Abstract
The “frozen elephant trunk” is a hybrid technique to treat aortic arch and proximal descending aortic pathologies in a single step. Despite its encouraging early and long-term results, some stent-graft-related adverse events have been reported. Here, we describe a possible treatment option to “re-freeze” the FET in case of loss of landing zone. We report a patient who developed significant kinking of the FET over the course of the first 2 postoperative years. The 1-year follow-up computed tomography angiography (CTA) showed significant kinking and proximal migration of the endograft portion of the FET, resulting in new thrombus formation. Due to kinking and thrombus progression in subsequent CTA follow-ups (2 years and 2½ years) with risk for peripheral embolization, a secondary endovascular repair was indicated. Transfemoral relining of the stent component with a thoracic aortic endovascular repair (Zenith®TX2®30142) stent-graft was performed. In the context of postoperative aneurysm sac shrinkage, the low radial force and lack of longitudinal stiffness of the hybrid graft may lead to proximal migration, thus secondary kinking, emphasizing the importance of an adequate degree of oversizing of the primary graft and an appropriate follow-up. Selection of a suitable graft for a particular pathology concerning the radial force and longitudinal stiffness is furthermore important.
Keywords
Introduction
Over the past 2 decades, the development of a single-step reconstruction of the aortic arch and proximal descending aorta using the frozen elephant trunk (FET) has simplified the treatment of complex aortic arch pathologies that would otherwise have required multiple operations. The introduction of commercially available one-piece hybrid arch FET grafts, such as E-vita OPEN PLUS (JOTEC, CryoLife, Hechingen Germany), the Thoraflex hybrid graft (Vascutek, Inchinnan, UK), and more recently E-vita OPEN NEO (JOTEC, CryoLife, Hechingen, Germany) have reduced the need for secondary procedures and increased the possibilities of treating aortic aneurysms affecting both the arch and the proximal descending aorta in single-stage procedures. Despite promising early and midterm results, these stent-grafts are not exempt from adverse events.1-4 Kreibich et al 5 recently reported 35 cases requiring aortic reinterventions after a FET procedure, with adverse events, including diameter progression (44%), endoleak (23%), distal stent-graft-induced new entry (11%), stent-graft kinking (5%), infection of the FET prosthesis (3%), and lower body malperfusion (3%). A significant number of patients undergoing FET required further endovascular or open surgical procedures, emphasizing the need to differentiate the application of the technique into those intended for single-step long-term durable repairs and those with a significant risk or even anticipated secondary downstream procedures.5,6 This case report presents a rare FET stent-graft-related adverse event, stent-graft kinking after a FET procedure. The aim is to discuss possible factors leading to this adverse event and to present a successful treatment strategy for such stent-graft stability problems after FET.
Case Report
We present a 71-year-old patient with a high cardiovascular risk profile, including arterial hypertension, type 2 diabetes mellitus and hyperlipidemia, who 2 years after undergoing FET with a multibranched hybrid graft (Vascutek Thoraflex 26/28/100 mm for repair of a 5.7 cm aortic aneurysm affecting both the arch and the thoracic aorta, developed significant kinking of the stent prosthesis. Follow-up computed tomography angiography (CTA) showed an early uncomplicated postoperative course with stable results (Figure 1) course of the following year, shrinkage of the aneurysm sac (initial maximum diameter 5.7 cm; 4.4 cm after 1 year) and subsequent kinking of the stent-graft were observed with retraction of the stent-graft (Figure 2A and B). The 2-year follow-up CTA (Figure 2C and D) and 2½ year CTA (Figure 2E and F) showed progressive kinking, with subsequent proximal migration of the endograft portion of the FET into the proximal descending aorta, resulting in new thrombus formation and stenosis. Due to the progression of the kink, as well as new intraluminal floating thrombus formation associated with risk of stent occlusion and peripheral thromboembolism, treatment with thoracic endovascular aortic repair (TEVAR) was indicated following 3 months of oral anticoagulation therapy with a direct oral anticoagulant (DOAC), to stabilize the thrombus and reduce thrombus progression, thereby reducing the possible pre- and intraoperative risk of peripheral thromboembolism.
