Abstract
Keywords
Introduction
Chronic limb-threatening ischemia (CLTI), the most severe presentation of peripheral artery disease (PAD), is associated with high rates of major amputation and mortality. Following the initial diagnosis of CLTI, mortality risk is 24% over 1 year and 60% over 5 years. 1 CLTI manifests clinically as lower extremity ischemic rest pain and/or ischemic tissue loss and is commonly linked to multilevel, long-segment atherosclerosis. 2 CLTI is highly prevalent in older patients with diabetes mellitus and/or chronic kidney disease and results in an estimated 150,000 amputations annually in the United States and more than 50,000 in Germany.3–5
Historically, up to 54% of patients in some regions underwent major amputation without a prior attempt at revascularization, prompting calls for renewed efforts at improving limb salvage in this population.6,7 As a result of lower procedure-related complication rates, shorter length of stay, and faster recovery, infrapopliteal percutaneous transluminal angioplasty (PTA) has become widely accepted as a primary therapy to establish a direct line of blood flow to the foot to assist wound healing and relieve rest pain, especially in patients at high perioperative risk. 8
Long lesions in small-diameter vessels with medial calcification add to the complex nature of PTA in the BTK arteries. The mechanism of PTA consists of plaque fracture, localized dissection, and permanent medial overstretching, often with dehiscence of the intima and media. 9 Post-PTA dissection adversely impacts clinical outcome and can be a predictor for infrapopliteal restenosis. 10 The lack of a dissection repair tool has been a deterrent to safe and effective infrapopliteal endovascular intervention for CLTI. These patients often undergo multiple reinterventions, and surgical outcomes are poor after failed endovascular attempts at revascularization.11–13 Thus, optimizing BTK PTA outcomes will provide durable vessel patency, reduce reinterventions, and promote limb preservation.
Materials and Methods
Study Design
The 4-F Tack Endovascular System (Intact Vascular, Wayne, PA, USA) is specifically designed for infrapopliteal post-PTA dissection repair. Because the Tack-Optimized Balloon Angioplasty (TOBA) II BTK study was the first investigational device exemption (IDE) study to evaluate a BTK implant for this indication, there was no approved implant to be compared in a randomized controlled trial. Instead, a prospective, multicenter, single-arm trial of the Tack Endovascular System was designed to be consistent with the safety objective performance goal (OPG) established by the Society for Vascular Surgery Critical Limb Ischemia Working Group’s guidance published in 2009.
14
Efficacy OPGs were derived from a systematic review of the BTK angioplasty literature. Details of the TOBA II BTK study design and the derivation of its OPGs have been described.
15
The TOBA II BTK study protocol was approved by all relevant institutional review boards of the 41 sites (Appendix 1) in the United States, Europe, and New Zealand. The trial was conducted in compliance with the Declaration of Helsinki, International Conference on Harmonization E6–Good Clinical Practice, and ISO 14155. Independent imaging core laboratories adjudicated all study angiograms (Yale Cardiovascular Research Group, New Haven, CT, USA) and duplex ultrasound scans (VasCore, Massachusetts General Hospital, Boston, MA, USA). Safety outcomes were adjudicated by an independent Clinical Events Committee (CEC), with patient safety and scientific integrity overseen by an independent Data Safety Monitoring Board. The trial was registered on the National Institutes of Health website (
Study Device
Tack implants are designed to treat dissections by applying a low outward radial force, thereby creating tissue apposition while minimizing the amount of metal in contact with the vessel wall. The 4-F Tack Endovascular System (Figure 1) is compatible with a 0.014-inch guidewire and preloaded with four 6-mm nitinol scaffolds that feature open cell geometry and a relatively low, flat force curve. This design allows each Tack implant to individually self-size across reference vessel diameters of 1.5 to 4.5 mm with a minimal metal footprint. Radiopaque markers and anchors are located around the centerline of each Tack implant to ensure accurate and secure placement. The Tack Endovascular System (4F) received premarket approval from the Food and Drug Administration in April 2020.
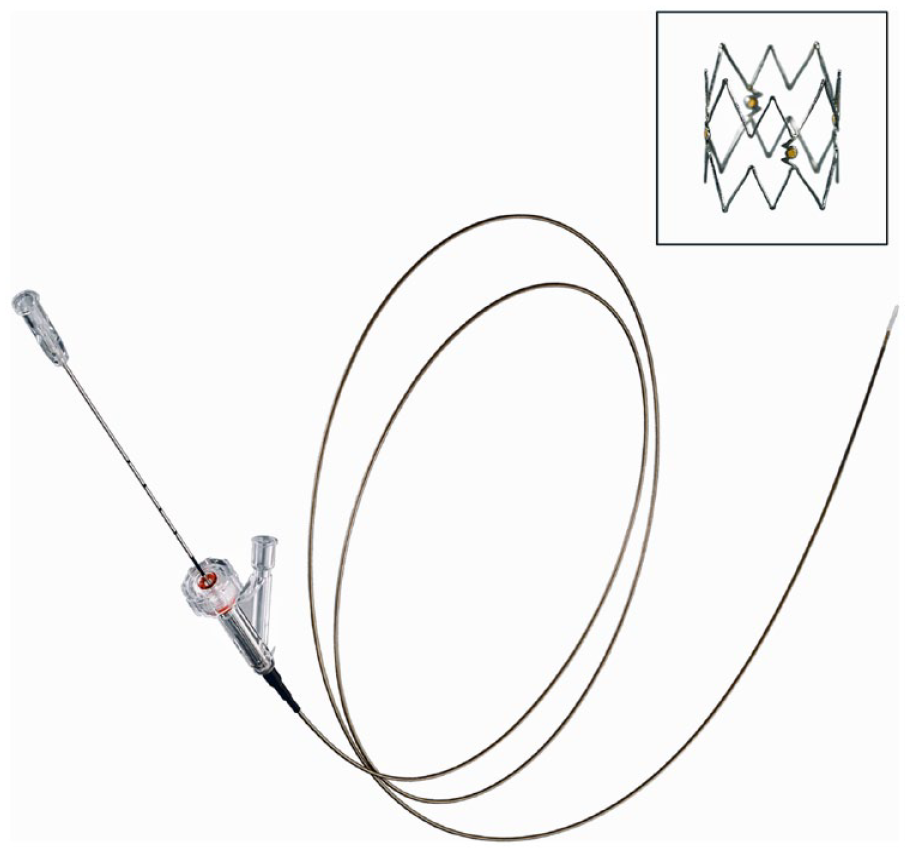
Tack Implant and Tack Endovascular System (4F).
