Abstract
Keywords
Introduction
Over the past 2 decades, thoracic endovascular aortic repair (TEVAR) has emerged as a less invasive therapeutic option compared to open repair and is now considered the first-line treatment strategy for several aortic pathologies.1–3 Despite the minimally invasive nature of the procedure, though, TEVAR is sometimes associated with significant morbidity, especially in cases of aortic arch involvement. The failure of the proximal stent-graft to conform to an angulated arch can create the bird-beak configuration and precipitate serious complications such as type Ia endoleak or stent-graft migration. 4 Other complications may be unintentional coverage of major aortic branches or even graft collapse. The complications often require adjunctive procedures or the extension of the proximal landing zone. Thus, besides sizing and planning, the optimal deployment of the endograft remains crucial for both the success and safety of the procedure.
The new delivery system of the Conformable TAG (CTAG) thoracic stent-graft (Gore Medical, Flagstaff, AZ, USA) was developed to provide a more precise deployment of the graft. This Active Control System (ACS) offers an optional angulation mechanism of the proximal stent end and 2-stage deployment for more controlled device positioning. The observational SURPASS registry was created to assess the performance of the CTAG with ACS in daily clinical practice, accumulating device-specific outcomes in the treatment of aortic disease. This article reports the analysis of the 30-day and 12-month outcomes from the registry.
Materials and Methods
Study Design
The SURPASS registry was an observational, prospective, single-arm, post-market, international study that enrolled patients undergoing TEVAR using the CTAG with ACS for both acute and chronic thoracic aortic disease between October 2017 and July 2018. Collaborators at the 20 investigative sites in Europe are listed in the Appendix.
Patients appropriate for treatment with the CTAG with ACS were eligible for the study. The presence of paraplegia or paraparesis at baseline, previous thoracic endovascular treatment, and a life expectancy <1 year were the main exclusion criteria (Table 1). A minimum of 2 follow-up visits at 30 days and 12 months after the initial procedure was specified by the protocol. Imaging follow-up was performed based on the standard of care at the participating centers, which included computed tomography angiography.
Inclusion and Exclusion Criteria.

Abbreviation: TEVAR, thoracic endovascular aortic repair.
All patients provided written informed consent and the trial was conducted according to the Declaration of Helsinki and the International Conference of Harmonization Good Clinical Practice guideline. The protocol was approved by the local ethics committee of each participating center. The trial was registered on the National Institutes of Health website (ClinicalTrials.gov; identifier NCT03286400).
Device Description
The CTAG with ACS is a flexible, self-expanding stent-graft constrained on a delivery catheter that is introduced over a dedicated 18- to 24-F DrySeal Flex sheath (Gore Medical). Stent-graft sizes range in diameter from 21 to 45 mm and in length from 10 to 20 cm. The ACS includes a nested handle delivery system that integrates 2 new features: 2-stage deployment of the stent-graft and an optional angulation mechanism that modifies only the proximal end of the device.
The 2-stage deployment option allows the stent-graft to be opened to its intermediate diameter from leading to trailing end to facilitate better positioning and correction. Of note, this feature permits continuous blood flow through the stent-graft lumen and around the device without any need for aggressive blood pressure reduction. During the second step of deployment, the stent-graft opens to its full diameter from the trailing to the leading end (Figure 1).
