Abstract
Purpose:
Anastomotic pseudoaneurysms of transplanted kidneys are a very rare complication encountered in less than 1% of cases. They may be devastating, leading to functional impairment, kidney transplantectomy, or death. Treatment has not been standardized, with open surgical repair considered the safest procedure even if it is often complicated by bleeding and graft loss. The purpose of this case report is to describe an endovascular treatment of this condition, consisting of the combination of coil embolization and arterial stenting.
Case report:
A 61-year-old woman developed an anastomotic pseudoaneurysm 2 months after kidney transplantation, causing acute kidney injury related to ab-extrinsic stenosis of the transplant renal artery (TRA) and external iliac artery. The pseudoaneurysm was successfully treated by coil embolization, and the arterial patency was restored by the stenting of TRA and external iliac artery. The patient completely recovered kidney function, and after a 6-month-follow-up, creatinine values were stable with normal renal perfusion.
Conclusion:
Endovascular repair through coil embolization and TRA stenting can be a safe and effective option to treat anastomotic pseudoaneurysm in kidney transplant.
Introduction
Anastomotic pseudoaneurysm following kidney transplant (KT) is a rare complication occurring in less than 1% of cases.1,2 Its consequences can be severe, leading to renal allograft loss or death. Many etiologies have been proposed in the development of these lesions, including infectious contamination, vascular calcifications, arterial injury, or immunological factors. 3 Conventional open surgical repair, and percutaneous and endovascular techniques have been reported in the literature for the treatment of this condition; however, the management is still challenging and there is not a gold standard option.
In this report, we describe the successful treatment of an anastomotic pseudoaneurysm causing ab-extrinsic stenosis of the transplant renal artery (TRA) in a KT recipient by coil embolization and TRA and external iliac artery stenting.
Case Report
