Abstract
Keywords
Introduction
Kissing stent (KS) treatment represents a well-accepted intervention for aortoiliac occlusive disease (AIOD). The technique has several established benefits over open surgery: shorter length of hospital stay, lower 30-day mortality and major morbidity, and comparable short-term outcome.1,2 The long-term outcome, however, has not matched that of open surgery due to a high rate of reinterventions to assure stent patency.3–6 Data published on the results of KS treatment usually include relatively low sample sizes (mean 63.2, range 18–215), 7 owing to the low prevalence of AIOD. Not only do these sample sizes hamper longer term analysis, but the heterogeneity of included patient groups reduces the comparability with respect to open surgery, though it does provide an opportunity for subgroup analysis. More recent results suggest that KS treatment can also be an appropriate choice in more severe lesions, classified as TransAtlantic Inter-Society Consensus (TASC) C and D.1,8–10 Also, while comparing the KS technique to other approaches, choices within the treatment protocol that could potentially influence stent patency are a much-debated topic (eg, balloon-expandable vs self-expanding stents, stent protrusion, and pre- and postdilation).
Our group recently published a systematic review 7 including 21 studies reporting results of KS treatment, but the lack of a proper meta-analysis based on individual participant data (IPD) prevented an in-depth examination of the aforementioned points. To this end, a consortium was established to collect the individual patient records on KS treatment and perform an IPD analysis. The goals were to evaluate short- and long-term technical and clinical outcomes after KS treatment in a large sample, identify possible predictors of failure/reintervention, and provide evidence-based protocol advice for KS treatment.
Materials and Methods
Data Sources
The search query built for an earlier systematic review 7 according to the guidelines from the Preferred Items for Systematic Reviews and Meta-Analysis (PRISMA) group 11 was re-executed on June 12, 2017, using the Scopus database to add the most recent publications. Execution of the IPD analyses followed the PRISMA-IPD statement. 12 The updated search query identified 22 articles.1–6,8–10,13–25 Authors of 19 articles with contact information were invited by electronic mail to join the consortium, and a communication log was kept to overview all responses. If the authors did not respond to the request, a second letter was sent; if this remained unanswered the study was excluded.
Eight authors responded, 2 could not locate the original data files (articles published before year 2000) and 1 did not have the resources to configure a database. Five authors3,10,16,21,25 agreed to join and signed clinical investigation and data transfer agreements to formalize the collaboration and expectations. The contributing authors supplied 6 anonymized databases (Sabri et al 21 included 2 separate groups) containing data on 605 (40.9%) patients out of the 1480 identified in the search (Figure 1).
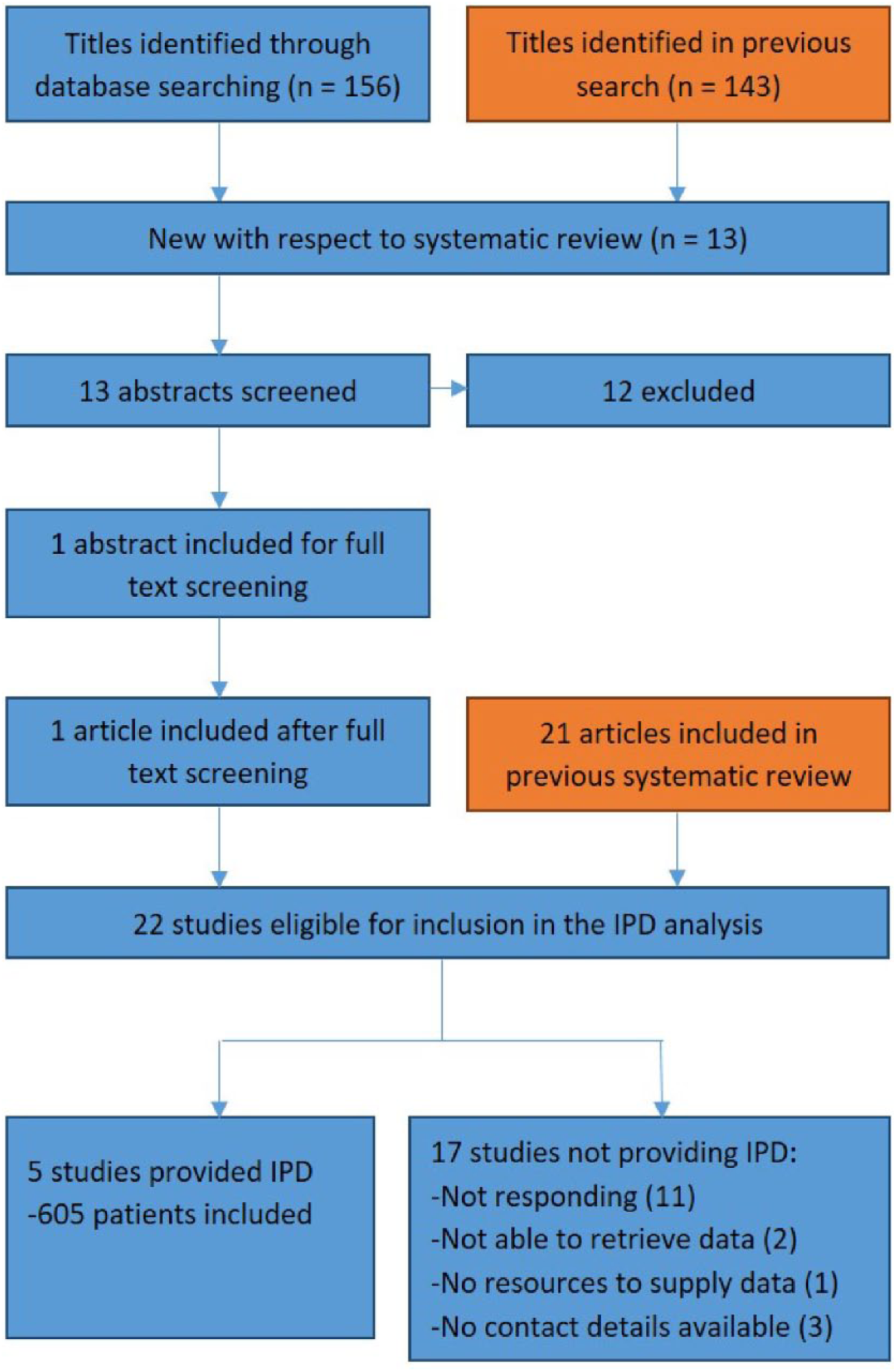
Individual participant data (IPD) inclusion flow diagram. The orange boxes indicate results used from a previous systematic review. 7
The original principal investigators had obtained ethics approval prior to inclusion in the IPD consortium; the analysis itself was approved by the Medical Ethics Committee Nijmegen (file number 2015-1752).
