Abstract
Purpose:
Despite encouraging early results, mid- and long-term follow-up of endovascular aneurysm sealing (EVAS) has shown increased rates of failure mainly associated with endoleak detection and progressive bag separation with aneurysm reperfusion.
Case Report:
We present the first case of a Nellix endograft stent fracture detected in a 91-year-old male patient, presenting with widespread abdominal pain, 7 years after elective treatment of an abdominal aortic aneurysm by EVAS. Considering the sudden and unexpected nature of the event, an in-depth analysis of the possible causes of this structural failure has been performed.
Conclusion:
Material fatigue could be another significant cause of late EVAS failure and should be carefully assessed in addition to endoleak detection during follow-up.
Clinical Impact
The case presented in this article further underlines the importance of a strict long term follow-up protocol in every patients who underwent EVAS.
Introduction
Endovascular aneurysm sealing (EVAS), performed with the Nellix endograft (Endologix Inc, Irvine, CA, USA), introduced an alternative approach to endovascular repair of abdominal aortic aneurysm (AAA) by obliterating the lumen of the aorta with the main objective to minimize the incidence of type II endoleak (EL). Even if preliminary data showed a good efficacy of the EVAS methodology in AAA treatment,1–5 unfortunately long-term results in the literature did not meet the expectations.6–9 We present the first case of Nellix rupture in a 91-year-old man admitted to the emergency department with a huge symptomatic infrarenal AAA previously treated with EVAS. Reviewing the English literature, we did not find other cases of Nellix rupture.
Case Report
A 91-year-old male patient presented to our hospital on January 2022 with widespread abdominal pain lasting from the previous day. He underwent elective AAA repair for an aneurysm of 63×57 mm maximum diameter by Nellix endograft in 2015 in our unit. Based on preoperative computed tomography (CT) scan, the index procedure was deemed fit for EVAS considering the original instructions for use (IFU); however, a ratio of maximum aortic aneurysm diameter to maximum aortic blood lumen diameter of 1.6 (> 1.4) has subsequently pushed the procedure outside the revised 2016 IFU. The anatomical features of the aneurysm and the preoperative and intraoperative images are shown in Figures 1 and 2. The patient was regularly controlled by periodic imaging based on our follow-up protocol consisting of a first CT scan at 3 months followed by duplex ultrasound (DU) every year. Considering the increasing evidence of progressive unexpected late device failures emerging from our experience and from literature,6–11 we started to perform additional CT scans after EVAS based on case-specific characteristics even in absence of clear images of EL or sac enlargement at the DU. The patient’s last CT scan was performed on May 2021 and did not show any relevant EL with a stable sac diameter (Figure 3A and B). His medical history was also remarkable for percutaneous transluminal coronary angioplasty in 2015, stage 3 chronic kidney disease, and subclinical hyperthyroidism under medical treatment. On admission, the patient resulted positive for infection from severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). He underwent urgent CT scan that revealed previous AAA exclusion by EVAS with an aneurysmal sac diameter of 91 mm, increased when compared with the previous CT scan of May 2021 (AAA maximum diameter of 65×63 mm), associated with a small posterior periprosthetic EL in the proximal area (IA) and a second one in the distal area of the sac with a clear image of rupture of the left stent in the distal portion (EL III; Figure 3C and D). The patient, being symptomatic due to the dislocation of the endoprosthetic branches and the endobag, in the presence of marked growth of the AAA, underwent emergent surgical endograft removal. Intraoperatively, within the aortic aneurysm, we observed the presence of recently coagulated blood (Figure 4B and C), and the rupture of the left stent Nellix endograft, in its distal portion (Figure 4D and F), was confirmed. Surgical endograft removal associated with an aorto-aortic graft bypass and a left aorto-iliac jump with a Dacron silver prosthesis (16 and 8 mm—Figure 4E) was performed. The patient died on the first postoperative day for multiorgan failure (MOF) probably as the result of intraoperative hemorrhagic shock.
