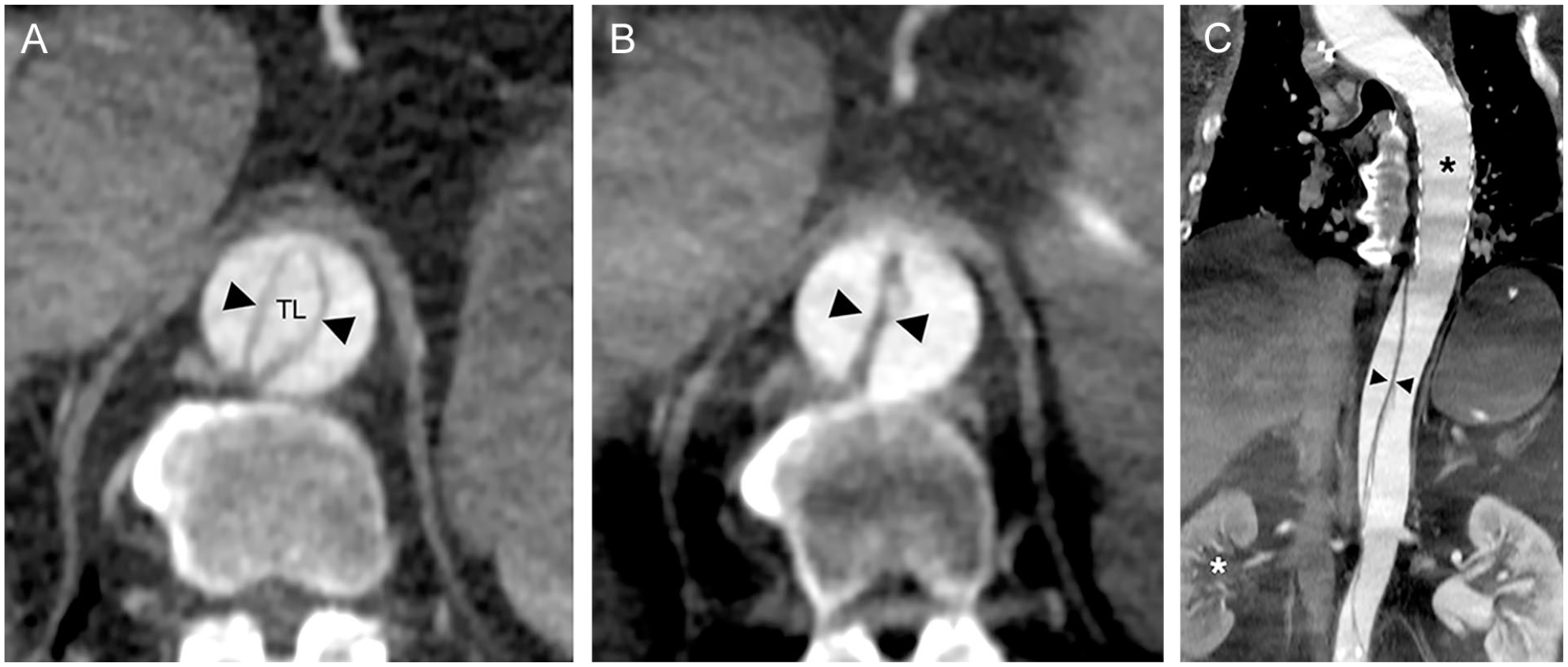
(A) Case 1. Pre index TEVAR procedure contrast-enhanced computed tomography angiography (CTA) shows dissection of the thoracoabdominal aorta. Arrowheads in the false lumen indicate the dissection membrane and the true lumen (TL).(B) Post TEVAR procedure CTA at the same level as (A) after erroneous TEVAR (in a patient without ischemia) into the FL depicts increased false lumen pressurization with subsequent slit-like complete collapse of the thoraco-abdominal TL (black arrowheads).(C) Curved multiplanar reconstruction illustrates inadvertent false lumen TEVAR (black asterisk) and subsequent visceral malperfusion apparent by low opacification of the right kidney (white asterisk). The left kidney is perfused by the pressurized large false lumen.
In both cases, the patients were transferred under general anesthesia from the referring hospital to our university medical center for further treatment. Due to the severely impaired clinical statuses, angiography for endovascular rescue was immediately initiated.
