Abstract
Purpose
To describe the use of large-diameter balloon-expandable stent-grafts (BeGraft aortic stent-graft, Bentley InnoMed GmbH, Hechingen, Germany) in the treatment of infrarenal penetrating aortic ulcer (iPAU).
Materials and Methods
Retrospective analysis of patients undergoing endovascular treatment with the BeGraft aortic stent-graft in 8 European centers from January 2017 to October 2020. Demographics, perioperative data, and midterm outcomes were collected. Endpoints of the study were technical feasibility, early mortality, and morbidity.
Results
A total of 40 patients were included. The mean age was 73.9±7.05 years and 63.2% were male. Indications for treatment included size and morphology (65%), presence of symptoms (29.5%), and contained ruptures (5.5%). Urgent treatment was performed in 5% of cases. Technical success was 97.5%. Median operation time was 58 minutes (19–170 minutes), with 27.5% of patients having additional procedures during the main intervention (1 additional repair with a C-TAG (W.L. Gore & Associates, Inc, Flagstaff, AZ, USA) thoracic endoprosthesis, 5 covered endovascular reconstruction of aortic bifurcation procedures, 3 extensions with proximal cuffs, and 2 percutaneous angioplasties of the common iliac arteries). Percutaneous femoral access was used in 72.5%, while groin cut-down was performed in 27.5%. Repair was successful with only 1 stent in 45% of cases, while 37.5% required 2 stents and nearly 17.5% required 3/4 stent-grafts. The 30-day mortality was 0%, with a 2.5% reintervention rate (1 patient required evacuation of an intra-abdominal hematoma). Median follow-up was 13.9 months (2–39 months), during which no vascular-related reinterventions or deaths were reported. In 4 patients, a type II endoleak was observed. No cases of graft migration, thrombosis, or stent-fracture were observed.
Conclusions
The treatment of iPAU with the BeGraft aortic stent-graft in a selective patient group is feasible with low rate of perioperative morbidity and mortality. Balloon-expandable stent-grafts offer the option to repair iPAUs with a shorter coverage of the aorta using low-profile sheath, that enables treatment in the presence of calcified access vessels and small diameter aortic bifurcations.
Keywords
Introduction
Penetrating aortic ulcers (PAU) were first reported by Shennan in 1934; however, it was not until 1986 that Stanson and colleges recognized PAU as a unique condition.1,2 They are currently classified under the term acute aortic syndrome (AAS), which also includes aortic dissection (AD) and intramural hematoma (IMH), and have been defined as a focal disruption in the arterial intima and elastic lamina that extends into the medial layer, likely resulting from erosion of atherosclerotic plaques and inflammatory changes in the aortic wall.3–5 In contrast to the other AAS, PAUs are characterized by being a focal lesion. This is thought to occur because, although the elastic and muscular portions of the aortic wall lose their strength, its progression is hampered by the calcification and inflammation present in the aortic wall of these patients.4–6
The true incidence of PAU is unknown since many of them are asymptomatic; however, they account for 2% to 7% of all AAS.3,6 Their clinical course is variable, with only 20% being symptomatic. They have an elusive natural evolution, with rates of progression to IMH and AD up to 30% and rupture rates of 40% in selected series. 4 They typically occur in the setting of advanced atherosclerosis and severe intimal disease and are more frequently seen in older and more diseased patients as compared with other AAS. The most frequent aortic location is the descending thoracic aorta, with over 60% of all PAUs occurring there. Abdominal PAUs make up 30% of the cohort aorta, with the remaining 7% to 10% occurring in the aortic arch. 7
Infrarenal PAUs (iPAUs) differ from thoracic PAUs. Specifically, around 45% are diagnosed in the form of isolated ulcers, while 9%, 29.5%, and 2.4% of cases are diagnosed in the context of IMH, saccular aneurysms or aortic ruptures, respectively. 8 A systematic review of iPAU from 2013 reports that around 30% of patients treated up to that point underwent open surgical repair, with 70% receiving endovascular treatment. However, endovascular treatment with stent-grafts designed for different pathologies can have a negative impact regarding their long-term results. Traditionally, endovascular solutions included the used of standard endovascular aortic repair (EVAR) stent-grafts or tubular thoracic/iliac extensions. 8 The durability of EVAR may be hampered by the heavy calcification and narrow anatomy frequently seen in these patients, while the use of straight tubular self-expandable endografts has been associated with up to 18% of type I endoleaks, probably due to limitations in sizes and lengths, as well as in discrepancy in proximal and distal diameter. 9
The BeGraft Aortic Stent-Graft System (Bentley Innomed GmbH, Hechingen, Germany) is a balloon-expandable, covered stent-graft that combines low foreshortening with a high radial force. Available diameters range between 12 and 24 mm, with lengths between 19 and 58/59 mm. One of the potential advantages of the use of the balloon-expandable stent system is their smaller introducing sheaths. The 12-mm BeGraft aortic stent-graft is mounted on a 9F introducer sheath, while the 14 and 16 mm diameters are mounted on a 12F introducer sheath. The remaining diameters, up to 24 mm, can be introduced over a 14F sheath. 10 This stent-graft has been specifically designed as a tubular stent-graft for treatment of aortic diseases associated with calcified atherosclerotic plaques and narrow diameters. Its use for treatment of iPAU has emerged as an alternative to EVAR and other tubular endografts, however.
