Abstract
Purpose:
To evaluate the clinical utility of the Gore Excluder iliac branch endoprosthesis (IBE) for Japanese patients with aortoiliac aneurysms.
Materials and Methods:
This was a multicenter retrospective cohort study (J-Preserve Registry). Patients undergoing endovascular aortic repair using the Gore Excluder IBE for aortoiliac aneurysms between August 2017 and June 2020 were enrolled. Data pertaining to the baseline and anatomical characteristics, technical details, and clinical outcomes were collected from each institution. The primary endpoints were technical success, IBE-related complications, and reinterventions. Secondary endpoints were mortality, aneurysm size change, and reintervention during follow-up. Technical success was defined as accurate deployment of the IBE without type Ib, Ic, or III endoleaks on the IBE sides on completion angiography. A change in aneurysm size of 5 mm or more was taken to be a significant change.
Results:
We included 141 patients with 151 IBE implantations. Sixty-five IBE implantations (43.0%) had at least one instruction for use violation. Twenty-two patients (15.6%) required internal iliac artery (IIA) embolization for external iliac artery extension on the contralateral side. Of 151 IBE implantations, 19 exhibited IIA branch landing zones due to IIA aneurysms. Mean maximum and proximal common iliac artery (CIA) diameters were 32.9±9.9 mm and 20.5±6.9 mm, respectively. The mean CIA length was 59.1±17.1 mm. The IIA landing diameter and length were 9.0±2.3 mm and 33.8±14.6 mm. The overall technical success rate was 96.7%. There were no significant differences in IBE-related complications (2.3% vs 5.3%, p=0.86) or IBE-related reinterventions (1.5% vs 5.3%, p=0.33) between the IIA trunk and IIA branch landing groups. The mean follow-up period was 635±341 days. The all-cause mortality rate was 5.0%. There were no aneurysm-related deaths or ruptures during the follow-up. Most patients (95.7%) had sac stability or shrinkage.
Conclusion:
The Gore Excluder IBE was safe and effective for Japanese patients in the midterm. Extending the IIA device into the distal branches of the IIA was acceptable, which may permit extending indications for endovascular aortic aneurysm repair of aortoiliac aneurysms to more complex lesions.
Clinical Impact
This study suggests clinical benefits of the Gore Excluder IBE for Japanese patients, despite 43% of the IBE implantations having at least one IFU violation.
Keywords
Introduction
Endovascular aortic aneurysm repair (EVAR) has become the gold standard treatment for abdominal aortic aneurysm (AAA) because it is less invasive and has better early outcomes than open surgical repair. 1 Approximately 25% to 40% of AAAs are associated with common iliac artery aneurysms (CIAAs).2,3 Although patients with AAA and concomitant CIAA require external iliac artery extension with internal iliac artery (IIA) embolization after EVAR treatment, there is a potential risk of pelvic ischemia, including buttock claudication, erectile dysfunction, and bowel ischemia. 4 Clinical guidelines from the European Society for Vascular Surgery and the Society of Vascular Surgery recommend preserving at least one IIA in these patients.5,6
In 2017, the Gore Excluder iliac branch endoprosthesis (IBE) was approved in Japan, and it can preserve the IIA when anatomically feasible. Two-year outcomes of 99 patients with CIAA or aortoiliac aneurysms treated with the IBE in the US investigational device exemption (IDE) trial showed safety and good internal iliac device patency of 94%. 7
A recent study also demonstrated satisfactory midterm outcomes of the Gore Excluder IBE using the division branches of the IIA as distal landing zones. 8
Asian patients are known to have smaller CIA diameters and shorter CIA lengths than those reported in published literature, 9 and only 25% to 31% of Japanese patients met the criteria for the IBE device.9,10 In addition, there is a paucity of data regarding the outcomes of IBE in Japanese patients because the use of this device has been limited to a small number of patients at a single center.
The present study aimed to clarify the clinical outcomes of EVAR with IBE in Japanese patients with aortoiliac aneurysms.
Materials and Methods
Study Design and Patient Selection
The J-Preserve Registry (Japanese experience of Gore Excluder Iliac Branch Endo
Data of baseline and anatomical characteristics, technical details, and clinical outcomes were collected from each institution. All procedures were performed by board-certified surgeons or interventional radiologists. The indication for using the IBE devices was decided by each physician. Most procedures were performed through groin incisions under general anesthesia to expose the common femoral artery. Standard bifurcated stent-grafts were implanted after the completion of the IBE procedure. In the case of IIA aneurysms, embolization of all branches from the aneurysm sac or extension of the internal iliac devices to the IIA branches was performed. Post-EVAR surveillance protocols included multidetector-row computed tomography (CT) with both early- and delayed-phase scans at 1 and 6 months postoperatively, and annually thereafter. Plain CT with duplex ultrasound was used for patients with allergy to contrast materials or severe renal dysfunction, instead of contrast-enhanced CT.