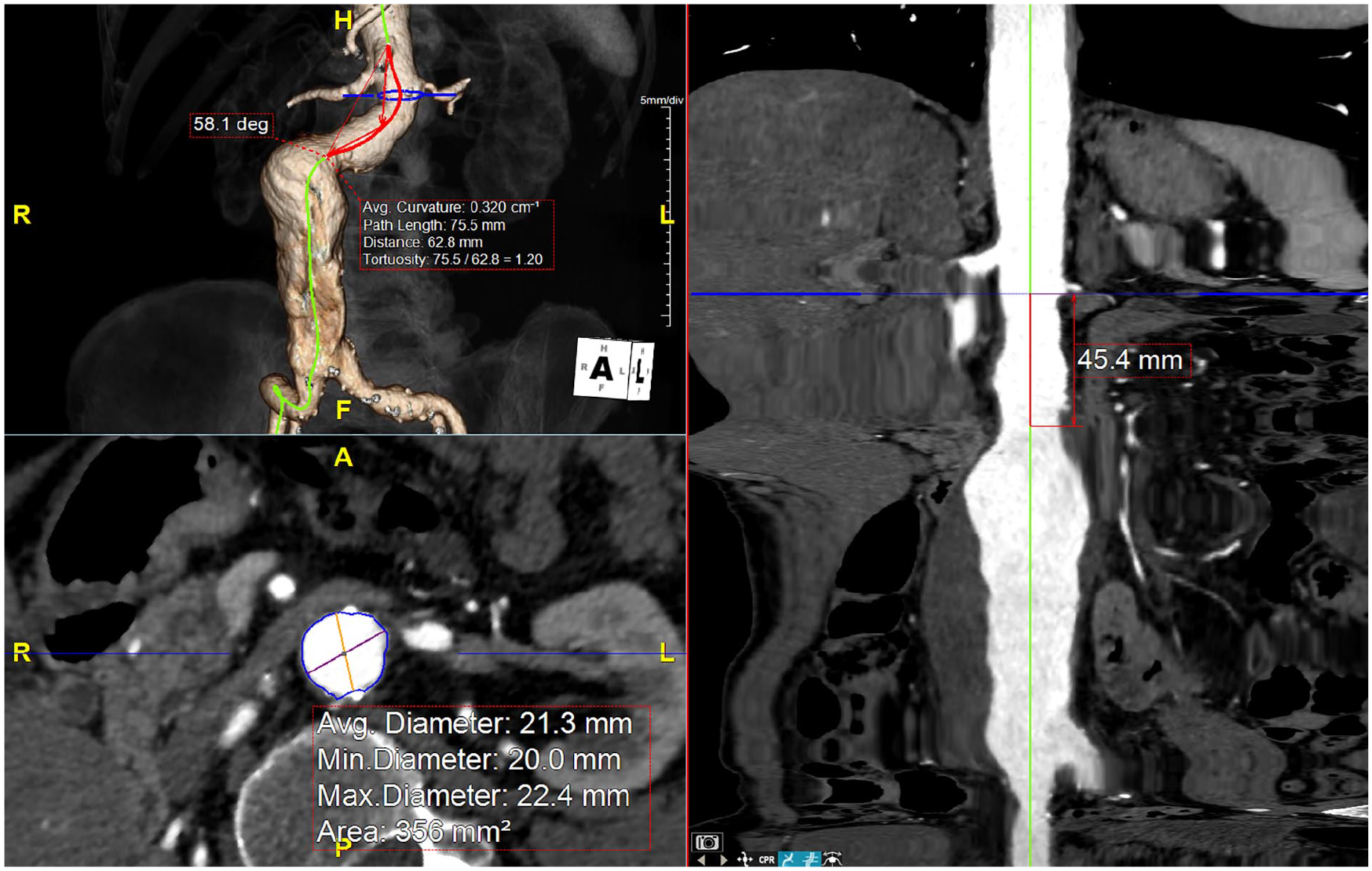
Preoperative planning of the index procedure based on 3D reconstruction of the computed tomography scan.