Interventional Techniques
To ensure access into the TL and not into the FL, pre- and post-index TEVAR CTA were reviewed to determine the distal extent of the dissection. Puncture of the right common femoral artery was performed using ultrasound guidance. For the procedure, a single transfemoral only access was used. Initially, a 70-cm 6F sheath and 5F pigtail catheter were stepwise advanced over a standard 0.89-mm/0.035” guidewire into the common iliac artery, infrarenal abdominal, thoracoabdominal, and thoracic descending aorta. In the absence of intravascular ultrasound (IVUS) at the time, correct TL localization along its entire length was meticulously verified at each level by digital subtraction angiography (DSA) after power injection of contrast agent (5-10 ml of non-diluted iomeprol 300 mg/ml at a flow rate of 20 ml/s, 4 images/s, field of view 42 or 32 cm), which provided a reliable and intense opacification of the corresponding vessel segment. Figures 2A and B and Supplementary Online Material video 1 illustrate this technique.
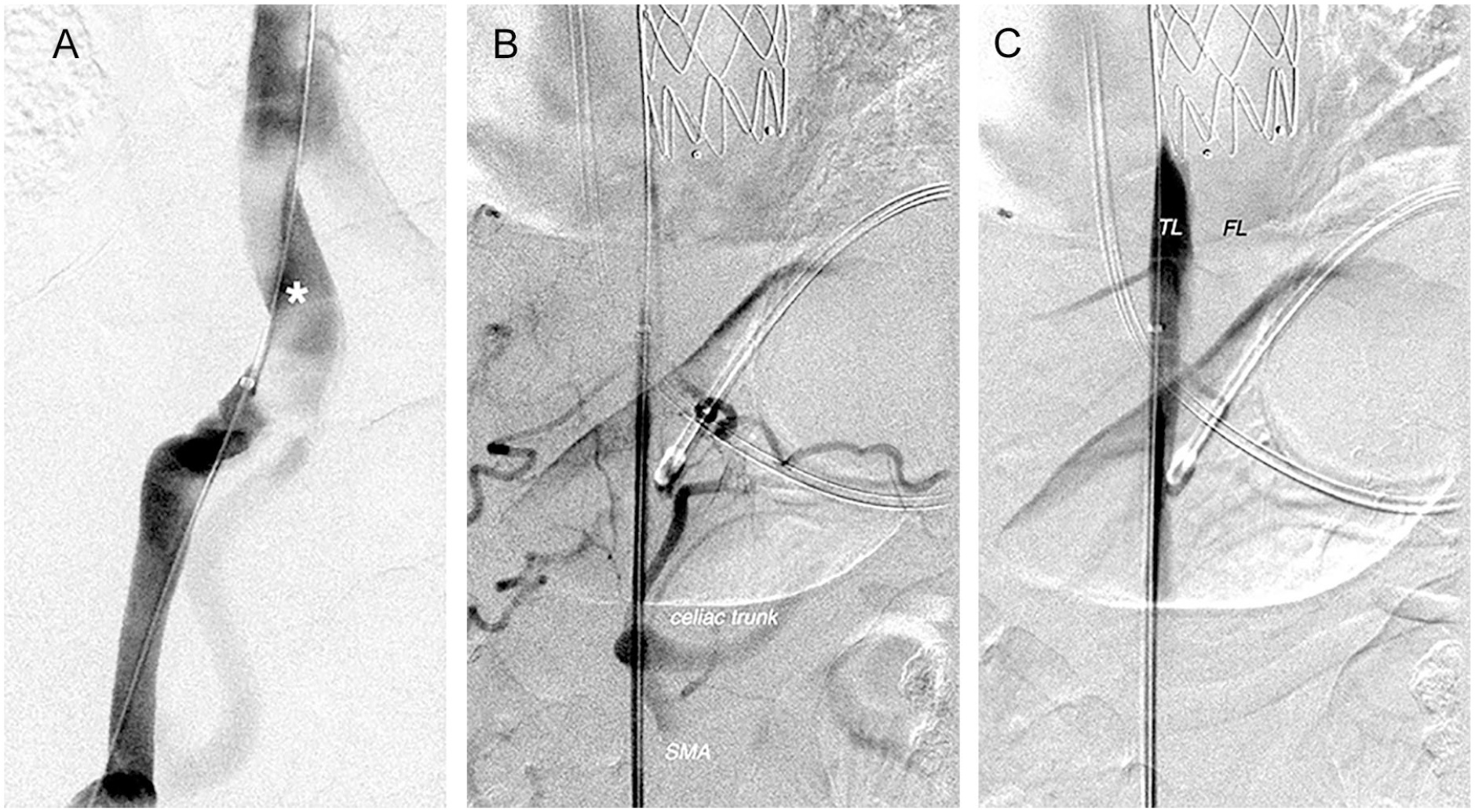
(A) Correct true lumen cannulation at the level of the common iliac artery (white asterisk) was achieved using stepwise advancement of catheter and sheath combined with repetitive digital subtraction angiography. (B) The characteristic floating viscera sign (superior mesenteric artery, SMA) is a result of aortic true lumen collapse and impaired visceral perfusion caused by a pressurized false lumen—and clearly confirms correct catheterization of the true lumen. (C) Angiography confirms complete true lumen (TL) compression as a result of erroneous stent graft implantation into the descending thoracic aorta false lumen (FL).
