Abstract
Fenestrated and branched aortic endograft (F/B EVAR) has become a widely accepted technique in the management of aneurysmal aortic pathology. However, intra-procedurally in some situations, there are F/B that remain unused because of target vessel occlusion or failure to cannulate the target artery. Leaving the F/B open will result in an ongoing endoleak, unless treated at the time. Herein we described the necessary considerations and several endovascular techniques to occlude these fenestrations and branches in this situation.
Keywords
Introduction
Fenestrated and branched aortic endografts (F/B EVAR) are now widely used as the main treatment option for a variety of aneurysmal aortic pathology from atherosclerotic degenerative aneurysm to chronic aortic dissection.1,2 The majority of these endografts are custom grafts where the F/B are designed according to the patient’s anatomy. In some countries, Cook Medical t-Branch® aortic endografts (Cook Medical), Gore TAMBE (Gore Medical), and Jotec E-nside TAAA (Cryolife) are available as off-the-shelf device branches for the renal and visceral arteries.
In most cases, all the fenestration or branches are utilized during the procedure, connecting to the target arteries. However, in some clinical scenarios, not all the F/B are utilized. These include failed cannulation of target artery, interval occlusion of target artery during graft manufacturing, or in the case of the “off-the-shelf” devices; the target vessel is already occluded and hence the branch is no longer needed. Leaving these unused fenestrations or branches patent without occluding them will lead to an endoleak, thereby leaving the aneurysmal pathology untreated.
Our institution has been using F/B devices for over 10 years with over 300 implantations. In this manuscript, we wish to share our experience in the endovascular management of these unused F/B in Cook Medical devices. However, similar techniques could be utilized for other device manufacturers.
Device
Branch Device
There are several manufacturers of branch devices but for illustration purpose we are using Cook Medical t-Branch® device (Figure 1). It is imperative that the operator understand the design of the branches. For Cook Medical t-Branch®, there are two different side branch diameters, 8 mm for the celiac and superior mesenteric artery (SMA) and 6 mm for the renal arteries. The length of the branch is 21 mm for celiac and 18 mm for SMA and renal arteries. Custom devices are also available and may have unique side branch designs but the vast majority are 6 and 8 mm in diameter and approximately 20 mm in length. These branches could be blocked by a variety of devices including:
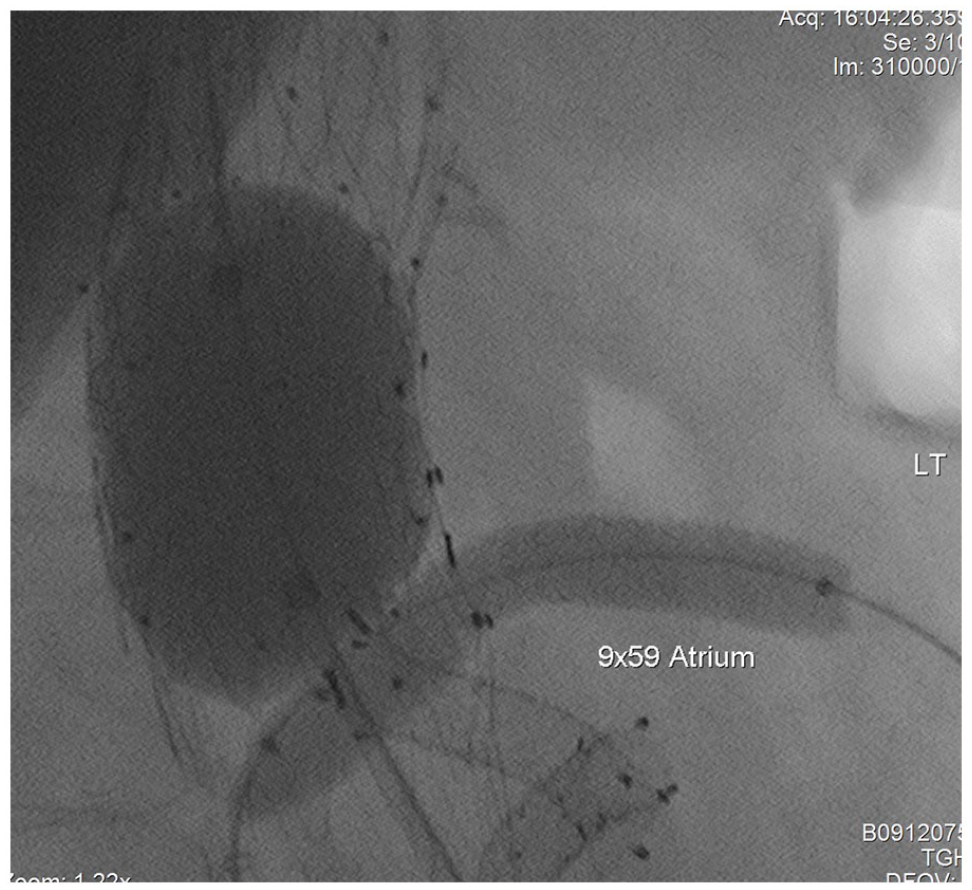
Amplatzer Vascular Plug (St. Jude Medical). This is a nitinol mesh plug with different configurations (Figure 2). From our experience, despite oversizing by 30% to 40%, we encountered 2 out of 6 cases of persistent flow (Figure 3) through the type 1 plug requiring additional embolization with ONYX (Medtronic) liquid embolic agent. Type 2 plug is a multilayered design with better occlusive property but it is longer in length which may protrude outside the branch. 3
MVP microvascular plug (Medtronic) (Figure 4). A 9 mm plug is suitable for both the 6 and 8 mm branch. This device is delivered through a 5F sheath and offers a theoretically superior embolic effect compared with the Amplatzer plug as the MVP is covered with PTFE fabric.