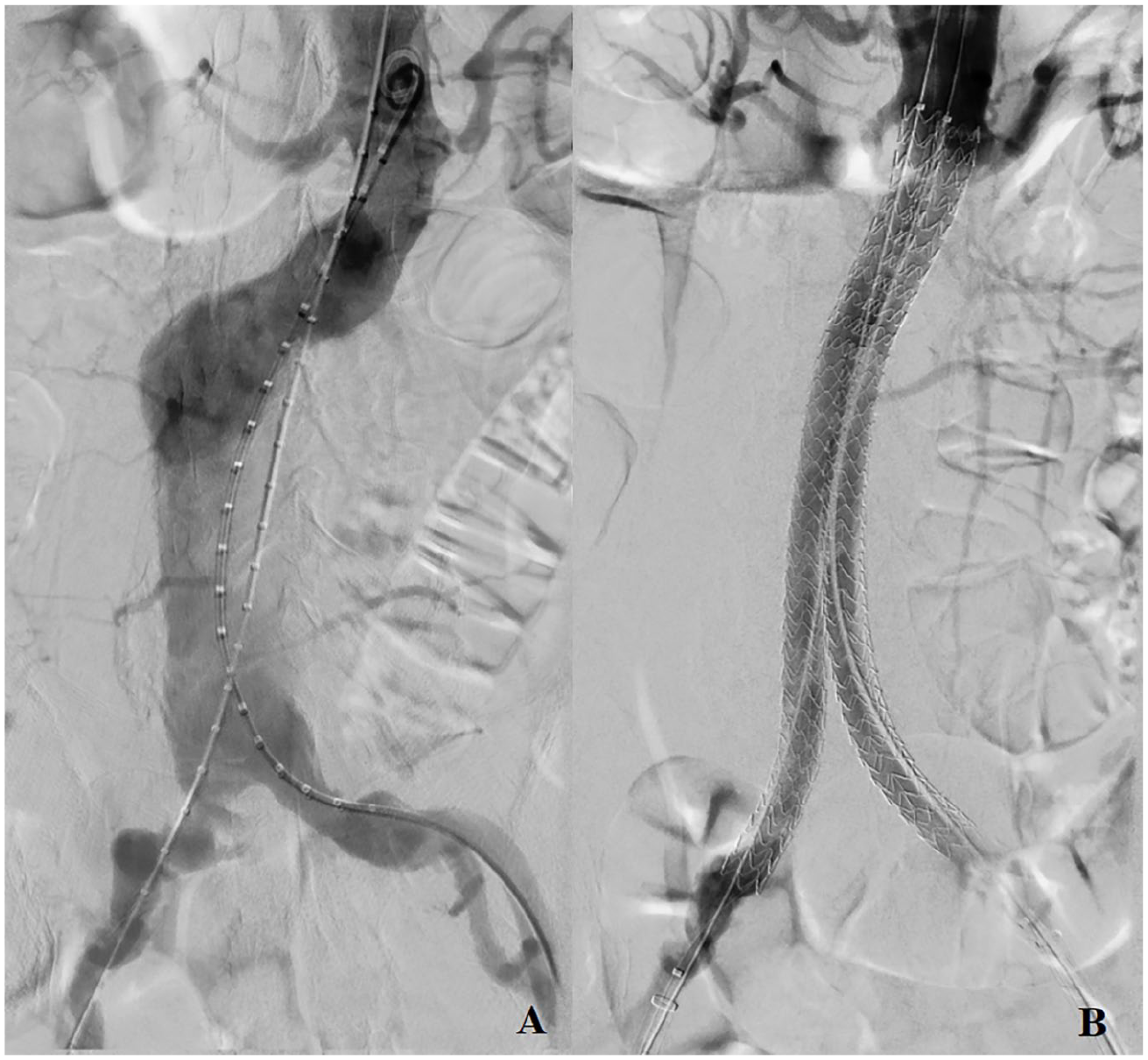
Images of the Nellix deployment: Preoperative angiography (A); control angiography (B).