Data Merge
Based on our prior review 7 and according to current reporting standards, 26 an IPD database was created using SPSS software (version 24; IBM Corporation, Armonk, NY, USA) prior to merging the individual datasets. In the conversion process, fields of each original study database were mapped to the prebuilt database (fields are listed in Supplemental File 1, which is available in the online version of the article). A conversion log was maintained for each study. When applicable a conversion scheme was also saved in the log, based on the value label defined for the original field and the IPD field. After initial mapping and conversion, the conversion log and data copying per study cohort were checked. Any discrepancies in the meaning of column headings or definitions were resolved by discussion with the author. A copy of the IPD database was saved for each database conversion; all copies were thereafter merged into one final database. Empty fields in the prebuilt IPD database were culled.
Definitions
Definitions for cardiovascular risk factors were based on the latest reporting standards, 26 if applicable. Units for length, diameter, blood pressure, and so on were predefined, and conversion was applied if needed. If no standard definition was available, labels per database were added if not yet available in the IPD database. Definitions of patency were used as specified by the authors. Clinical improvement was defined as an increase of the ankle-brachial index (ABI) by at least 0.1 and a 1-level improvement in the Rutherford category between baseline and the first postoperative measurement and subsequent follow-up visits.
Outcomes
The primary study outcome was the cumulative patency at 24 months. Secondary outcomes were patency at 30 days, 6, 12, and 60 months; complication rate (major/minor); and improvements in Rutherford category and ABI. Additionally, the predictive value of protrusion length, pre- and postdilation, stent type, and patient demographics on primary patency was evaluated.
Statistical Analysis
Continuous variables are presented as median and interquartile range (IQR; Q1, Q3) since the data were not normally distributed (tested using the Kolmogorov-Smirnov test). Categorical variables are presented as numbers (percentage).
Pre- and postprocedure comparisons, as well as changes in continuous variables at follow-up intervals (30 days, 1 year, 2 years, and 5 years), were analyzed using analysis of variance (ANOVA) for repeated measures. Dichotomous variables were compared with a Fisher exact test, and the chi-squared test was used to compare nominal categorical variables. The Kruskal-Wallis test and ANOVA were employed to evaluate categorical and continues parameters, respectively, between the study groups. Differences were considered significant at the p<0.05 level.
Primary and secondary patency rates were estimated on an intention-to-treat basis with Kaplan-Meier analyses; estimates are reported with the 95% confidence intervals (CI). Patency curves in the subgroup analyses were compared using the log-rank test.
Variables were examined with univariate correlation analysis; those with a significant univariate effect (p<0.05) were combined in a Cox proportional hazards model, stratified by research group. Variables were subjected to multiple regression analysis based on current knowledge with respect to patency. The following parameters were included in the model for primary patency: age scaled to the IQR, hypertension, hyperlipidemia, diabetes, smoking, cardiovascular disease, preoperative ABI, stent crossing, and TASC II category (A+B and C+D). For secondary patency the variables were age scaled to the IQR, hypertension, diabetes, smoking, cardiovascular disease, previous (endo)vascular interventions, and stent type. Results of the Cox models are presented as the hazard ratio (HR) with 95% CI.
Results
Study Characteristics
Of the 5 articles3,10,16,21,25 selected, 4 publications reported single-center data; Dorigo et al 25 published data collected from 3 centers. All patients were treated between April 1995 and February 2014. Patient demographics in the IPD database are presented in Table 1. Table 2 gives an overview of demographic parameters and outcome variables per study.
Patient and Lesion Characteristics. a

Abbreviations: ABI, ankle-brachial index; BA, balloon angioplasty; TASC, TransAtlantic Inter-Society Consensus.
Continuous data are presented as the median (interquartile range Q1, Q3); categorical data are given as the number/sample (percentage).