A 61-year-old woman with a history of end-stage renal disease due to scleroderma renal crisis received a KT from a deceased donor. The left donor kidney with a single renal artery on an aortic patch and a single renal vein was implanted in the right iliac fossa through end-to-side anastomosis. The patient received basiliximab and methylprednisolone as induction therapy and tacrolimus, mycophenolate mofetil, and prednisolone as maintenance immunosuppression. Two days after intervention, she underwent a re-laparatomy and the kidney was replaced in the right iliac fossa, due to a Doppler ultrasonography (US) showing allograft hypoperfusion. The transplant had a delayed graft function and the patient underwent kidney biopsy that revealed an acute antibody rejection. She was successfully treated with intravenous immunoglobulins and eculizumab and was discharged with a creatinine of 1.3 mg/dL.
At 2 months after transplantation, the patient presented with oliguria and hypertension. Laboratory tests revealed acute kidney injury with serum creatinine of 6.1 mg/dL. Doppler US showed very damped flow signals on the intrarenal blood vessels with low resistant index (RI) (0.40–0.45) and a reduced flow at the TRA associated with high peak systolic velocities (PSVs) at the iliac artery before the anastomosis. A pseudoaneurysm of 22 mm×20 mm originated from the anastomosis and tightly compressed the TRA (Figure 1A and B). A computed tomography (CT) scan confirmed kidney hypoperfusion secondary to an ab-extrinsic arterial stenosis caused by the pseudoaneurysm of a diameter of 22 mm and a neck length of 2.5 mm (Figure 1C). Blood cultures excluded mycotic infection. However, due to positivity at beta-
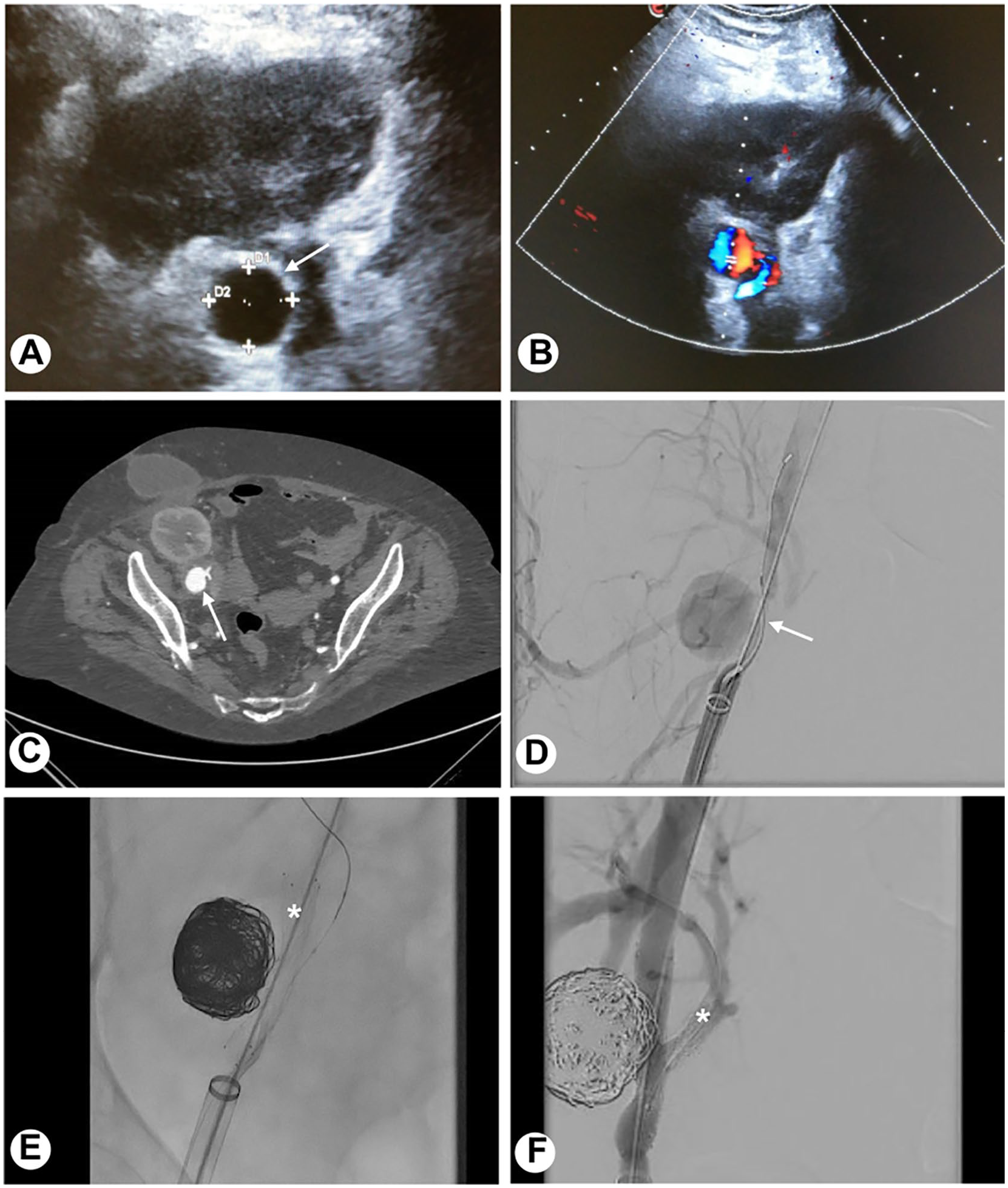
Radiological imaging in the diagnosis and management of anastomotic pseudoaneurysm. (A) B-mode ultrasonography revealed a round-shaped anechoic lesion adjacent to the transplant renal artery (arrow). (B) Color Doppler sonography demonstrated the perfusion within the pseudoaneurysm and the ab-extrinsic stenosis of the transplant renal artery (TRA). Doppler signal in the kidney transplant was very damped. (C and D) Computed tomography (CT) scan and angiography confirmed the anastomotic pseudoaneurysm (arrows) and kidney transplant hypoperfusion due to TRA and external iliac artery stenosis. (E) A stent was placed in the external iliac artery (asterisk) and the pseudoaneurysm was packed with 12 detachable coils. (F) After the deployment of a stent also in the TRA (asterisk), the final angiography revealed the stenosis resolution and the complete occlusion of the pseudoaneurysm.