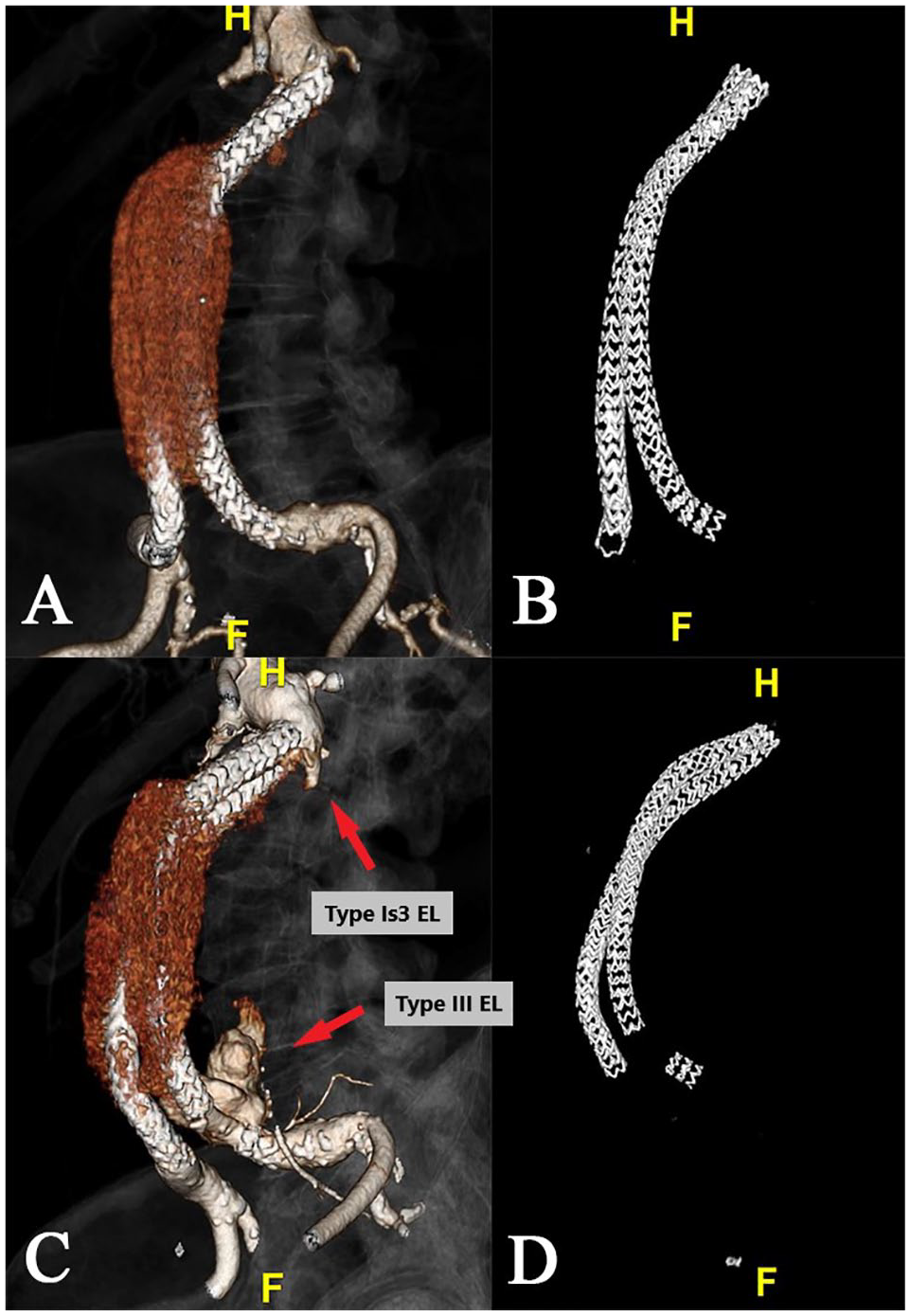
The 3-dimensional reconstruction of May 2021 computed tomography scan showing no relevant endoleak (EL) or stent fracture (A and B). The 3-dimensional reconstruction of January 2022 computed tomography scan showing the ELs and the stent fracture (C and D).