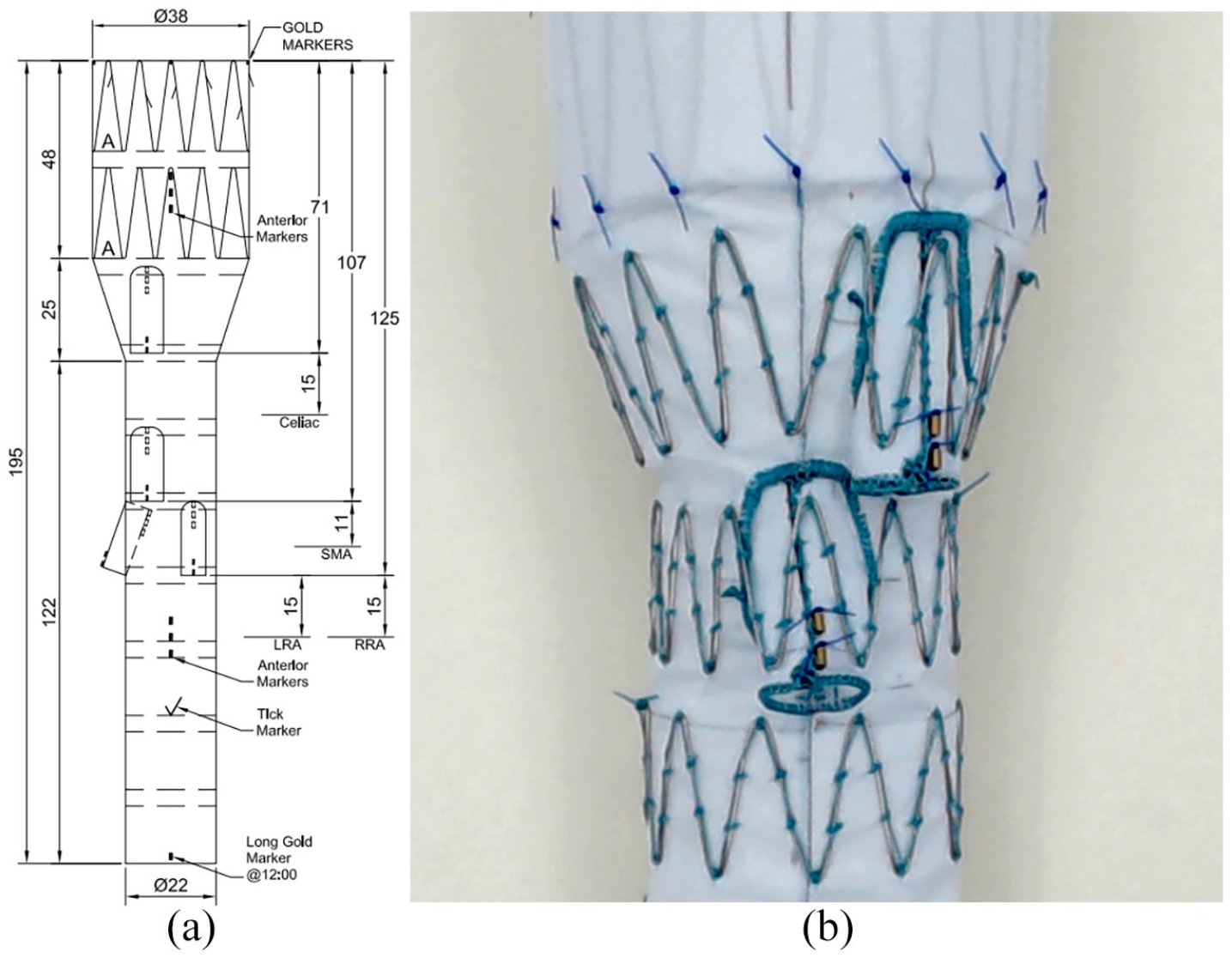
Cook medical t-Branch®. (a) With four side branches for celiac, SMA, and renal arteries. (b) Magnified view of 2 branches.
