Abstract
Purpose:
The Medyria TrackCath Catheter (MedTCC) is an innovative, thermal convection–based blood flow velocity (BFV) tracking catheter that may be used during complex aortic endovascular procedures for identification and catheterization of target orifices. The ACCESS Trial analyzes the safety and performance of the MedTCC for targeted vessel catheterization to generally evaluate the feasibility of thermal convection–based BFV.
Materials and Methods:
We performed a first-in-human, proof-of-concept, prospective single-arm multicenter clinical trial between March 2018 and February 2019 in patients who underwent endovascular aortic procedures at 4 high-volume centers. During these procedures, the MedTCC was advanced over a guidewire through the femoral access. The D-shape was enfolded in the reno-visceral part of the aorta and target orifices were identified and catheterized with a guidewire via the side port of the MedTCC through BFV tracking. BFV measurements were performed at baseline (Baseline-BFV), alignment to the orifice (Orifice-BFV), and following catheterization (Confirmation-BFV) to prove correct identification and catheterization of target orifices. The procedural success rate, the catheterization success rate, procedure-related parameters, and (serious) adverse events ((S)AE) during the follow-up were analyzed.
Results:
A total of 38 patients were included in the safety group (SG) and 26 in the performance group (PG). The procedural success rate was 89% (PG), the MedTCC catheterization success rate was 98% (PG). The MedTCC reliably measured BFV changes indicated by significant differences in BFV between Baseline-BFV and Orifice-BFV (p<0.05). Median (interquartile range; IQR) fluoroscopy time per orifice was 5.0 (1.5–8.5) minutes [total surgery 49 (26–74) minutes], median (IQR) contrast agent used per orifice was 1.0 (0–5.0) mL [total surgery 80 (40–100) mL], and median (IQR) MedTCC-based procedural time was 3.0 (2.0–6.0) minutes. There was no device-related SAE.
Conclusions:
The ACCESS Trial suggests that BFV measurement allows for reliable target orifice identification and catheterization. The use of MedTCC is safe and generates short fluoroscopy time and low contrast agent use, which in turn might facilitate complex endovascular procedures.
Introduction
Complex aortic procedures remain challenging. As such, thoracoabdominal (TAAA) and/or short-necked, juxtarenal, and suprarenal abdominal aortic aneurysms (AAA) are increasingly treated by fenestrated (fEVAR) or branched (brEVAR) stent-grafts constituting the sealing zone into a more proximal and stable aortic segment.1–3
In general, the catheterization of target vessels such as renal, visceral, and lumbar arteries is involved in various complex aortic procedures. For instance, to yield satisfying procedural success rates, fEVAR and/or brEVAR requires renal and/or visceral artery catheterization to deliver bridging stent-grafts into the target vessels. 4 Other aortic procedures, such as type II endoleak prevention with coil embolization may also require reliable native target vessel catheterization. 5 Furthermore, novel approaches, such as contrast-free duplex-assisted EVAR, also involve the catheterization of the renal arteries with a guidewire. 6 For all the aforementioned procedures, the identification and catheterization of the target vessels can be challenging due to the diversity of anatomies and/or stenotic vessels necessitating prolonged fluoroscopy time and high volumes of contrast agent. 7
Despite the constantly expanding application of complex endovascular aortic procedures, exposure to radiation and contrast dye are potentially harmful, as they increase the lifetime cancer risk for patients and the surgical team and can cause renal impairment.8,9 Thus, the development of new devices is warranted aiming to reduce both radiation exposure and volume of contrast agent, while providing a safe and reliable method of identifying and cannulating target vessels during complex endovascular aortic procedures.
The ACCESS (Clinical application of the Medyria TrackCath system in endovascular repair of abdominal aortic aneurysms) Trial (ClinicalTrials.gov Identifier: NCT04190979) is a first-in-human, proof-of-concept, multicenter clinical trial investigating the safety, performance, and overall feasibility of thermal convection-based blood flow velocity (BFV) measurement as a novel concept for target vessel identification using the Medyria TrackCath Catheter (MedTCC) in patients with TAAA or complex AAA. The study also aimed at analyzing specific procedure-related outcome parameters, such as the procedure duration, the contrast agent consumption, and the radiation exposure.
Materials and Methods
The Medyria TrackCath (MedTC) System
The MedTC System is composed of the MedTCC and the MedTC Equipment. The MedTCC is a D-shaped catheter with nonhydrophilic coating that provides a pathway for delivering contrast agent, guidewires, and catheters to selected target sites in the aorta. The length of the MedTCC is 860 mm (sensor position) and has options for 2 D-shaped dimensions of 30 mm and 50 mm width (the ACCESS Trial used the 30 mm version of the MedTCC), which potentially allow for delivering the catheter through an 8F sheath over a transfemoral and/or transaxillary access. The MedTCC has an inner diameter of 4F, and guidewire delivery can be performed via a 4F side port (Figure 1A). The MedTC System is used during endovascular procedures which require identification and catheterization of aortic side branches and can be combined with modern fusion imaging.
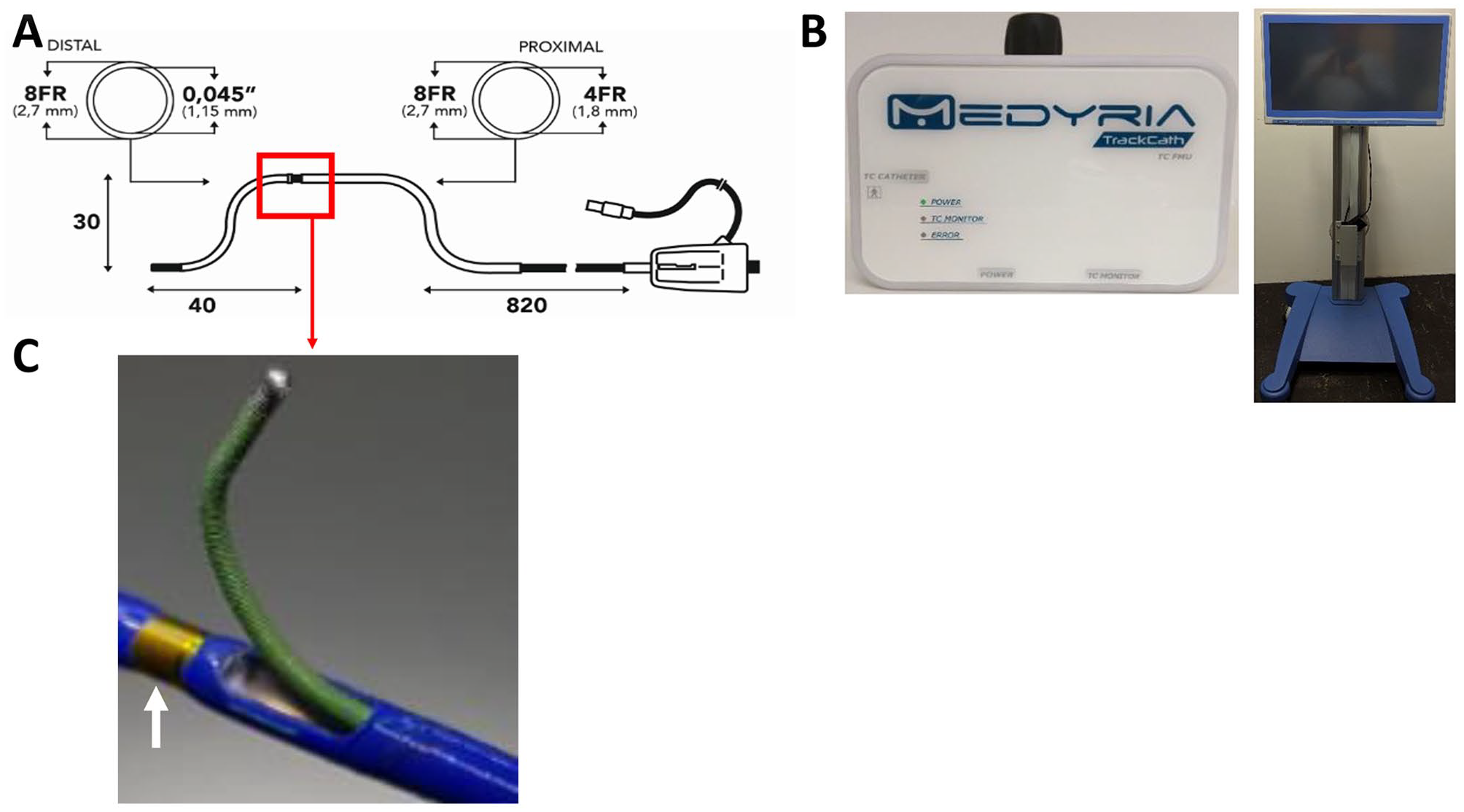
The Medyria TrackCath (MedTC) System [Medyria TrackCath catheter (MedTCC) with equipment]. (A) Schematic illustration of the MedTCC dimensions. The MedTCC has a proximal hub, Luer lock connector, and connector for the MedTC equipment. The MedTCC has an outer diameter of 8 French (F) and an inner diameter of 4F. To ensure providing applicability for different vascular configurations and anatomies the MedTCC is available in 2 different sizes, a 30 mm (D30) and 50 mm (D50) version. The position of the sensor in relation to the distal tip is also variable and is available in 40 mm (short tip—model S, depicted), 100 mm (long tip—model L) length. (B) The MedTC equipment. The flow measurement unit (FMU) (left) and the user interface on the cart (right). The FMU can read the temperature and the blood flow velocity (BFV). (C) The flow sensor is mounted on the shaft close to the exit side port of the guidewire (white arrow).