The aim of this study is to report early and midterm results of the BeGraft Aortic Stent-Graft System for treatment of iPAU in multiple European institutions. The indication for use of the BeGraft Aortic Stent-Graft System is for treatment of coarctation of the aorta and iliacal lesions. The use of the BeGraft Aortic Stent-Graft System for treatment of iPAU is “off-the-label.”
Materials and Methods
A retrospective analysis was conducted of patients undergoing iPAU treatment with the BeGraft Aortic Stent-Graft System in 8 European centers between January 2017 and October 2020. Demographic data, including comorbidities and preoperative imaging were included. The distance from the lower renal artery to the beginning of the lesion, and from the distal end of the lesion to the aortic bifurcation, were collected. Operative data, including total number of stents required, additional procedures, complications, operating time, fluoroscopy, and contrast-volume used were also collected. Thirty-day outcomes evaluated included mortality, reintervention rate, and complication rate. Midterm variables collected were mortality and reinterventions, as well as the final imagining study and the rate of endoleaks and stent-graft complications observed.
The primary objective of the study was to evaluate the feasibility and short-term results of treatment of iPAU with the BeGraft aortic stent-graft. Secondary objectives included rate of endoleaks and other stent-graft-related complications (rupture, migration, fracture, and thrombosis) as well as mid- or long-term reintervention rates.
Statistical analysis was performed with Stata 14.2. The Shapiro-Wilk test was used to evaluate if variables had a normal distribution. Means, standard deviations, and parametric tests were used for normally distributed variables. Medians, ranges, and nonparametric tests were used for nonnormally distributed variables. Kaplan-Meier survival analysis was performed with SPSS 26 and used for evaluation of midterm results.
Results
Patient Cohort
A total of 40 patients were included. The mean age was 73.9±1.14 years, 63.2% were male. Most prevalent comorbidities are presented in Table 1. None of the patients had prior aortic repair, although 2 patients had received prior iliac treatment (1 transluminal angioplasty, 1 iliac bypass).
Patient Demographics (N=40).
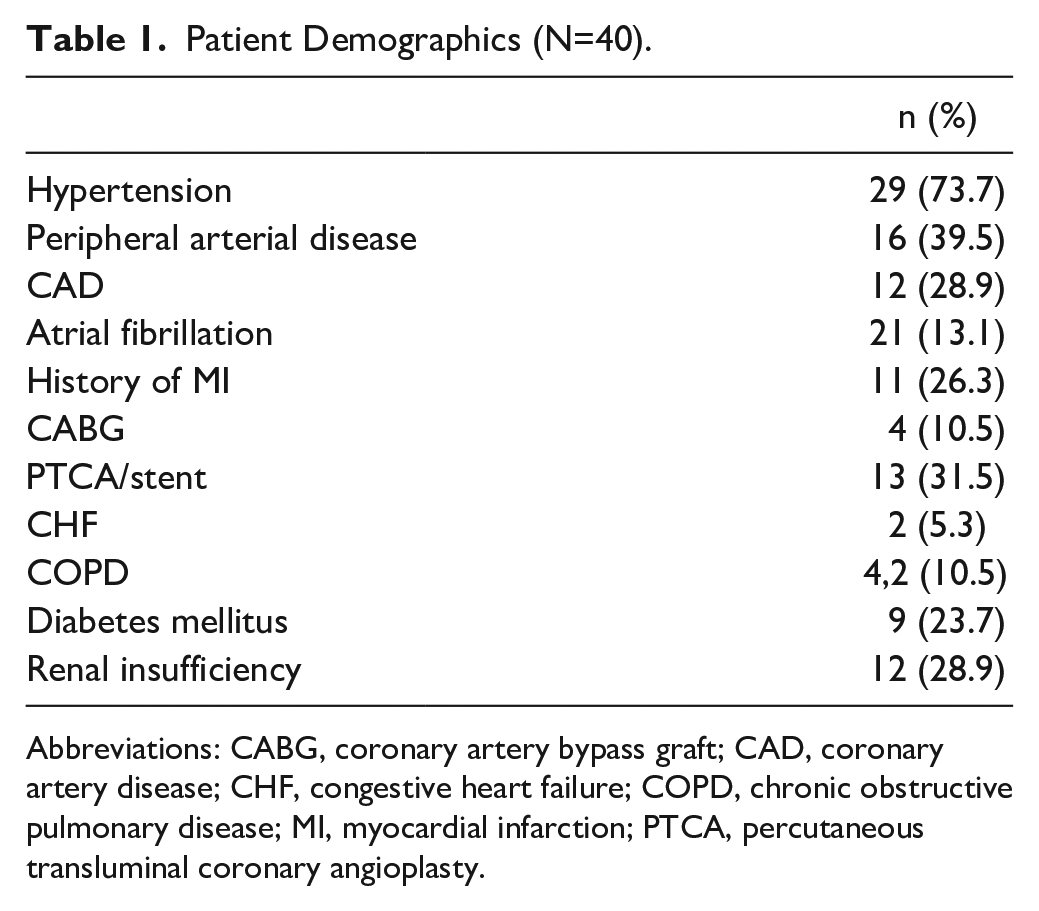
Abbreviations: CABG, coronary artery bypass graft; CAD, coronary artery disease; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; MI, myocardial infarction; PTCA, percutaneous transluminal coronary angioplasty.