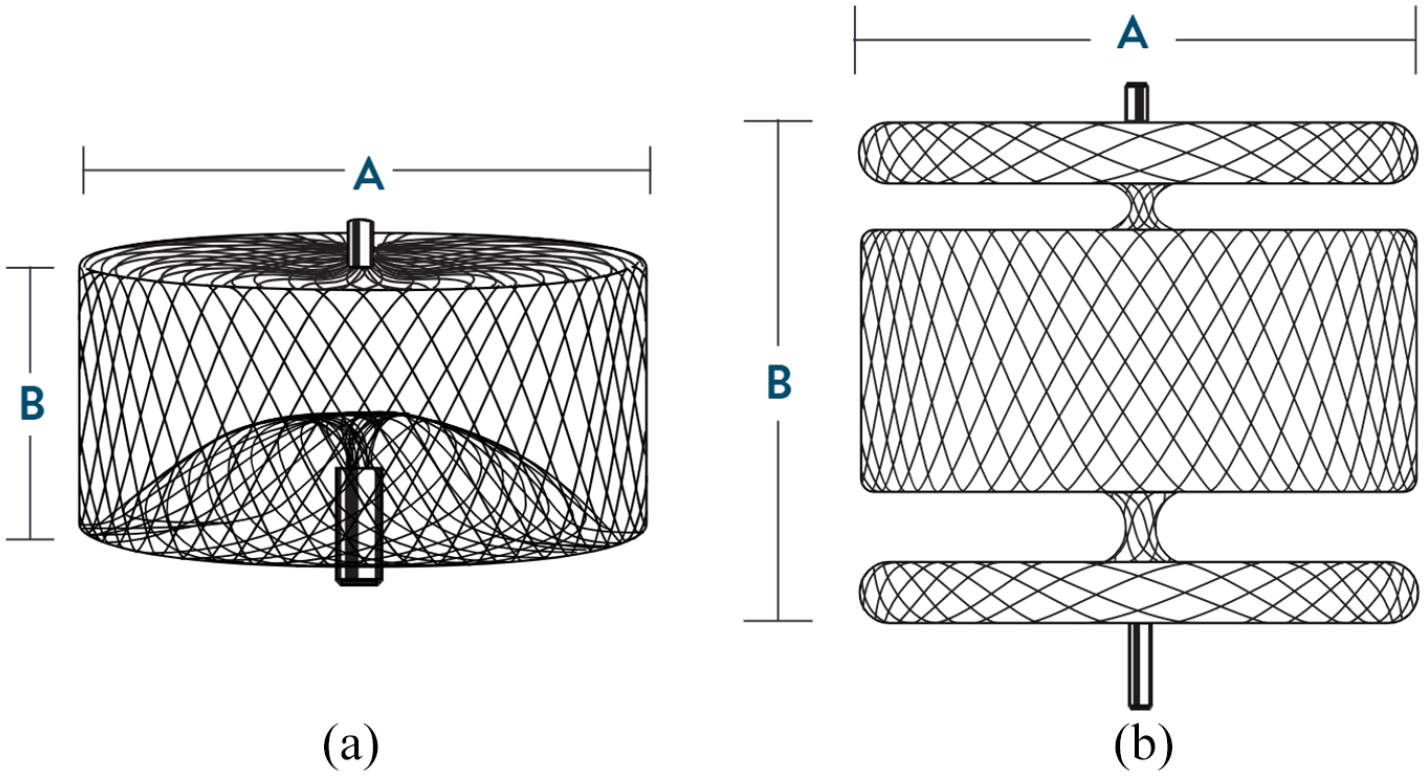
(a) Single lobe Amplatzer Vascular Plug 1. (b) Multilobe Amplatzer Vascular Plug 2.
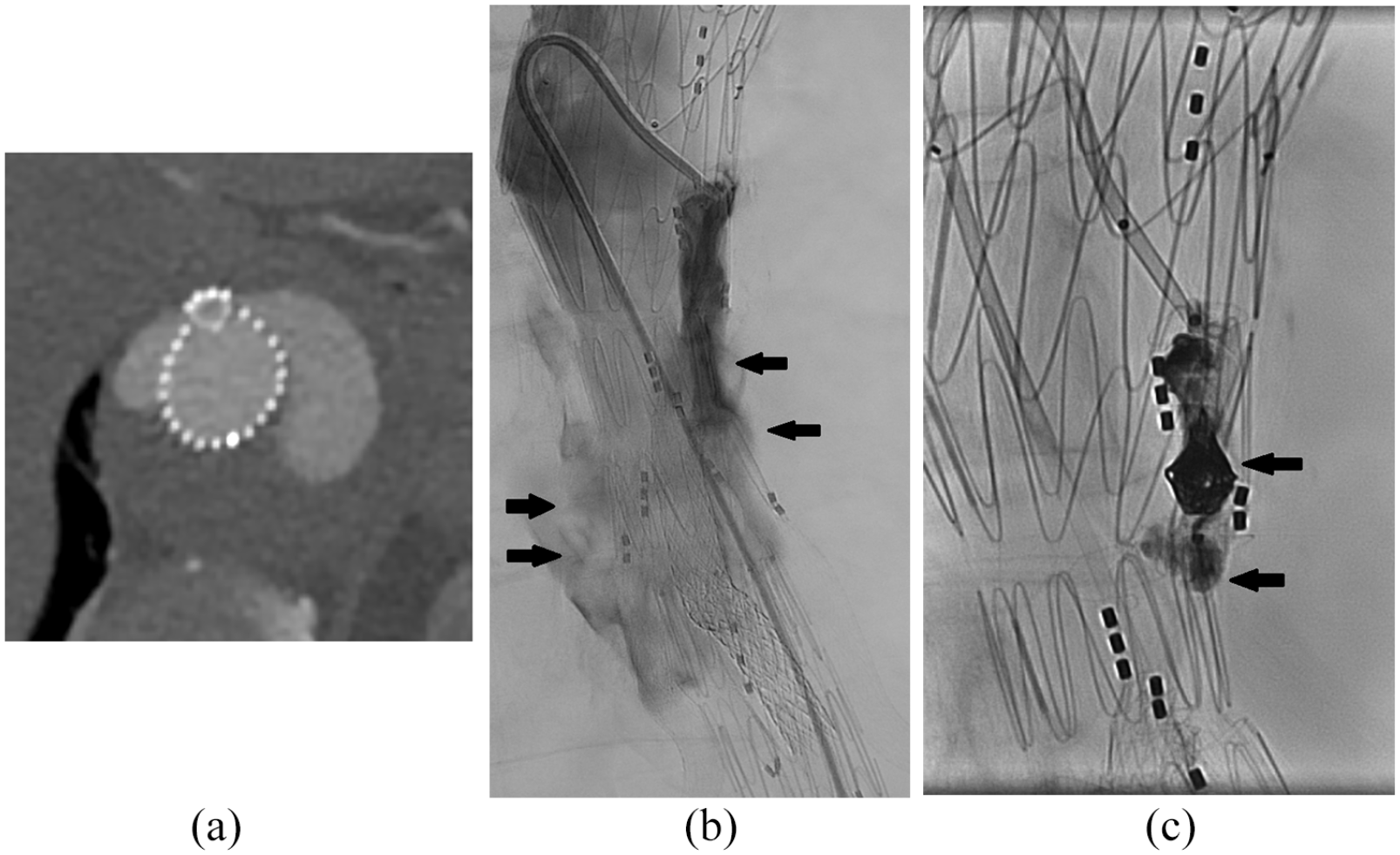
Endoleak through type 1 plug in celiac side branch. (a) Contrast in the plug in CT