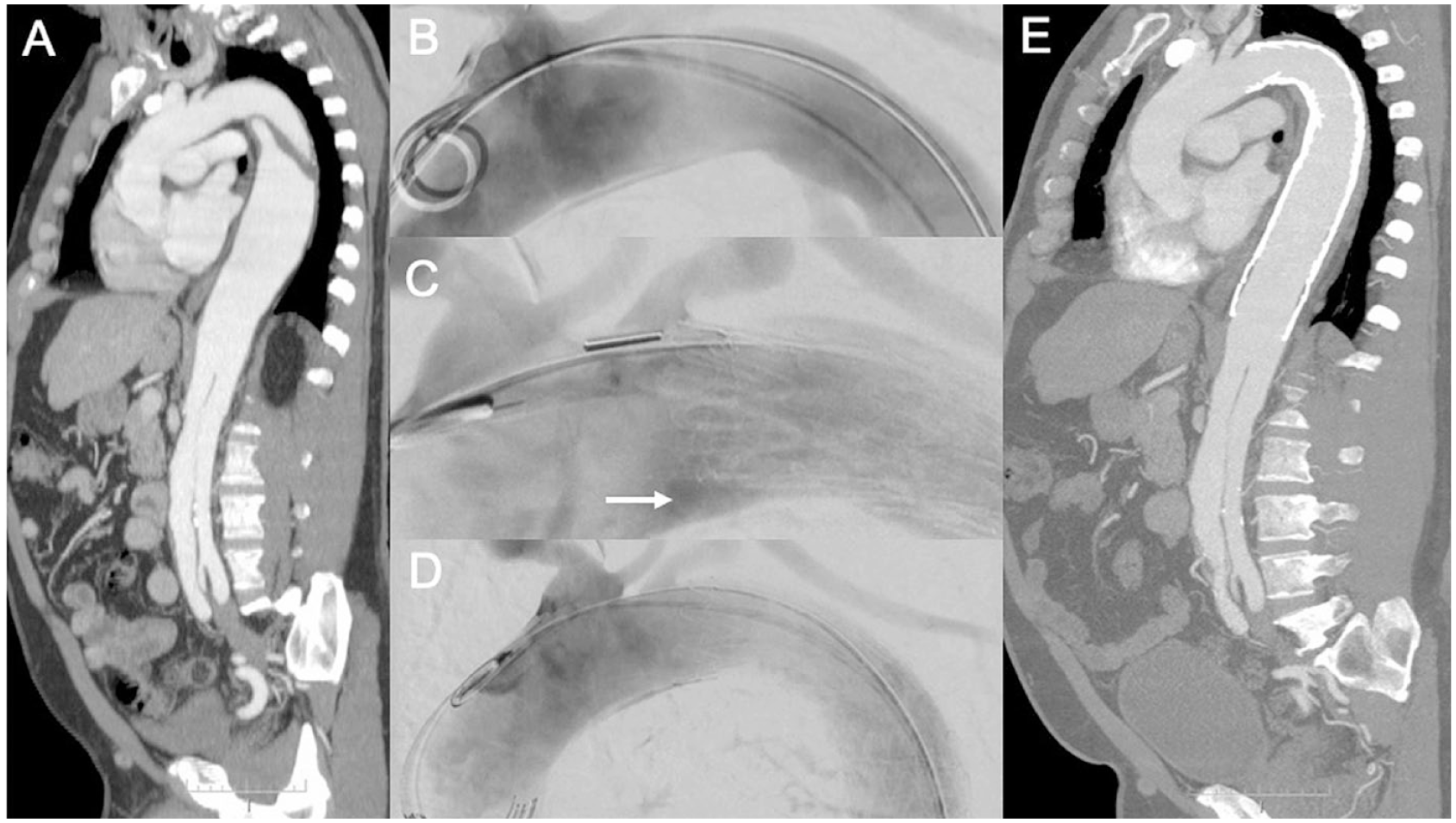
A 54-year-old man with multiple comorbidities had a type B aortic dissection diagnosed in 2013 (4 years prior). (A) The last computed tomography angiogram (CTA) showed aneurysm growth to 6 cm maximum diameter in the descending thoracic aorta. (B) After angiography, a TGM 45-45-20E CTAG device was placed just distal to the left subclavian artery. (C) After deployment, a bird-beak configuration (white arrow) was detected. (D) The angulation control mechanism was used for a more complete apposition to the inner aortic curvature. In total, 3 devices were implanted, leaving the celiac trunk patent. (E) In the control CTA scan 12 months later, the stent-graft was aligned with the aorta and the aneurysm sac had shrunk.
The angulation control dial is accessible after expanding the stent-graft to its intermediate diameter. Modifying the angulation is optional and executed by rotating the dial to adjust the leading end for a more harmonious alignment to the inner aortic curve. After full deployment of the stent-graft, the angulation mechanism remains accessible. Adjusting the angulation can achieve better apposition of the device to the aortic wall; however, the angulation is non-reversible.