Indications for treatment included size/morphology of the iPAU (67.5%), presence of symptoms (27.5%), and contained ruptures (5%). Urgent treatment was performed in 5% of cases. The depth of the PAU in asymptomatic patients was 19±11.7 mm, in the symptomatic patients was 19±11.2 mm and in patient with contained rupture was 23±3 mm.
Preoperative Anatomic Characteristics
All patients included had an available preoperative computed tomography angiography. The preoperative anatomical characteristics are presented in Table 2.
Preoperative Anatomical Characteristics.
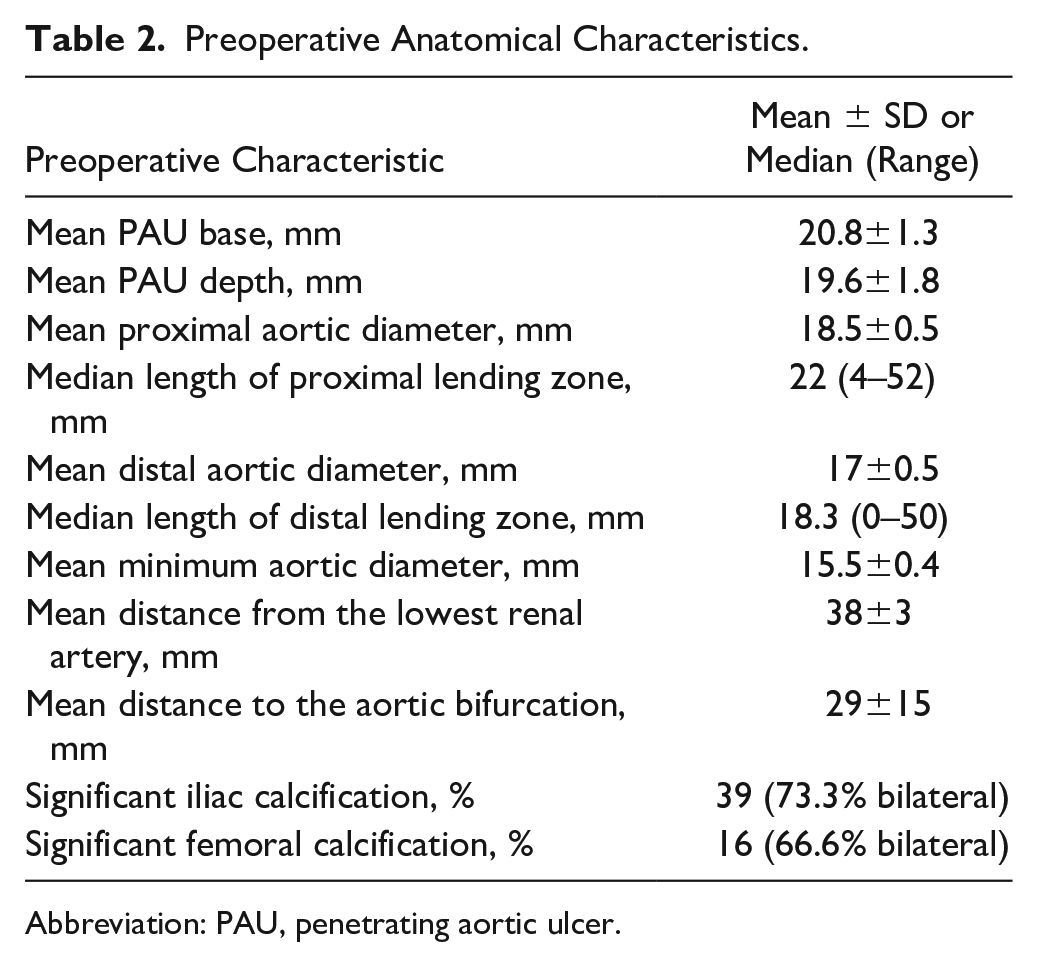
Abbreviation: PAU, penetrating aortic ulcer.
Perioperative Data
Technical success was 97.5%: One patient with a symptomatic PAU suffered an aortic rupture after deployment of the second BeGraft due to extensive oversizing in a severely calcified aorta. An additional procedure was needed in 27.5% of cases. The patient with the acute rupture required additional repair with a C-TAG (W.L. Gore & Associates, Inc, Flagstaff, AZ, USA) thoracic endoprosthesis. Because of insufficient distal landing zone in 5 cases (12.5%) a covered endovascular reconstruction of aortic bifurcation (CERAB) procedure was performed. To achieve proximal sealing and avoid an overflaring of the balloon expandable stent a heavily calcified proximal lending zone in 3 patients (7,5%) a proximal cuff was used. In 2 cases (5%), an angioplasty of the iliac artery was needed to treat an endoleak type Ib. The preoperative data are presented in Table 3.
Perioperative Data.