The primary endpoints of this study were technical success, IBE-related complications, and reinterventions. Secondary endpoints were mortality, aneurysm size change, and reintervention during the follow-up. As for the IBE-related complications or reinterventions, they were further compared between IIA trunk landing and IIA branch landing groups. Regarding the criteria for choosing IIA branch landing sites, larger diameter, greater length, or less kinking of the branches was generally selected.
The study complied with the principles of the Declaration of Helsinki, and the study protocol was reviewed and approved by the institutional review board (IRB) of each institution (approval number: 5128). Because this study was a retrospective review of charts, the IRB waived the need for informed consent from the patients, and the opt-out method was used.
Definitions
Technical success was defined as accurate IBE deployment without type Ib, Ic, or III endoleaks on the IBE sides on completion angiography. The main aneurysm was defined as an indication for treatment with the largest diameter of aortoiliac aneurysms. A change in aneurysm size of 5 mm or more was taken to be a significant change. Renal dysfunction was defined as an estimated glomerular filtration rate of <60 mL/min/1.73 m2.
Statistical Analysis
Statistical analyses were conducted using JMP software (version 14; SAS Institute Inc., Cary, North Carolina). Comparison of variables was performed using the chi-square test or the Mann-Whitney test, with p<.05 considered to indicate a statistically significant difference.
Results
Patient Demographics and Lesion Characteristics
A total of 141 patients treated with EVAR using IBE were included in this study (Table 1). Mean patient age was 76.2±7.2 years, and 77% of the patients were male. The most frequent comorbidity was hypertension (70.9%), followed by smoking (64.5%) and renal dysfunction (49.6%). The main aneurysms were AAA (37.6%), CIAA (51.8%), and internal iliac artery aneurysm (IIAA) (10.6%), and the maximum aneurysm size in diameter was 46.6±10.5 mm.
Patient Demographics and Lesion Characteristics.

Abbreviations: AAA, abdominal aortic aneurysm; ASA, American Society of Anesthesiologists; CAD, cardiovascular disease; CIAA, common iliac artery aneurysm; COPD, chronic obstructive pulmonary disease; CVD, cerebrovascular disease; IIAA, internal iliac artery aneurysm.
Anatomical Characteristics and Instruction for Use Violation on the IBE Sides
Nineteen patients (12.6%) had distal landing of IBE at the IIA branch due to the coexisting IIAA or IIA dilatation. Mean maximum, minimum, and proximal CIA diameters on the IBE sides were 32.9±9.9 mm, 18.0±6.1 mm, and 20.5±6.9 mm, respectively. The mean CIA length was 59.1±17.1 mm. The IIA landing diameter and length in the IIA branch group were significantly smaller (9.2±2.2 mm vs 8.1±3.0 mm, p=0.004) and shorter (34.8±13.1 mm vs 28.2±22.3 mm, p=0.006), respectively, than those in the IIA trunk group.
Sixty-five IBE implantations had at least one instruction for use (IFU) violation (43.0%). Among the anatomical criteria, proximal CIA diameter of <17 mm was the most common limitation (30.5%), followed by IIA landing diameters (11.9%) and short lengths from the lowest renal artery to the CIA bifurcation (7.9%). There were no short IIA lengths of less than 10 mm, and all but one had favorable EIA diameters (Table 2).
Anatomical Characteristics and IFU Violation on the IBE Sides.

Abbreviations: CIA, common iliac artery; EIA, external iliac artery; IBE, iliac branch endoprosthesis; IFU, instruction for use; IIA, internal iliac artery; RA, renal artery.
p<0.004. **p<0.006.