Demographics and Outcomes per Study. a
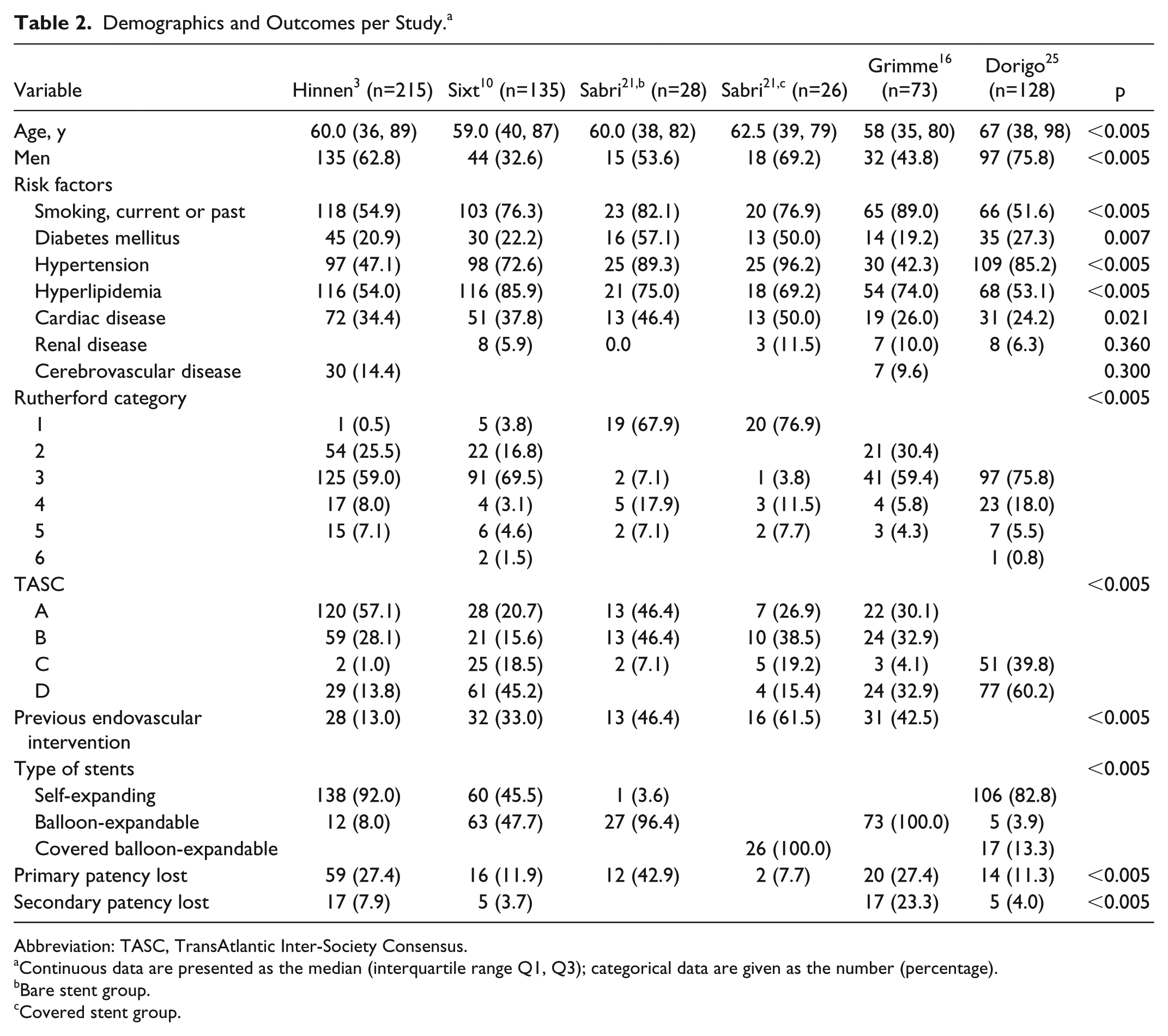
Abbreviation: TASC, TransAtlantic Inter-Society Consensus.
Continuous data are presented as the median (interquartile range Q1, Q3); categorical data are given as the number (percentage).
Bare stent group.
Covered stent group.
The indication for intervention in 594 of 605 patients for whom this variable was reported was intermittent claudication in 500 (84.2%) compared to 94 (15.8%) patients with critical limb ischemia. Lesions were classified as TASC A or B in 317 (52.8%) and TASC C or D in 283 (47.2%). Previous vascular interventions (either endovascular or surgical, both aortoiliac and more distal) were performed in 120 (27.3%) of 439 patients.
Procedure Results
The overall technical success rate was 99.1% (527/532). Procedure details are given in Table 3. The vast majority of cases (485, 91.9%) were treated with bare metal stents, of which 305 (57.8%) were self-expanding models. Covered balloon-expandable stents were used in 43 (8.1%) cases. The stents protruded into the distal aorta and crossed in 233 (67.3%) of 346 patients. In 43 (12.8%) of 269 patients, an additional treatment was performed along with the primary KS treatment.
Procedure Details. a
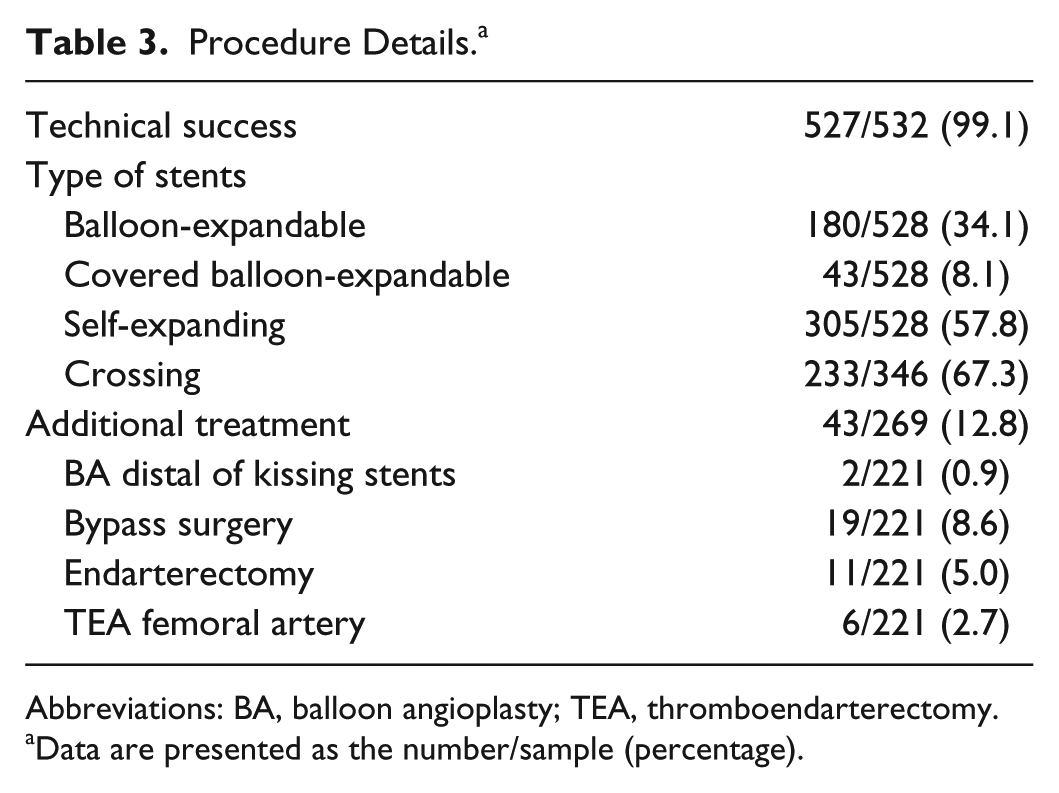
Abbreviations: BA, balloon angioplasty; TEA, thromboendarterectomy.