After a multidisciplinary meeting including nephrologists, infectious disease specialists, vascular surgeons, and transplant surgeons, the patient underwent endovascular intervention. By a surgical cut down on the right groin, the femoral artery was cannulated, followed by a 2500 IU endovenous heparin. Angiography confirmed the anastomotic pseudoaneurysm with the stenosis of TRA and external iliac artery (Figure 1D). A Terumo stiff guidewire (Terumo Corporation, Tokyo, Japan) and a 16F sheath were advanced in the external iliac artery distally to the TRA. The guidewire was maintained in the iliac axis for stent deployment. Coaxially, a 0.014 inch guidewire (Command guidewire; Century Medical, Inc., Tokyo, Japan) was placed into the pseudoaneurysm and a microcatheter (Rebal-18 Micro Catheter, EV3) was advanced. A stent (Protégé RX Self-expanding Peripheral Stent; Medtronic Inc., Minneapolis, Minnesota) of 8 mm diameter and 40 mm length was deployed at the stenosis and the pseudoaneurysm was embolized by 12 micro-spirals (Concerto Coils; Medtronic Inc.) through a microcatheter (Figure 1E). Through the cells of the stent placed in the external iliac artery, the TRA was cannulated by a 0.014 inch guidewire (Command guidewire; Century Medical, Inc.), and after cell dilatation with a 3 mm balloon (Armada balloon; Abbott, Chicago, Illinois), a stent (5 mm diameter and 18 mm length, RX Herculink Elite Renal Stent System; Abbott) was deployed in the TRA, with resolution of the stenosis and perfusion of kidney.
The angiographic control showed the complete occlusion of the pseudoaneurysm and the restored patency of TRA and external iliac artery (Figure 1F).
After the procedure, Doppler US revealed a satisfactory kidney perfusion with RI of 0.75–0.80, a successful coiling of the pseudoaneurysm with no evidence of TRA stenosis. Seventy-two hours after the intervention, the diuresis improved and the kidney function started to ameliorate, until a complete recovery of creatinine values (1.2 mg/dL). After completion of 2 weeks of antimycotic therapy, the patient was discharged. Inflammatory markers were negative and the kidney function was good. A double antiplatelet therapy (clopidogrel and aspirin) was administered for 3 months; then, she was maintained with aspirin monotherapy.
At 6-month follow-up, creatinine values were stable at 1.2 mg/dL. The patient was evaluated with both CT scan and Doppler US. The CT scan shows the patency of the stents and the persistent occlusion of pseudoaneurysm. At Doppler US, kidney perfusion was normal, with RI of 0.60–0.65 and PSV of 100 cm/s.
Discussion
Vascular complications after kidney transplant are rare and include TRA stenosis or thrombosis, and transplant renal vein thrombosis. 2 Pseudoaneurysms involving the TRA, the iliac artery, or their anastomosis are uncommon vascular complications and are etiologically classified as infective or noninfective. Infective pseudoaneurysms are the most frequent and are especially related to mycotic infections. 3 Noninfective pseudoaneurysms can result from arterial wall injury or suture defects.4–6
These lesions may be asymptomatic or present with kidney dysfunction, pain at transplant site, or lumbar plexopathy due to compression and malignant hypertension. 7 In the case of rupture, it can lead to hemorrhagic shock and death. 3 Diagnosis is performed by Doppler US and confirmed by CT or magnetic resonance. 1
There are many controversies regarding indications for the treatment of pseudoaneurysms, probably due to their difficult management and the risk of graft loss. In fact, some authors propose a close follow-up for asymptomatic and small pseudoaneurysms, whereas those with a size greater than 2.5 cm or showing a rapid growth or those secondary to infections need to be treated. Treatment options include open surgical repair, percutaneous thrombin injection by US guide, and endovascular treatment with stent-graft or embolization. 4
Surgical procedures may be considered the most prudent treatment option in the presence of hemodynamic instability or in the case of infective pseudoaneurysms, even if they have a high risk of kidney loss due to vascular bleeding or the need of renal artery ligation.1,7
Three case reports have described the treatment of pseudoaneurysms in kidney transplant by percutaneous US-guided thrombin injection. The first case was reported in 2002 by Reus et al, 8 which successfully treated a giant pseudoaneurysm (7 cm in diameter) of the common iliac artery in a patient with a nonfunctioning kidney transplant. In 2006, two other similar cases9,10 have been reported; in both cases, the authors were able to treat the TRA pseudoaneurysms without compromising the kidney function. Thus, the authors believe that this procedure is suitable and safe with the most significant risk identified in the formation of emboli in the systemic arterial circulation, including the TRA. The risk is reduced by placing the needle tip as far as possible from the neck of the pseudoaneurysm and injecting small amounts of thrombin under continuous control with the color Doppler.