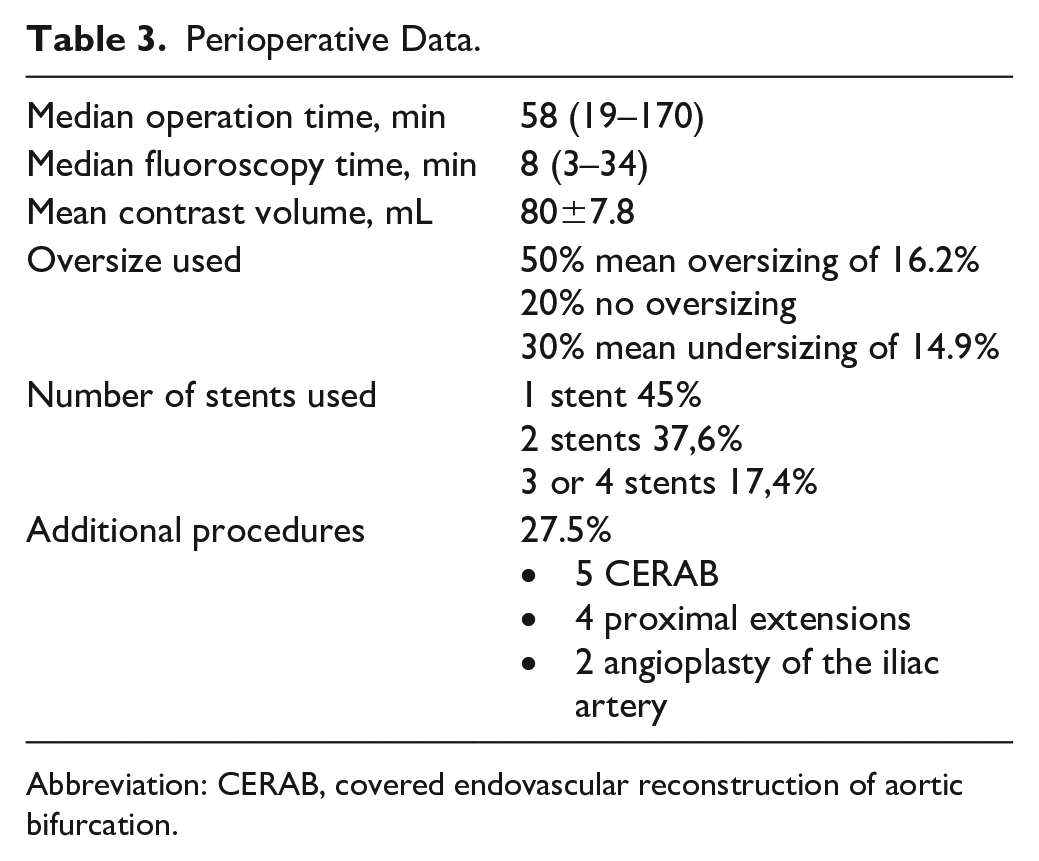
Abbreviation: CERAB, covered endovascular reconstruction of aortic bifurcation.
Thirty-Day Outcomes
The 30-day mortality was 0%, with 0% aortic reintervention rate, a 2.5% general reintervention rate (1 patient required hematoma evacuation) and a 12.5% postoperative complication rate (n=5). Of the 5 patients who developed postoperative complications, one was the patient with intraoperative rupture, who developed acute renal failure and required temporary dialysis. Of the remaining 4, 1 presented transient renal impairment (intentional coverage of polar renal artery), 1 developed a urinary tract infection, 1 had a gastrointestinal complication, and 1 developed a false aneurysm, which was treated with thrombin injection. No cases of bowel ischemia, secondary reinterventions due to access problems, or acute conversions were reported. The median postoperative length of stay was 2 days (interquartile range, 2–3 days).