Data are presented as the number/sample (percentage).
Clinical Outcomes at 30 Days
At discharge the ABI [median 0.99 (IQR 0.23, 1.38)] had significantly improved with respect to the baseline measurements [median 0.73 (IQR 0.09, 1.24); p<0.001]. The shift in Rutherford class between baseline and discharge is presented in Figure 2. Clinical improvement (based on both the discharge Rutherford category and ABI) was established in 222 (66.5%) of 334 cases. In 112 (33.5%) cases of no clinical improvement, the ABI improved but the Rutherford classification did not in 73 (65.2%), the Rutherford classification improved but the ABI did not in 17 (15.2%), and both the ABI and the Rutherford did not improve in 22 (19.6%). The 30-day complication rate was 8.2% (46/558 patients); the most frequently observed complication was perioperative bleeding (2.3%). Details regarding the type of complication are depicted in Table 4.
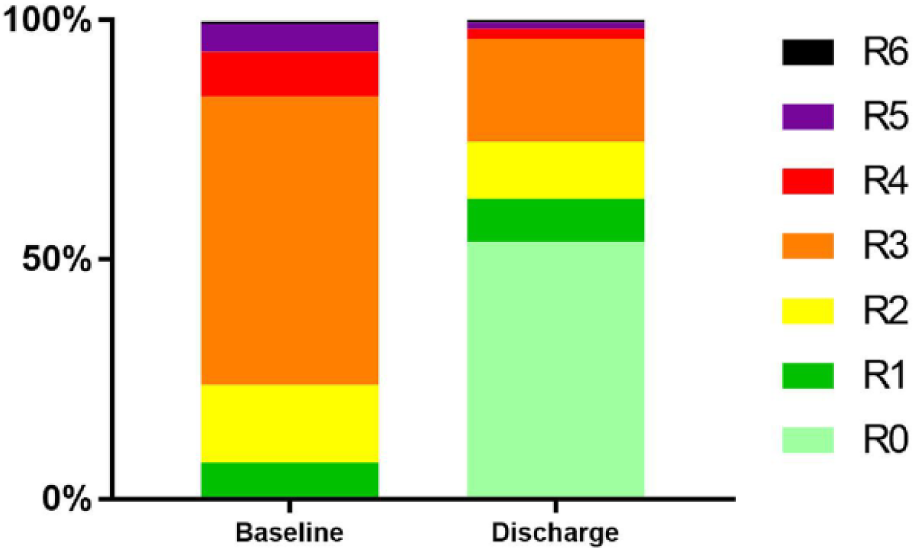
Overview of Rutherford distribution at baseline and discharge.
Clinical Outcome at 30 Days. a
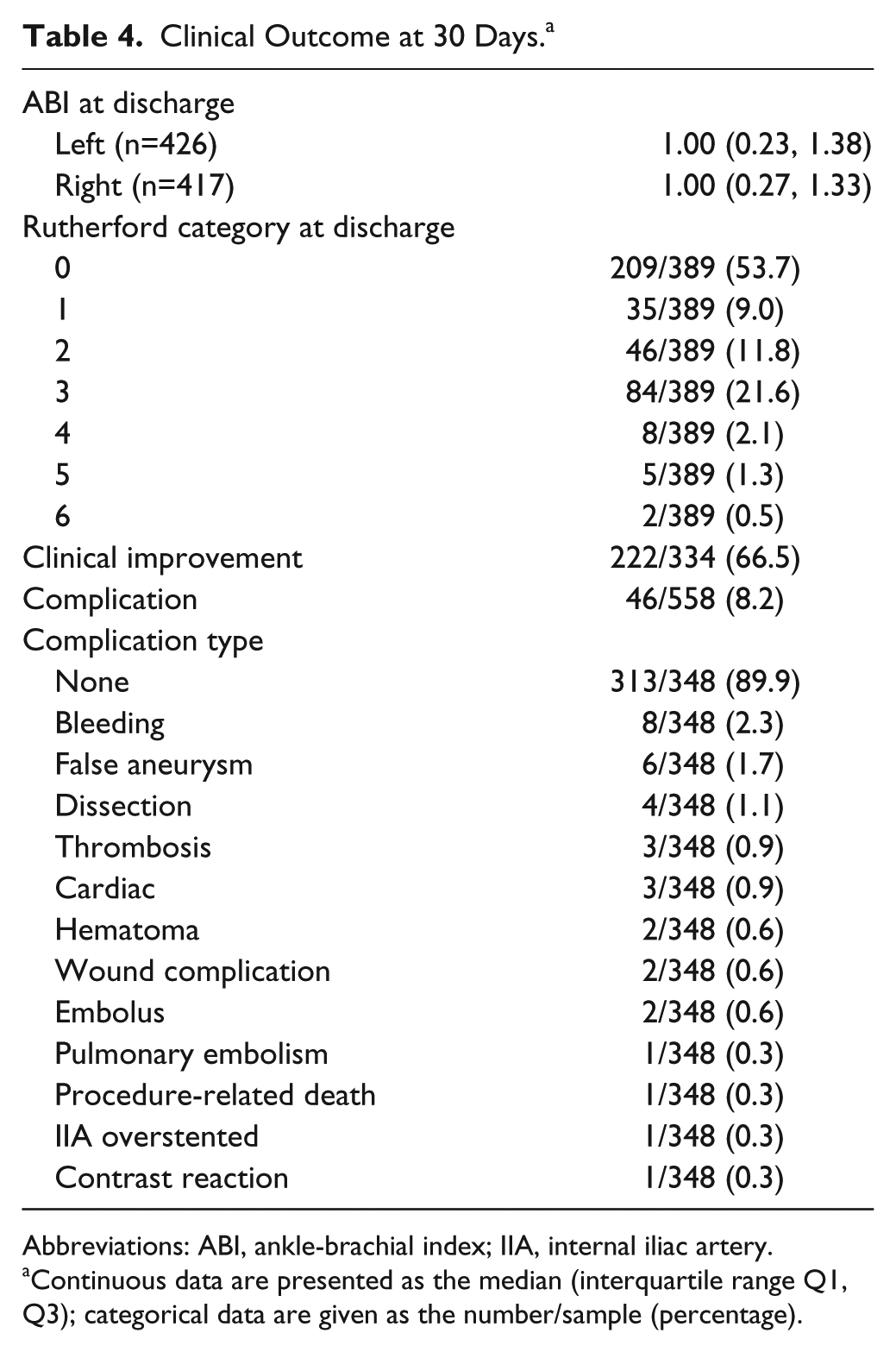
Abbreviations: ABI, ankle-brachial index; IIA, internal iliac artery.