Patient Population
During the observation period, 127 patients (mean age 67.1±12.1 years, range 27–86; 92 men) were enrolled in the registry. Table 2 summarizes the baseline characteristics of this cohort. An array of aortic pathologies was treated, including chronic and acute lesions and 4 ruptured descending thoracic aneurysms. Aortic arch morphology was demanding: 25 patients (19.7%) had a type I arch, 62 (49.2%) a type II arch, and 39 (31.0%) a type III arch; in 1 case, the arch type was not recorded.
Characteristics of the 127 Patients in the Study. a
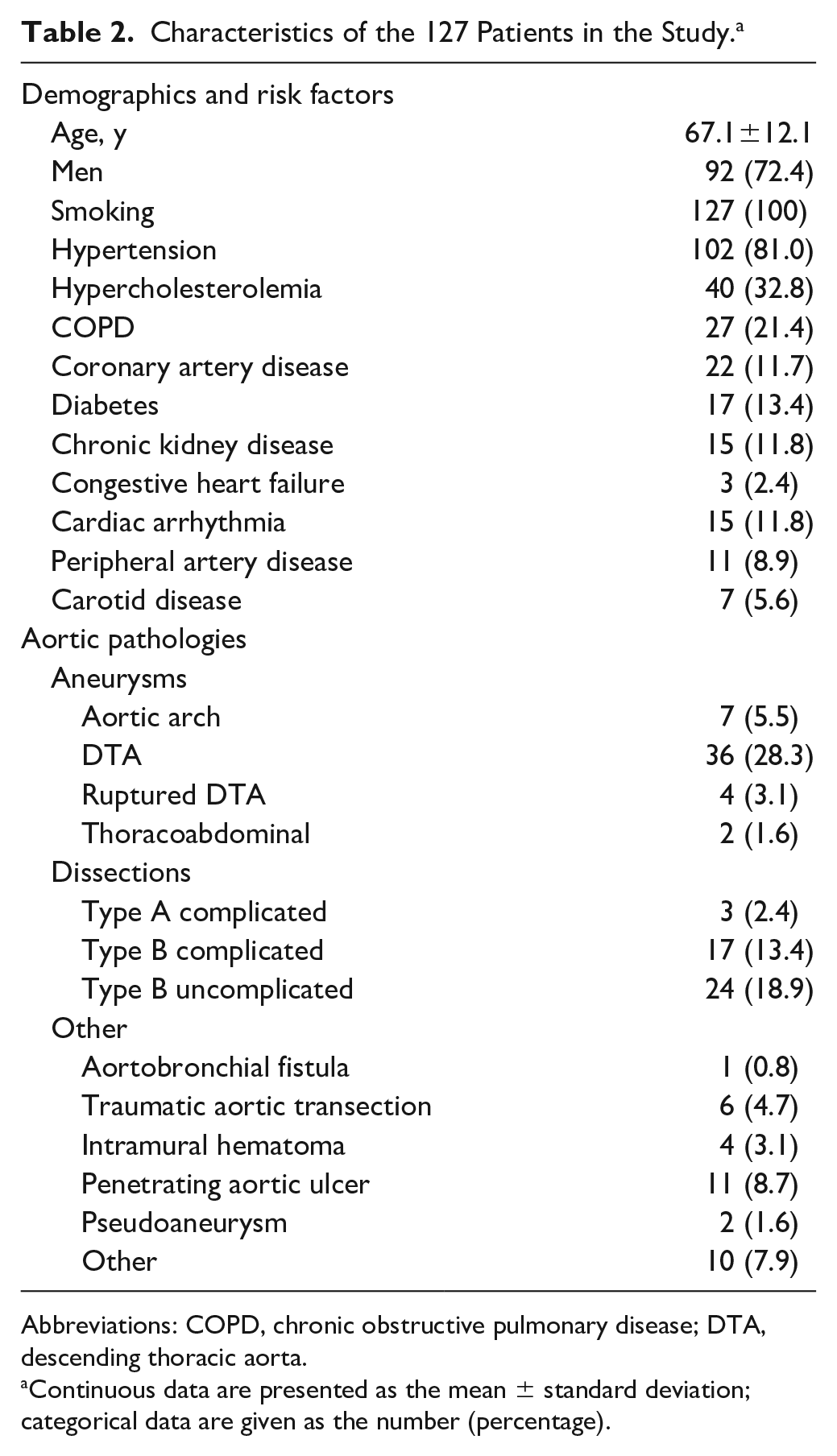
Abbreviations: COPD, chronic obstructive pulmonary disease; DTA, descending thoracic aorta.