Procedural Outcomes
The procedural details are summarized in Table 3. Ten patients (7.1%) underwent bilateral IBE implantations. Of the 19 IBE implantations in the IIA branch group, 15 were landed into the superior gluteal artery (SGA) and 4 into the inferior gluteal artery (IGA). The IIA branch landing was caused by an IIA aneurysm in 18 patients and an IIA trunk with wall irregularity in 1 patient. Twenty-two patients (15.6%) required IIA embolization for contralateral EIA extension. Technical success was achieved in 146 of 151 IBE implantations (96.7%). Of the 5 IBE implantations with technical failure, 1 had an iliac branch component (IBC) device malpositioned distally during deployment, and the internal iliac component (IIC) gate had to be deployed into the EIA. The other 4 patients had type Ib or III endoleaks on the IBE sites on completion angiography, although these endoleaks disappeared spontaneously during follow-up. There was no type II endoleak in the IIA aneurysms. No intraoperative was observed. The mean operative time was 191±62 minutes. Intraoperative complications were observed in 10 patients (6.6%). Access injuries were the most common complications, and 2 patients had SGA/IGA injuries during IBE device insertion treated with embolization and balloon angioplasty, respectively.
Procedural Outcomes.
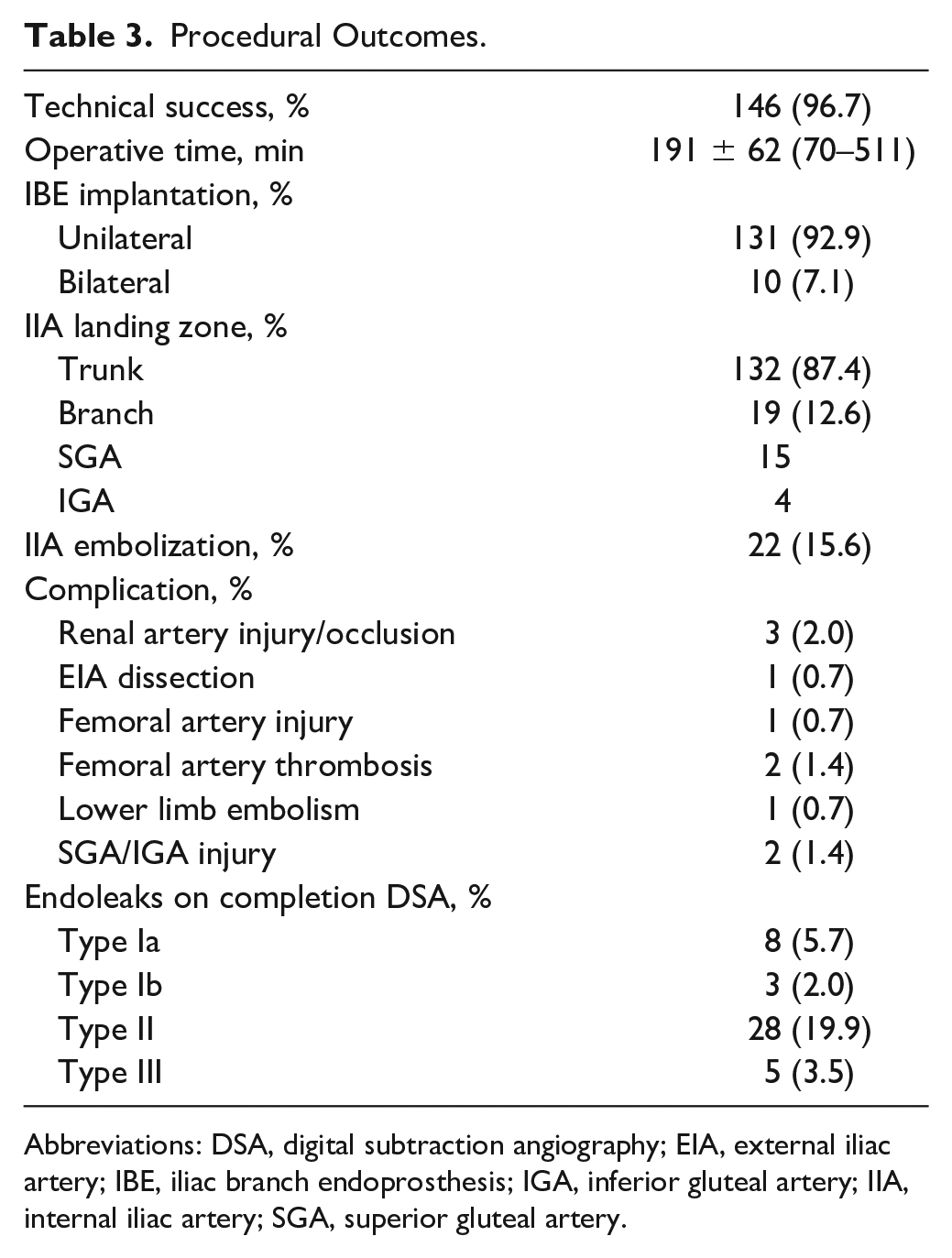
Abbreviations: DSA, digital subtraction angiography; EIA, external iliac artery; IBE, iliac branch endoprosthesis; IGA, inferior gluteal artery; IIA, internal iliac artery; SGA, superior gluteal artery.