angiography. (b) Contrast leaking (arrows) into aneurysm sac. (c) Managed by additional embolization with Microcoil and Onyx (arrows).
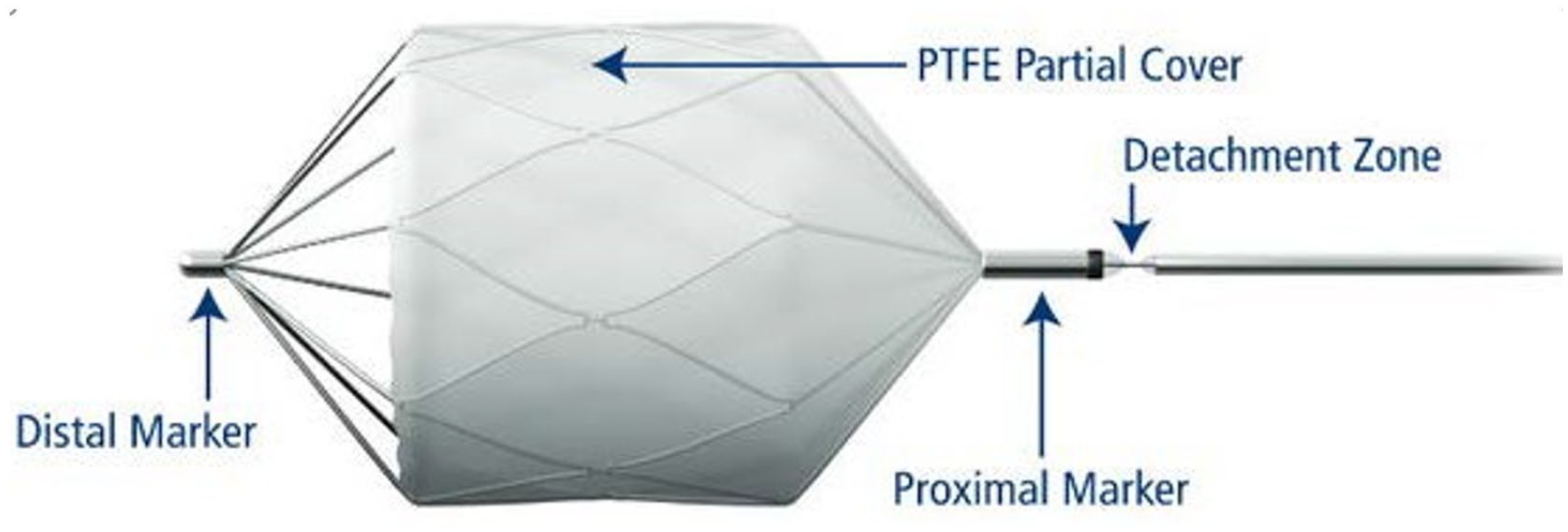
MVP microvascular plug with partial PTFE cover on the proximal cone and body of the device.