Continuous data are presented as the mean ± standard deviation; categorical data are given as the number (percentage).
Outcomes and Definitions
Reporting of all outcome variables followed the definitions of the latest Society for Vascular Surgery reporting standards on TEVAR. 3 The primary outcome of this study was technical success, which was defined as successful access and deployment of the stent-graft at the intended location resulting in a patent device in the absence of death within 24 hours, surgical conversion, or type I or III endoleak detected on angiography.
The main secondary outcome was clinical success, which encompassed freedom from procedure- or aneurysm-related death, type I or III endoleak, aneurysm-related expansion or rupture, stent-graft occlusion, infection or migration, conversion to open repair, failure to arrest the original pathologic process, or a new thoracic aortic pathology. A further secondary endpoint was major adverse events, including paraplegia or paraparesis, stroke, myocardial infarction, acute kidney injury, bowel ischemia, or major blood loss.
Access and procedure details were recorded. The numbers of 2-stage device deployments and applications of the angulation mechanism were noted, along with the reasons for use of this option.
Statistical Analysis
Continuous variables are expressed as the mean ± standard deviation or median [interquartile range (IQR) Q1, Q3]. Categorical variables are given as the number (percentage). The analysis was performed using the statistical SAS software (version 9.4; SAS Institute, Cary, NC, USA).
Results
Outcomes at 30 Days
Technical success was 97.6%. Three left common carotid arteries (2.4%) were partially covered unintentionally. The length of the proximal landing zone in these cases was <20 mm each. All 3 vessels were patent on post-implantation angiograms and CTA scans. In 2 cases of these cases, angulations were optimized during deployment.