Eligibility
Patients presenting with Rutherford category (RC) 3–5 lower extremity ischemia underwent screening for trial participation using the inclusion and exclusion criteria summarized in Supplemental Table 1 (available in the online version of the article). To ensure that the study population included predominantly CLTI patients, enrollment of RC 3 patients was discontinued several months into the study after 38 RC 3 patients had been enrolled. Baseline data collection included standard demographic data along with history and physical examination, laboratory testing, ankle-brachial index (ABI) and toe-brachial index (TBI) measurements, RC status, WIfI (wound, ischemia, and foot infection) classification,16,17 the EQ-5D-3L quality of life (QoL) questionnaire, 18 and the Walking Impairment Questionnaire (WIQ). 19
Patients underwent BTK intervention after successful screening, baseline data collection, and informed consent. These patients were eligible for enrollment during the index procedure if angioplasty resulted in dissection(s) of the mid/distal popliteal artery segments, tibioperoneal trunk, or the anterior tibial, posterior tibial, and/or peroneal arteries that the investigator would have otherwise treated with prolonged repeat angioplasty, stent deployment (off-label in the United States), or bypass surgery. There was no limit to the number or length of ipsilateral BTK target lesions that could be enrolled. Tacks were not permitted to be implanted within 1 cm above the radiographic tibiotalar joint.
Procedure
In keeping with the institutional standard of care and the manufacturer’s instructions for use, angioplasty was performed using a standard uncoated PTA balloon sized 1:1 with the vessel diameter (visual estimate). Post-PTA dissections were identified using the angiographic core laboratory protocol, which required imaging in at least 2 planes with ≥45° difference and use of magnification. Dissection(s) were graded by the operator using the National, Heart, Lung and Blood Institute (NHLBI) classification system. 20 Patients were eligible for enrollment if the investigator concluded that the dissection(s) required repair. A patient was enrolled when the Tack delivery system was advanced through the introducer sheath.
Study implants were deployed under fluoroscopic guidance, beginning with the most distal dissection and working proximally. The center markers of each implant were aligned with the intended treatment site and the implant was deployed by retracting the outer sheath. Tack implants were not permitted to overlap. Placement of additional implants at the operator’s discretion was performed by repeating the process, moving in a proximal direction, and leaving a recommended gap of ≥4.0 mm between individual Tacks. Each implant was postdilated using a new balloon of size and inflation pressure that did not exceed initial angioplasty. Final angiography was performed to confirm acute procedural success, including dissection resolution. Clinical follow-up was performed at 30 days, 6 months, and 12 months with planned follow-up to 36 months.
Patient Population
Patient enrollment and follow-up through 12 months is depicted in Figure 2. Between February 2017 and December 2018, the study enrolled 233 patients (mean age 74.4±10.0 years; 157 men). Table 1 summarizes patient and lesion characteristics at baseline. Half (50.2%) of the patients (117/233) were classified as RC 5, 33.5% (78/233) as RC 4, and 16.3% (38/233) as RC 3. Consistent with the predominance of CLTI, considerable rates of diabetes (153/233, 65.7%), smoking (145/233, 62.2% current or former), and chronic kidney disease (57/232, 24.6%) were present.
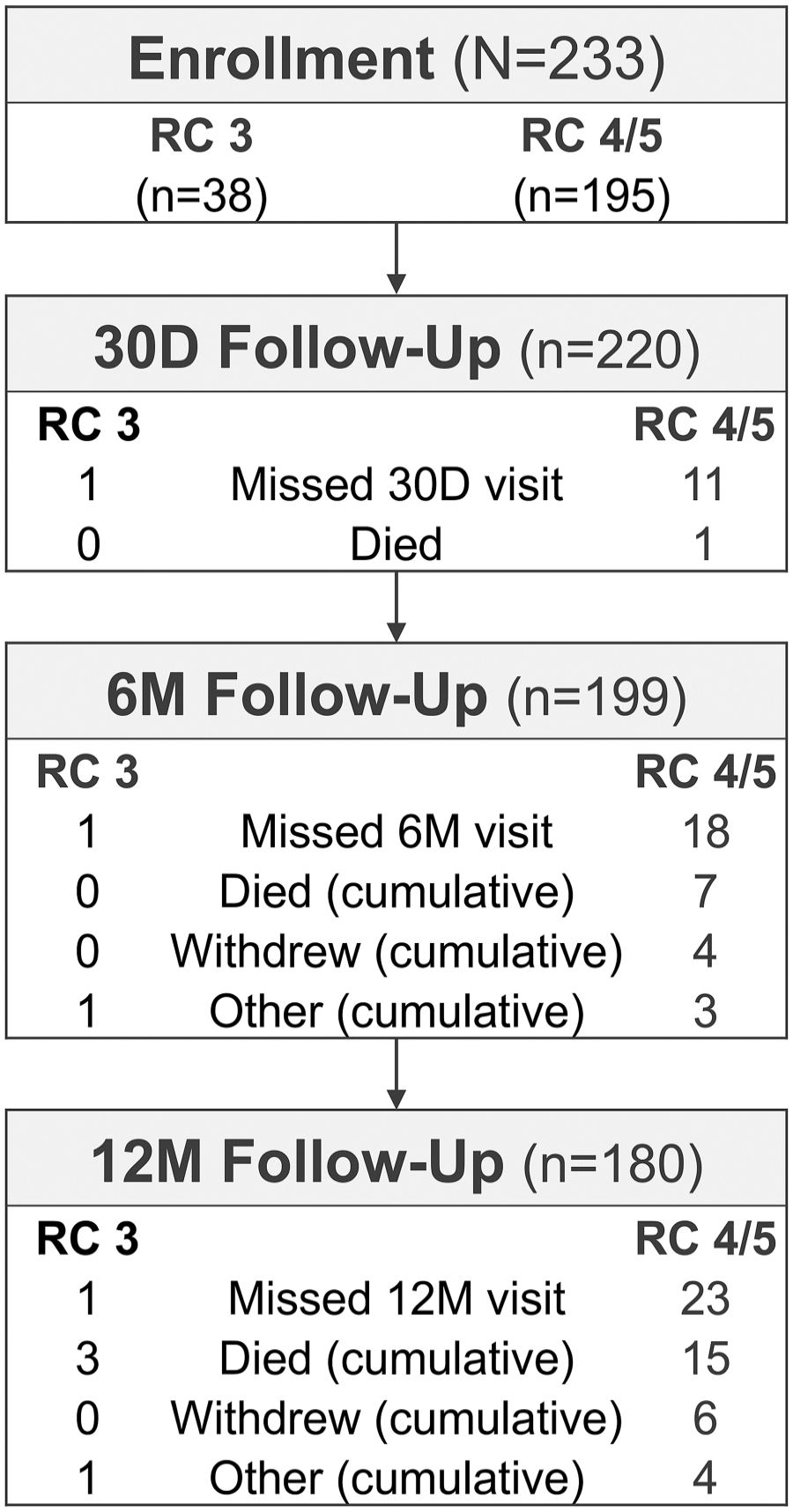
Patient enrollment and follow-up. D, day; M, month; RC, Rutherford category.