The MedTCC is equipped with a proprietary BFV sensor system based on thermal convection and features a side port that can be used to catheterize target vessel orifices with guidewires. At the distal end, the catheter has a hub with a standard Luer fitting. The BFV sensor is embedded in the distal section of the catheter and transmits signals to the MedTC equipment. The flow measurement unit (FMU) activates the BFV sensor, provides signal conditioning, and streams the BFV data to the equipment’s user interface (Figure 1B and C). During its clinical application, the BFV is measured along 2 directions, radial and longitudinal with respect to the aorta. As such, the BFV measurements were used to identify flow features typical for aortic side branches, including an increase in flow magnitude and change of BFV direction from longitudinal to radial.
In the ACCESS Trial, the MedTCC was used to mark or catheterize native target vessels, fenestrations, and side branches of aortic stent-grafts during complex aortic procedures. In detail, the catheterization of native target vessels was performed during EVAR, fEVAR, and brEVAR. Here, the MedTCC was advanced over a guidewire through a femoral percutaneous access to the region of interest (Figure 2A). Next, the guidewire was removed so that the MedTCC enfolds its D-shape (Figure 2B). Under constant BFV tracking the MedTCC was aligned with the target vessel orifice by rotation and vertical movement, indicated by a spike signal on the MedTC screen (Figure 2C and D). Once aligned, a guidewire was advanced through the side exit port of the D-shape of the MedTCC to catheterize the target orifice (Figure 2E–G). The 4F side port exit allows a catheter to follow into the target vessel for successful catheterization of native target vessels (Figure 2H). To further explore the capability of the thermal convection–based BFV measurement to catheterize fenestrations and side branches of aortic stent-graft utilizing the MedTCC, a slightly different approach was used in a subset of patients. Here, the catheterization of fenestrations or side branches was performed following aortic stent-graft placement using the same approach for MedTCC delivery within the aortic stent-graft. For those procedures, the catheterization of fenestrations and side branches of stent-grafts was considered as procedural success. The catheterization of native target vessels, fenestrations, and side branches of aortic stent-grafts was performed during a total of 15 EVARs, 17 fEVARs, and 6 brEVARs. Of note, the MedTCC has to be removed while its D-shape stays enfolded regardless of the type of target orifice at the end of the catheterization procedure, applying a certain friction force to the guidewire in the target vessel.
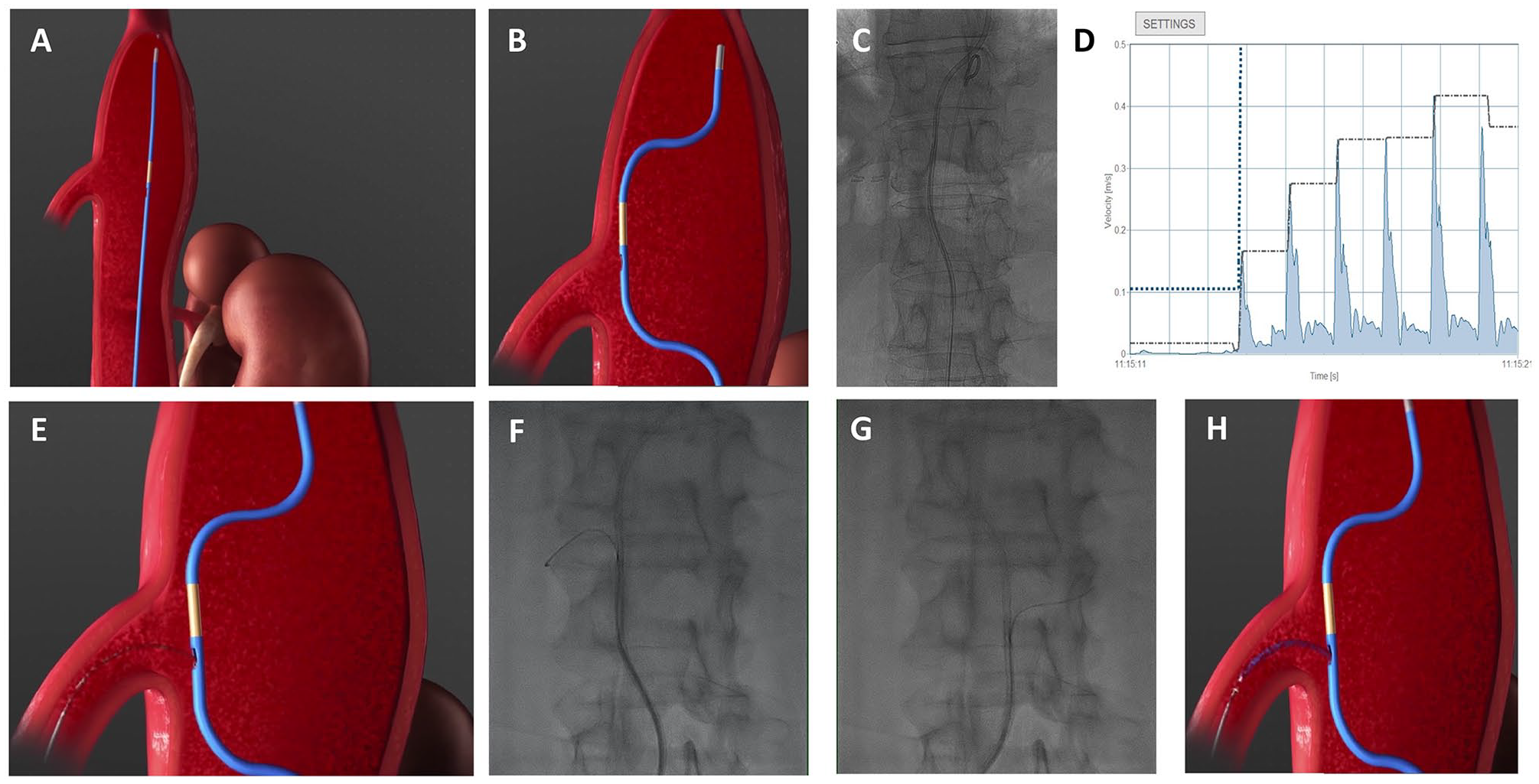
Application of the Medyria TrackCath Catheter (MedTCC). (A) The MedTCC is advanced over a 0.035-inch guidewire to the region of interest. (B and C) Following removal of the 0.035-inch guidewire, the MedTCC unfolds its D-shape configuration. Rotation and vertical movement allow for correct alignment of the MedTCC to the target orifice. (D) Constant tracking of thermal convection–based blood flow velocity (BFV) displays a sharp peak at the point of correct alignment with the target orifice [eg, the superior mesenteric artery (SMA)]. (E–G) A guidewire is advance through the exit port at the side of the D-shape of the MedTCC for cathterization of the target orifice. (H) The guidewire can be followed by any 4F catheter to confirm correct catheterization.