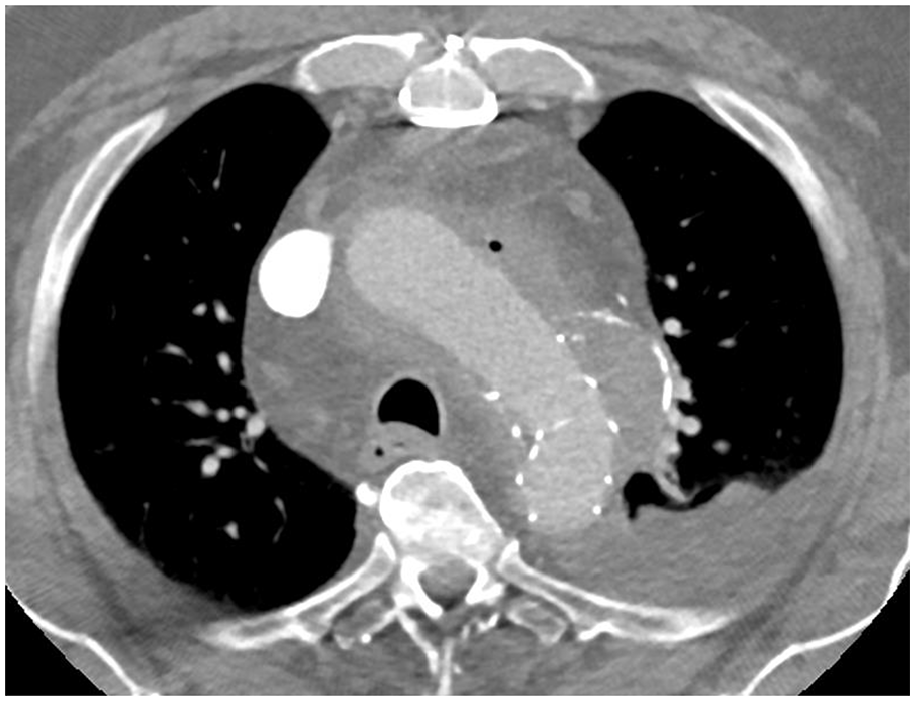
Early-follow-up computed tomography angiography, 10 days after FET showing the 5.7 cm aortic aneurysm affecting both the arch and the thoracic aorta and depicting the original localization of the distal end of the FET.
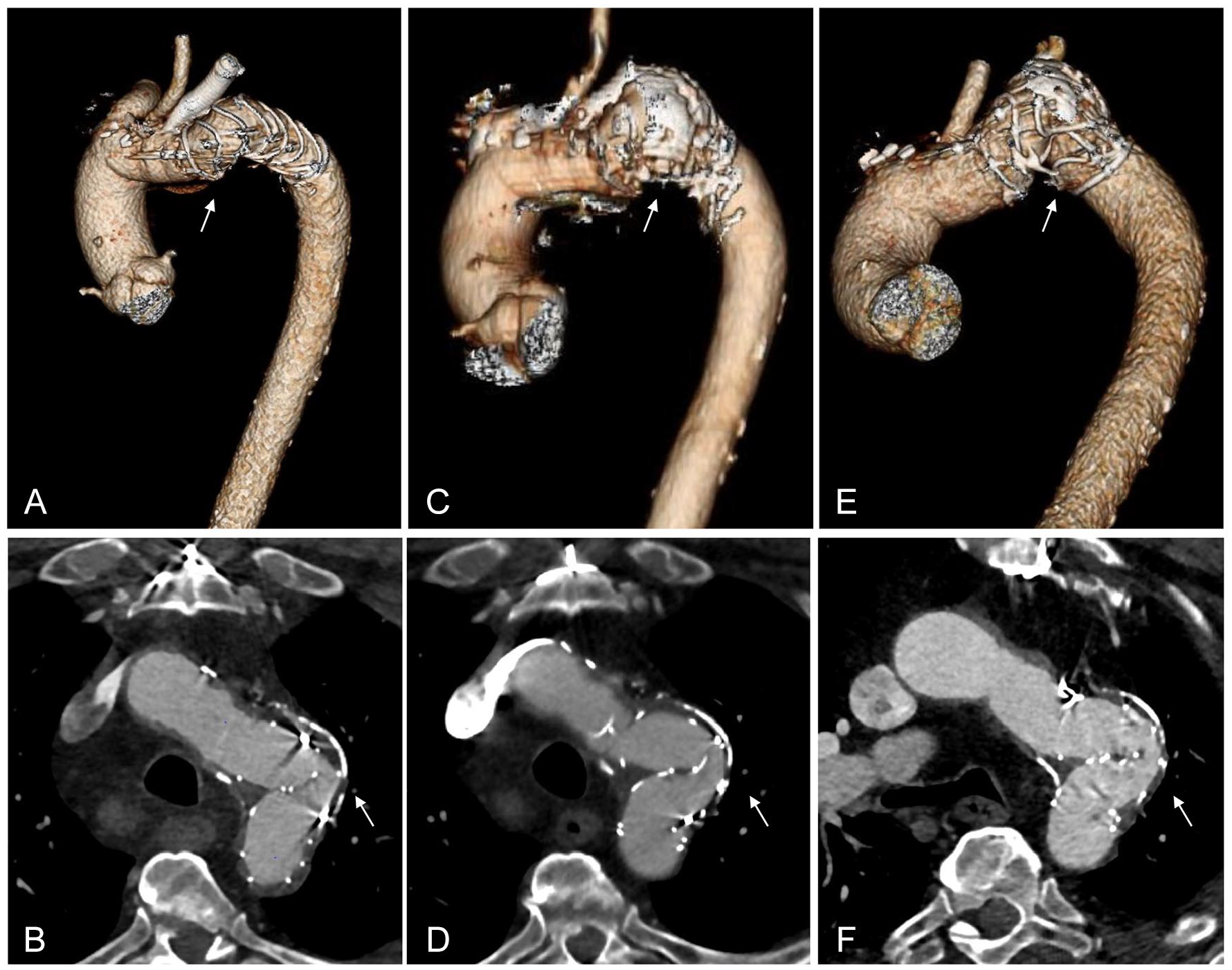
Follow-up computed tomography angiography after the original FET procedure (A) and (B) 1-year follow-up CTA, (C) and (D) 2-year follow-up CTA, and (E) and (F) last CTA before endovascular treatment showing the progression of the kinking over 2 years. The white arrow shows kinking of the Thoraflex hybrid graft (Vascutek, Inchinnan, UK).
Ultrasound-guided percutaneous puncture of both femoral arteries was performed. Two percutaneous closure devices were applied in the right femoral artery, whereas the left femoral artery was used for a 5Fr angiographic catheter. Systemic heparinization was administered until an activated clotting time of >250 seconds was achieved.
After correctly cannulating the kinked FET and placing of an extra-stiff double-curve thoracic Lunderquist® (Cook Medical, Blooming, IN, USA) in the ascending thoracic aorta, the kinked portion of the FET was pulled down and ballooned twice with a 46 mm compliant balloon (Cook Medical, Bloomington, IN, USA) (Figure 3A and B) in order to straighten the graft fabric, thereby increasing the available length graft length and finally increasing the potential overlap between the FET and the TEVAR. Additionally, besides increasing the overall proximal landing zone length, we wanted to avoid landing the TEVAR in an area with a high curvature to avoid bird-beaking and malposition. After ballooning, the FET was then relined with a 30 mm proximal diameter and 142 mm long Zenith® TX2® (Cook Medical, Bloomington, IN, USA) (Figure 3C) to support the longitudinal stiffness of the hybrid Thoraflex endoprosthesis (Vascutek, Inchinnan, UK) and increase the radial force. Intraoperative angiography showed a satisfactory result with expansion of the kinked portion of the FET and an improved wall apposition (Figure 3D).