Key Points
Correct indication for TEVAR: Presence of complicated type B dissection, that is, organ or peripheral malperfusion.
Thorough assessment of guidewire position in the true lumen using IVUS or transesophageal echocardiography, alternatively step-by-step DSA including focused visceral angiography.
Control DSA after TEVAR to verify successful restoration of visceral and peripheral branching arteries perfusion.
Clinical re-evaluation after TEVAR, that is, presence of femoral pulses or peripheral vascular ultrasound and continuous urinary excretion assessed using a transurethral catheter.
Further wire advancement into the aortic arch was impossible due to TL compression by the FL TEVAR, as shown in Figure 2C. TL reperfusion was established 1 cm distal to the stent graft using retrograde fenestration from the TL into the FL, as shown in Figure 3A and Supplementary Online Material video 2.
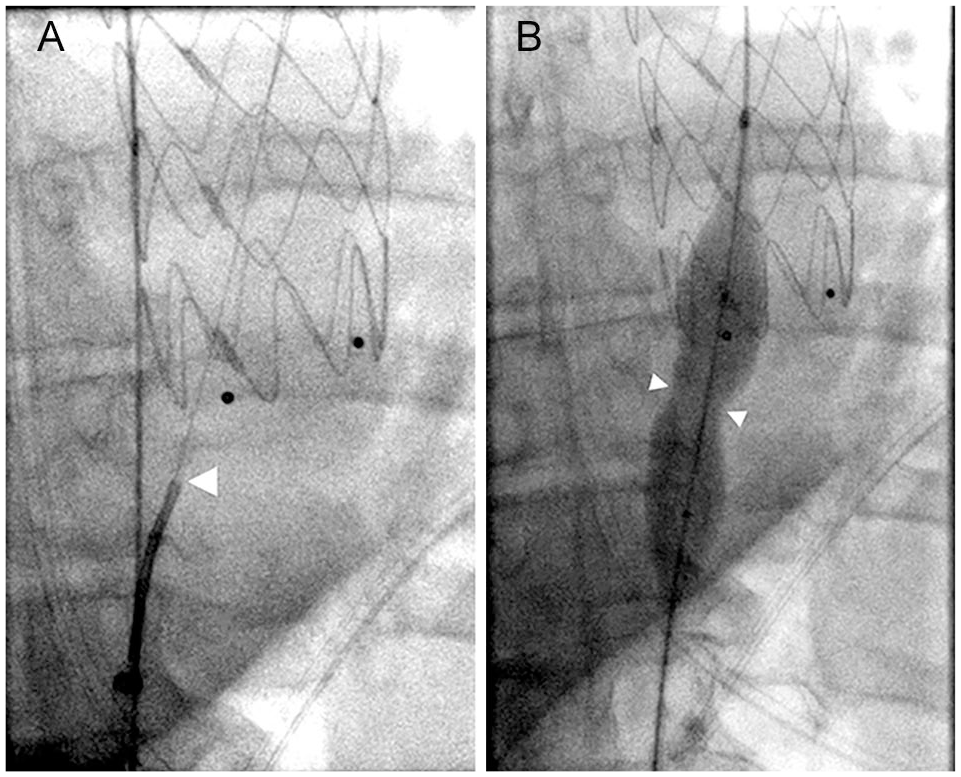
(A) Fluoroscopy illustrates fenestration of the aortic dissection about 1 cm distal to the incorrectly positioned aortic stent graft using a 5F catheter and the acute proximal stiff and sharp end of an 0.018” guidewire (arrowhead). (B) Balloon dilatation (25/40 mm) for additional expansion of the fenestration (arrowheads).