Study Design
The ACCESS Trial was conducted as part of CE-mark approval. It is a single-arm, prospective uncontrolled multicenter proof-of-concept trial conducted in 4 major academic medical centers. The study design allowed participating centers to use the MedTCC in 2 to 3 early-phase roll-in patients for training purposes, which resulted in 11 roll-in patients.
Study Population
Between March 2018 and February 2019, 40 patients with asymptomatic TAAA and short-necked, juxtarenal, and suprarenal AAA planned for endovascular repair were enrolled in the study. Major inclusion criteria were as follows: informed consent, ≥18 years, need for standard or complex aortic procedures following current guidelines. Major exclusion criteria were as follows: need for emergency surgery, dissecting aneurysms, stroke within the past 6 months, myocardial infarction within the past 3 months, acute or chronic kidney failure with creatinine serum levels >2 mg/dL (Supplemental Table 1). Two patients were not treated with the MedTC System, since either intraprocedural inclusion/exclusion criteria were not met prior to use or physical unavailability of the MedTC System. Therefore, both patients were excluded from further analysis. The remaining 38 patients were included for safety analysis (Safety Group, SG). After the exclusion of 1 screening failure case and 11 early-phase roll-in patients, 26 patients were considered for further performance analysis (Performance Group, PG) (Figure 3).
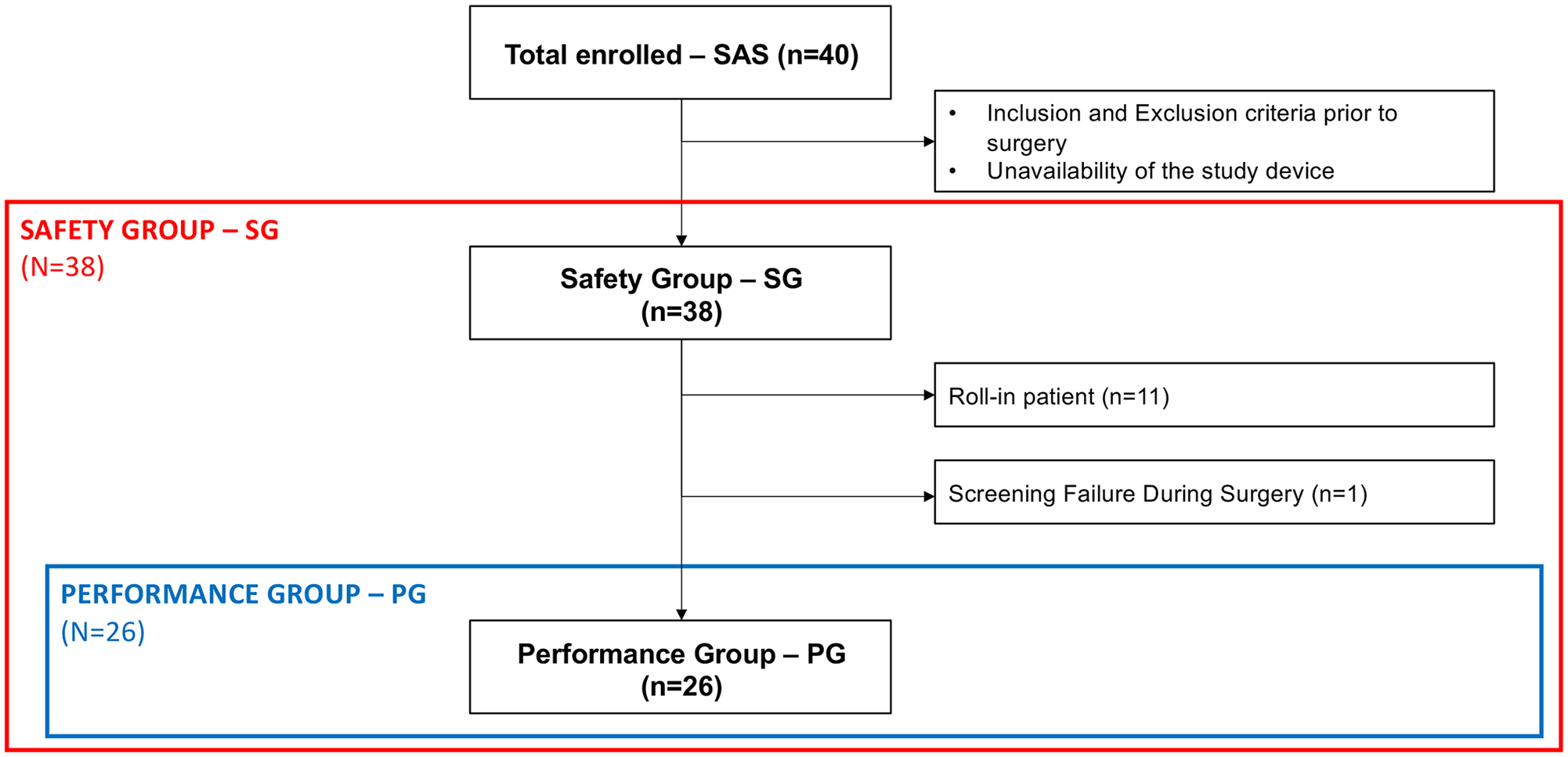
Definition of patient groups for analysis. In total 40 patients were enrolled in the study. The Safety Group (SG) was defined as any patient with contact to the Medyria TrackCath (MedTC) System. After exclusion of 11 roll-in patients and 1 patient resulting to be a screening failure during surgery, 26 patients remained in the Performance Group (PG) for further analysis.