Fenestrated Endograft
This device is could be “off-the-shelf” or custom made depending on the patient’s anatomy. The majority of the fenestrations are 6 or 8 mm in diameter. Fenestration with 10 mm is rarely used (Figure 5). In comparison to branches, these fenestrations are harder to block as there is often insufficient space to place conventional plug-shaped occluder devices. The optimal device should not protrude much into the stent-graft lumen and must be stable and have good occlusive property. The possible techniques are:
If there is sufficient length between the fenestrations, an aortic cuff could be placed covering the fenestration without interfering with other fenestration or branches. This scenario is rarely encountered because of the close proximity between the fenestrations with the exception of occasional celiac fenestration which could be >1 cm above the SMA fenestration. In this case, an aortic cuff could be used to cover the celiac fenestration. However, we would recommend placing a protective balloon in the SMA fenestration stent during molding of the aortic extension cuff (Figure 6). If the distance between is celiac and SMA is less than 1 cm, PDA device would be a better choice.
Patent Ductus Arteriosus (PDA) Amplatzer Duct Occluder (Figure 7). This is a nitinol wire mesh with polyester fabric to induce thrombosis. This occluder measures 7 to 8 mm in length (D measurement) depending on diameter chosen. We oversize the device by 2 mm therefore a 6 mm fenestration would need an 8 mm device (A measurement), and an 8 mm fenestration would need a 10 mm device. Placement of this device is feasible if there is 3 to 4 mm of space from the fenestration to the aortic wall. If there was no space, the device could protrude inside the endograft lumen and potentially obstruct blood flow.