To achieve retrograde fenestration, a curved 5F catheter combined with the sharp back end of a 0.45 mm/0.018” wire (Boston Scientific ControlWire® V-18™) was orthogonally advanced toward the dissection membrane. To avoid fatal aortic wall perforation, cautious positioning of the wire was confirmed by multiple angulated views of the X-ray C-arm before puncture. Wire passage into the FL was realized by firmly overcoming the typical tender resistance of the dissection membrane. Free maneuverability of the catheter tip within the stent graft and DSA from the ascending aorta verified successful TL-FL fenestration. Expansion of the fenestration using a 25-mm balloon is shown in Figure 3B and Supplementary Online Material video 3.
Ballooning alone was inadequate to obtain sufficient TL reperfusion. Therefore, in case 1, distal extension of the FL stent graft into the descending aorta was performed. Another thoracic stent graft (W.L. Gore & Associates, TAG® Conformable Thoracic Stent Graft with ACTIVE CONTROL System 40/40/200 mm) was used, resulting in prompt visceral reperfusion and delayed partial FL perfusion (Figure 4A).
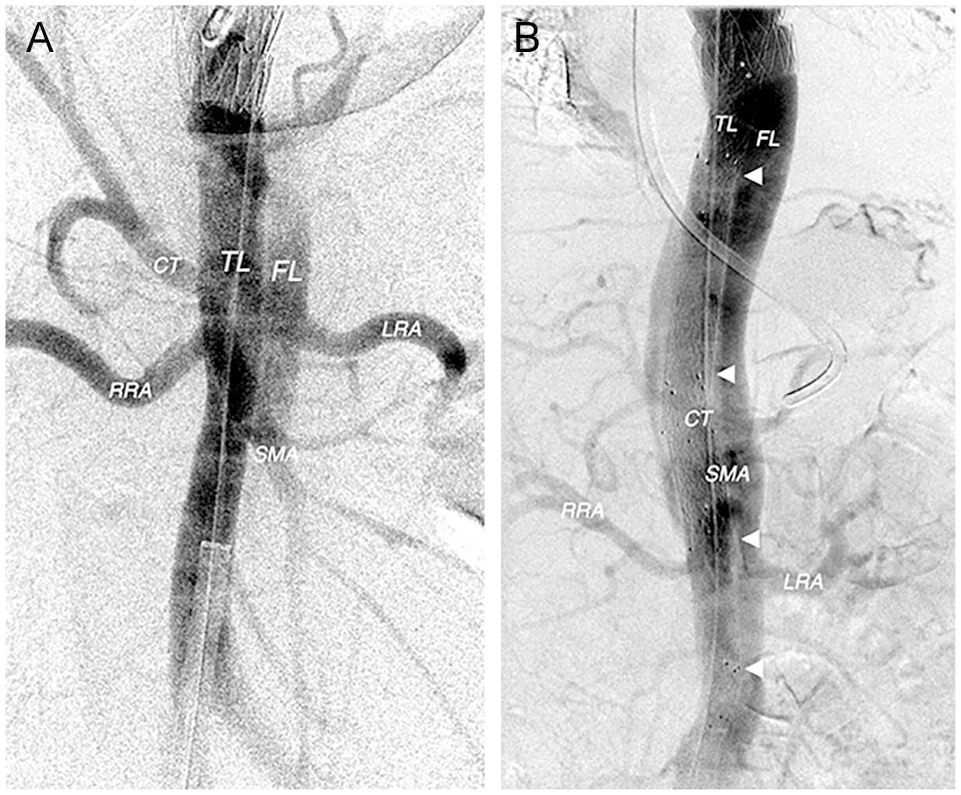
(A) After thoracoabdominal stent graft extension via a transfemoral 22F sheath, prompt visceral reperfusion and delayed partial false lumen perfusion is apparent. (B) After intentionally undersized thoracoabdominal bare metal stent extension (arrowheads), prompt visceral and peripheral reperfusion via the true and false lumen is apparent.TL, true lumen; FL, false lumen; CT, celiac trunk; SMA, superior mesenteric artery; RRA, right renal artery; LRA, left renal artery.