Endpoints
The primary safety endpoint was defined as any serious adverse device effect (SADE) until discharge (SG). The primary performance endpoints were the procedural success rate, defined as successful delivery of the guidewire to target orifices, which were intended to be catheterized during surgery, and the MedTCC catheterization success rate, defined per protocol as successful delivery of a guidewire through the distal end or through a 4F side port–exited catheter to the selected targeted orifice(s) once the MedTCC has been positioned correctly for BFV measurement (PG, SG). Secondary endpoints included all adverse events until discharge (SG), and procedural-related parameters such as the volume of administrated contrast agent and fluoroscopy time (PG, SG). The MedTCC-based procedure time was considered as the sum of the time needed for identification and catheterization of the target orifice. The identification time was measured from the time the MedTCC was deployed above the targeted orifice until correct positioning of the MedTCC toward the target orifice, confirmed by BFV peaks. The catheterization time was considered as the time from correct positioning until the guidewire catheterized a target orifice. In addition, analysis of device-specific BFV measurements was performed at given locations and/or time points during the procedure (SG). Baseline-BFV—BFV prior to side branch identification and appropriate positioning; Orifice-BFV—BFV at the time when the flow sensor was positioned in front of the target orifice; Confirmation-BFV—BFV after catheterization of the target vessel. The difference between Baseline-BFV and Orifice-BFV was used to confirm the correct identification of the target orifice; the absence of a difference between Orifice-BFV and Confirmation-BFV was used to validate the correct identification of the target orifice (validation was achieved by a nonsignificant difference, as after catheterization the MedTC sensor and the target orifice were mechanically aligned). BFV analysis was performed for the SG group due to the limited sample size.
Statistics
All analyses were performed using SAS 9.4 (SAS Institute Inc, Cary, NC, USA) except for analyses of BFV changes, which were realized using Prism GraphPad 8.4.3. Statistical tests were deliberately reviewed to the full level of significance (α=0.05).
Patient characteristics are reported as mean ± standard deviation, categorical data are presented as absolute and relative frequencies. Procedural data are presented as median with interquartile ranges (IQR). A Wilcoxon signed-rank test was applied to analyze BFV changes between the Baseline-BFV and Orifice-BFV through the MedTCC. The procedural and the MedTCC catheterization success rate are reported as percentages with frequency distribution per orifice. The device-related SAE rate is reported with its exact upper confidence limit according to Agresti-Coull (α=0.05, one-sided).
Ethics
The investigational plan, informed consent, all other specific study documents and amendments were reviewed and approved by the ethic committee of the Medical Faculty of the Heinrich-Heine-University Düsseldorf (MPG-LKP-50) and competent authority (“Bundesinstitut für Arzneimittel und Medizinprodukte”; BfArM) (10916) prior to patient enrollment.
Results
Study Population
Patients mean age was 70.6±8.4 years, 30 patients were male. There were 12 TAAA and 26 AAA at a mean diameter of 5.7±1 cm. Cardiovascular, renal, and pulmonary comorbidities were present in 94.7%, 26.3%, and 21.1% of the patients, respectively. The aneurysmatic aortic segments included a total of 16 celiac trunks (CT), 19 superior mesenteric arteries (SMA), 45 renal arteries [right renal artery (RRA); left renal artery (LRA)], and 17 inferior mesenteric arteries (IMA) (Table 1).
Baseline Patients and Aneurysm Characteristics.
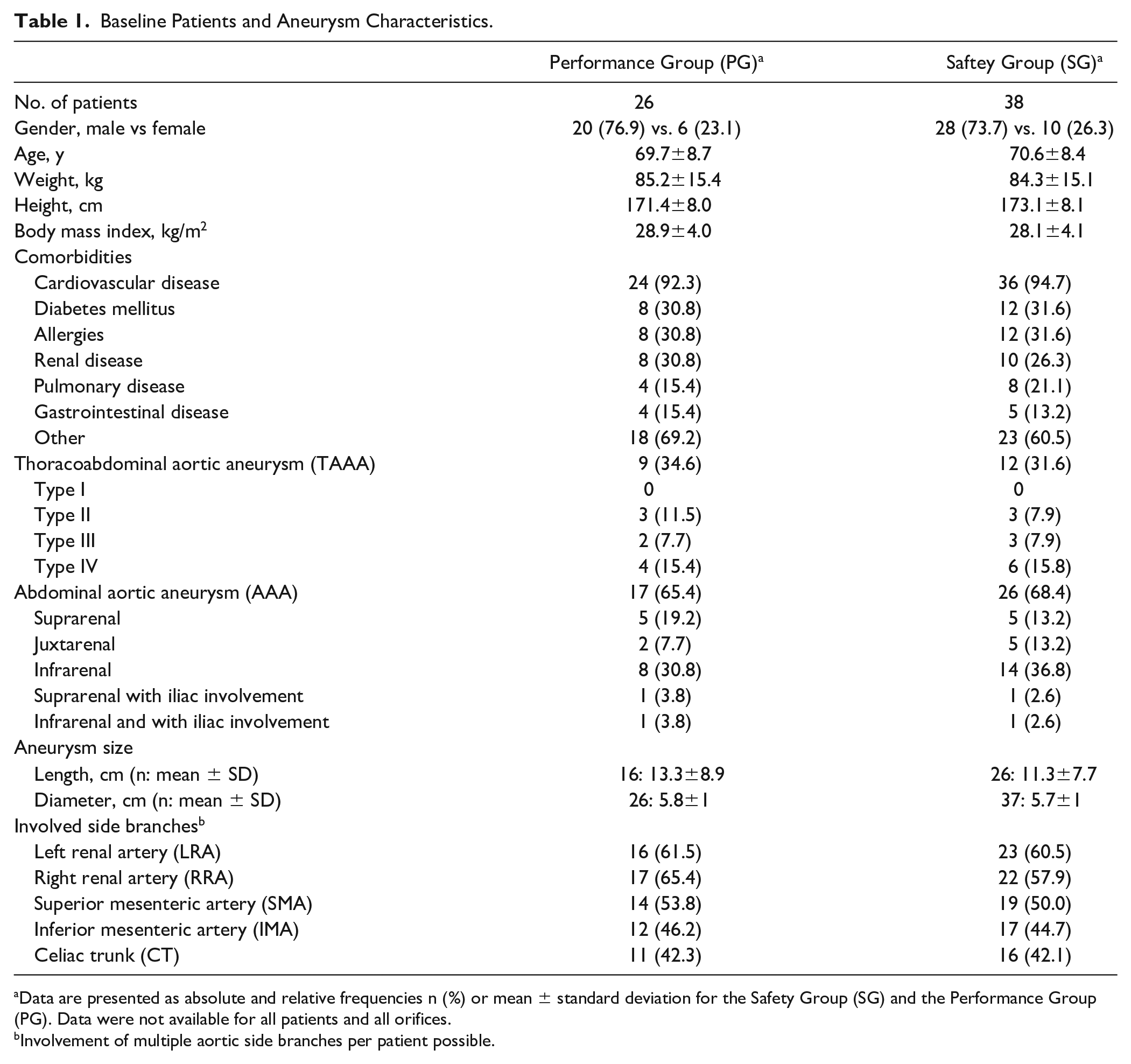
Data are presented as absolute and relative frequencies n (%) or mean ± standard deviation for the Safety Group (SG) and the Performance Group (PG). Data were not available for all patients and all orifices.
Involvement of multiple aortic side branches per patient possible.
Performance
The MedTCC catheterization and the procedural success rates are reported for the PG and SG. There were 54 orifices planned for catheterization prior to surgery for the PG and 78 orifices for the entire SG. During surgery, there were 2 additional orifices targeted, one orifice in the PG and one in the SG, of which both had successful catheterizations. That being said, there were a total of 50 orifices targeted during surgery in the PG and 67 orifices in the SG. Of the targeted orifices, there were 4 catheterizations of fenestrations and 5 side branches of aortic stent-grafts in the PG, while there were 7 fenestrations and 9 side branches targeted and catheterized in the SG. The author experienced 49 successful target orifice catheterizations in the PG group and 65 in the SG. Consequently, the per-protocol-defined MedTCC catheterization success rate was 98% (49/50 orifices) for the PG and 97% (65/67 orifices) for the SG (Table 2). Of note, an unsuccessful attempt of catheterization occurred in a patient with a TAAA type IV due to a low flow signal. In that patient guidewire delivery utilizing the MedTCC was successful for the SMA, while the RRA failed successful catheterization. Such failed attempt occurred in a native target vessel. The other failed catheterization also occurred when targeting the RRA in an early-phase roll-in patient. In this particular case, the authors could not detect any flow signal from the RRA side branch of an aortic stent-graft although the MedTCC was correctly aligned.