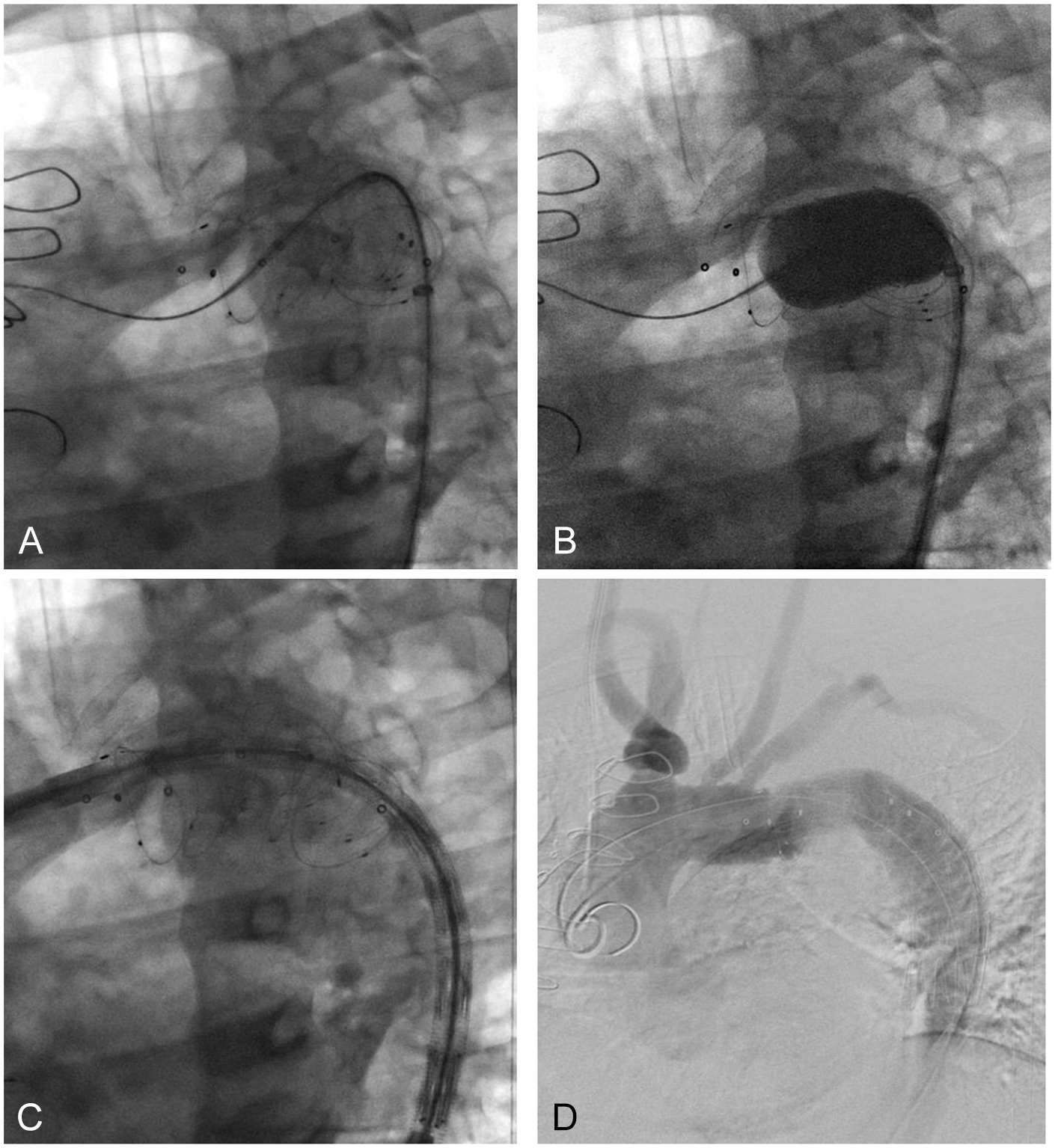
Intraoperative angiogram (A) introducing a 46 mm compliant balloon in the kinked proximal descending aorta. (B) Dilatation of the kinking twice. (C) Introducing the thoracic endoprosthesis device in the descending proximal aorta. (D) Final admission with the thoracic endoprosthesis, Zenith TX2 dissection endovascular graft (ZDEGP-30-142-PF) in the descending aorta.
The sheaths and endovascular material were then removed. Hemostasis of the right femoral artery was achieved through the percutaneous closure devices, and manual compression of the left femoral artery was performed. The procedure time was 49 minutes and was performed without any cardiac output reduction technique given a proximal landing zone in Ishimaru Zone 3. The patient remained during the entire procedure under general anesthesia cardiopulmonary stable.
The patient was taken to the intensive care unit, where he remained for 24 hours. Two weeks following the procedure, the patient who had been taken off the DOAC presented with a deep vein thrombosis and pulmonary embolism, which were managed with anticoagulation therapy and compression stockings, without additional complications. The early control CTA showed correct expansion of the FET, without any recurrence (Figure 4).