Baseline Characteristics for the 233 Study Patients With 248 Below-the-Knee Target Lesions. a

Abbreviations: BMI, body mass index; P1, proximal popliteal artery; PVI, peripheral vascular intervention; RVD, reference vessel diameter; SFA, superficial femoral artery; TBI, toe-brachial index.
Continuous data are presented as the mean ± standard deviation (sample size) [minimum, median, maximum]; categorical data are given as the number/sample (percentage).
Coagulopathy, hypercoagulable state, bleeding diathesis, or other.
Reported per lesion; adjudicated by angiographic core laboratory.
Most lesions (257/274, 93.8%) were de novo as reported by the investigator (248 lesions adjudicated by the core laboratory). Lesions were located in each of the BTK arteries, with the majority in the anterior tibial (103/248, 41.5%), posterior tibial (55/248, 22.2%), and peroneal arteries (53/248, 21.4%). Almost half (118/248, 47.6%) of the lesions were core laboratory–adjudicated as total occlusions. Mean baseline target lesion length was 80±49 mm (Table 2). Using the Peripheral Academic Research Consortium (PARC) classification, 21 moderate calcium was defined as ≥180° (both sides of the vessel at the same location) and less than one-half of the total lesion length and severe was defined as >180° (both sides of the vessel at the same location) and greater than one-half of the total lesion length. Moderate to severe calcium was present in 35.8% (89/248) of lesions.
Details of Procedures to Treat 248 Lesions. a
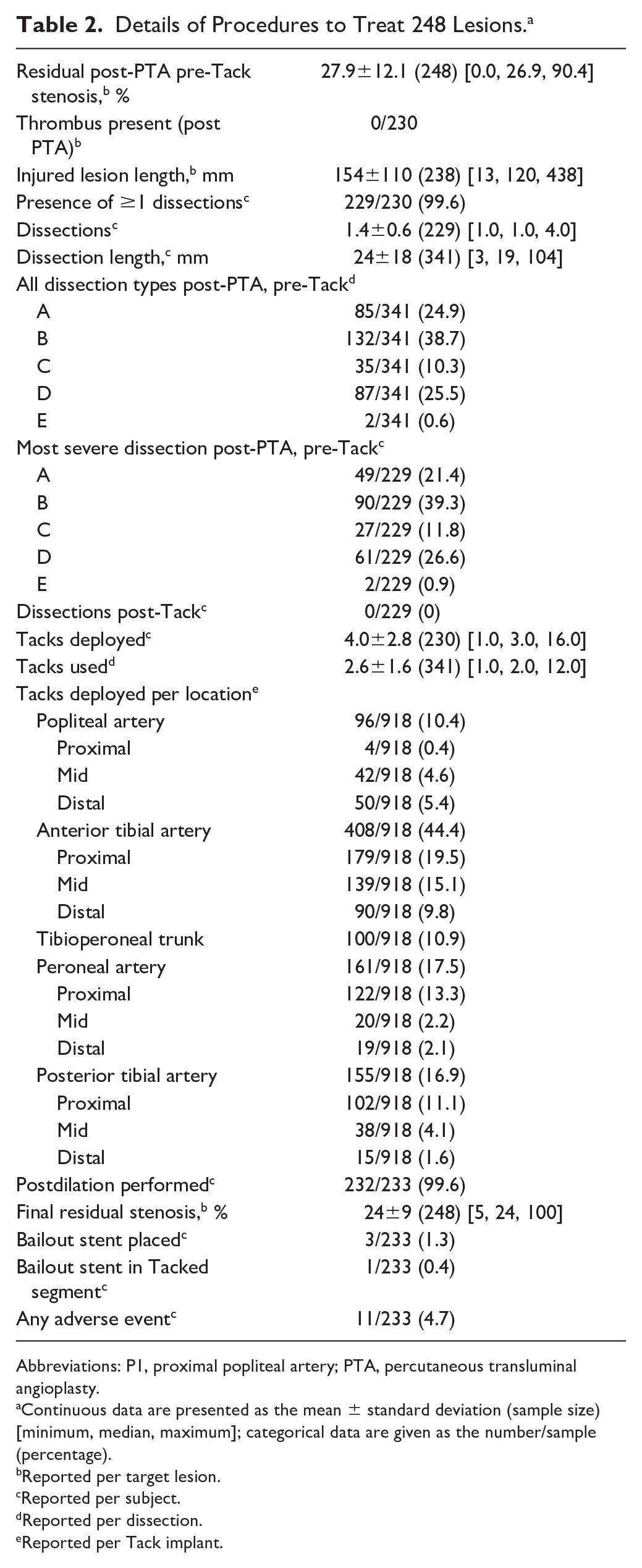
Abbreviations: P1, proximal popliteal artery; PTA, percutaneous transluminal angioplasty.
Continuous data are presented as the mean ± standard deviation (sample size) [minimum, median, maximum]; categorical data are given as the number/sample (percentage).
Reported per target lesion.
Reported per subject.
Reported per dissection.
Reported per Tack implant.
Study Outcomes and Definitions
The 30-day primary safety endpoint was a composite of major adverse limb events (MALE) and postoperative death (POD). MALE was defined as any above-ankle target limb amputation and/or major target lesion reintervention (defined as placement of a new bypass graft, jump/interposition graft revision, or performance of thrombectomy/thrombolysis). The primary efficacy endpoint was a combination of freedom from MALE at 6 months and 30-day POD. Safety and efficacy endpoints were assessed at 12 months as observational endpoints.