Follow-up Clinical Outcomes
The clinical outcomes are summarized in Table 4. Although 7 patients (5.0%) died during follow-up, there were no aneurysm-related deaths or ruptures. The mean follow-up period for the entire cohort was 635±341 days. Regarding aneurysm size change, most patients (95.7%) had no change or a decrease in size, whereas only 6 patients had an increase in size (4.3%), possibly due to type II endoleaks in 4 and endotension in 2. There were only 2 cases of type Ia endoleaks. No type Ib or III endoleaks were observed after discharge. Although 51 patients (36.2%) had type II endoleaks, only 1 patient required embolization. Major adverse events within 30 days were observed in 2 patients (acute renal failure and retrograde type B aortic dissection), both of which were treated conservatively.
Follow-up Clinical Outcomes.

Abbreviations: CT, computed tomography; IBE, iliac branch endoprosthesis; IIA, internal iliac artery; PTA, percutaneous transluminal angioplasty; TAE, transcatheter arterial embolization.
Buttock claudication occurred in 12 patients (8.5%), and 9 of them were due to IIA embolization on the contralateral side. The remaining 3 cases of buttock claudication on the IBE sides were caused by IIA device occlusion (2 cases) and IIA reconstruction failure due to IBC device malposition during deployment (1 case). No bowel ischemia or erectile dysfunction was observed. Reinterventions were needed for 9 complications. Of these, 2 were associated with endoleaks, 3 with iliac limb, 3 with renal arteries, and 1 with stent-graft infection. Of the patients with a small proximal CIA outside the IFU, 1 had a proximal CIA diameter of 11 mm, which resulted in iliac limb stenosis treated with stenting 11 days postsurgery. Internal iliac device occlusions were present in 3 cases. Although all 3 cases followed appropriate oversizing of the IIA devices for landing diameters, 1 had stenosis of the IIA orifice with calcification. Two of them had buttock claudication on the IBE side, but no additional treatment was performed.
Comparison of IBE-Related Outcomes Between IIA Trunk and IIA Branch Landing Groups
The comparison is shown in Table 5. There was no significant difference in technical success rates between the groups (96.2% vs 100%, p=0.86). The mean operative time in the IIA branch group was significantly longer than that in the IIA trunk group (189±65 minutes vs 218±57 minutes, p=0.016). Viabahn or VBX was more frequently used as additional IIA stent-grafts in the IIA branch group. Iliac branch endoprosthesis–related complication (2.3% vs 5.3%, p=0.86) or reintervention (1.5% vs 5.3%, p=0.33) did not significantly differ between the groups. Iliac branch endoprosthesis–related reinterventions were needed in 3 complications: balloon touch-up for type Ic endoleak, iliac limb thrombectomy in the IIA trunk group, and iliac limb stenting in the IIA branch group.
Comparison of IBE-Related Outcomes Between the Groups.