A femoral approach was the access of choice in 122 patients (96.1%), while an iliac conduit was needed in 5 cases (4%) because of excessive tortuosity or severe calcification of the access vessels (Table 3). A total percutaneous approach was utilized in 58 cases (45.7%).
Procedure and Deployment Data. a
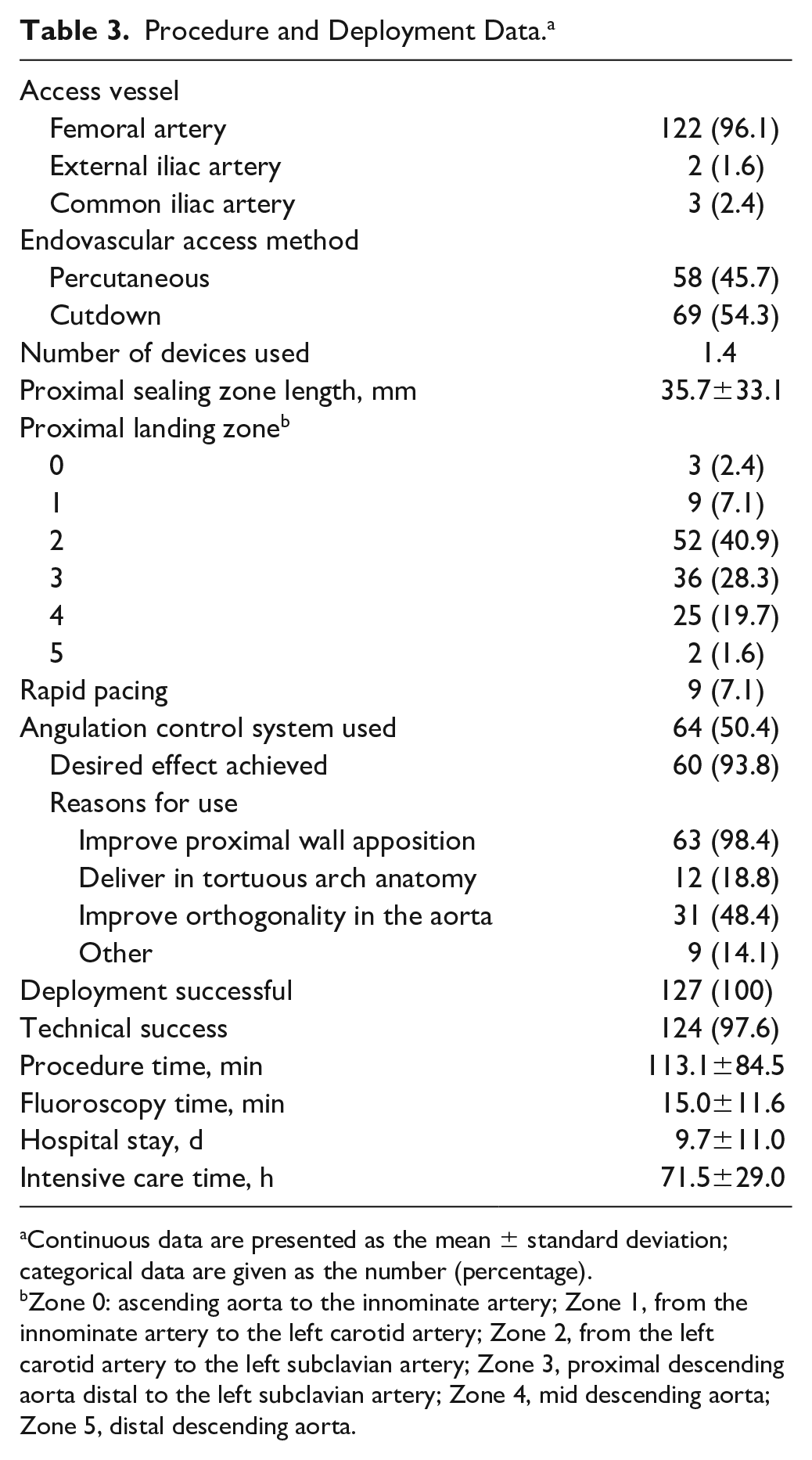
Continuous data are presented as the mean ± standard deviation; categorical data are given as the number (percentage).
Zone 0: ascending aorta to the innominate artery; Zone 1, from the innominate artery to the left carotid artery; Zone 2, from the left carotid artery to the left subclavian artery; Zone 3, proximal descending aorta distal to the left subclavian artery; Zone 4, mid descending aorta; Zone 5, distal descending aorta.
The mean number of CTAGs deployed was 1.38 (range 1–3). Of note, 64 patients (50.4%) required device implantation in the proximal landing zones. In 79 patients (62.2%), the stent-graft was repositioned at its intermediate diameter. The angulation feature was applied in 64 cases (50.4%): 23 (35.9%) at the device’s intermediate diameter, 17 (26.6%) at its full diameter, and 24 (37.5%) at both diameters. The main reasons for using the ACS were to improve proximal wall apposition (63, 98.4%) and orthogonality in the aorta (31, 48.4%). The desired effect was achieved in 60 cases (93.8%). There were no reports of device compression, bird-beak configuration, fracture, or graft occlusion.
Rapid ventricular pacing during deployment was used in 9 procedures (7.1%). A blood transfusion was required intraoperatively in 6 cases (4.7%), with a mean blood loss of 140.1±162.2 mL. The mean procedure time was 113.1±84.5 minutes, while the mean fluoroscopy time amounted to 15.0±11.6 minutes, during which a mean 115.5±73.4 mL of contrast agent were used. The mean hospital stay was 9.7±11.0 days.