Observational Endpoints
Patency of Tacked segment(s) was defined as presence of prograde blood flow through the treated arterial segment as confirmed by duplex ultrasound. Loss of patency was indicated by either absence of blood flow by duplex or by angiography during reintervention. A Tacked segment was defined as a Tack implant plus 5 mm of artery proximal and distal; if implants were deployed within 10 mm of each other, this was considered a single Tacked segment for the analysis. Target limb salvage was defined as freedom from any above-ankle target limb amputation. Additional longer-term observational endpoints included 12-month amputation-free survival (AFS), freedom from clinically-driven target lesion revascularization (CD-TLR), freedom from clinically-driven target vessel revascularization (CD-TVR), patency of target vessel and target lesion, and changes from baseline in clinical and QoL measures.
Statistical Analysis
All patients who had the Tack Endovascular System advanced through the introducer sheath were assigned to the intent-to-treat (ITT) population. ITT patients were included in all endpoint analyses. Endpoints were analyzed using descriptive statistics based on evaluable assessments. Continuous variables are presented as the mean ± standard deviation (sample size) or the median for skewed data; categorical variables are reported as the number/sample (percentage). Kaplan-Meier time-to-event analyses were used to estimate freedom from MALE + POD, patency, target limb salvage, and AFS; estimates are presented with the 95% confidence intervals (CI). Changes from baseline in RC, ABI, TBI, and QoL questionnaires were evaluated using the Wilcoxon signed-rank test or the McNemar test. Patients missing the 12-month visit (n=53) were compared to 180 patients evaluated at 12 months for differences in baseline variables using chi-square or
Results
Procedure Outcomes
Standard PTA was performed in the 248 target lesions. The PTA treated length of 154±110 mm was longer than the adjudicated mean 80-mm lesion length due to a longer site-reported visual estimate (116±100 mm) and to the use of longer balloons to completely cover the target lesion with a proximal and distal margin of angiographically normal vessel. Following angioplasty, 341 post-PTA dissections requiring repair were identified. The severity and length of each post-PTA dissection was evaluated angiographically (visual estimate) during the index procedure followed by angiographic core laboratory adjudication. Dissection grades ranged from A to E (Table 2).
All patients were treated with at least 1 Tack implant, averaging 4.0±2.8 Tacks per patient. The angiographic core laboratory adjudicated 100% dissection resolution with Tack treatment (341/341). The 918 Tack implants were deployed across all BTK arteries, from mid popliteal to 1 cm above the tibiotalar joint. Per the angiographic core laboratory, 35.0% (321/918) of Tack implants were deployed in the distal two-thirds of the tibial vessels. One patient (1/233) required a stent to bail out a Tacked segment; the overall bailout stent rate was 1.3% (3/233). There was no implant fracture, distal embolization, or migration in 146 cases adjudicated by the core laboratory.
Outcome Analyses
Patients treated with the Tack Endovascular System had favorable safety and effectiveness outcomes at 12 months, as summarized in Table 3. To observe the differences in clinical outcome between claudicants and patients with CLTI, results were stratified post hoc with respect to RC status at baseline. Freedom from the composite MALE at 12 months + 30-day POD was found in 93.4% (170/182) When analyzed separately, all 34 RC 3 patients and 136 (91.9%) of 148 RC 4/5 patients were free of the endpoint. The Kaplan-Meier curves for freedom from MALE at 12 months + POD across RCs are shown in Figure 3.
Outcomes Across Rutherford Categories. a
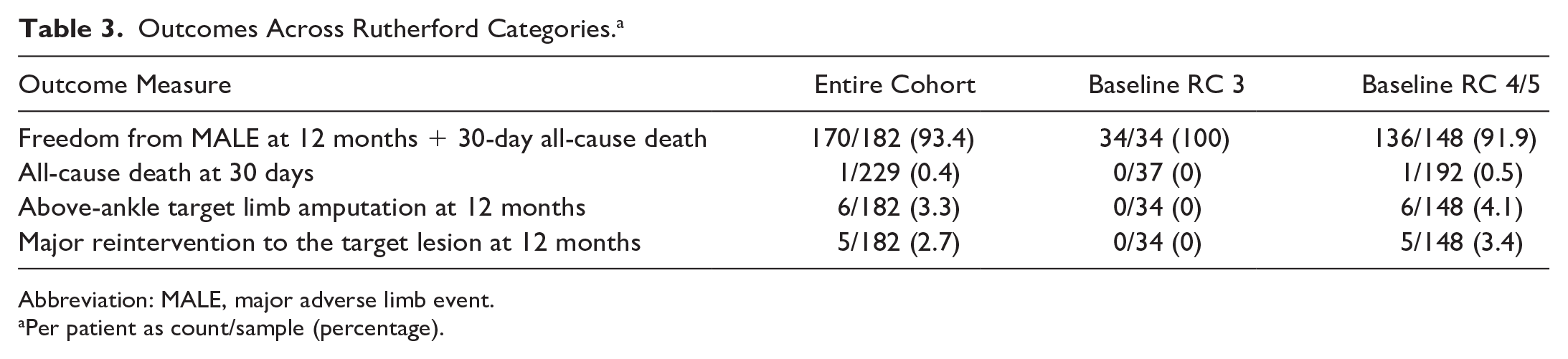
Abbreviation: MALE, major adverse limb event.
Per patient as count/sample (percentage).