Midterm Results
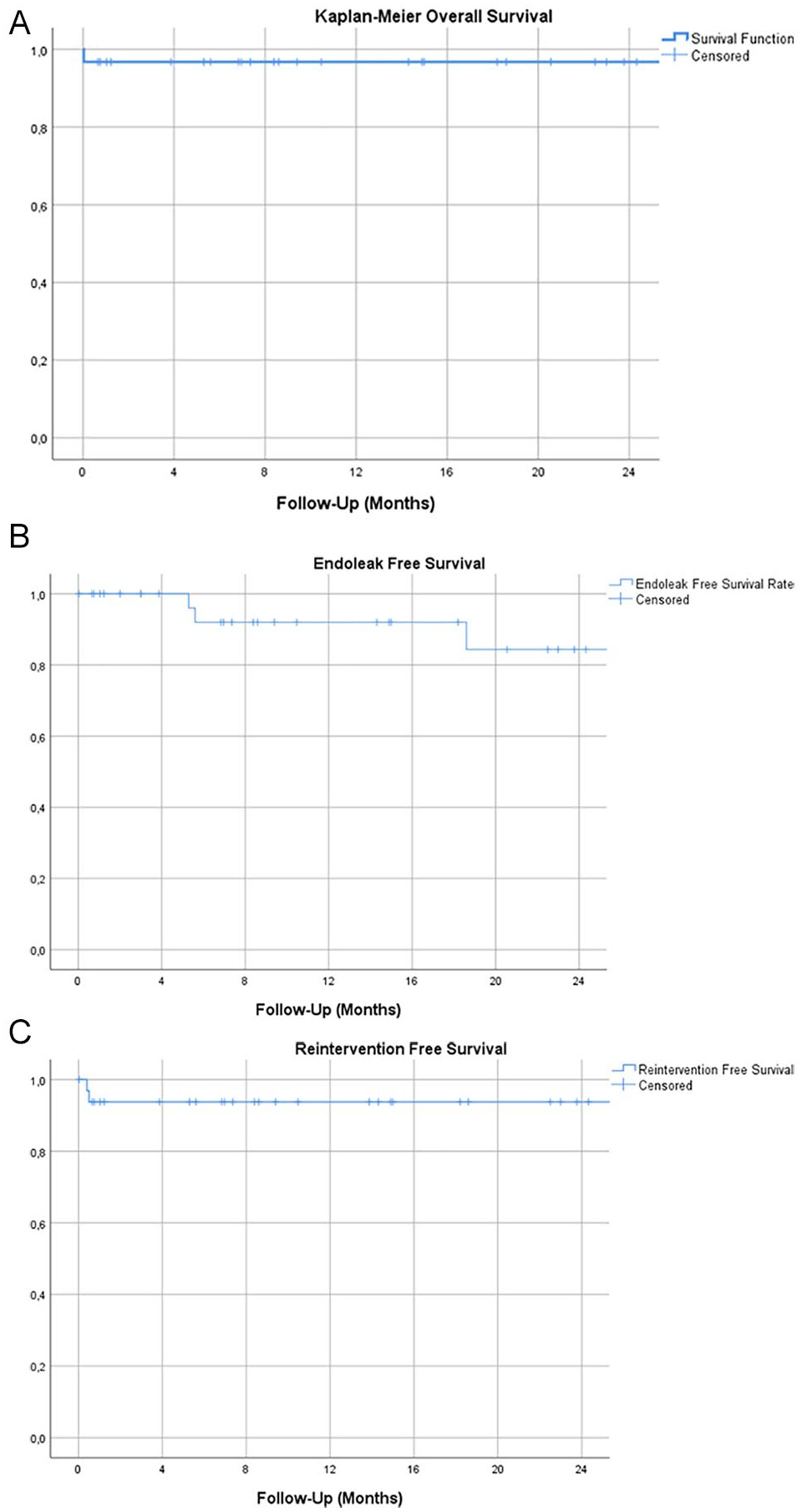
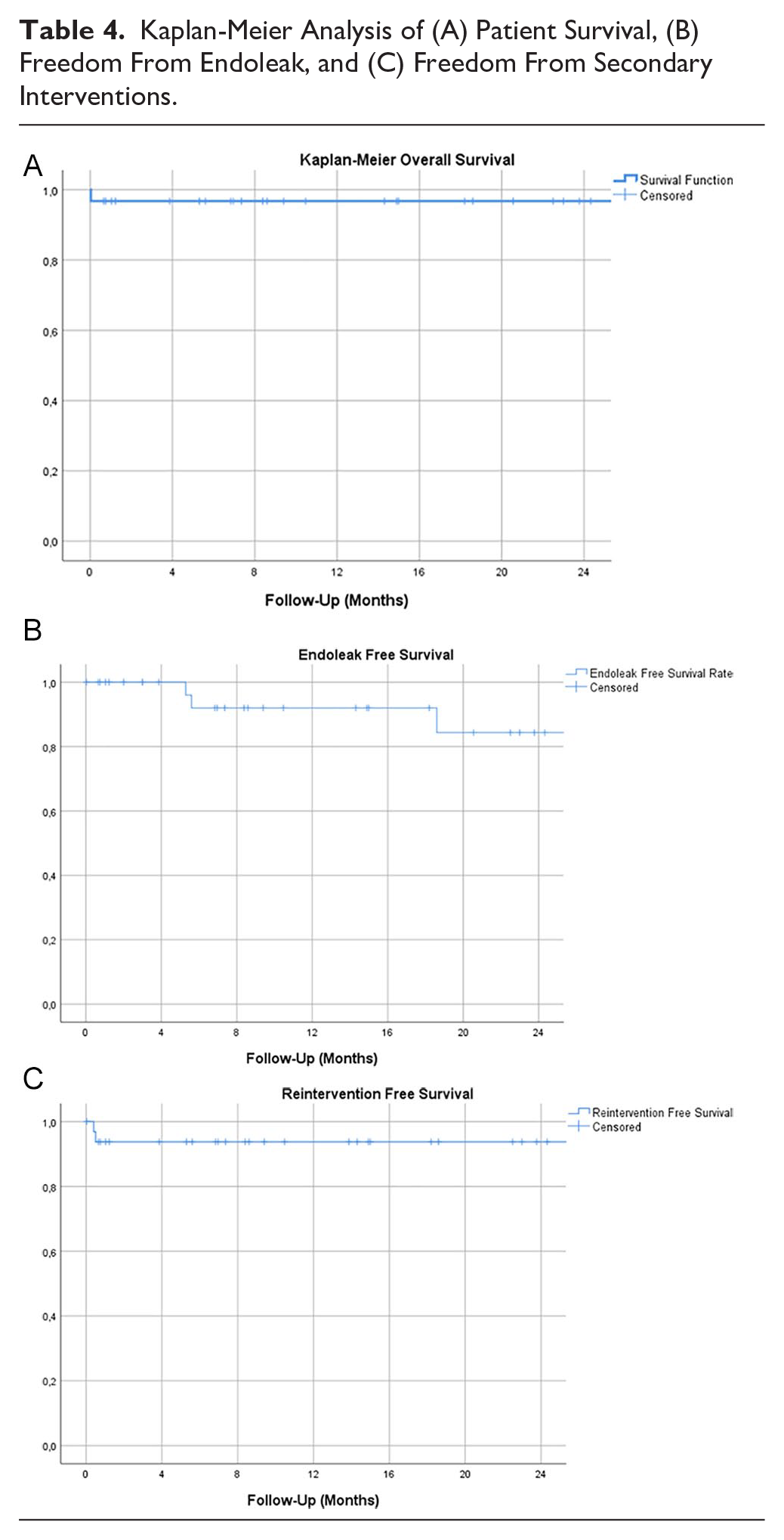
The median follow-up time was 13.9 months (2–39 months). During this period, no vascular reinterventions or vascular-related deaths were reported. One patient died during the follow-up from an undetermined cause. Only 19 patients (47.5%) had long-term postoperative computed tomography angiography. In 4 patients, a type II endoleak was observed. During the follow-up, growth of 2 iPAU was described; 1 as a result of a type II endoleak and 1 of an undetermined cause. No cases of type I or III endoleaks, graft migration, thrombosis, or stent-fracture were observed. Kaplan-Meier survival analysis was used for evaluation of midterm results for patient survival, freedom from endoleak, and freedom from secondary interventions. The results are presented in Table 4.