One or multiple complications were observed in 28 patients (22%; Table 4). The access-related complication rate was 2.4% (n=3), including 2 cases (1.6%) of bleeding (1 did not require any treatment) and 1 pseudoaneurysm (0.8%). Other systemic complications were categorized as cardiac (0.8%), renal (3.2%), pulmonary (3.2%), and miscellaneous (7.9%). Neurological complications were observed in 5 patients (3.9%), including paraplegia or paraparesis in 4 patients (3.2%) and an ischemic stroke in an additional subject (0.8%) treated for a ruptured descending thoracic aneurysm.
Procedure-Related Adverse Events. a
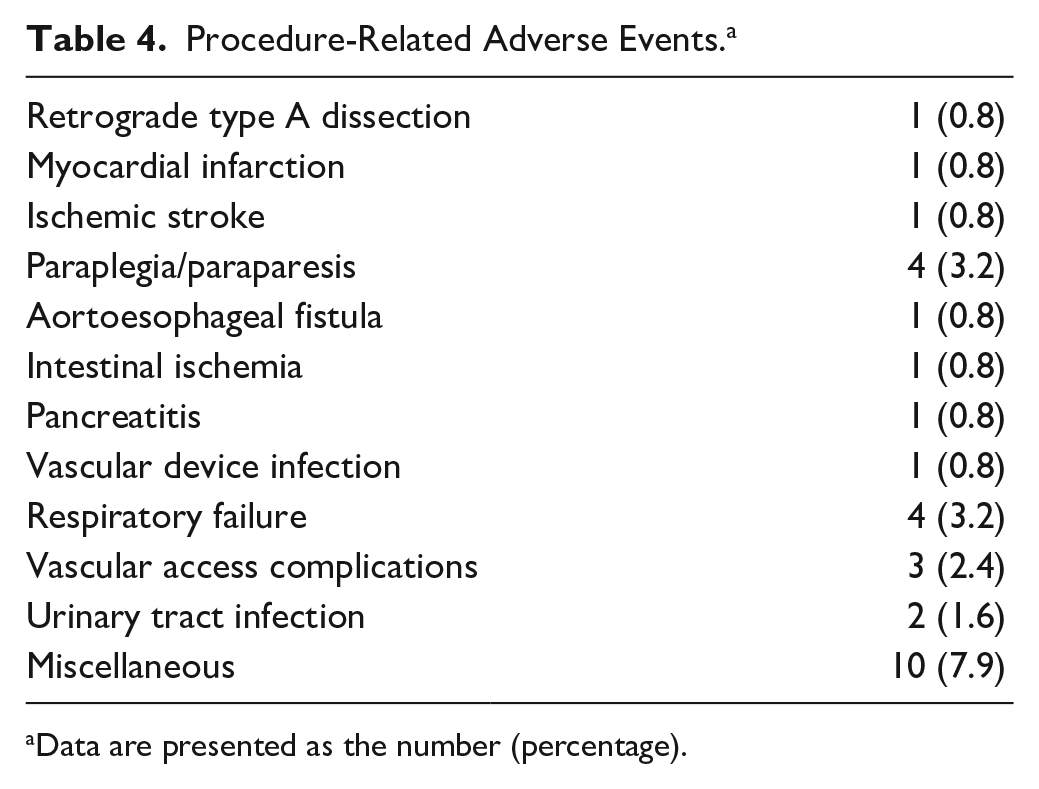
Data are presented as the number (percentage).
Primary clinical success was 97.6% owing to 3 deaths during the first 30 days after the initial procedure. One retrograde type A aortic dissection (0.8%) was observed in a patient treated for a complicated type B dissection 8 days after implantation of a 40-mm-diameter stent-graft into a 36.5-mm-diameter zone 1 landing site (9.6% oversizing). In another patient, paraplegia occurred 24 hours after emergent implantation of a single stent-graft covering 20 mm of thoracic aorta; no cerebrospinal fluid drainage was performed prior to the procedure. The patient expired on the seventh postoperative day (POD). The third patient died after laparotomy for bowel ischemia 4 days after treatment of an uncomplicated type B dissection.