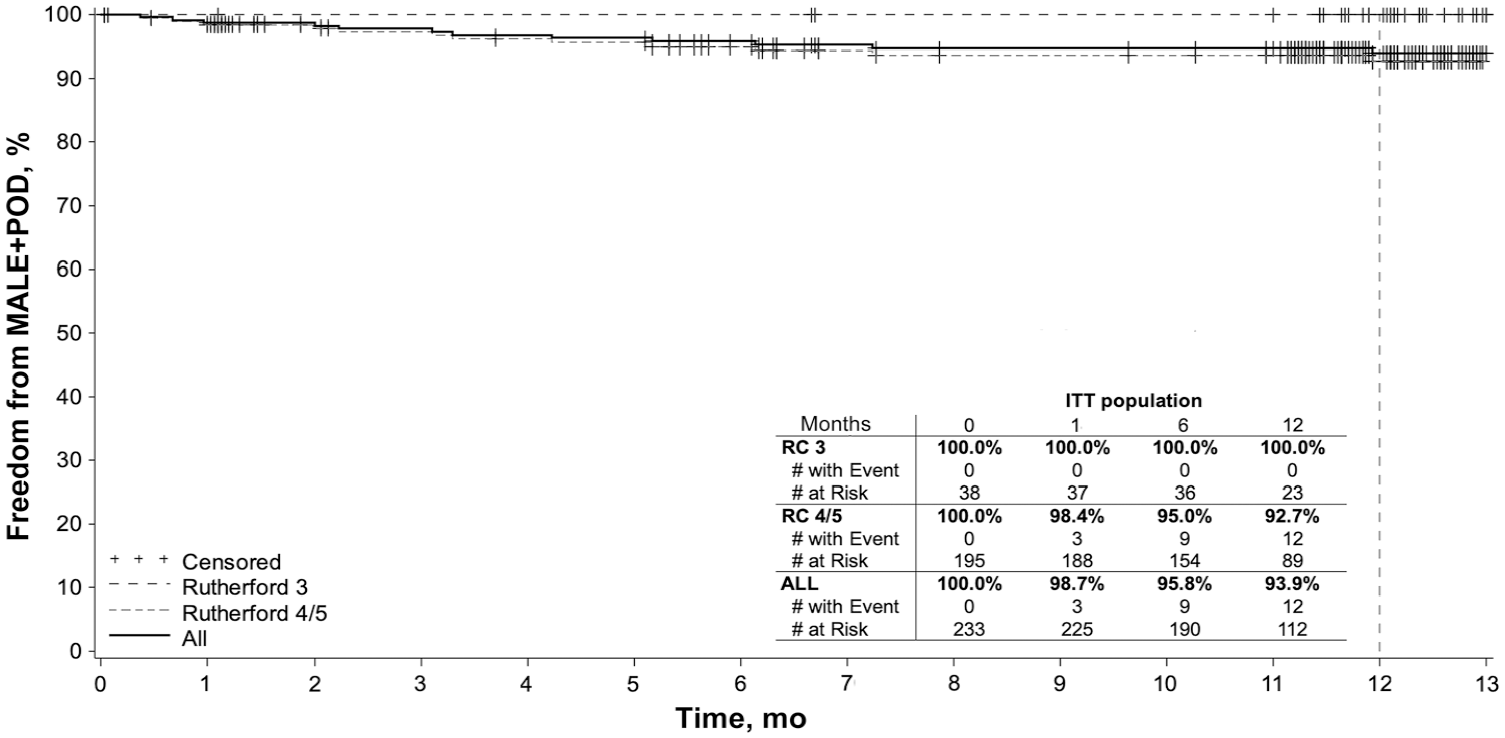
Kaplan-Meier curves for freedom from major adverse limb events (MALE) + postoperative death (POD) in intent-to-treat (ITT) subjects. RC, Rutherford category.
Kaplan-Meier curves for 12-month Tacked segment patency and target lesion patency in the entire cohort and stratified by baseline RC status are shown in Figure 4A and B, respectively. The 12-month Tacked segment patency was 81.3% (95% CI 76.7% to 86.0%) for all patients, 96.0% (95% CI 90.6% to 100.0%) for RC 3 patients, and 78.0% (95% CI 72.5% to 83.5%) for RC 4/5 patients.
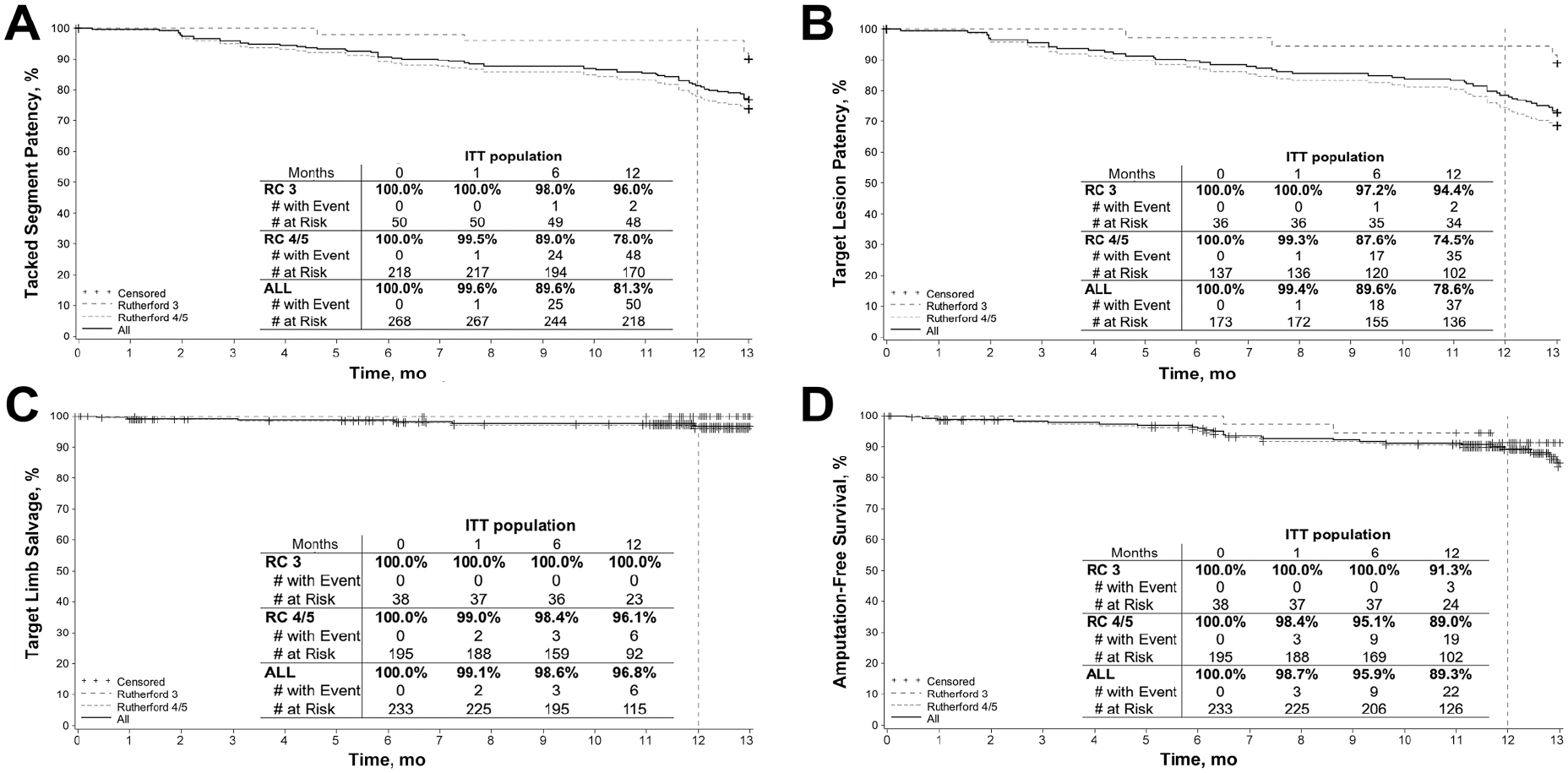
Kaplan-Meier curves for (A) Tacked segment patency, (B) target lesion patency, (C) target limb salvage, and (D) amputation-free survival in intent-to-treat (ITT) subjects up to 12 months. RC, Rutherford category.