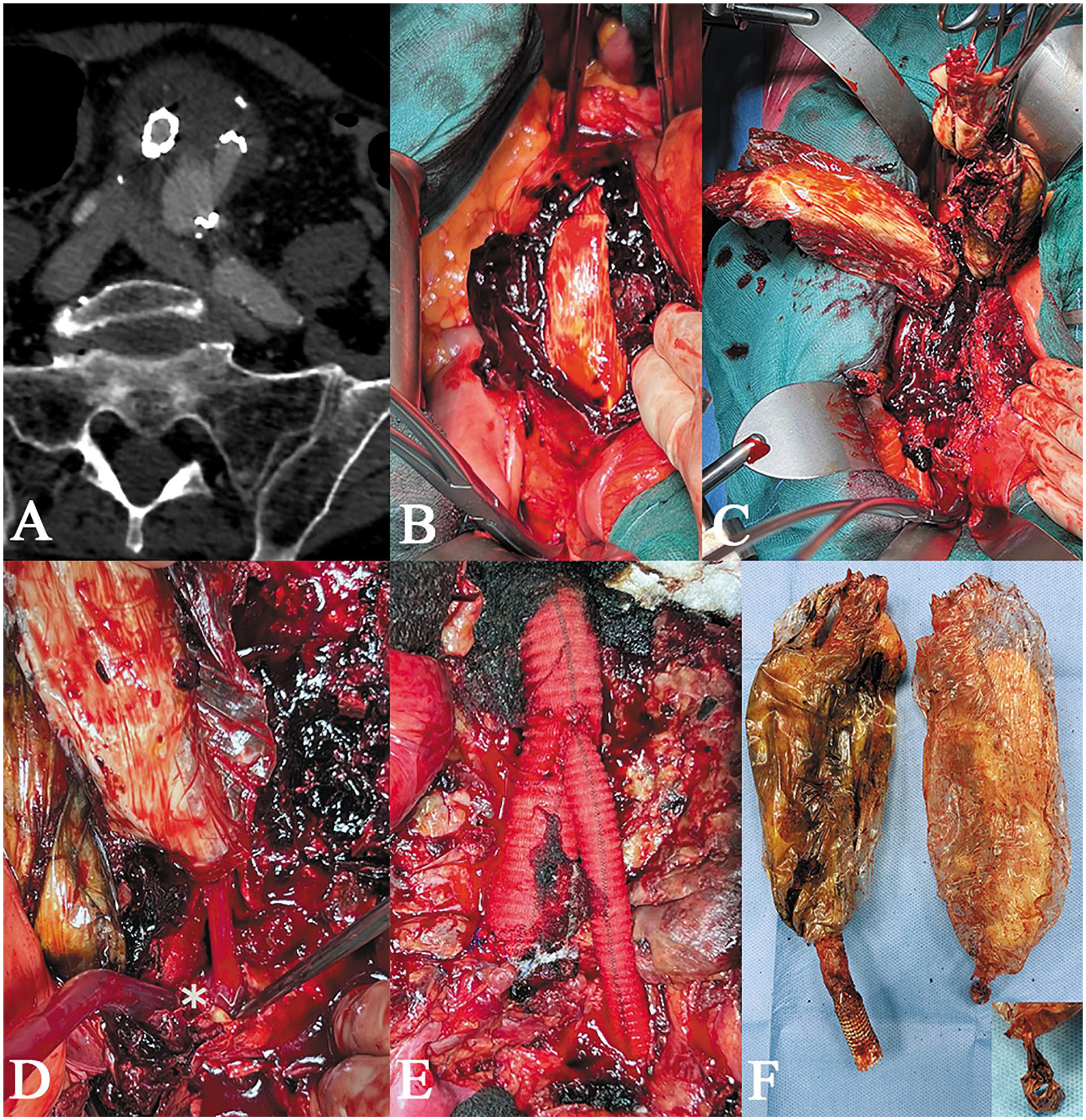
Computed tomography scan of January 2022 with type III endoleak (A). Intraoperative image of the aortic aneurysm with the presence of recently coagulated blood (B). Intraoperative image of the surgical endovascular aneurysm sealing removal (C). Intraoperative image of the left stent rupture in its distal portion (D). Aorto-aortic graft bypass and a left aorto-iliac jump with a Dacron silver prosthesis (E). Nellix endograft removed with rupture image in its distal portion (F).