Continuous data are presented as the median (interquartile range Q1, Q3); categorical data are given as the number/sample (percentage).
Follow-up
The median follow-up was 24 months (absolute range 0–120). Follow-up was available at 1, 12, 24, and 60 months in 561, 399, 330, and 113 cases. Overall survival estimates at these time intervals were 98.7% (95% CI 97.3% to 99.4%), 95.9% (95% CI 93.6% to 97.4%), 91.5% (95% CI 88.4% to 93.9%), and 76.9% (95% CI 71.2% to 81.6%). Table 5 describes the available data per period. An increase in stent failure (loss of primary patency), definitive stent failure (loss of secondary patency), and reinterventions were observed as the number of patients available for follow-up decreased.
Follow-up Outcomes per Interval. a
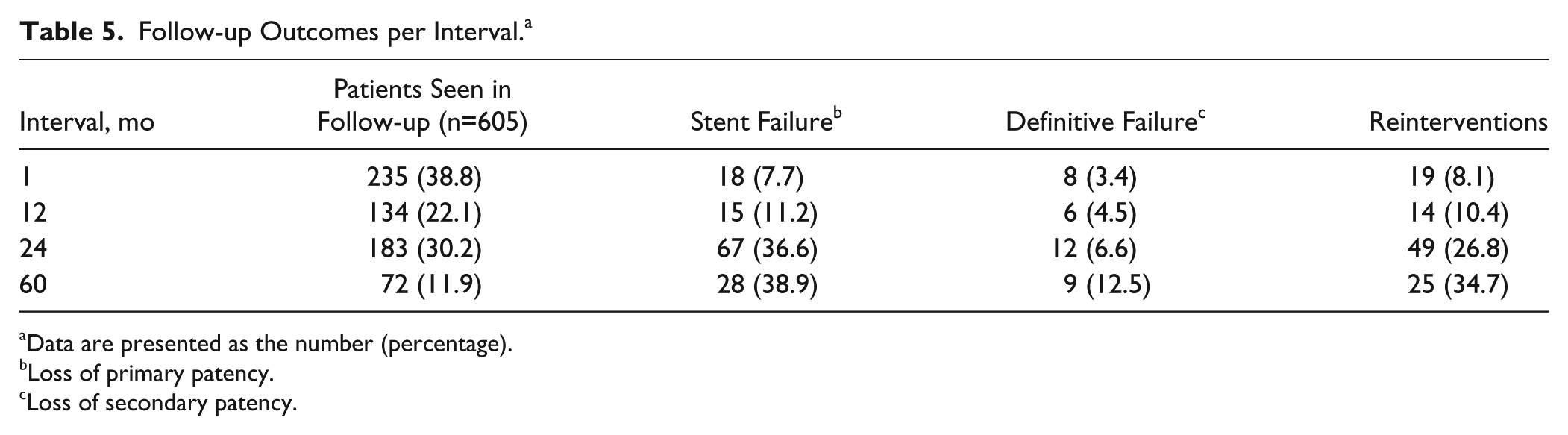
Data are presented as the number (percentage).
Loss of primary patency.
Loss of secondary patency.
Patency
The included studies used different definitions for primary patency; 3 studies10,17,21 defined primary patency as the absence of restenosis (<50% based on duplex or peak systolic velocity ratio <2.5), while 2 others3,25 defined primary patency as the absence of a reintervention on the target lesion. Kaplan-Meier estimates of primary and secondary patency (Figure 3A) are summarized in Table 6. The overall primary and secondary patency estimates were 81% (95% CI 77% to 84%) and 93% (95% CI 90% to 95%), respectively, at 24 months and 73% (95% CI 68% to 78%) and 89% (95% CI 85% to 93%) at 60 months. Figure 3B shows the patency estimates per site. Primary patency per TASC classification (A+B vs C+D) was better (p=0.047; Figure 3C) for C+D lesions at 48 months.