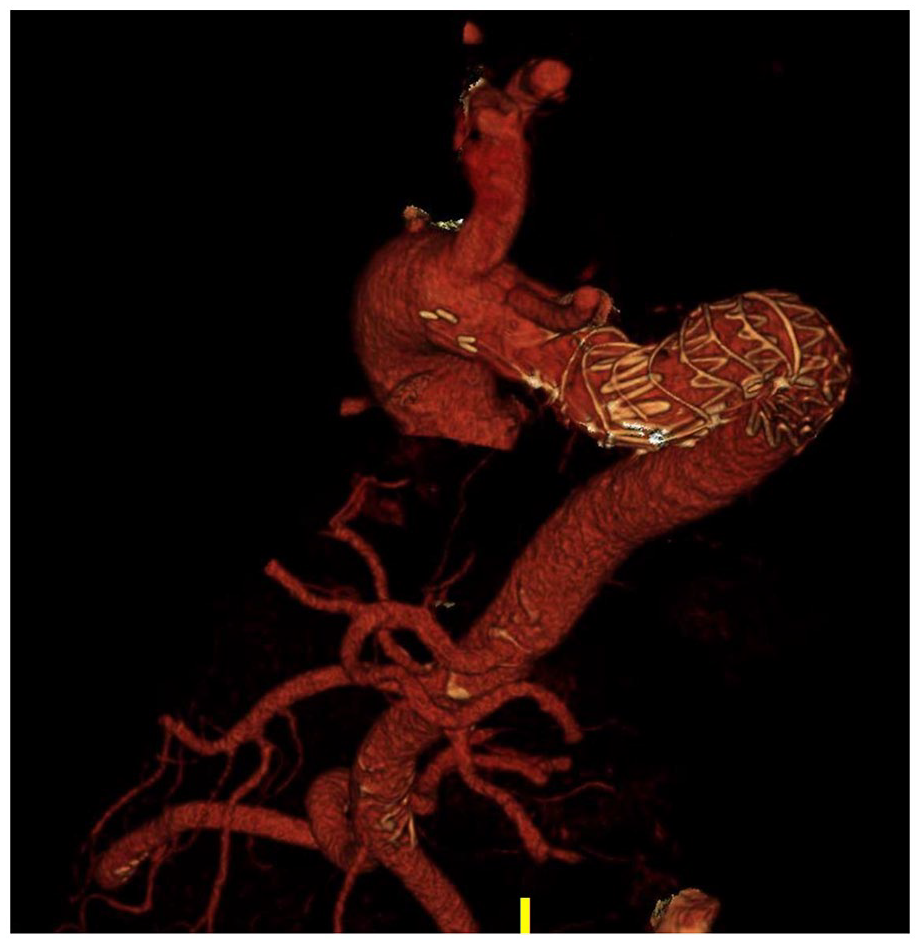
Early follow-up computed tomography angiography, 15 days after implantation of a thoracic endoprosthesis, Zenith® TX2® dissection endovascular graft (Cook Medical, Bloomington, IN, USA) (ZDEG-P-30-142-PF) in the descending aorta.
Discussion
In general, the FET is indicated for both aneurysms and dissection. Of course special consideration needs to be given to the anatomy of the proximal descending aorta where the stent of the FET is to land, to account for special features of the stent of the FET and avoid late complications. Resch et al 7 have associated the proximal migration and the subsequent possible kinking of the distal end of a stent prosthesis placed in the thoracic aorta with vector forces acting in this area, and to morphological changes in the aneurysm sac after endovascular aneurysm exclusion. Due to its curvature, fixation to the thoracic wall and location near the heart, the proximal descending thoracic aorta is subject to more dynamic and unstable forces as compared to the abdominal aorta. Accordingly, increased vector forces are exerted on stent- grafts implanted in this area, so the need for an adequate radial force to overcome the external tensions generated in this area and achieve sufficient stability becomes even more important. Thus, the question arises: May a lack of radial force lead to thawing of the “frozen elephant” in certain cases such as the one presented here?