Kaplan-Meier Analysis of (A) Patient Survival, (B) Freedom From Endoleak, and (C) Freedom From Secondary Interventions.
Discussion
The primary objective of this retrospective multicenter study was to evaluate the feasibility of the BeGraft aortic stent-graft in the treatment of iPAU and its short-term results. The most common indication to use BeGraft aortic stent-graft instead of a bifurcated device were localized infrarenal lesions of a short extent at the mid-part of the infrarenal aorta. Thirty-nine of the 40 patients were successfully treated, with 0% 30-day mortality and a short-term 2.5% reintervention rate. The single failure observed in a symptomatic patient was most probably due to extensive oversizing in a heavily circular calcified landing zone leading to rupture.
The general demographics of the patients included in this study do not differ greatly to those reported in prior literature, with elderly patients associated to a heavy atherosclerotic disease burden being the norm.11,12 Symptomatic patients made up 27.5% of the cohort. This is a slightly higher incidence of symptomatic patients as compared with systematic reviews of the literature. 4 A significant number of patients were treated due to the size or configuration of the aneurysm, estimated at higher risk of rupture. However, a careful balance between rupture risk and operative risk in this aged group of patients is required, especially since clear cutoff values for indicating repair of iPAU have not yet been established. It is generally accepted that PAUs have a higher rupture risk compared with dissection alone and an urgent treatment is often advocated.13,14
Endovascular therapy is the treatment of choice in this patient cohort, with options including standard bifurcated endografts, iliac limbs, as well as thoracic tube endografts. All of these have shown good results in selected patients. 12 In our opinion, bifurcated endografts have an important role in managing PAUs that lie close to the aortic bifurcation, and in which the aortic and iliac diameters are sufficiently large to allow for their use. In cases in which the PAU is located in the mid-part of the infrarenal aorta, bifurcated grafts, especially those with a suprarenal fixation stent, may be an overtreatment of very limited lesions. The CERAB technique, which has not been found to associate a higher risk of type III endoleaks represents a value alternative to the use of bifurcated endografts in patients with PAUs close to the aortic bifurcation who present narrow aortic bifurcations and/or iliac arteries. Thoracic endografts are another endovascular option; however, they associate longer lengths (frequently too long for the infrarenal aorta) as well as bigger diameters (both of the stent-grafts and their introducer sheaths), limiting their use in the infrarenal and heavily calcified aortas. Iliac limb extensions are usually too narrow in diameter and are wider at the bottom end, contrary to what needed for iPAUs, where the larger diameter is needed at the top. Use of a balloon-expandable stent-graft for iPAUs represents a different approach. The potential higher radial force of the balloon-expandable stent and the low profile have a benefit in treatment of heavily calcified, small-diameter aortas, which was also the case in this study. To better understand the morphological characteristics of the iPAU, we also collected the data regarding the distance of the PAU from the renal arteries and to the aortic bifurcation. The relatively short mean length of base of the PAU of just 20.8±1.3 mm has a positive side effect, namely that the mean distance from the lowest renal artery to the upper edge of the PAU was 38±3 mm and the mean distance from the lower edge to the aortic bifurcation was 29±15 mm. By using the shorter lengths of the BeGraft aortic stent-graft, we were able to achieve a sufficient sealing and still preserve the patency of the inferior mesenteric artery in 26.3% of cases (Figure 1). In this sense, one of the main advantages of using the BeGraft aortic stent-graft becomes especially evident when treating PAUs that are limited to the infrarenal aorta, with enough landing zone to achieve proximal and distal with a maximum of 1 or 2 stents.