Amplatzer Muscular Ventricular Septal Defect (VSD) Occluder (Figure 8). This device has polyester fabric discs on both sides connected by a short waist (C). This device is suitable in cases where there is space between the aortic wall and the fenestration for the external disc to form. Sizing of this device is more complicated in comparison to PDA device. The A disc is the part attached to the delivery wire hence should be located in the stent-graft lumen and the B disc should be in the aortic sac outside the fenestration. A 6 mm waist device has 14 mm disc on both flange and a waist length of 7 mm. This device is suitable for both 6 and 8 mm fenestrations as the discs are 14 mm in diameter. However, if the distance between fenestrations is less than 4 mm, one has to careful that the internal disc does not encroach into the adjacent fenestration.
Placing a balloon expandable covered stent such as Atrium/iCast (Maquet) in the fenestration followed by an occluding device (Figure 9). In this technique, a covered graft is deployed from the fenestration into the aneurysm sac. The lumen of the graft is then subsequently embolized with a vascular plug. 4 This technique is useful if there is large amount of space available between the fenestration and the wall of the aneurysm sac. The main advantage is that the devices used are generally available in most vascular/interventional unit. The disadvantages however include: (a) large amount of space needed for placement of stent-graft between fenestration and aortic wall and (b) multistep procedure of stent-graft placement, flaring of stent-graft to prevent type 3 endoleak between the covered graft and the fenestration and then deploying a vascular plug in the stent-graft.
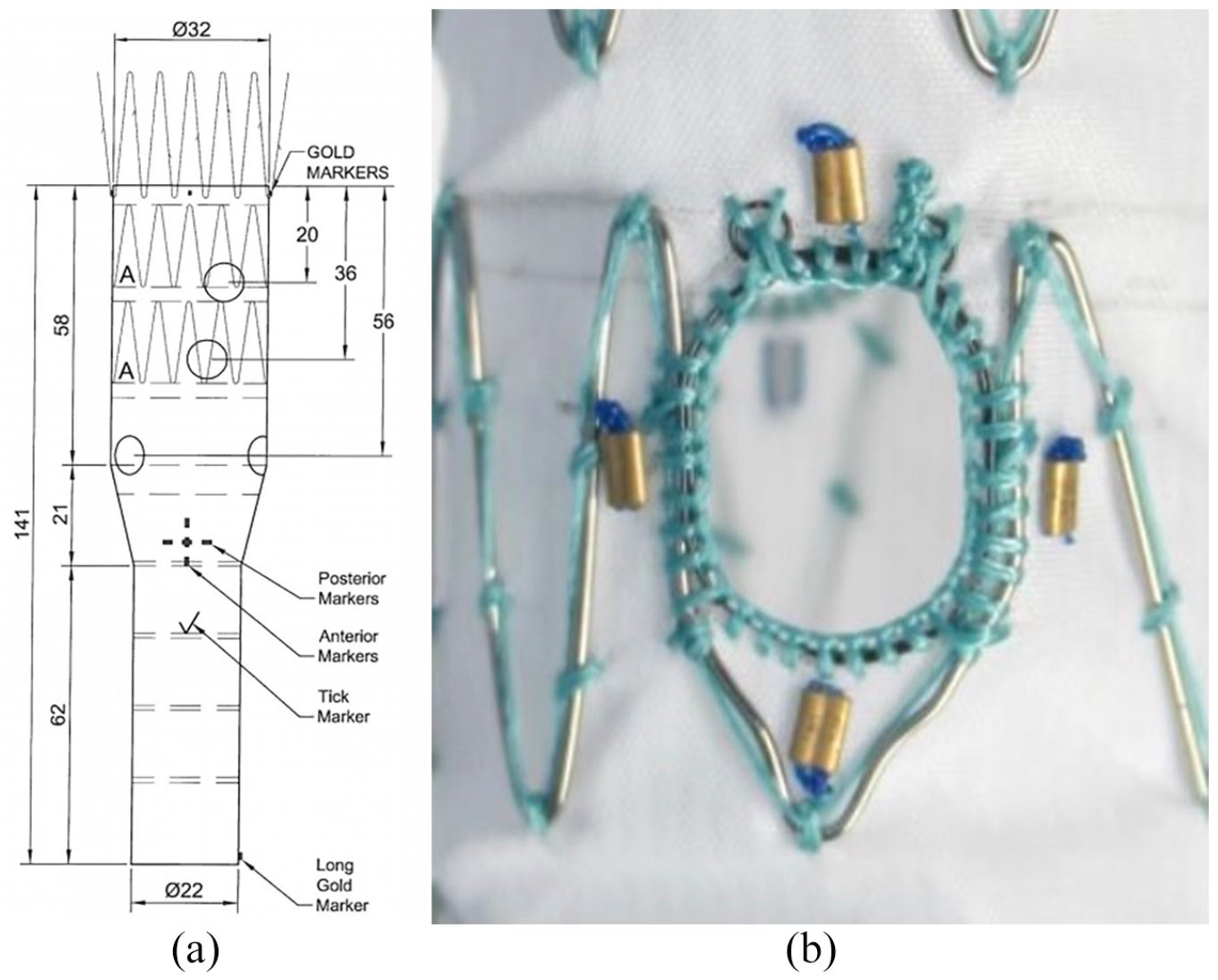
Cook medical fenestrated graft. (a) Four-vessel custom graft plan. (b) Magnified view of a 6 mm × 8 mm fenestration outline by 4 gold marker.
Placement of aortic cuff and molding of cuff with balloon protection of SMA bridging stent.
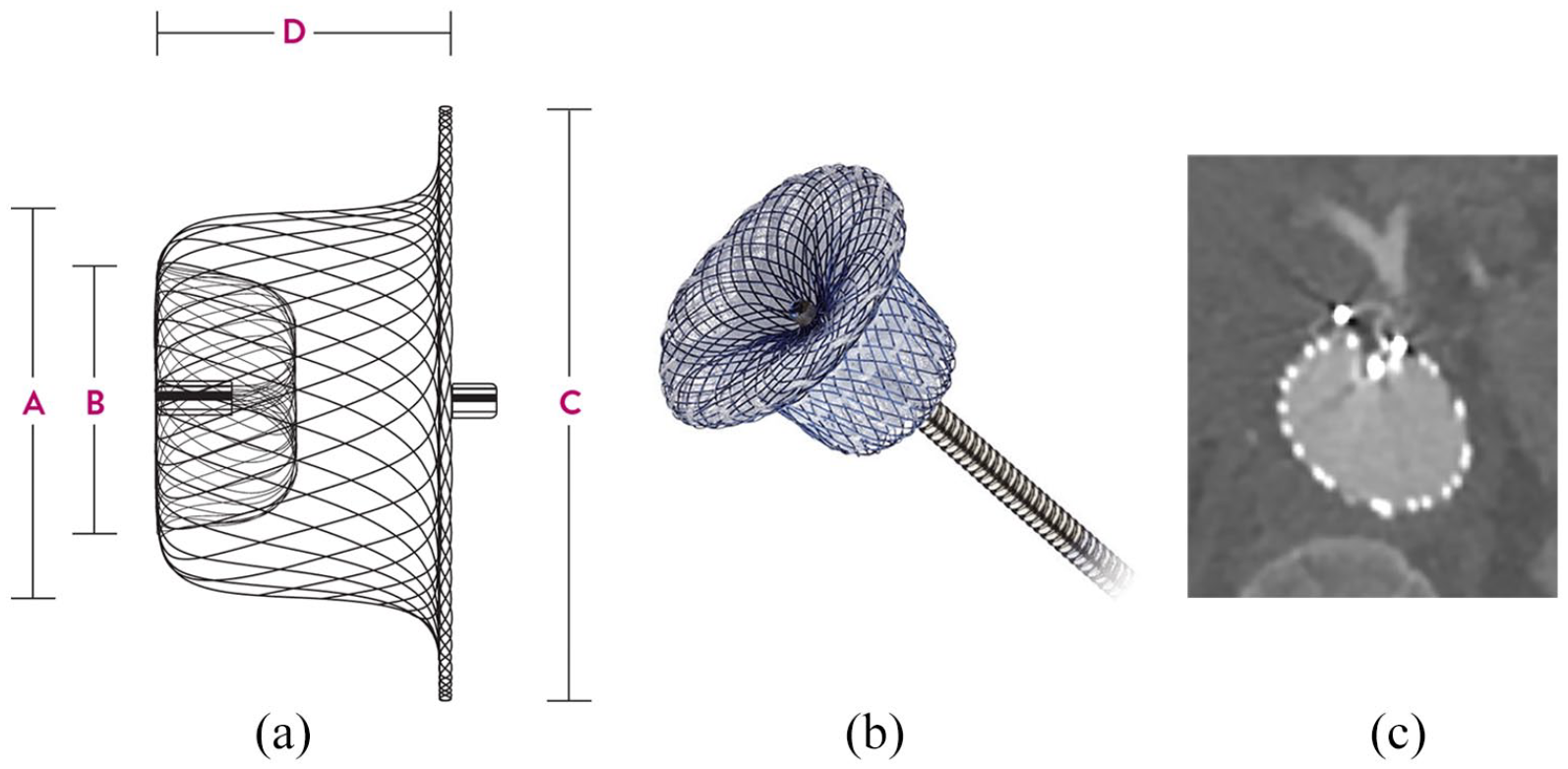
(a) Schematic diagram of the PDA occlude. (b) Magnified view of occlude. (c) PDA occluder in celiac fenestration with the disc component in the sac and the body in the fenestration with small portion protruding into endograft lumen.