Targeted Orifices for the Performance Group (PG) and the Safety Group (SG).
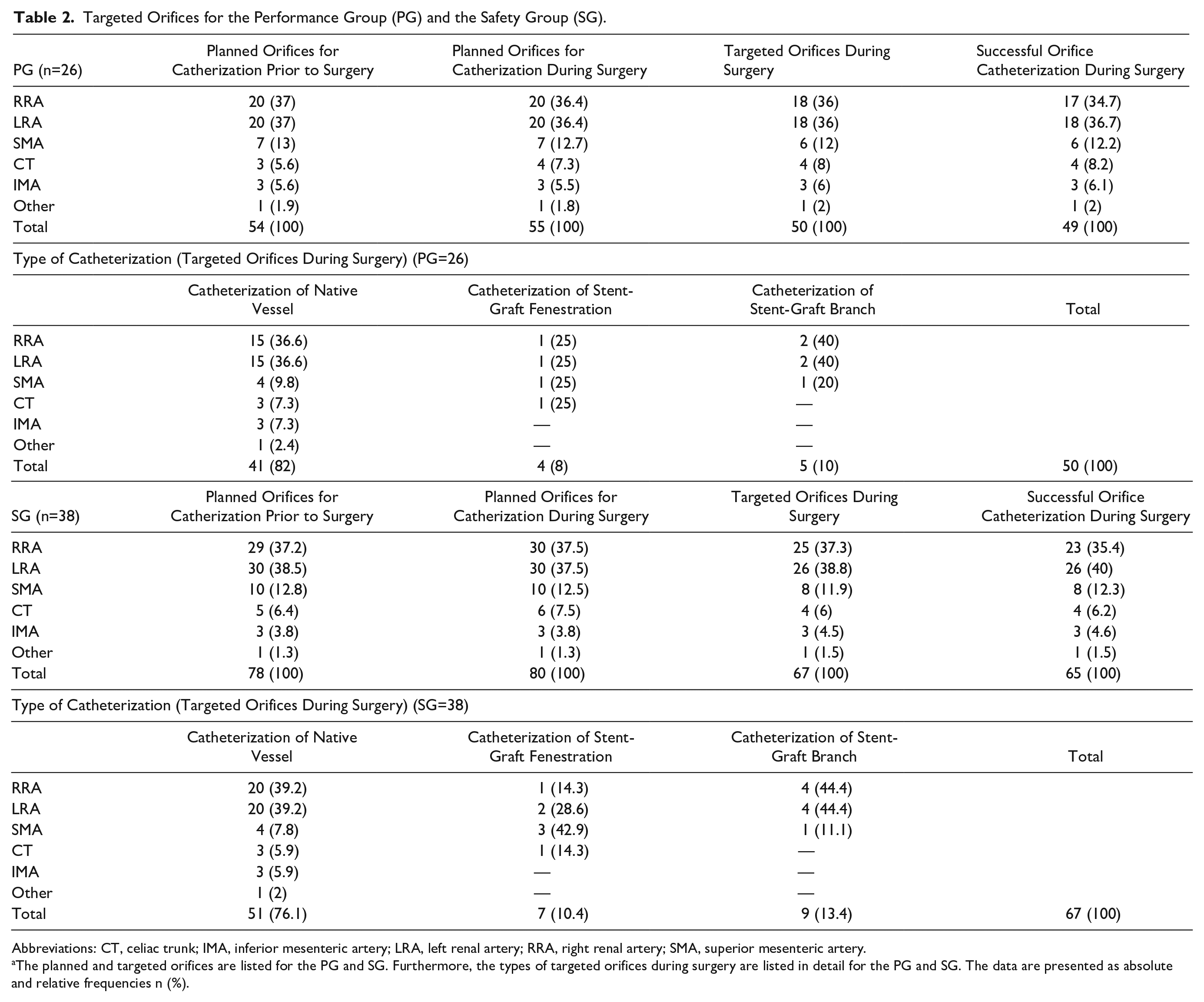
Abbreviations: CT, celiac trunk; IMA, inferior mesenteric artery; LRA, left renal artery; RRA, right renal artery; SMA, superior mesenteric artery.
The planned and targeted orifices are listed for the PG and SG. Furthermore, the types of targeted orifices during surgery are listed in detail for the PG and SG. The data are presented as absolute and relative frequencies n (%).
The procedural success rate was 89.1% (49/55 orifices) for the PG and 81.3% (65/80 orifices) for the SG (Table 2); most of the cases for not attempting catheterization of target orifices occurred in the roll-in patient group, suggesting a significant learning curve during the use of the MedTCC. Lack of BFV signal at the beginning of the procedure (1 patient, 4 orifices) and a screening failure (1 patient, 1 orifice) suggested technical software issues. Furthermore, tortuous anatomies that did not allow for correct positioning of the MedTCC occurred in 2 patients (1 orifice each). In another case, the distance from the stent-graft fenestration to the aortic bifurcation was too short so that the MedTCC could not target the lowest renal artery orifice (1 patient, 1 orifice). In the PG, the reasons for not attempting catheterization were tortuous vascular access (1 patient, 2 orifices), time constraint (1 patient, 1 orifice), occluded side branch (1 patient, 1 orifice), and misalignment of the main endograft with the target vessel (1 patient, 1 orifice). In the latter two cases, the MedTCC was able to confirm the orifice occlusion and endograft misalignment by indicating no flow in correspondence of the stent-graft fenestration.
Procedural Parameters
Procedural parameters are reported for the PG and the SG. The median procedure time was 173 (IQR 114–283) minutes (PG). Femoral vascular access was performed in all cases. When utilizing the MedTCC, the median MedTCC-based procedure time was 3.0 (IQR 2.0–6.0) minutes [identification time was 2.0 (1.0–4.0) minutes and catheterization time was 1.0 (<0.1–2.0) minutes]. Median fluoroscopy time was 49 (26–74) minutes overall, and 5.0 (1.5–8.5) minutes per orifice. The median contrast agent consumption was 80 (40–100) mL overall, and 1.0 (0–5.0) mL per orifice. Median length of in-hospital stay was 7.5 (5.0–12.0) days. Procedural data for the SG were similar (Table 3).
Procedural Parameters by Patient and by Orifice for the Performance Group (PG) and Safety Group (SG).