The majority of the reports on endovascular treatment involves the use of covered stents alone with good results. However, stenting across an anastomosis can be technically challenging, due to anastomosis angulation. Thus, the risk of graft thrombosis is high.1,5,11,12 The main papers reporting endovascular treatment of extrarenal pseudoaneurysms are summarized in Table 1.
Main Papers Reporting Endovascular Treatment of Extrarenal Pseudoaneurysm.
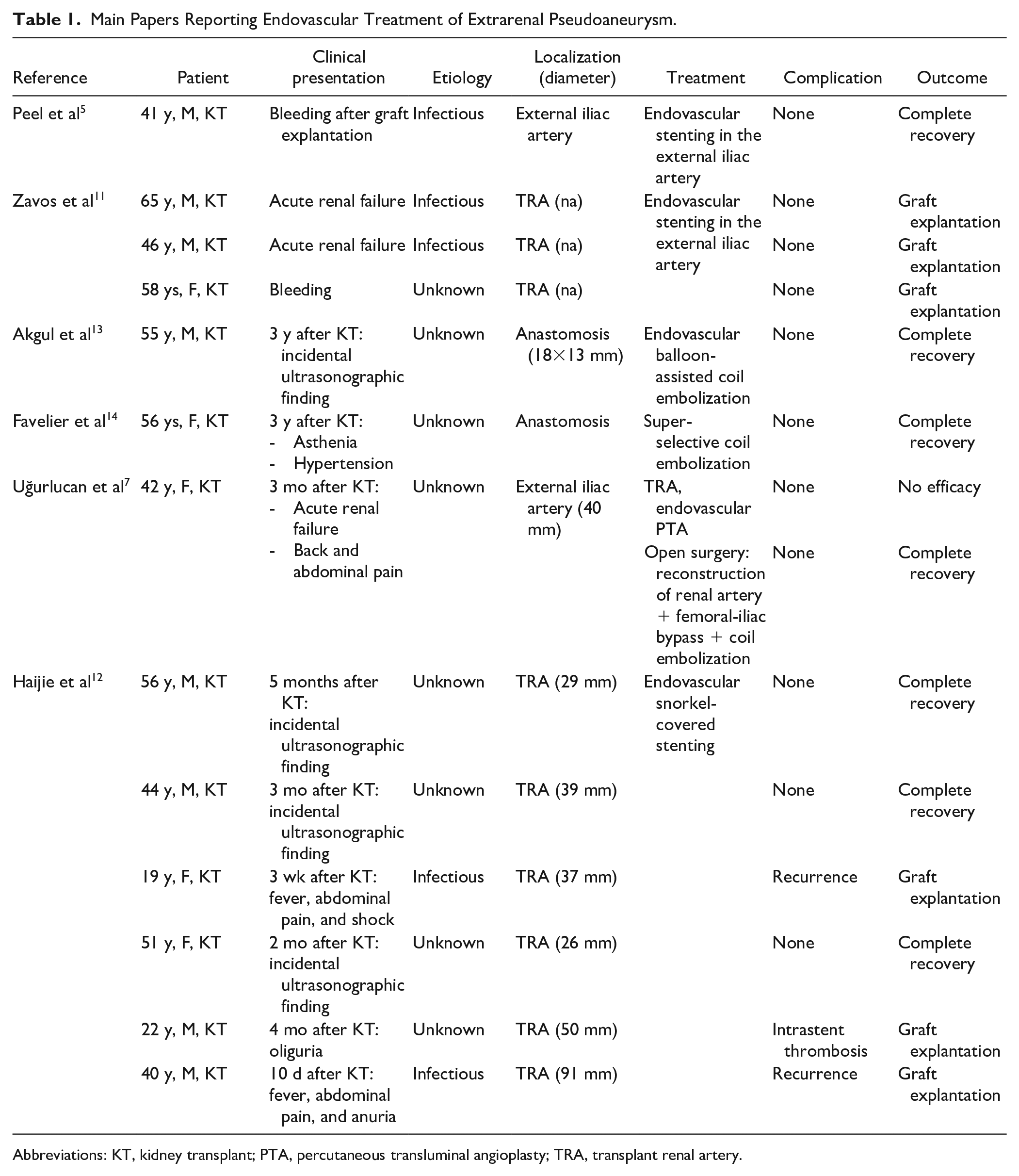
Abbreviations: KT, kidney transplant; PTA, percutaneous transluminal angioplasty; TRA, transplant renal artery.
Detachable coil embolization of pseudoaneurysms in KT has been described in 2012 by Favelier et al. 14 The authors successfully used this endovascular technique in a patient who developed a wide-necked anastomotic pseudoaneurysm 3 years after KT. The technique is the same as that used for the treatment of intracranial aneurysms, and it often involves the application of a stent to avoid coil protrusion into the artery lumen. The risk related to this procedure is the possibility that coils packed into a pseudoaneurysm will get loose, unlike in the case of a true aneurysm, because of loose wall. However, data from cases about the treatment of other vascular pseudoaneurysms (ie, cerebral post-traumatic or aortic arch) and from few other patients with pseudoaneurysm of the kidney transplant artery have confirmed the efficacy of coil embolization.7,13,15,16
Our patient had an anastomotic pseudoaneurysm leading to renal and external iliac artery stenosis. Thus, she suddenly developed acute renal failure. We could not rule out that the origin of the pseudoaneurysm was infective even if, at the time of kidney transplant, there were no signs of donor infection and of preservation fluid contamination, and the patient was