The Kaplan-Meier 12-month target limb salvage (Figure 4C) estimate for all patients was 96.8% (95% CI 94.1% to 99.4%): 100% for the RC 3 patients and 96.1% (95% CI 92.9% to 99.3%) for RC 4/5 group. Freedom from CD-TLR was 83.1% in all patients (95% CI 77.7% to 88.4%) and 81.8% (95% CI 75.8% to 87.9%) in CLTI patients. Kaplan-Meier freedom from CD-TVR at 12 months was 82.7% (95% CI 77.4% to 88.0%) and 81.4% (95% CI 75.4% to 87.5%) in all patients and CLTI patients, respectively.
In terms of mortality, there were 17 patient deaths through 12 months, all of which were reviewed by the CEC. No deaths were attributed to the Tack Endovascular System. At 12 months, Kaplan-Meier survival was estimated as 91.7% (95% CI 87.9% to 95.5%) in all patients and 91.9% (95% CI 87.8% to 96.0%) in RC 4/5 patients. There were 6 major amputations at 12 months, none of which occurred in the baseline claudicant population. The estimated AFS (Figure 4D) was 89.3% (95% CI 85.1% to 93.6%) overall and 89.0% (95% CI 84.3% to 93.7%) in RC 4/5 patients.
Changes in Wound Status
Assessment of all wounds present at study entry was performed by the investigator and classified as healed, improved, unchanged, worsening, or amputated (if applicable). At baseline, 111 patients (47.6%) presented with 146 wounds. At 12 months, 22 of those index wounds were not evaluable due to patient death, study withdrawal, or missed visit. Thus, 124 index wounds were assessed at the 12-month interval (Table 4). Investigators reported 85 of 124 wounds (68.5%) as healed and 5 (4.0%) as improving. A total of 25 index wounds were removed by amputation, 17 of which were removed with minor amputation. A post hoc analysis to assess the amputation sites revealed that 17 wound sites had healed and 1 was improving. The remaining sites were not able to be assessed for the same reasons as described above.
Changes in Functional Status
Significant and sustained hemodynamic improvement was seen in ABI and TBI from baseline through 12 months. The average TBI at 12 months was 0.61±0.28, compared with the baseline mean of 0.43±0.23 (p<0.001), with similar improvement in ABI (0.91±0.19 at 12 months vs 0.74±0.27 at baseline; Figure 5). Significant improvement was reported in the WIQ in all categories at 12 months compared with baseline values (p<0.001). Sustained improvement in the RC (Figure 6) was reported in 146 of 177 patients (82.4%), with 107 (62.4%) improving ≥3 categories (p<0.001). In 143 patients with CLTI at baseline, 114 (79.8%) improved, with the majority (101, 70.7%) improving ≥3 levels (p<0.001).
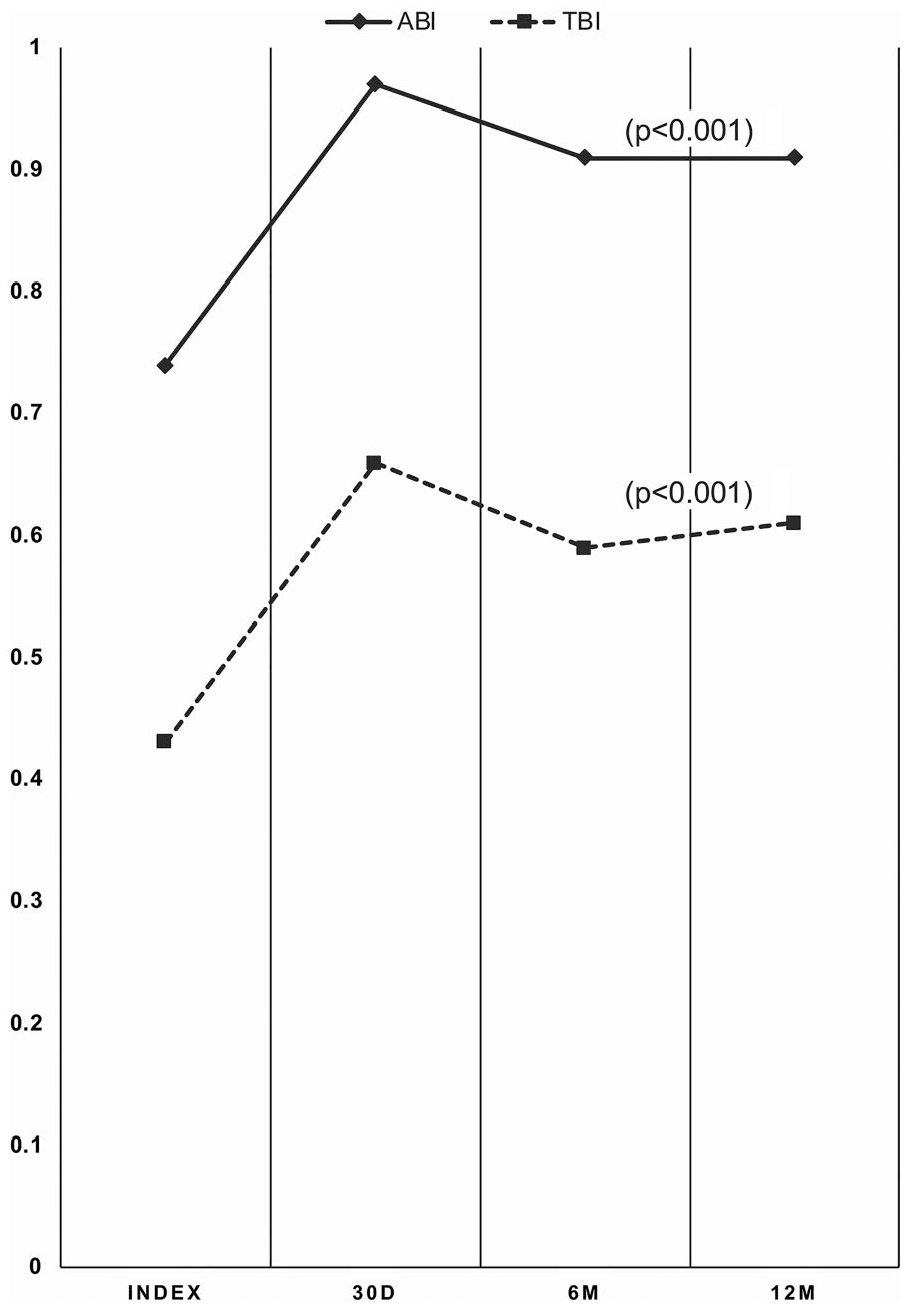
Sustained improvement in the ankle-brachial index (ABI) and toe-brachial index (TBI). D, day; M, month.