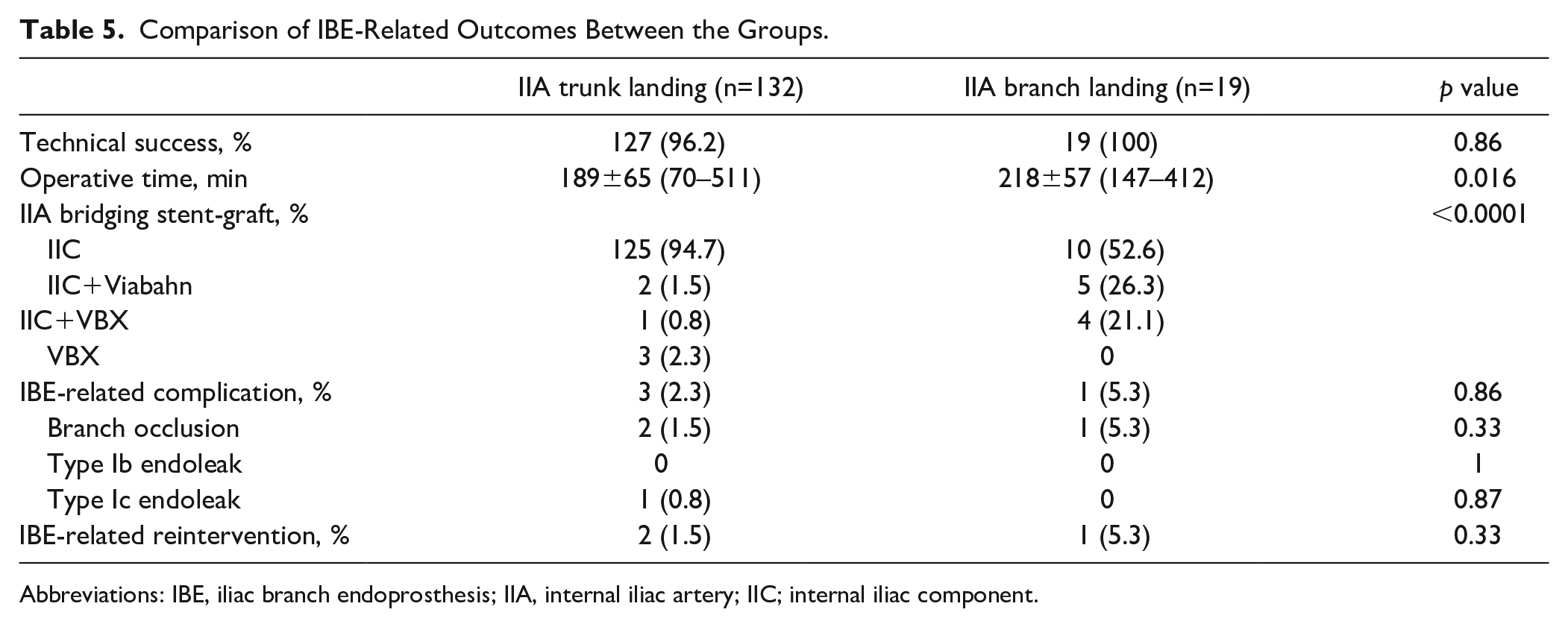
Abbreviations: IBE, iliac branch endoprosthesis; IIA, internal iliac artery; IIC; internal iliac component.
Discussion
This is the first report of a multi-institutional study of EVAR using a Gore Excluder IBE device for Japanese patients with aortoiliac aneurysms. This study suggests both early and midterm benefits of IBE for Japanese patients, despite 43% of the IBE implantations having at least one IFU violation.
Two-year outcomes of the IDE trial and 6-month outcomes of the GREAT registry have shown that aortoiliac aneurysm repair with the Gore IBE device is safe and effective, and internal iliac device patency was 93.6% over 2 years in the IDE trial. 7 Thrombotic event reintervention was required in 2 patients with IDE trial (2.1%) and 1 patient with GREAT registry (1.6%). These data are comparable with our results. However, there was a report where the IIA branch patency decreased from 97.8% at 1 year to 88.5% at 3 years. 11 In this report, patients with IIA branch occlusion presented with severe calcification and stenosis at the IIA orifice. There have been other reports stating that the cause of the IIA branch occlusion includes excessive oversizing of the IIA devices, severe calcification, ostial stenosis, and tortuosity.12,13 Indeed, 1 of 3 patients with IIA branch occlusion also had stenosis of the IIA orifice with calcification. Thus, special attention should be paid for patients with these lesion characteristics.
There is still debate regarding the clinical utility of bilateral IBE implantation and selection criteria. Recent studies have reported that contralateral IIA embolization leads to ischemic complications, including buttock claudication and erectile dysfunction.14,15 Our results also showed that 41% of patients with IIA embolization had buttock claudication and only 2.5% of cases in the IBE sides. On the contrary, the pELVIS registry from 8 European vascular centers evaluated the data of 96 patients with bilateral IBE implantation and showed a technical success rate of 100% with low rates of IIC occlusion (2.2%), buttock claudication (5.2%), or erectile dysfunction (2.1%). This high rate of technical success with promising safety is comparable with that of other reports of unilateral IBE implantation. 16 These findings suggest that bilateral IIA preservation may produce better outcomes and we believe that bilateral IBE implantation should be considered when its advantages are beyond the disadvantages such as longer operation time with fluoroscopy time, or higher costs. Specifically, preservation of the bilateral IIA would be advisable for patients with high physical or sexual activities, or for those with previous, concomitant, or planned extensive aortic repair to minimize the risk of spinal cord ischemia when anatomically feasible.16,17
In the present study, 43% of the IBE implantations had at least one IFU violation. A previous study on the anatomic suitability of iliac branch devices for Chinese patients with aortoiliac aneurysms showed that only 20% of cases met the inclusion criteria of either Gore or Cook devices, and IIA diameter was the most common cause of failure. 18 By contrast, our results showed that a smaller proximal CIA diameter was the main reason for being outside the IFU. Other Japanese studies on the anatomical suitability of the Gore IBE also demonstrated that approximately 40% to 50% of patients had a smaller CIA diameter outside the IFU, which was the most common barrier to the use of the IBE.10,19 However, of those patients, only one with a proximal CIA diameter of 11 mm required iliac limb stenting in the present study. Therefore, a small CIA diameter outside the IFU may not affect the clinical outcomes of IBE.