In case 2, there was intense FL supply to the left renal and iliac arteries 2 in addition to the TL collapse in the thoracoabdominal aorta. Therefore, a different strategy was used in this patient in order to stabilize the dissection membrane and maintain perfusion to both kidneys and lower extremities. Four undersized (half of the aortic diameter) self-expanding bare metal stents (BMS) with high radial force and flexibility (Opti-Med, Medizinische Instrumente; Sinus XL flex 16 mm) were used for distal extension of the incorrectly positioned thoracic stent graft, resulting in prompt reperfusion of the visceral arteries (Figure 4B). Furthermore, there was patent outflow to the right iliac arteries via the TL, and uncompromised flow to the left renal and iliac arteries via the FL.
After completion angiography confirmed visceral vessel patency and a stable hemodynamic situation, the patients were immediately transferred to the intensive care unit for postoperative care. Overall fluoroscopy times were 25.4 minutes (case 1) and 36.2 minutes (case 2). Patients were administered intravenous antihypertensive therapy (urapidil perfusor), pain medication (morphine), and vital signs were controlled.
Despite excellent acute hemodynamic results, the patient with additional stent graft extension died 12 hours later due to multiorgan failure. The patient with the BMS extension showed improvement in the severe ischemia. However, he also died 5 days later due to large cerebral hemorrhagic infarction.
Discussion
Our case studies highlight five key aspects of an endovascular bailout of inadvertent FL stenting. First, we used a single percutaneous transfemoral approach for controlled access to the TL, which is minimally invasive, fast, and safe.
Second, we employed repetitive high-frame rate DSA with automated power injection for continuous confirmation of correct TL cannulation. While we recognize that IVUS may be the ideal tool to confirm catheterization of the TL, IVUS is not uniformly available and our DSA technique is an alternative option that may be more accessible or applicable to interventionalists faced with similar challenges. We acknowledge the drawback that multiple contrast injections are not ideal in patients that already have some degree of renal malperfusion as a result of their TBAD and the aortic stent graft location in the FL. However, we felt this technique was the best course of action in these emergency cases and in the absence of IVUS at the time, considering the life-threatening circumstances.
Third, we employed a standard technique for sharp recanalization from the TL to the FL for rescue fenestration of the incorrectly positioned TEVAR. Thorough evaluation of imaging studies is essential for successful puncture and to avoid fatal aortic wall perforation. We crossed into the FL approximately 1 cm distal to stent graft, which was as close as possible to the incorrectly positioned stent graft; further wire advancement was not possible due to TL compression by the FL TEVAR. There was limited space between the distal end of the stent graft and the visceral arteries. The TEVAR stent graft acted like a counterfort and the dissection membrane was fixed in a stable position to allow successful puncture against an otherwise very mobile dissection membrane. This technique and situation obviated the need for an additional balloon. 3 Successful TL-FL fenestration must be verified before ballooning. During ballooning downward traction was omitted in order to avoid further unpredictable damage to the dissected aorta.
Fourth, we recognized the visceral and lower limb perfusion profile unique to each case and executed our bailout strategy accordingly. We were mindful to consider the location of the visceral artery ostia and that stent graft extension in a malperfused patient could have triggered spinal cord ischemia. In case 1, we chose another stent graft for distal extension because visceral and peripheral malperfusion was a consequence of the completely collapsed TL. With the fenestration and stent graft extension in place, almost normal hemodynamic reperfusion of the expanded TL was apparent, along with discontinued FL pressurization. In case 2, we intentionally did not use another aortic stent graft because the left renal and iliac arteries were perfused via the FL. Use of another aortic covered stent graft would have potentially reduced or eliminated patency of these peripheral arteries. Based on our prior experience in the index treatment of aortic dissections, we chose flexible self-expanding nitinol BMS with high radial force and a diameter half of the original aortic diameter. 2 This intentional undersizing allowed complete stent expansion in the TL, resulting in a circular shape and consequent optimal radial force. In an aortic dissection, the forces of the dissection membrane are sufficient to secure an undersized stent in place. With the fenestration and BMS extension in place, hemodynamic reperfusion of the expanded TL was apparent, along with continued patency of the left visceral and iliac arteries via the FL.
Finally, despite immediate technical success, both patients died in the following days. This was most likely due to the time delay (24-36 hours) between TL collapse caused by index erroneous stent graft placement and our rescue attempt. Therefore, timely recognition of the error and initiation of the rescue is critical to success.