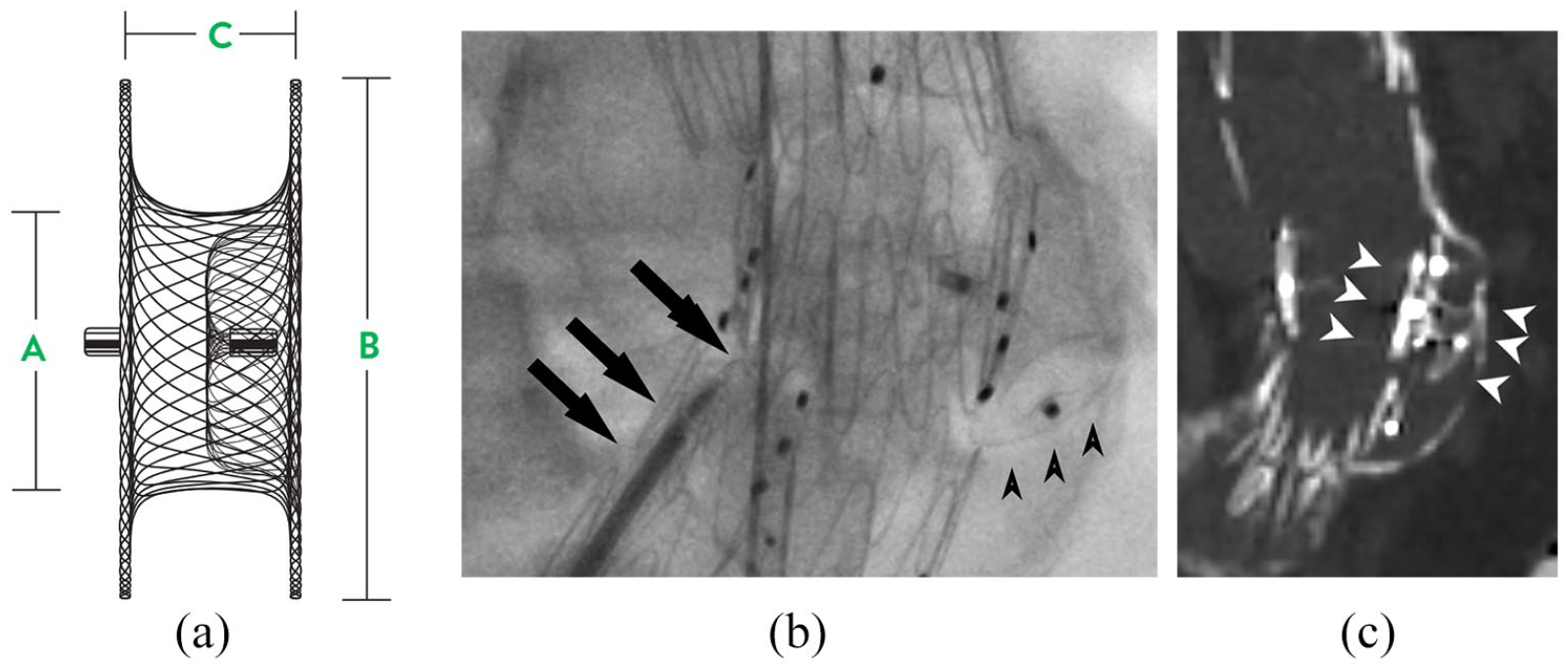
(a) Diagram of the VSD with A represent waist diameter, B disc diameter, and C waist length. (b) Deployment of device in renal fenestration (4 vertical gold marker), arrowhead indicates outer disc, arrow indicates delivery device. (c) CT post showing location of the discs (arrowhead) in relation to aneurysm wall and fenestration.