On the contrary, there were only 8% of patients with shorter aortoiliac lengths in our study, although the Japanese are noted to be of a shorter average height. A possible explanation is that patients with shorter aortoiliac lengths may be excluded when bridging the device between the main body and the IBC device cannot be adapted for preoperative planning. Another Japanese study included 29% of patients with shorter aortoiliac lengths outside the IFU, 19 and they were treated by pushing up the bridging device. This may be an option for treating these patients, if anatomically possible. Although various techniques can help in successful IBE implantation, future designs of iliac branch devices are needed to treat a wider range of patients who have IFU violation.
Extension to EIA with IIA embolization was the only option when a large size or aneurysm of the IIA trunk existed before the introduction of iliac branch devices. A recent study evaluated the outcomes of the Gore IBE using the IIA branch compared with the IIA trunk as distal landing zones. 8 There were no significant differences in technical success, mortality, or major adverse events between the groups, although the branch-related reintervention rate tended to be higher in cases of the IIA branch (13%) than in those with the IIA trunk (2%). When extending the IIA device to the IIA branch, device occlusion is concerning because of the longer length of stent-grafts and the small size of the IIA branches. Regarding IIA device selection, a self-expanding stent-graft would theoretically be better suited to kinking or tortuous anatomy, whereas balloon-expandable stent-graft would be favorable for stenosis or hard plaque.20,21 The pELVIS registry evaluated outcomes of different IIA branch devices including self-expanding stent-graft, balloon-expandable stent-graft, and any stent-graft relining with a bare metal stent, 22 and showed no significant differences in survival, branch device patency, or branch-related reintervention. They concluded that the IIA device selection should be determined based on individual anatomy. Our study also showed similar results, although 47% of patients in the IIA branch group used either Viabahn or VBX as additional IIA branch devices, whereas only 2.3% in the IIA trunk group. However, these studies did not include morphological details and issues of appropriate iliac device selection remain. Therefore, further examination is required.
This study has several limitations. First, this study had a retrospective design and, therefore, subject to recall bias. The small sample size, although larger than those in most published IBE reports, in the present study limits statistical methods such as competing risk models.
Furthermore, CT imaging data were not accumulated for one location; thus, additional analysis was impossible. Finally, the mean follow-up period was relatively short, and the long-term outcomes are unknown.
Conclusion
This is the first report of a multi-institutional study of EVAR using a Gore Excluder IBE device for Japanese patients with aortoiliac aneurysms. This study suggests both early and midterm benefits of IBE for Japanese patients, although 43% of the IBE implantations had at least one IFU violation. This study also demonstrated that extending the IIA device into the distal branches of the IIA was acceptable, which may permit extending indications for EVAR of aortoiliac aneurysms to more complex lesions.
Footnotes
Acknowledgements
The authors would like to thank Takuya Tsuruoka, Yohei Kawai, Shuta Ikeda, and Tomohiro Sato, Division of Vascular and Endovascular Surgery, Department of Surgery, Nagoya University Graduate School of Medicine; Koichi Morisaki, Department of Surgery and Science, Kyushu University Graduate School of Medicine; Riha Shimizu, and Yuri Murakami, Department of Vascular Surgery, Saitama Cardiovascular and Respiratory Center; Akiyuki Kotoku and Hiroki Horinouchi, Department of Radiology, National Cerebral and Cardiovascular Center; Hiroyuki Otsuka, Department of Surgery, Kurume University School of Medicine for their contributions to this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: N.F. receives consulting fees from Cook Medical, Endologix, Medtronic, and W.L. Gore. H.N. activities not related to this article and is employed by Dantex. All other authors declare no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