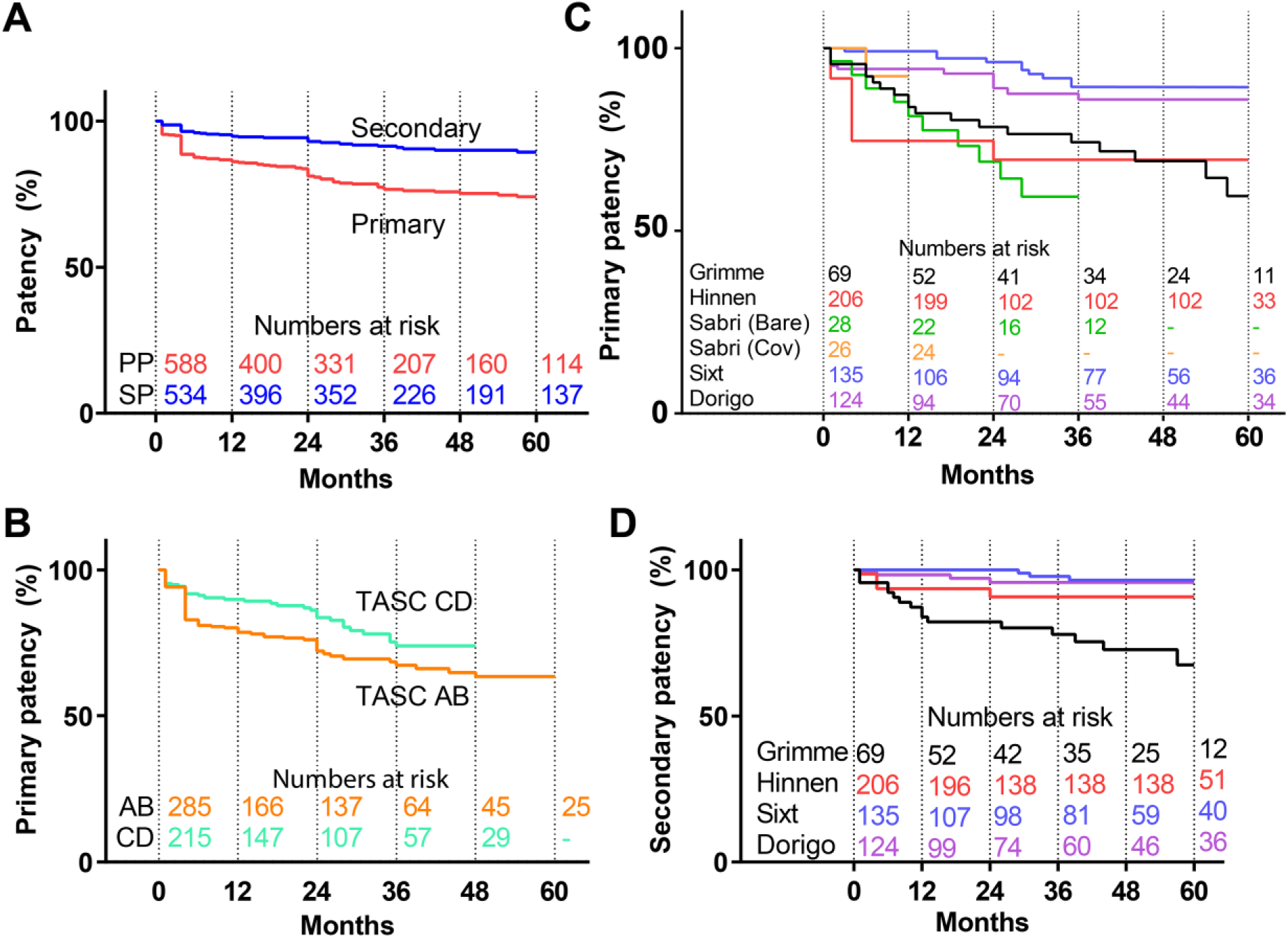
Kaplan-Meier curves for patency (A) overall, (B) by TransAtlantic Inter-Society Consensus (TASC) classification (p=0.047), and (C and D) by site (p<0.005). Standard error (SE) does not exceed 10% at 60 months in A. In B, the SE exceeds 10% for primary patency at 57 months for the TASC C/D group. In C and D, the SE exceeds 10% for primary patency at 78 months for Grimme et al 16 and 28 months for Sabri et al 21 (bare). Dashes indicate group size <10.
Kaplan-Meier Estimates of Primary and Secondary Patency.
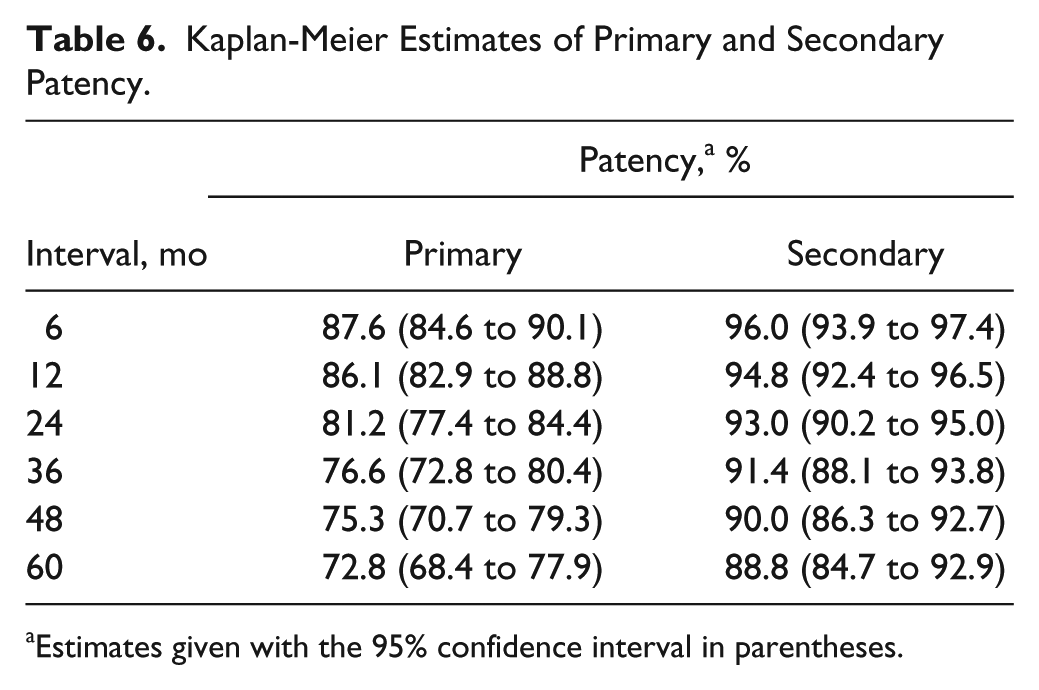
Estimates given with the 95% confidence interval in parentheses.