In order to be able to answer this question, the stent design, on which the radial force depends, should be examined in more detail. The Thoraflex hybrid graft (Vascutek, Inchinnan, UK), used for treatment of the patient herein presented, comprises of a proximal 4-branched gelatin-sealed prosthesis for the reconstruction of supra-aortic vessels (3 branches for the supra-aortic trunks and 1 perfusion side branch) and a distal nitinol supported (ringed) Dacron stent-graft for the repair of proximal descending aorta. The arch and proximal part of the hybrid prosthesis, which is fixed by a proximal and a distal anastomosis, may be more stable than the distal stent-graft of the hybrid prosthesis, which is only held in place by the radial force provided by the nitinol rings in the descending thoracic aorta. The unique design of the stent-graft with the nitinol rings offers an excellent adaptation in the area of the aortic arch, but has a lower radial force compared to conventional Z-shaped nitinol stents. 1 This has been seen as considerable advantage in acute aortic dissection. However, in aneurysmal disease, the lower radial force may be too small to counteract the vector forces in the area of the distal stent-graft, leading to the destabilizing effect of postoperative aneurysm sac regression. This may have led to the kinking of the stent-graft in this case. In order to ensure stability of the stent-graft and prevent migration and kinking, a sufficient radial force against external vector forces should be provided by an adequate size hybrid prosthesis selection, taking into consideration the degree of oversizing, the ring design, and the properties of the target vessel to be treated, as reported by Senf et al. 8 The Thoraflex hybrid graft (Vascutek, Inchinnan, UK) is available in different sizes (distal FET diameters from 26 to 40 mm and lengths from 100 to 150 mm), which enables an individual selection of the prosthesis size for each patient. Recommendations, including ring design, are currently limited. Accordingly to early clinical outcomes, Chu et al 1 recommended a graft-oversizing of 10% to 20% and a distal sealing zone of 3 to 4 cm for a successful exclusion of thoracic aneurysm with Thoraflex hybrid graft (Vascutek, Inchinnan, UK). This is in parts explained by the fact that a ring stent does not exert its radial force in a linear fashion with the achieved percentage of the nominal diameter.
Resch et al 7 further suggested that the fixation of 3 longitudinally interrupted interconnecting wires in the stent-graft may provide additional longitudinal support and thus represent a possible solution for reducing migration and kinking of a stent-graft with Z-shaped stainless-steel stents. These connecting wires could also be considered for the distal nitinol-ringed stent portion of the Thoraflex hybrid graft (Vascutek, Inchinnan, UK) to increase the stability in the longitudinal axis.
Another consideration while treating patients with pathologies including the distal aortic arch is to timely extend the repair with a more distal repair to the FET ahead of the occurrence of possible complications. Despite this being a matter of debate, Czerny et al 9 recently described how the FET stent resulted in distal stent-graft-induced new entry (dSINE) in 5.3% of patients in the acute setting and in 6.5% of patients in the chronic setting. Kozlov et al, 10 concluded in their comparative study of patients who underwent FET versus extended FET with additional TEVAR within 30 days of the index procedure, that more favorable remodeling was achieved downstream of the graft reaching as far as the abdominal aorta in the second group. Low-risk secondary procedures could prevent complications, such as the one described in this case report. This strategy could even potentially reduce the need for more extensive distal repairs, such as fenestrated-branched repairs, which are always associated with increased morbidity and mortality. 11
Despite satisfactory early post-interventional results, as in this case, TEVAR can cause stent-graft-related or systemic complications in up to 38% of cases, including endoleaks in up to 4% to 15% of the cases, and much less frequently endograft migration (1%–3%). 12 Another relevant systemic complication associated with TEVAR is spinal ischemia, which can occur in up to 12% of cases in the early post-interventional phase, requiring an adequate risk stratification of spinal ischemia prior to TEVAR. The degree of aortic coverage, prior infrarenal aortic repair, coverage of left subclavian artery, long procedure durations, and renal insufficiency are the risk factors associated with high risk for spinal ischemia. In this case, preoperative spinal fluid drainage was not indicated, given adequate perfusion of all supra-aortic trunks and short aortic coverage. 12 Finally, the frequency of late endograft-related events after TEVAR makes lifelong imaging follow-up necessary. However, determining the adequate follow-up regiment for young patients, such as this case, can be difficult. In this patient, we recommended a post-interventional CTA at 30 days and then yearly thereafter for the first 3 years. If correct apposition of the FET and TEVAR is observed, after 3 years we will increase the time span between follow-up CTAs.
Conclusion
Thoracic endovascular aortic repair (TEVAR) offers a successful treatment strategy to resolve stent-graft stability problems following the FET.
Footnotes
Authors’ Note
This work has not yet been presented elsewhere.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