Discussion
EVAS is achieved through a combination of stent fixation and adhesion to the abdominal aortic wall with a polymer-filled bag. This should have ensured a reduction of the EL rates and a reliable AAA sealing. However, recent studies suggested that EVAS, at a long-term follow-up, is at risk of migration and/or EL with consequent AAA rupture.6–9 The mechanisms underlying this condition are not fully understood yet, but some hypotheses could be postulated. One hypothesis could refer to the fact that in 2016 the manufacturer has revised the IFU of the device reducing its applicability for the treatment of AAA; some cases performed prior to this revision, like the patient of our report, may not have been adequate for treatment with EVAS, resulting in an increased incidence of ELs and migration as reported by Zerwes et al. 10 However, other studies7,11 showed no statistically significant differences in terms of overall mortality, freedom from aneurysm-related mortality, or any differences in the incidence of any complications between the in-IFU and off-IFU groups, suggesting that the cause of the complications is not related to the IFU’s change. Another hypothesis to understand EVAS failure considers the hemodynamics changes developing in the AAA neck region after endograft implantation involving increased pressure values and altered wall shear stress in the juxtarenal portion of the aorta, these phenomena have been measured in the EVAS system after lateral migration of the endobags, thus indicating the onset of pathological conditions.12,13 A study by Boersen et al 14 demonstrated that the wall shear stress, in the caudal wall of the renal artery after aortic endograft deployment, presents an increased shear rate after EVAS than after endovascular aneurysm repair. Assuming, therefore, that one of the weaknesses of Nellix is the remodeling occurring at the proximal neck, we do not believe that it can be the only cause of complications. As already reported by van den Ham et al, 15 it is necessary to adopt a new classification to correctly evaluate the ELs following the EVAS implant: an incorrect assessment could underestimate a serious EL that would require re-intervention, by endograft explantation or endovascular treatment.9,15 Therefore, an unrecognized EL type IA could induce the migration of one or both endobags. This, as suggested by Battista et al, 13 is supposed to be related to the existing differential pressures acting in the gap formed between the two, which could go on pushing the two branches away from each other, causing aneurysm re-activation and ELs. Our case was characterized by a double issue: two ELs, one from the top and one from the bottom. Using the classification of van den Ham, we can describe the EL from the top as a type Is3 (appearance of contrast or fresh thrombus between the endobags inside the aneurysm sac—Figure 3C) associated with a type III (Figure 3C). This finding was also confirmed intraoperatively. During the Nellix removal maneuver, it can be seen that part of the stent has remained attached to the left common iliac artery and is disconnected from the rest of the body of the endograft (Figure 4D). So what was the cause of the AAA’s increase? The EL from above or from below? Analyzing the CT scans of May 2021 and the previous ones, we noticed that using the Van den Ham classification, a negligible EL from above (Is1) had been present for several years and remained stable over time. Therefore, it could be hypothesized that the EL Is1 has transformed into an EL Is3, in the last 8 months, resulting in a displacement of the Nellix prostheses with subsequent fracture of the stent. Or it could be hypothesized that following the fracture of the Nellix stent, the stability of the endograft was altered with consequent formation of EL Is3, but with origin from below. Despite the relative high occurrence of Type I EL and endobags separation after EVAS described in literature articles, we have not found reports of stent fracture as its direct consequence.7,9 Furthermore, comparing the CT scan of May 2021 with the preoperative one, it can be seen that the proximal portion of the Nellix stents is placed in the same position as the initial release, without lateral dislocation of both stents at proximal level or clear guttering from above. We are thus convinced that in our case the second hypothesis, namely that the genesis of the complication was generated by the type III EL, is more plausible. Analyzing the English literature, we did not find other similar articles relating to a spontaneous detachment of part of the Nellix stent resulting in the displacement of the endoprosthesis with consequent reperfusion of the aneurysm sac; we have only found one Nellix endobag rupture. 16
Conclusion
We present the first case of Nellix stent rupture. This event should certainly not cause alarm, as it is a single case. Further attention should however be paid, in EVAS follow-up, not only to the EL types but also to the possible presence of structural alterations of the endograft.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