discharged with negative inflammatory markers, negative microbiology screening, and normal Doppler US. In addition, the patient had at least 3 risk factors for vascular complication related to arterial wall injury or anastomotic suture failure. The first risk factor was re-laparotomy occurred 2 days after transplant, with the repositioning of the kidney in the right iliac fossa due to graft hypoperfusion. 6 The second one is the history of systemic sclerosis, an autoimmune disease with frequent and early macrovascular involvement, due to endothelial disfunction, atherosclerosis, and arterial stiffness. 17 Third, the patient experienced acute rejection, which is a risk factor for acute vascular complications. 6 Considering the hemodynamic stability and the good quality of the kidney, a plan for graft salvage was made. Surgical option was excluded because of high risk of nephrectomy, bleeding, and hemorrhage shock. The percutaneous US-guided thrombin injection was not suitable due to the depth of the lesion and the necessity to correct the arterial stenosis. Thus, we felt that endovascular repair through coil embolization and TRA stenting, associated with empiric antifungal therapy, was a better solution.
Conclusion
In conclusion, anastomotic pseudoaneurysm is a rare vascular complication in kidney transplant, still challenging to treat. Infective origin should always be ruled out even if it is difficult to confirm. Noninfective pseudoaneurysms are rarer but should be considered especially in those patients with vascular or immunological risk factors. In both cases, a conservative approach through endovascular procedures associated with antibiotic or antimycotic therapy can be a safe and effective option.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by research fundings from the Italian Health Ministry.