One-Year Outcomes
The clinical success rate was 92.9% at 12 months; 1 patient was lost to follow-up. Among the 3 late deaths, one patient suffered aneurysm rupture at POD 40; no endoleaks had been reported, but the patient had initially been treated for an infected aneurysm and developed an aortoesophageal fistula on POD 7. One patient expired on POD 78 due to Salmonella sepsis after treatment of mycotic aortic aneurysm. The third patient died of multiple organ dysfunction syndrome on POD 48.
During follow-up, there were 2 type Ia endoleaks (1.6%); 1 was due to stent-graft migration (0.8%). Both required implantation of an additional stent-graft. There also was 1 type III endoleak (0.8%) that required placement of another stent-graft. Two patients (1.6%) had a type II endoleak, but no reintervention was performed. No additional retrograde dissections or neurological complications were reported during the 12-month follow-up period.
Discussion
The SURPASS registry assessed the safety and efficacy of the CTAG with ACS for endovascular repair of a variety of thoracic aortic pathologies from dissections to aneurysms. Stent-graft conformability to the inner aortic arch curve and long-term stability of the stent-graft are important contributors to the durability of endovascular repair.5,6 The use of the CTAG’s intuitive staged delivery system facilitates accurate deployment and conformability of the stent-graft in the aortic arch, minimizing the risk of inadvertent coverage of supra-aortic branches and the windsock effect. Opening the stent-graft to its intermediate diameter during positioning directs blood flow through the device, which is especially important in more proximal landing zones where blood pressure dynamics are more demanding. In this registry, rapid pacing was used in only 7% of the cases, reducing the complexity of the procedure. This benefit also avoids aggressive pharmacological reduction of blood pressure, increasing hemodynamic stability.
Another significant advantage of the staged delivery is the absence of complete contact between the device and the aortic wall during intermediate deployment and refinements of the graft position. In nearly two-thirds of the cases, the graft position was adjusted, optimizing deployment accuracy and reducing the risk of the windsock effect. It might also be hypothesized that the low stroke rate in this study population was also due to a reduced risk of cerebral embolization because of less mechanical trauma to the aortic wall during staged deployment.
The second unique feature of the CTAG with ACS is the optional in situ angulation control of the leading stent for more accurate apposition to the aortic wall. This is especially important at the inner curve where incomplete conformance can create a bird-beak configuration and ultimately result in type Ia endoleak. In this study, the angulation was corrected in more than half of the patients and was particularly helpful in the treatment of aortic pathologies in type II or III aortic arches.
Previous studies reported high rates of bird-beak configurations after TEVAR.7,8 Although the literature is not conclusive, a bird-beak phenomenon and a poor apposition of the stent-graft in a highly angulated aortic arch can increase the risk for periprocedural complications.9–11 A recently published meta-analysis demonstrated an association of bird-beak configuration and an increased risk of type Ia endoleak and stent-graft migration. 4 Risk factors include the treatment of traumatic aortic rupture and the deployment of the graft in landing zone 2. Both features of the new deployment mechanism can significantly improve the apposition of the graft, especially in case of a hostile proximal landing zone. Accordingly, no bird-beak configuration was observed in the current cohort, and the rate of type Ia endoleak was low even though half of the endografts were implanted in zones 0 to 2.
Mariani et al 12 reported the early results of the CTAG in 30 patients; they achieved a 100% technical success rate without type Ia endoleak, bird-beak configuration, or retrograde aortic arch dissection. The Conformable GORE TAG Endoprosthesis European Registry for thoracic arch pathologies found a comparable technical success of 92% and a low (4%) endoleak rate. 13 In that registry, the stroke rate was 11%, in contrast to the 0.8% stroke rate in the SURPASS registry with the same stent-graft design. Again, it might be hypothesized that to some extent the new delivery system allows less traumatic positioning due to the intermediate deployment option.
Regarding the outcomes of other available devices, the 92-patient TRAVIATA registry assessed the performance of Medtronic’s Valiant Stent Graft in the treatment of thoracic aortic disease.14,15 The technical success was 86.9% with 1-year survival of 95.5%. Type I endoleaks were detected in association with 4 aneurysms and 2 dissections, and graft migration occurred in 1 patient from each of these groups. Of note, the TRAVIATA registry did not focus on pathologies involving the aortic arch, with less than a third of the grafts deployed in this region. On the other hand, three-quarters of the devices in the present study were deployed in landing zones 0 to 3.