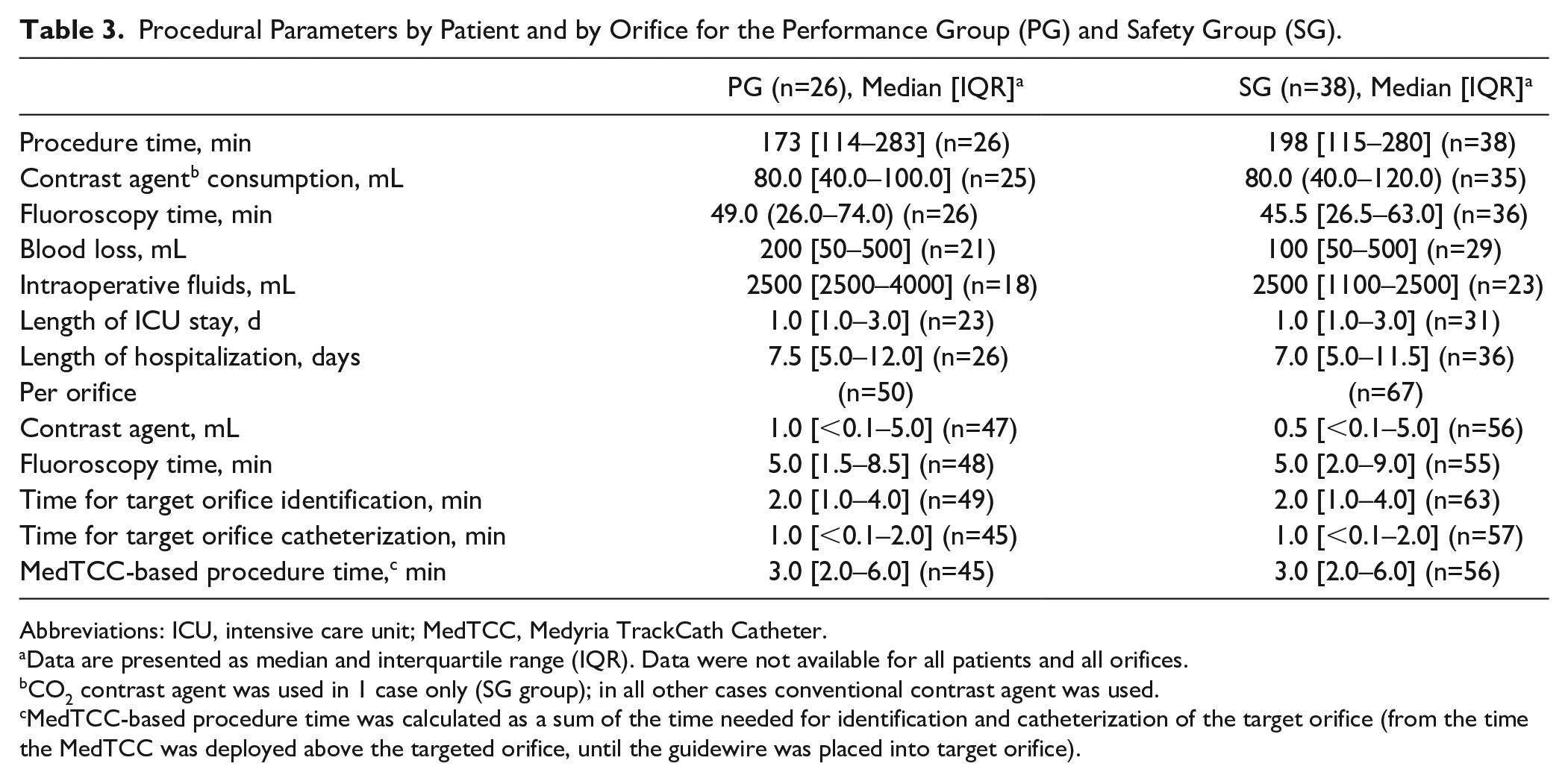
Abbreviations: ICU, intensive care unit; MedTCC, Medyria TrackCath Catheter.
Data are presented as median and interquartile range (IQR). Data were not available for all patients and all orifices.
CO2 contrast agent was used in 1 case only (SG group); in all other cases conventional contrast agent was used.
MedTCC-based procedure time was calculated as a sum of the time needed for identification and catheterization of the target orifice (from the time the MedTCC was deployed above the targeted orifice, until the guidewire was placed into target orifice).
Analysis of the BFV Changes
BFV data are reported for the SG. Our results revealed a significant difference in BFV between Baseline-BFV and Orifice-BFV for the SMA, the RRA, and the LRA, illustrating the capability of the MedTCC to reliably detect BFV changes for major target vessels (p<0.05). Of note, the difference in BFV for the CT trended in the same direction; however, results failed statistical significance, probably due to the limited sample size. As expected, we observed similar Orifice-BFV and Confirmation-BFV. While the limited sample size makes a valid statistical comparison somewhat imprecise, our data still suggest that tracked BFV changes through the MedTCC originated from target vessels rather than from random artifacts (Figure 4).
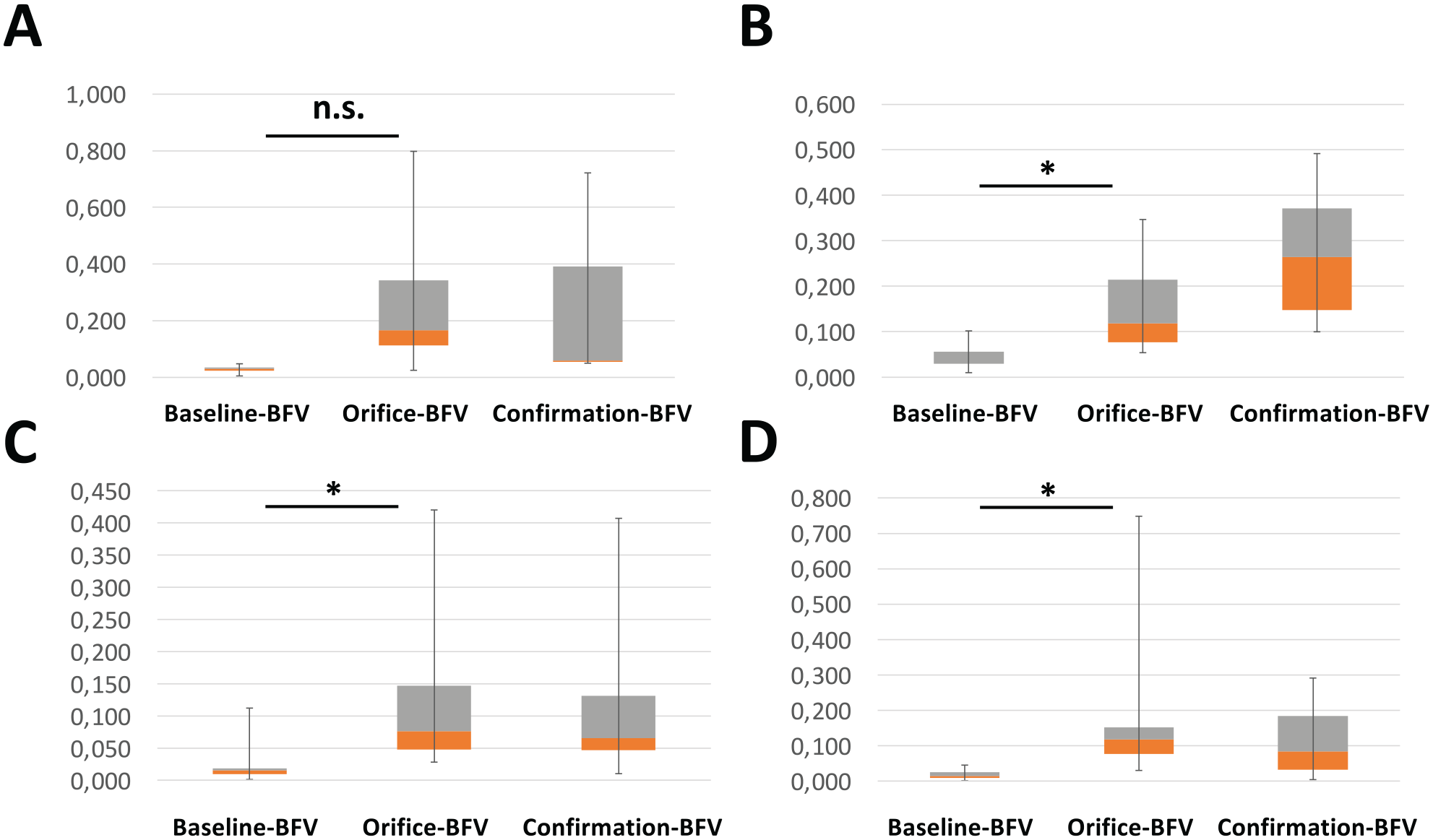
(A) Overview of blood flow velocity (BFV) measurements: Baseline-BFV, Orifice-BFV, and Confirmation-BFV. Baseline-BFV is measured prior to side branch identification and appropriate positioning, Orifice-BFV is measured at the time when the flow sensor is positioned in front of target orifice, Confirmation-BFV is measured after catheterization of the target vessel. The BFV is presented for the celiac trunk (CT) (n=4) (A), the superior mesenteric artery (SMA) (n=7) (B), the left renal artery (LRA) (n=18) (C), and right renal artery (RRA) (n=20) (D). Data are presented as boxplots with median, interquartile range (IQR), and minimum-maximum range. The change in BFV between the Baseline-BFV and Orifice-BFV suggests effective identification of target vessels. Data were analyzed for the Safety Group (SG) and was extracted from the MedTC flow measurement unit. A Wilcoxon signed-rank test was applied to prove valid measurements of BFV changes through the Medyria TrackCath Catheter (MedTCC) (Baseline-BFV vs. Orifice-BFV) in consecutive measurements. *p<0.05 Baseline-BFV vs. Orifice-BFV.