Half of the procedures were performed before 2005. Only the patients from Dorigo et al 25 were predominantly treated after 2005 (121/128, 94.5%); for the other groups the procedures were divided 50/50 over the period before and after 2005. This excluded the data from Sabri et al, 21 as their ethics board prohibited the exchange of dates. When comparing the patency rates, the primary patency before 2005 was significantly lower compared with the results obtained after 2005 (p=0.005; Figure 4A).
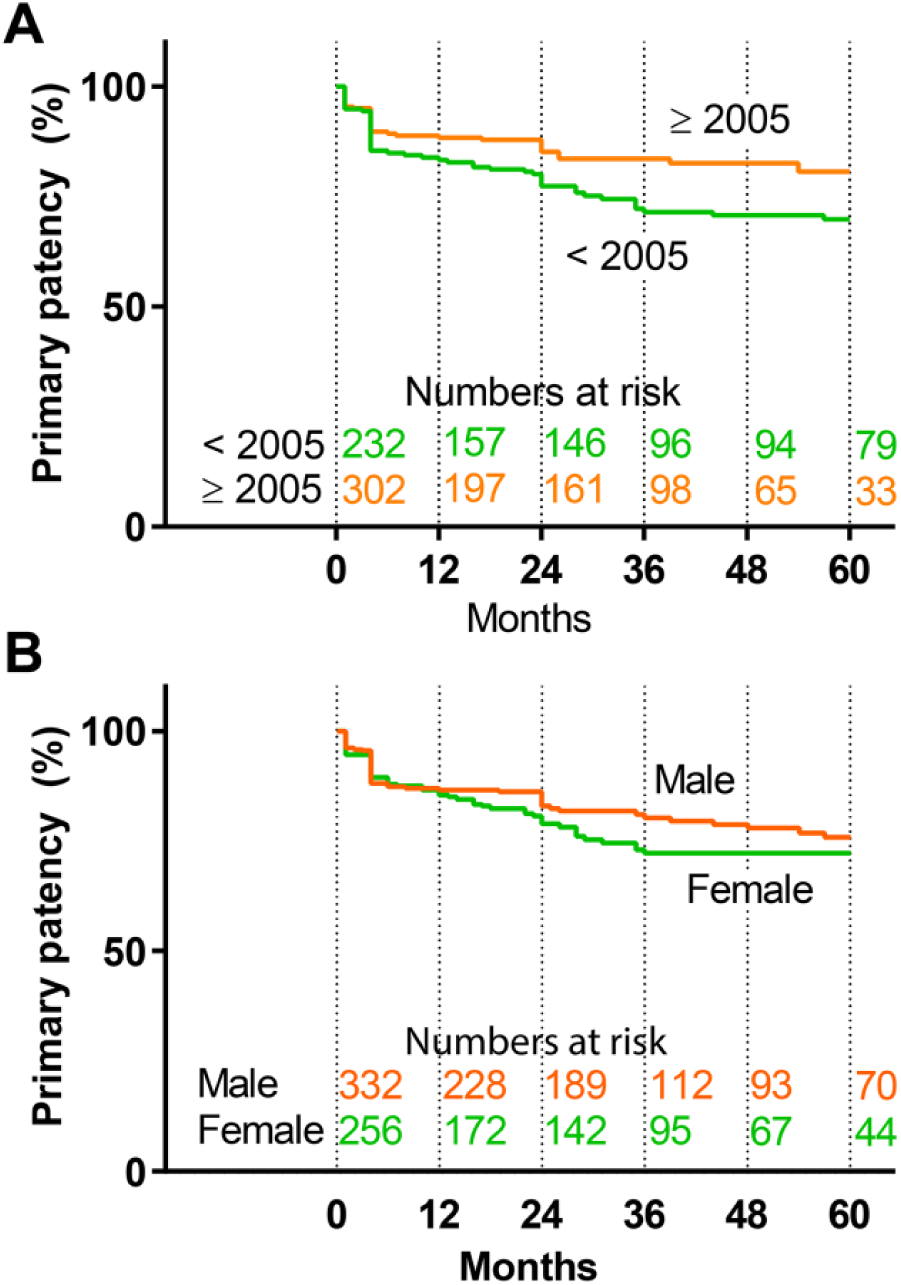
Patency values (A) before and after 2005 (p=0.005) and (B) by sex (p=0.08). Standard error does not exceed 10% up to 60 months.
Analysis of patency based on sex (Figure 4B) showed a difference in favor of men, mostly in late follow-up (over 60 months); however, this did not prove to be significant. This difference was not observed for secondary patency.
Predictors of Primary and Secondary Patency
Cox proportional hazards analysis was applied to identify factors influencing primary and secondary patency. Although significantly correlating with primary patency, preoperative ABI and crossing of stents were removed from the models due to high numbers of missing data (up to 70%). Pre-/postdilation did not have a significant univariate effect and was therefore not included in the multiple regression modeling. Age scaled to the IQR (15 years) was the only variable that significantly predicted sustained primary patency [hazard ratio (HR) 0.60, 95% CI 0.44 to 0.80, p<0.005). A previous (endo)vascular intervention (HR 2.52, 95% CI 1.20 to 5.30, p=0.02) was a significant predictor of lost secondary patency, while a history of cardiovascular disease (HR 0.27, 95% CI 0.08 to 0.82, p=0.04) predicted sustained secondary patency.
Discussion
The current study included the IPD of 605 patients with AIOD treated with the KS technique, which is the largest group presented in the literature to our knowledge. Despite improvements in endovascular techniques and equipment there still seems to be a gap between results from open and endovascular surgery, with an average 2-year primary patency of 93% (range 87%–98%)27,28 for open cases compared with 81% from this IPD analysis.