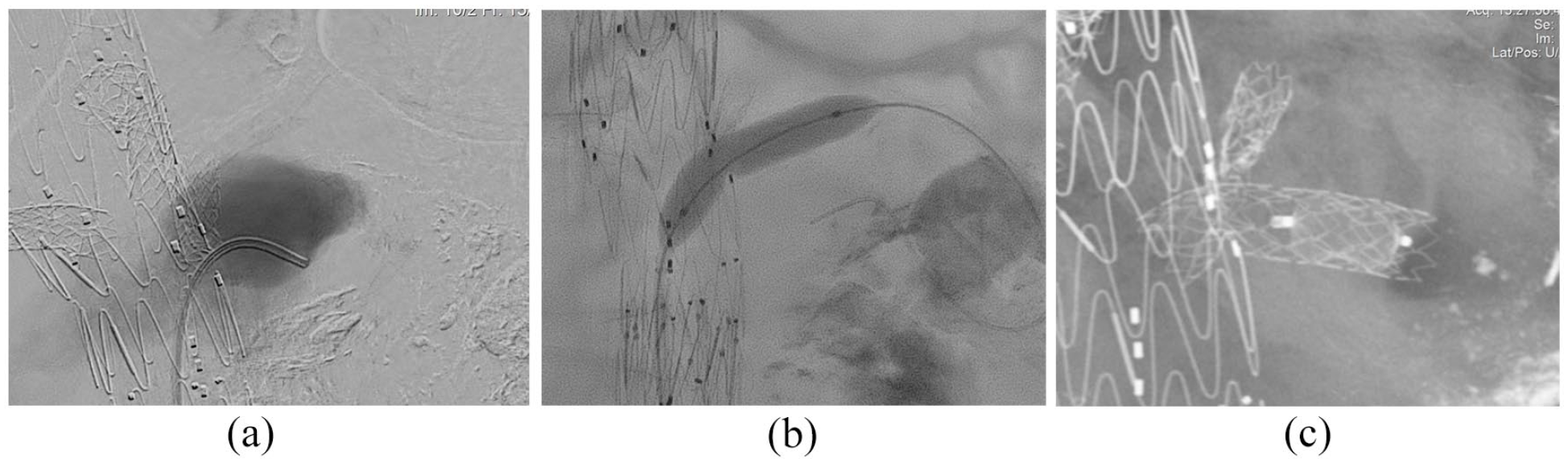
(a) Large endoleak from the renal fenestration secondary to a dislocated stent-graft with occluded renal artery. (b) Placement of a balloon expandable stent-graft. (c) Amplatzer type 1 Plug in the balloon expandable stent-graft.
Discussion
Custom aortic endografts technology is maturing with several medical devices company manufacturing these endografts according to physicians’ requirement. Conceptually, all these devices have similar design based on fenestration or branches of various sizes and diameters. As these devices become more widely available, physicians who implant them must be prepared for unexpected endovascular scenario. One of the challenging situation is failure to cannulate the target vessel and to manage unused fenestration or branch. Currently, there is no specific device that is dedicate for the closure of the fenestration/branch.
For the closure of a side branch, generally there is ample space for the placement of an occluder device. If required, a short stent-graft could be deployed into the branch to extend the overall length prior to placing an occluder device as described by Hongku et al. 3
Occluding a fenestration, however, is much more challenging due to physical property of the fenestration, which is a small ring enforced with a nitinol wire. Conventional Amplazter vascular plug is not suitable as there is no space to place the plug in the ring. We find that cardiac vascular plug is the best device because it has shortest physical length and entirely covered with polyester fabric for complete occlusion of flow. The deployment sequence is similar to conventional Amplatzer vascular plug which most interventionalists would be familiar with. Furthermore, most of these devices could be delivered through a 6 or 7 F vascular sheath. Muscular VSD devices have double discs which sandwich the fenestration. We use this device if there is sufficient space for the external disc to deploy between the aortic endograft and the aneurysm wall. Another alternative is the PDA plug which is a bullet-shape plug encased with polyester fabric. We find that this device is easy to use with good occlusive property.
In rare circumstances, an unstented fenestration might oppose tight against the aortic wall with no endoleaks. In this case, we will not attempt to place a vascular plug into the fenestration. However, from experience, careful monitoring is required, as the neck might dilate over time and hence the fenestration no longer opposed against the aortic wall, resulting in an endoleak.
To date, from our experience, none of the patients had vascular plug failure/recanalization during follow-up once occlusion was achieved.
Summary
As F/B devices are used more frequently for the treatment of aortic aneurysms, interventionalists should be familiar with endovascular options for the management of unused fenestration or branches. Generally, from our experience we find that Amplatzer Vascular Plug are best suited for branch occlusion, and VSD or PDA cardiac plug for fenestration occlusion. Understanding the physical properties and deployment techniques of these devices allow for secure occlusion of unwanted fenestrations and branches.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