Safety
Safety data are reported for the SG. There were no SADEs. This resulted in an upper confidence limit according to Agresti-Coull of 7.94%. A total of 9 SAEs were reported in 8 patients, while 44 AEs occurred in 17 patients (Table 4). Of note, 2 patients presented an acute kidney injury (AKI), of whom 1 patient required medical treatment. None of the (S)AEs was related to the MedTCC or led to death.
Serious Adverse Events (SAEs) for the Safety Group (SG).
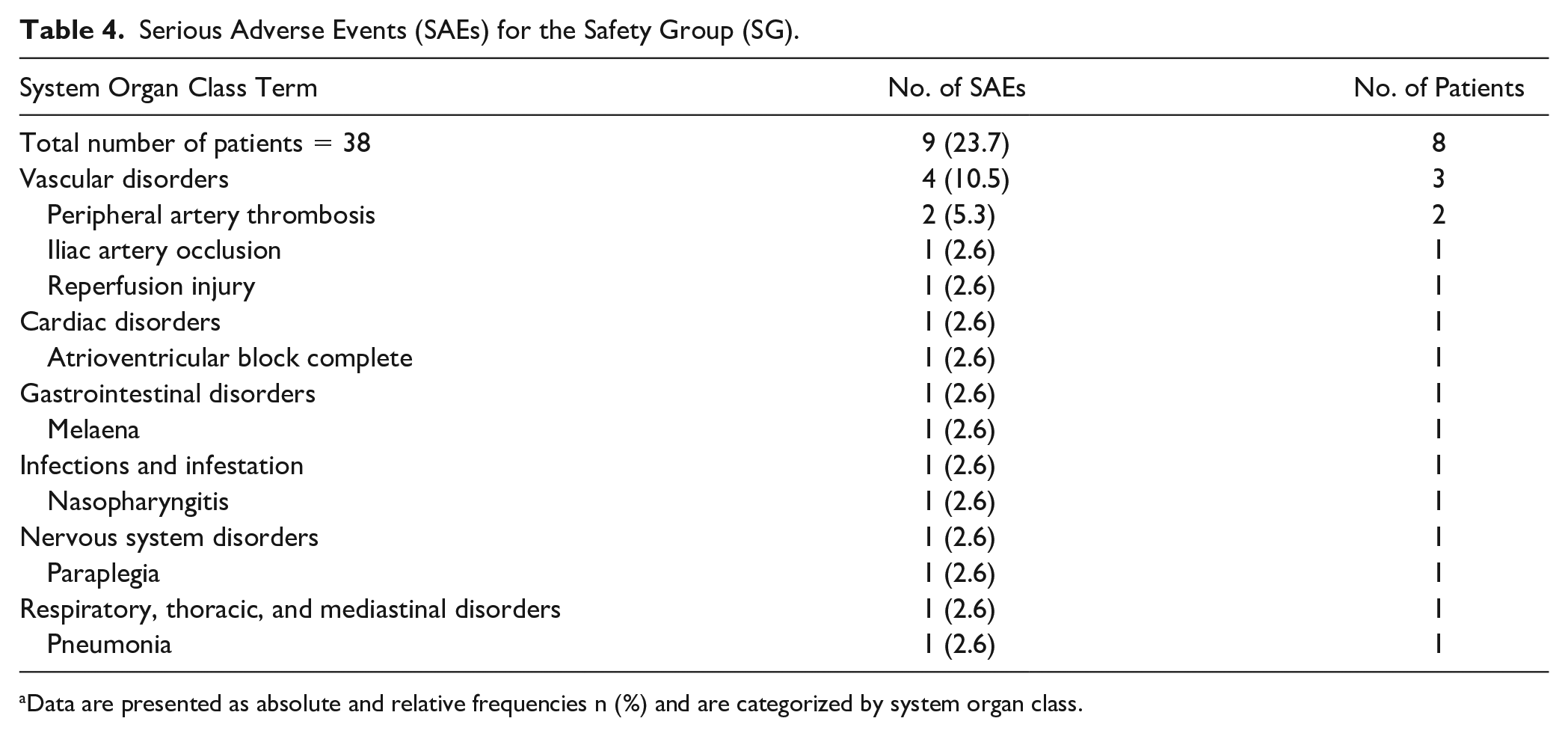
Data are presented as absolute and relative frequencies n (%) and are categorized by system organ class.
Discussion
The ACCESS Trial is the first multicenter prospective in-human single-arm trial for establishing clinical evidence on the safety and the efficacy of the MedTCC in delivering devices (eg, 4F catheter or guidewire) to target orifices in patients with TAAA or short-necked, juxtarenal, and suprarenal AAA requiring complex endovascular procedures. Comprehensively, our findings support the capability of the MedTCC to safely identify target orifices, thus facilitating catheterization during complex endovascular procedures, while tortuous anatomy was the most frequent reason for an unsuccessful use. Furthermore, the ACCESS Trial demonstrates a learning curve during the clinical use of the MedTCC, since both the MedTCC catheterization and the procedural success rate improved when exclusively analyzing the PG and not considering the early-phase roll-in patients. In contrast, the same learning curve is not true for most of the key procedural parameters, since the majority of these do not change between the PG and SG.
Catheterization of the visceral and/or renal arteries or the according side branches of stent-graft consumes a significant portion of the procedural time during fEVAR and brEVAR. As reported by Ullery et al, 10 the time required for the catheterization of a renal artery ranges from 10.9 to 32.8 minutes depending on the renal artery angulation. Furthermore, Cochennec et al 11 stated that, while utilizing an automated steering guiding sheath with 2 bending degrees of freedom, the catheterization time for the same artery was as low as 4.2 minutes on average. When utilizing the MedTCC, our data reveal a median time of 3 minutes from deploying the MedTCC until the guidewire was placed into the side branch. The authors are aware that the definition of the reported catheterization times in the literature differs from this study making valid comparisons somewhat difficult. Also, the authors might emphasize that other methods, such as 3-dimensional (3D) fusion computed tomographic imaging, have proven to effectively reduce procedure time, contrast agent use, and fluoroscopy time in some cases.12,13 As of today, however, fusion imaging approaches might still require manual realignment to achieve full accuracy in most cases causing an increase in the overall procedure time. 14 Albeit a direct comparison between fusion imaging approaches and BFV measurement for target vessel cannulation is yet to be performed, the authors underline that the MedTCC can be combined with fusion imaging, which might expand the application range. The MedTCC has the potential to reduce orifice catheterization time during complex endovascular procedures, although the authors are aware that the reported total fluoroscopy time of the ACCESS Trial is comparable to those reported by others for similar patient cohorts and procedures.9,15 Of note, this might support observations from Ruz et al 16 who suggested that rather than the anatomic complexity, the design of the device, or the number of components inserted, the surgeon’s individual skills are more critical in minimizing radiation exposure.