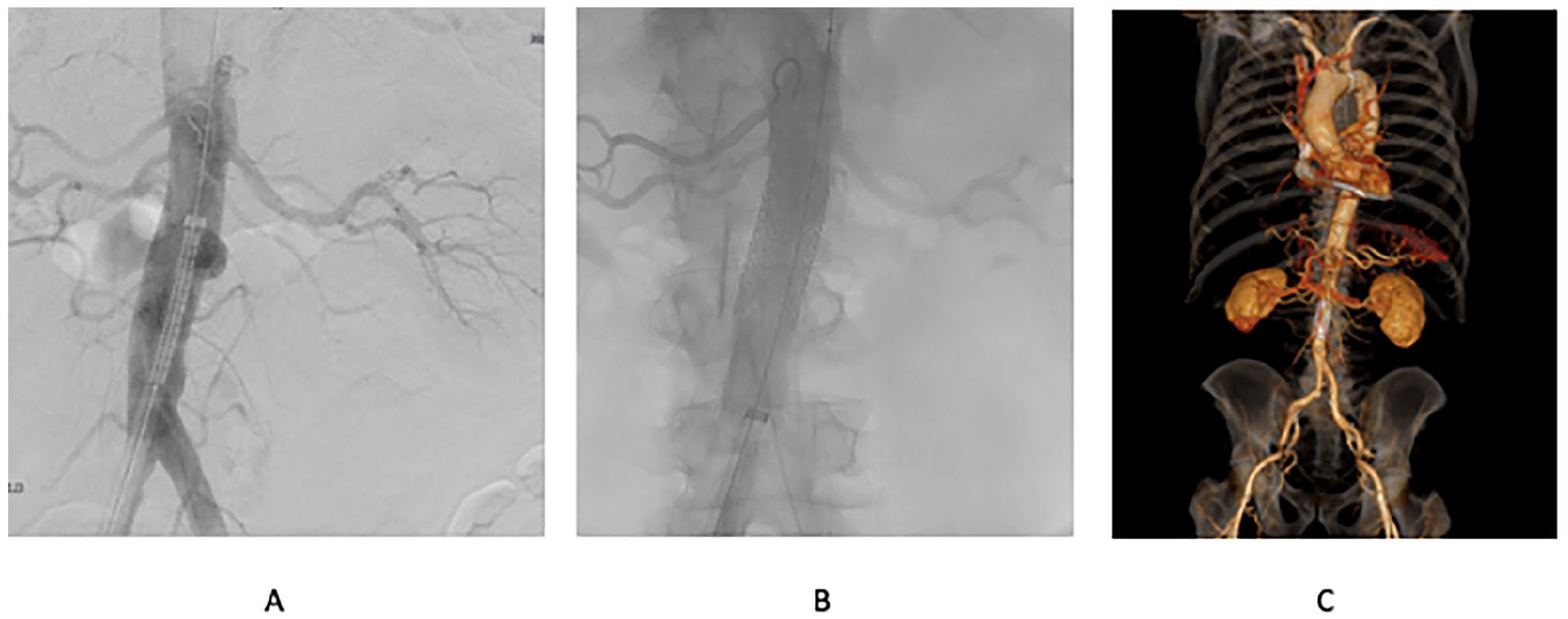
(A) Infrarenal penetrating aortic uncer (iPAU) before exclusion. (B) Angiography after stent placement. (C) One-year follow-up computed tomography scan.
The BeGraft aortic stent-graft allows treatment of aortas with diameters between 12 and 24 mm. The 24-mm stent can be post dilatated up to 30 mm, which is important to adjust the stent size to the diameter of the landing zone if needed. Clear requirements for proximal and distal landing zones for exclusion of iPAU have also not been defined in the literature. Applying the same general concepts as those traditionally used in EVAR, one may assume that for a durable result, free from migration and type I endoleaks, at least 10 to 15 mm of healthy proximal and distal aortic tissue are necessary for exclusion of the PAU. In our cohort, the mean proximal diameter of the infrarenal aorta was 20.8±1.3mm, indicating that in these patients iPAU was not related with an aortic dilatation, contradictory to findings observed in the thoracic aorta. 7 On the contrary, the mean minimum infrarenal aortic diameter observed in these patients was 15.5 mm, with the smallest being 9 mm. These diameters make it technically challenging to perform standard EVAR and cause a significant compromise to its long-term durability. Furthermore, the cost of the therapy is reduced when avoiding using a bifurcated device.
The BeGraft aortic stent-graft allows treatment with either single or multiple stents, in a tubular-straight or bifurcated configuration with the use of the CERAB technique. Regarding the number of used stents, the results have shown that in 82.6% of cases one or two stents were enough to sufficiently exclude the iPAU. A positive effect of this tubular design is that the aortic bifurcation does not need to be stented, especially important in cases of narrow aortic bifurcations (Figure 2). Expanding the treatment of patients with a short distal landing zones, over the aortic bifurcation was successfully done in 5 cases, requiring however multiple stents to be used. To extend the repair in the iliacs, a technique similar to CERAB was used, what enabled treatment of small diameter aortic bifurcations.