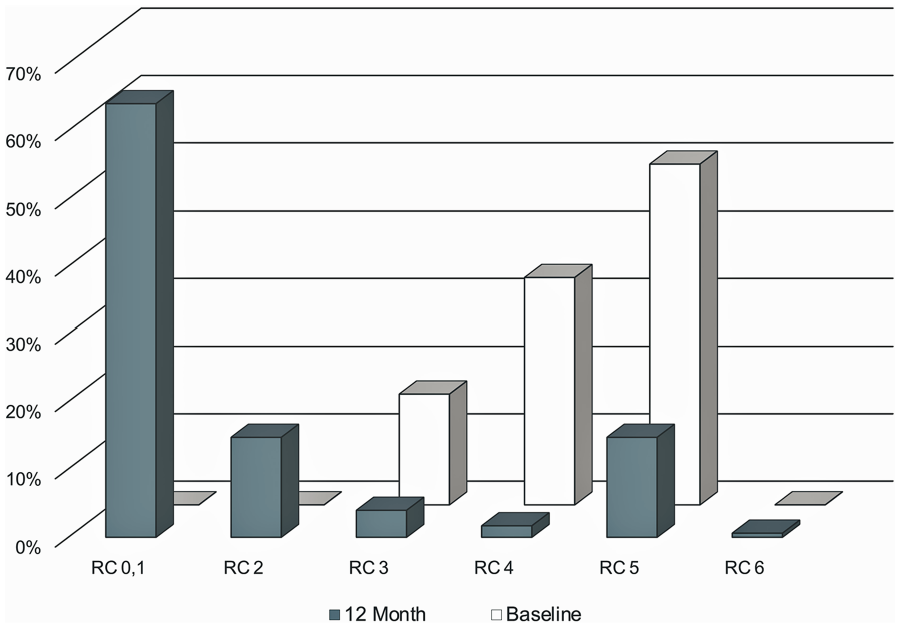
Sustained improvement in the Rutherford category (RC) status.
Changes in QoL Measures
Patients reported improvement in QoL over 12 months in the EQ-5D-3L assessment, with decreased limitations in mobility and pain/discomfort (p<0.001) and increased overall health in the EQ-visual analogue scale scores (p<0.001).
Post Hoc Analysis
In a post hoc analysis of outcomes by initial dissection grade, Tack outcomes were grouped by the severity of the initial dissection (NHLBI grades A-B vs C-F). Initial angiographic (procedural) success with regards to dissection resolution was excellent and unaffected by the dissection severity. Post hoc analyses of various outcome measures through 12 months (Figure 7) showed a significant decrease in Tacked segment patency and nonsignificant decreases in target lesion patency, freedom from MALE + POD, and target limb salvage in patients treated for more severe (grades C-F) dissection.
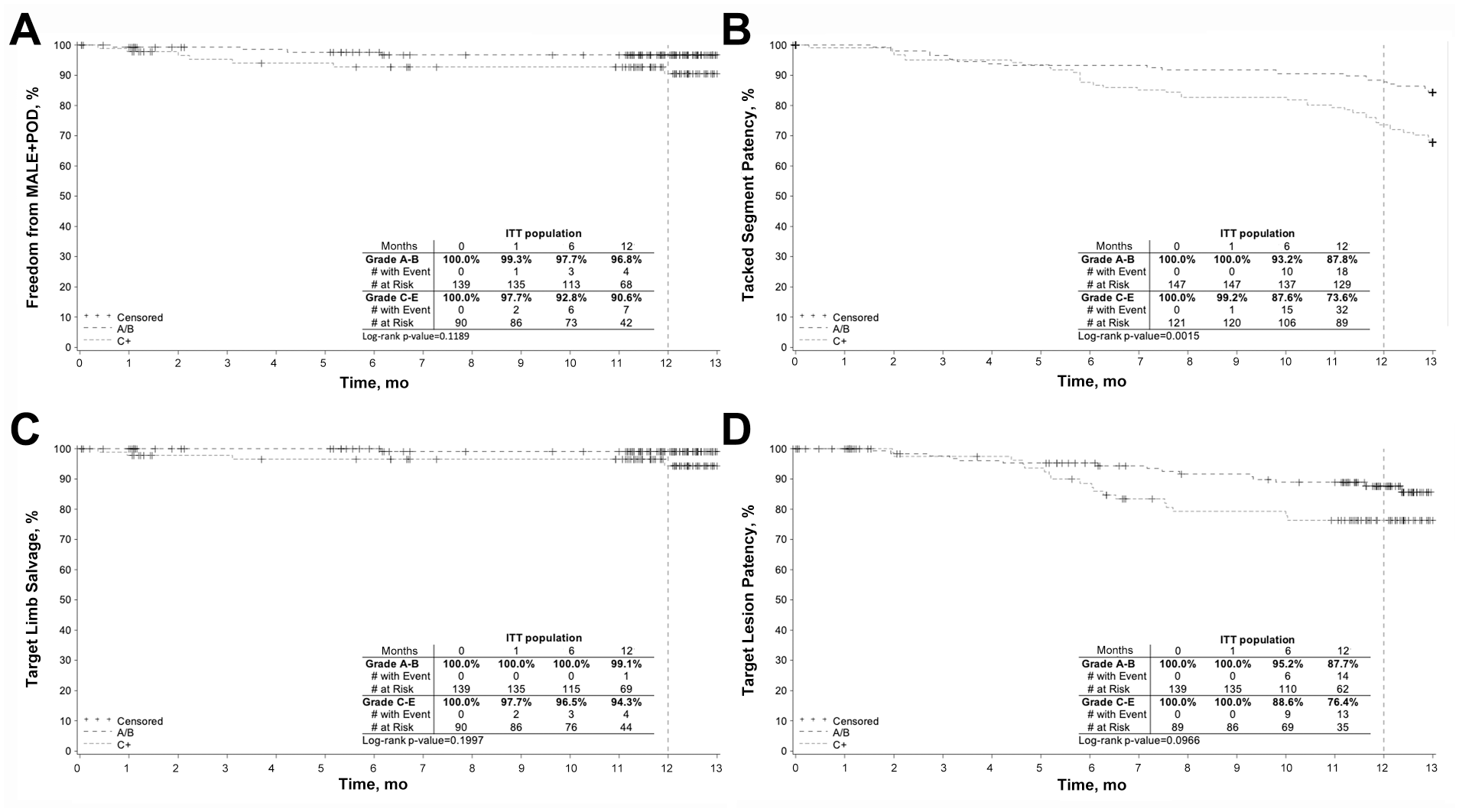
Post hoc Kaplan-Meier analyses in intent-to-treat subjects by dissection grade of (A) freedom from major adverse limb events (MALE) and perioperative death (POD), (B) Tacked segment patency, (C) target limb salvage, and (D) target lesion patency.