Since fEVAR and brEVAR are complex endovascular procedures, they require the use of higher amounts of contrast compared with standard EVAR. Applying contemporary criteria for AKI such as from the Acute Kidney Injury Network (AKIN), Sailer et al and Ducasse et al both reported similar AKI incidences ranging from 28% to 32% following fEVAR and/or brEVAR.17–19 The authors of this study experienced 2 patients with AKI following fEVAR or brEVAR; however, we did not apply the aforementioned criteria. As a consequence, a comparison with the current literature regarding this endpoint is impossible. To this end, carbon dioxide (CO2) angiography may provide an alternative during these procedures in patients with chronic kidney disease (CKD).20,21 Given its nonubiquitous availability, this technique remains an option in certain centers; however, further evidence is required to establish the essential skill set for safe use of this approach.
Considering the association between AKI and long-term renal dysfunction, the aim to reduce contrast agent use to achieve the best clinical outcomes seems justified.18,22 That said, the present study reports a median procedural contrast agent use of 80 mL, which is comparable to literature-derived observations for similar procedures. As such, Sailer et al 15 described the use of contrast at a mean of 199 mL without using fusion imaging, while Scali et al 23 claimed a contrast use ranging from 41 and 245 mL when using surgeon-modified endovascular aneurysm repair in acute visceral aortic pathologies. Although our data do not allow to determine whether the MedTCC reduces contrast agent consumption during fEVAR and/or brEVAR, the reported data seem encouraging.
The rate of SAE and mortality of this study did not exceed the perioperative mortality and morbidity rates reported in a comprehensive study on type II and type III TAAA in 354 patients and a recent meta-analysis with clearly defined inclusion criteria encompassing 185 patients following the screening of 370 articles.24,25 There were no specific device-related risks noted during this clinical trial since there were no SADEs, so the authors conclude that utilizing the MedTCC during complex endovascular procedures is safe.
Target vessel catheterization remains a critical and time-consuming step during fEVAR and/or brEVAR. Recent technical advances in catheter design and application have been reported to facilitate this substep. As such, robotic steerable endovascular (micro)catheter systems provide remote-controlled catheter tip reflection and advancement and have been reported to shorten procedure times.26–28 We report another innovative method for target vessel catheterization based on the convective heat exchange between heated resistance and a fluid temperature allowing for thermal convetion-based BFV tracking by a single sensor scheme. Notably, we illustrate that this method is effective for target vessel identification and catheterization and does not cause procedural-related complications.
Besides the aforementioned promising results, the authors may want to describe some potential improvements to the MedTCC based on their experience. It is well accepted that tortuous anatomy is challenging during most endovascular procedures. 29 To address this issue, hydrophilic coating might be a significant technical improvement. Likewise, the D-shape design with the option to advance a 4F catheter through a side port might set some procedural challenges, since it applies a friction force to the guidewire during the removal from the MedTCC. Also, the D-shape of the catheter might limit its use in small lumen anatomies.
In general, thermal convection–based BFV could be integrated into various catheter designs. Here, a straighter catheter design that allows for appropriate wall apposition while still enabling effective BFV measurement together with a soft tip may overcome such an issue. More to this, such design would allow for direct MedTCC advancing into the target vessel following guidewire catheterization. It may also be worth mentioning, that besides target vessel identification and catheterization, the BFV measurement technology might have broader applications. For instance, the identification of misaligned implants, segmental arteries, and stenosed target vessels might expand the range of use. Considering this, the authors may want to emphasize that the technology may hold great potential in facilitating procedures to prevent type II endoleaks following aortic stent-graft placement or spinal cord ischemia through ischemic preconditioning with the necessity to catheterize aortic side branches, such as the inferior mesenteric artery or segmental arteries for coil embolization. 30
The study has various limitations. This multicenter prospective trial is not controlled, restricting comparisons of outcomes to reported literature results. Also, divergent definitions between the current literature and the present study regarding performance parameters make valid comparisons somewhat at least imprecise if not impossible. Adverse vascular anatomies have been an exclusion criterion albeit being the most frequent reason for an unsuccessful use. That being said, the reported procedural success rates may be lower in unselected patient cohorts. The number of participants in this trial is limited and most patients had several comorbidities suggesting significant confounding. As of today, the sales price of the MedTCC may be high so that further research on its potential benefits is required prior to market launch.
Conclusion
This first-in-human trial illustrates the general feasibility of thermal convection-based BFV measurement for target vessel identification and catheterization during complex endovascular aortic procedures. As such, the MedTCC may be flexibly applicable for delivering contrast, guidewires, and other devices to target orifices; however, more research is required to better characterize the performance of convection-based BFV in a variety of possible surgical scenarios, such as its capability in calcified or stenosed target orifices.
Supplemental Material
sj-docx-1-jet-10.1177_15266028211030536 – Supplemental material for First-in-Human Clinical Application of the Medyria TrackCath System in Endovascular Repair of Complex Aortic Aneurysms (ACCESS Trial): A Prospective Multicenter Single-Arm Clinical Trial
Supplemental material, sj-docx-1-jet-10.1177_15266028211030536 for First-in-Human Clinical Application of the Medyria TrackCath System in Endovascular Repair of Complex Aortic Aneurysms (ACCESS Trial): A Prospective Multicenter Single-Arm Clinical Trial by Nikolaos Floros, Tilo Kölbel, Nikolaos Tsilimparis, Alexander Oberhuber, Daniel Kindl, Johannes Kalder, Drosos Kotelis, Andrej Schmidt, Daniela Branzan, Daniela Adolf, Hubert Schelzig and Markus Udo Wagenhäuser in Journal of Endovascular Therapy
Footnotes
Acknowledgements
The authors thank all employees at the participating study centers for their help.
Authors’ Note
Parts of the work were presented at the annual meeting of the European Society for Vascular Surgery 2019, Hamburg, Germany and at the VEITH Annual Symposium on Vascular and Endovascular Issues 2019, New York, USA.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NF has not conflict of interest to disclose. TK serves as consultant to Medyria AG, Winterthur, Switzerland. NT has no conflict of interest to disclose. AO receives payment by Medyria AG, Winterthur, Switzerland for testing the catheter in the animal lab, helping finalize the catheter design and setting up the basic study design. DK has no conflict of interest to disclose. JK receives payment by Medyria AG, Winterthur, Switzerland for testing the catheter in the animal lab, helping finalize the catheter design and setting up the basic study design. DK receives grants and grants speaker’s honorarium from Medtronic, Bentley Innomed, and Siemens Healthcare. AS receives consulting/speakers honorarium from C.R. Bard, Cook Medical, and Cordis. DB receives grants from BD Bard, Bentley Innomed, Bayer Medical, and Terumo Aortic. DA receives fees by Medyria AG, Winterthur, Switzerland for statistical support. HS and MUW have no conflict of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided Medyria AG, Winterthur, Switzerland.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