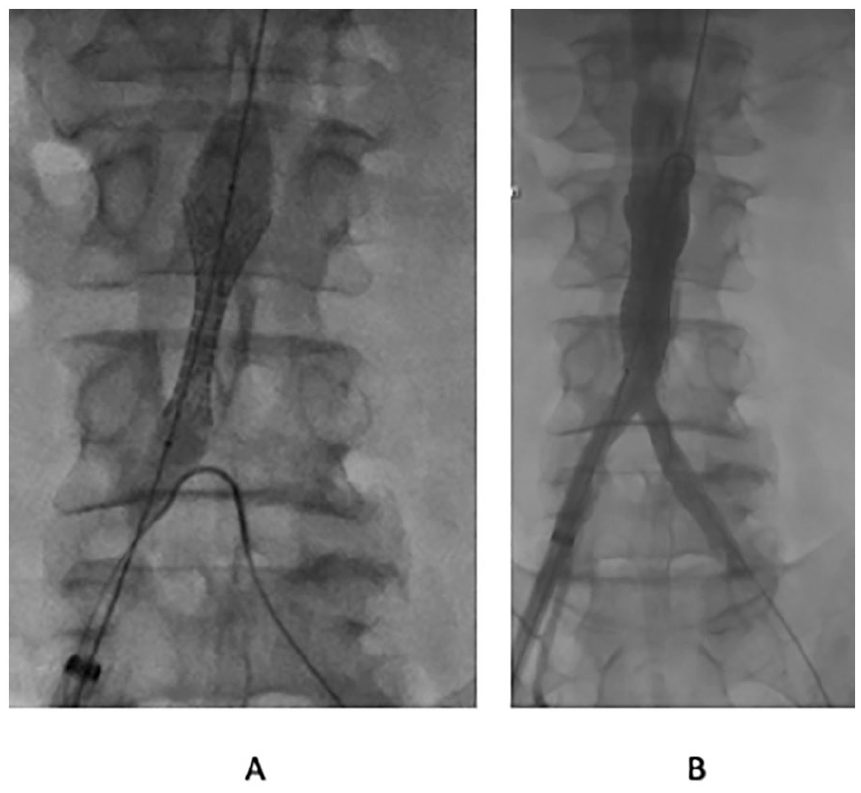
Example for treatment of infrarenal penetrating aortic ulcer (iPAU) with a short distal landing zone. (A) Shoulder of the balloon opening in the common iliac artery. (B) Postimplantation angiography.
One of the goals of this study was to observe the impact of oversizing on the rate of postoperative endoleaks, especially type Ia and Ib endoleaks. In up to 20 cases, a mean value of 16.2% oversizing was used, 8 cases had no oversizing, and in 12 cases an average of 14.9 % undersized stents were used. These results reflect a degree of uncertainty regarding the optimal stent diameter. A similar trend was observed regarding the landing zone length, with a mean proximal landing length of 22 mm (range, 4–52 mm) and a mean distal landing zone length 18.3 mm (range, 0–50 mm). An issue with short distal landing zones is that the shoulder of the balloon will open up in the iliac artery. This can potentially lead to rupture of the artery due to massive oversizing of the balloon. To overcome this problem, one possibility is to partially deploy the stent-graft, move the balloon out of the iliac artery, and then fully deploy the stent. Given also the ruptured case in this series, it seems that avoiding extensive oversizing is a key point in choosing the right endograft size.
Two possibilities exist to achieve a proper sealing. The first is the use of 10% to 30% oversizing of the chosen stent, thereby taking the higher risk of rupturing the aorta. A second technique is to use a nonoversized stent, which can be flared to the proximal or distal diameter of the aorta as needed. One of the disadvantages of this technique is that most of the large diameter balloons have a large shoulder, which reaches over the top of the covered area. However, an additional advantage is that this can be applied in tapered aortas, where a dilation proximally with an appropriate balloon would ensure proper oversizing proximally and distally. We would suggest a 10% oversizing at the level of the narrowest lending zone and a subsequent postdilatation with a noncomplaint balloon at the wider aortic segments. CERAB can also be used to address proximal or distal diameter differences. Because proximal cuffs lose the benefits of the small introducer sheaths, their use should be limited to cases in which one wants to achieve additional tapering or to avoid deployment of balloon-expandable stents in a heavily calcified part of the aorta.
The completion angiography showed a primary success of 97.5%, without a significant higher rate of endoleaks in compartment of these 3 groups. In 3 cases, the BeGraft aortic stent-graft was extended proximally with a proximal cuff as part of the treatment concept to overcome tapering issues of the infrarenal aorta. Another potential benefit of straight cuffs would be to avoid the use of stent-grafts with a higher radial force (balloon-expandable) in heavily calcified aortas. Two percutaneous transluminal angioplasties of the iliacs were also required. Moreover, 4 type II endoleaks were reported in the follow-up in which one resulted in iPAU growth.
One limitation of this study, besides its retrospective nature, is the short mean follow-up of 13.9 months, as well as the various technical approaches in the use of the BeGraft aortic endograft in different centers. Because of the low number of computed tomography angiographies controls in the follow-up, only limited statement of the midterm results about endoleaks, stent migration and fractures of the BeGraft aortic stent-graft can be made. Long-term results are needed to accurately assess the performance of balloon-expandable stent-grafts for treatment of iPAU, as well as the determination of the optimal oversizing to prevent endoleaks in the long run.
Conclusion
For selected patients and anatomies, the treatment of iPAU with the BeGraft aortic stent-graft is feasible with low rate of perioperative morbidity and mortality. Balloon-expandable stent-grafts offer the option to repair iPAUs with a simple technique and a shorter coverage of the aorta. The low-profile sheaths necessary enable the treatment through calcified access vessels and of small diameter aortic bifurcations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Department of Vascular Surgery Ludwig-Maximilians University Hospital, Munich, Germany received travel and institutional research grants from Bentley InnoMed GmbH, Hechingen, Germany.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.