Moreover, the pivotal results of the Valiant EVO global clinical trial evaluating Medtronic’s novel Valiant Navion stent-graft in 81 patients reported an overall endoleak rate of 2.5% (1 type Ia and 1 type II). However, the anatomy of the included patients was not particularly demanding in terms of landing zone, tortuosity, or calcification. 16
In a midterm evaluation of a single-center experience of 70 patients treated with Cook’s Zenith Alpha thoracic stent-graft, ongoing clinical success was 87.1% at a mean follow-up of 22.3 months. 17 However, there is no information on the exact localization of the aortic pathologies. Endoleak rates were comparable to the results of the present study, as was the rate of access complications (4.3% vs 2.4% in the present study), but the low-profile device allowed a less invasive access strategy (87.1% totally percutaneous access vs 45.7% in the present study).
The RESTORE registries evaluated the use of Bolton Medical’s RELAY thoracic stent-graft.18,19 The rate of aortic arch involvement (44% landing zone 0 to 2), technical success (97.7%), and type I endoleak (3.9%) in RESTORE I was comparable to our cohort. 18 Similar results were reported in RESTORE II. 19 Migration rates were low at 2.6% and 2.3% for RESTORE I and II, respectively.
Retrograde aortic dissection can be a devastating complication of TEVAR. A meta-analysis of 50 studies with a total of 8969 patients reported an overall 2.5% incidence of retrograde type A aortic dissection after TEVAR. 20 Factors such as the underlying pathology, the proximal landing zone, and the proximal stent-graft configuration can play a role in the development of this uncommon but challenging complication. In the present cohort, only a single retrograde aortic dissection occurred.
Limitations
This study carries the well-known limitations of nonrandomized studies. All adverse events were assessed by the investigator, and no provision was made for a clinical event committee. The results are preliminary; long-term results are required.
Conclusion
Endovascular treatment of aortic pathologies involving the arch can be very challenging. In the SURPASS registry, the CTAG with ACS was associated with promising outcomes. The new delivery system offers a more controlled deployment of the stent-graft, allowing the treatment of aortic disease with challenging anatomy.
Footnotes
Appendix
Collaborators in the SURPASS Registry are Manuel Alonso Pérez, Hospital Universitario Central de Asturias, Oviedo, Spain; Dittmar Böckler, University of Heidelberg, Germany; Stefano Bonardelli, ASST Spedali Civili di Brescia, Italy; Jan Brunkwall, University of Cologne, Germany; Nabil Chakfé, CHU Strasbourg, France; Giovanni Dialetto, Vincenzo Monaldi Hospital Napoli, Italy; Jorge Fernandez Noya, University Hospital of Santiago de Compostela, Spain; Robin Heijmen, St Antonius Hospital, Nieuwegein, the Netherlands; José Antonio Lechón Saz, Hospital Universitario Miguel Servet, Zaragoza, Spain; Ian Loftus, St George’s University Hospital, NHS Trust, London, UK; Nicola Mangialardi, Azienda Ospedaliera San Camillo Forlanini, Rome, Italy; Simon McPherson, Leeds Teaching Hospital, NHS Trust, Leeds, UK; Bijan Modarai, Guy’s and St Thomas, NHS Foundation Trust, London, UK; Karin Pfister, University of Regensburg, Germany; Jean Picquet, Centre Hospitalier Universitaire d’Angers, France; Artai Pirouzram, Örebro University Hospital, Örebro, Sweden; Giovanni Torsello, St Franziskus Hospital Münster, Germany; Hence Verhagen, Erasmus Medical Center, Rotterdam, the Netherlands; Anders Wanhainen, Uppsala University, Uppsala, Sweden; and Kak Khee Yeung, VU Medical Center, Amsterdam, the Netherlands.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The SURPASS Registry was supported by a grant from W.L. Gore & Associates.