Discussion
The incidence of CLTI is likely to increase over time as a result of the diabetes pandemic and an aging population. 22 Atherosclerotic BTK arteries are typically diffusely diseased and accompanied by high resistance outflow. The most common treatment, PTA, functions by creating potentially obstructive dissections and lumen-occupying tissue planes. These dissections are reported in up to 30.7% of BTK angioplasty, though they can be difficult to detect and thus are likely underestimated in frequency and severity. 23 Post-PTA dissection adversely affects clinical outcome and is a predictor of infrapopliteal restenosis. 11 Decreasing the rate of immediate technical failure is integral to limb preservation and improving patency. 24
The clinical benefit of dissection repair with the Tack Endovascular System has been previously reported. In the femoropopliteal segment, excellent outcomes were documented for dissection repair following standard and DCB PTA.25–27 The TOBA BTK pilot study demonstrated improved patency (78.4%) and freedom from CD-TLR (93.5%) at 12 months compared with angioplasty alone. 28 Significant (p<0.001) improvement from baseline in RC status was observed, as 75% of patients improved by 4 or 5 categories. All patients enrolled had dissected vessels, and 88.9% of dissections were repaired by Tacks. 28 In a single-center registry of 51 patients, post-PTA dissection repair was performed with Tack implants both above and below the knee in complex lesions. 29 Technical success was achieved in 98.3% (59/60) dissections and no major adverse event or device-related complication occurred during the procedures. 29
The 12-month outcomes of the TOBA II BTK study provide a robust validation of the promising results seen in the earlier BTK pilot studies. TOBA II BTK enrolled a challenging cohort characterized by long lesion lengths, a high proportion of chronic total occlusions (47.6%), and the ubiquitous presence of post-angioplasty dissection flaps. It is thus gratifying to see that CLTI patient outcomes in this study were commendable, despite the disadvantages posed by lesion complexity. The finding of a trend toward reduced 12-month patency in patients treated for more severe dissection grades is not surprising, as the higher dissection grade reflects the greater degree of arterial wall injury sustained at the index angioplasty and thus the predisposition of that vascular bed to subsequent restenosis over time.
Post-PTA BTK dissection can also be treated using coronary bare metal (BMS) or drug-eluting (DES) stents in an attempt to improve patency and reduce restenosis, depending on the length and location of the lesion. In the US, the use of these devices is currently off-label, and there is no direct comparator device in the literature. The Tack Endovascular System (4F) is the only approved scaffold for use in BTK arteries in the United States.
Limitations
Our study was subject to the characteristic limitations of a single-arm, nonrandomized design. The safety and effectiveness endpoints of this study were consistent with the proposed trial endpoints. 14 Wound status was not adjudicated by the core laboratory and thus was subject to investigator bias. Patients with major tissue loss (RC 6) were excluded from this study, potentially skewing limb salvage outcomes.
Conclusion
The TOBA II BTK study met both its primary safety and efficacy endpoints. After treatment with Tack implants, all dissections were resolved per the core laboratory review. Positive patient outcomes were associated with the Tack-optimized angioplasty procedures as evidenced by low rates of MALE, significant improvements in symptom status, hemodynamic parameters, and patient-reported QoL metrics. The 12-month trial results demonstrate satisfactory midterm safety and efficacy of the Tack Endovascular System for post-PTA dissection repair in patients with advanced BTK atherosclerotic disease.
Supplemental Material
20-0334_supplementary_material – Supplemental material for Twelve-Month Results of Tack-Optimized Balloon Angioplasty Using the Tack Endovascular System in Below-the-Knee Arteries (TOBA II BTK)
Supplemental material, 20-0334_supplementary_material for Twelve-Month Results of Tack-Optimized Balloon Angioplasty Using the Tack Endovascular System in Below-the-Knee Arteries (TOBA II BTK) by Ehrin J. Armstrong, Patrick J. Geraghty, George L. Adams, Andrej Schmidt, Michael Lichtenberg, Christian Wissgott, Ehrin J. Armstrong, Klaus Hertting in Journal of Endovascular Therapy
Footnotes
Appendix 1
Acknowledgements
The authors thank the TOBA II BTK study investigators. The authors acknowledge Shannon Matricardi, RCIS, MA, of Intact Vascular, Inc, for editing and review of the manuscript.
Authors’ Note
The data in this study were presented at VIVA Virtual (Late-Breaking Clinical Trials) (June 25, 2020; Las Vegas, NV, USA).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: George L. Adams is a consultant/advisory board member for Abbott Vascular, Bard, Boston Scientific, Cook Medical, W.L. Gore & Associates, Medtronic, and Cordis, a Cardinal Health Company. Ehrin J. Armstrong is a consultant/advisory board member for Abbott Vascular, Boston Scientific, Cardiovascular Systems, W.L. Gore & Associates, Intact Vascular, Janssen, Medtronic, and Philips Healthcare. Patrick J. Geraghty is a consultant/advisory board member for Bard Peripheral Vascular, Boston Scientific, and Ascension Ventures; he has an equity investment in Euphrates Vascular. Michael Lichtenberg is a consultant/advisory board member for Intact Vascular. Andrej Schmidt is a consultant/speaker for Abbott Vascular, Bard/BD, Cook Medical, Reflow Medical, Upstream Peripheral, and Cordis, a Cardinal Health Company. Christian Wissgott is a consultant/advisory board member for Intact Vascular.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Intact Vascular, Inc.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