While not performed at our hospital, we reflect on the key errors or things that may have been overlooked during the index procedures. First, correct indication is essential. In case 1, initial clinical symptoms associated with TBAD were only pain and hypertension. Signs of a complicated TBAD were absent; there were no signs of organ or peripheral malperfusion. Therefore, indication for TEVAR was only based on CT imaging findings. In general, images themselves should not be treated, rather only in the case of associated symptoms.
A single aortography of the aortic arch and descending thoracic aorta is not sufficient to fully characterize the hemodynamic profile of a complicated TBAD. Preprocedural visceral angiography is mandatory to fully characterize the hemodynamic profile of the case. We believe the index misplacement of the stent graft into the FL was the consequence of either primary iliaco-aortic exit from the TL into the FL or exit of the guidewire from the TL into the FL and again re-entry into the TL while approaching the aortic arch from distal to proximal. IVUS or transesophageal echocardiography may be helpful for identifying fenestration and aortic branches and/or repetitive verification of the guidewire localization.4–6 Furthermore, the guidewire should be securely placed in the ascending aorta after meticulous verification of correct TL catheterization.
Post index stent graft angiography showed apparently patent, normal looking thoracic aortas with regular vessel diameters. However, impaired or missing depiction of visceral or peripheral branching arteries on DSA is highly suspicious and indicates absent or insufficient restoration of the TL perfusion. This should be considered a trigger for immediate re-evaluation of the given pathoanatomic situation. Checking for a femoral pulse or the use of peripheral vascular ultrasound are also convenient ways to confirm peripheral perfusion.
There are several reports of endovascular bailouts in the literature. In 2016, Han et al reported an endovascular rescue fenestration approach, starting with a surgical cut-down of both femoral arteries for TL and FL access. 7 A snare was positioned into the FL directly below the mispositioned aortic stent graft. From the TL, the proximal stiff end of a 0.014” wire was directed to the snare in the FL and then into the FL stent graft. Finally, another stent graft implantation from the FL stent graft into the TL resulted in successful re-expansion of the downward TL, unimpaired perfusion of the visceral and aorto-iliac vasculature with complete clinical recovery.
In 2020, Li et al reported endovascular repair of an intraoperative FL stent graft deployment after open repair of Stanford A dissection 2 years prior. 8 Numerous intimal tears presenting as multiple entries and re-entries of the dissected aorta sustained a FL-TL perfusion to the visceral arteries. Therefore, fenestration was not necessary for repair. An overlapping stent graft extending from the FL stent graft through the dissection membrane into the TL was used to redirect blood flow back to the TL. Additional four-fenestrated thoracoabdominal stent graft implantation was required for visceral artery reconstruction.
In 2020, Chen et al reported that inadvertent placement of an elephant trunk into the FL was repaired by implantation of a stent graft using a right-brachial to femoral through-and-through wire approach. 9 A femoral guidewire was advanced from the TL into the FL via a preexistent intimal tear without necessity for fenestration. Snaring of the wire from an additional transbrachial access secured successful FL-TL redirection.
More generally, in the absence of an intimal tear, fenestration of the dissection membrane may be performed using a Rösch-Uchida needle, radiofrequency puncture needle,10,11 or a dedicated re-entry catheter.12–14 In 2014, Bertoglio et al also described neofenestration using a standard or IVUS-guided reentry device in either the TL or FL, while a balloon simultaneously inflated in the opposite lumen to stabilize the lamella provided needle counterforce, and prevented displacement of the lamella away from the reentry needle. 3
In our rescue cases, a transbrachial antegrade endovascular access to the aorta may have reduced contrast exposure and procedure time using a through-and-through brachial-femoral wire to enable retrograde delivery of the TEVAR extension across the aortic septum. However, fenestration from the stented FL to the almost completely collapsed TL would have borne high risk for fatal aortic perforation.
The unique points of our endovascular rescues were the single transfemoral and percutaneous approach, the repetitive DSA-based confirmation of TL cannulation, and the case-specific choice of stent extension in order to regain and maintain complete visceral and lower limb perfusion.
Conclusion
Meticulous care is mandatory to verify correct TL catheterization in the endovascular treatment of an aortic dissection. To avoid inadvertent stent graft placement into the FL, it is insufficient to only check the point of entry at the puncture site and the most proximal position of the guidewire in the aorta for correct placement in the TL. In the case of erroneous FL stenting, prompt recognition of the error of the utmost importance to avoid disastrous malperfusion. Endovascular salvage is a feasible option for restoration of correct perfusion and must be immediately identified and treated.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