Nearly a third of the current KS cohort consisted of TASC D lesions, which had an 88% primary patency estimate at 2 years, similar to TASC C lesions (89%). In fact, the TASC C and D lesions performed significantly better than TASC A (primary patency 77%) and B lesions (primary patency 71%), which is surprising and contrary to previous studies that have shown no difference in outcome between TASC lesion category and primary patency.1,10,13,28 Furthermore, the TASC II consensus document 29 states that generally the outcome gradually worsens with increasing extent of the disease. This observation may be related to an increased learning curve or improvements in devices, as more complex lesions are likely to be treated by more experienced interventionists drawing on a wider stent selection. The observation that more complex lesions were treated in the latest time interval seems to support this assumption.
To investigate any change in outcome over time, the cohort was split into groups treated prior to 2005 or in or after 2005. This threshold was somewhat arbitrarily based on the fact that 2005 was the median year of procedures in the IPD cohort. This analysis showed that there is a significant difference in patency results obtained prior and after 2005, but stratifying based on the 2005 cutoff for treatment year within the Cox model did not identify interactions with primary or secondary patency. After 2005 more self-expanding stents were deployed (67% vs 56%), and stents were placed less often in a configuration that protruded into the aorta (74% vs 87%). The latter is, however, contradictory as TASC D lesions, which were more prevalent in the after-2005 subgroup (40% vs 28%), include lesions that cross the bifurcation; when treating these lesions from healthy-to-healthy tissue, stent protrusion is often necessary.
Lesions treated with self-expanding stents that do not protrude into the aorta may influence the improved patency results after 2005. 30 The recently published ICE trial 31 documented superior results of self-expanding stents for the treatment of lesions in the iliac vasculature with respect to restenosis rate. Our current dataset shows that primary patency is not significantly influenced by procedural choices, such as type of stent, protrusion, pre- and postdilation, and patient demography. In the BRAVISSIMO trial 32 the use of kissing stents that protruded into the distal aorta was an independent predictor of primary patency.
In the regression analysis, only aging was a protective factor for sustained primary patency, which was also reported by the BRAVISSIMO trial. 32 Younger (<50 years) patients might have a more “aggressive” type of atherosclerosis, becoming symptomatic at an earlier age and being more prone to disease progression after treatment and subsequent patency loss. 33 Therefore, the endovascular-first decision might be less obvious in young patients.
The multiple regression analysis also showed that any previous vascular intervention was a predictor of secondary patency loss, which could relate to the fact that patients have an advanced form of atherosclerosis. On the other hand, history of cardiovascular disease predicted sustained secondary patency, which was not expected. Possibly these patients benefitted from secondary preventive measures that were already taken.
Since a previous endovascular intervention predicts loss of secondary patency, close follow-up is needed after restoring patency in a patient with a history of endovascular treatment. However, based on these results no change in the recommendations for future treatment of patients with the KS technique can be established. Other (imaging) modalities might be needed to identify lesions at risk for restenosis and grafts for failure.
After analysis of the follow-up per interval, the results showed an increase in the number of primary failures and reinterventions at 2 years. Apart from primary failures there was also an increase of definitive failures at 2-year follow-up. This justifies an extra visit between 12 and 24 months if patients need open surgery to restore blood flow, bringing about extra patient burden and costs.
When reporting follow-up results, the studies that were included in this KS IPD analysis mainly used ABI and Rutherford category as a basis for clinical outcome. During follow-up, the ABI was seldom reported, and patency was mostly based on duplex ultrasound and observed symptoms. This choice of clinical outcome parameters may be justified by the fact that no clinical improvement was mainly due to unimproved Rutherford category, making it an important marker.
The KS technique is not the only endovascular option for AIOD. Covered endovascular reconstruction of the aortic bifurcation (CERAB) is another means of treating severe AIOD. Initial primary patency results at 2 years are around 87%, in primarily TASC D lesions, 34 which is in line with the results from this IPD analysis. The Taeymans study, 34 however, included all patients from the first in human, and a learning curve bias was likely. In the current study, 195 lesions were classified as TASC D, but the majority of these (138, 70.7%) were from the study groups of Sixt (n=61) 10 and Dorigo (n=77), 25 which may indicate a heterogeneity in treatment strategy between study groups. These results call for a one-to-one comparison of the CERAB and KS techniques for severe AIOD.
Limitations
This IPD has several limitations, mostly due to the fact that all groups focused on a different set of parameters in their databases. They coded variables using different definitions, which limited the number of parameters available for merging and introduced missing data. Two different definitions were used for primary patency, and this could not be recoded due to lack of data during the follow-up intervals. Not all data could be retrieved, highlighting the importance of proper storage of research data. Furthermore, symptoms were not regularly reported during the follow-up period, therefore clinically-driven reinterventions could not be analyzed. No information about serious adverse events was available, and detailed data regarding complications were reported in only about half of the cases. The aforementioned shortcomings hampered the construction of models (due to exclusion in case of missing data) that can correctly predict failure of primary patency.
The technical success rates have to be regarded with care as definitions were not always reported and it was not always clear how a technical failure was handled. The number of studies included was less than half of the data that were available in the literature (5 of 19 studies); more than half of the corresponding authors did not reply to our request. In a field where patient cohorts are usually small, this may hamper robust conclusions and may limit the advancement of the KS treatment and knowledge regarding the KS technique and outcome. Unfortunately, the present IPD analysis did not allow us to draw robust conclusions on choice of patient categories that can be best treated with the KS technique.
Conclusion
The current KS IPD analysis has shown that acceptable patency rates can be obtained at 2-year follow-up, even in TASC D lesions, with high levels of technical success and acceptable complication rates. Guidelines with respect to treatment choices could not be developed based on the IPD dataset. However, outcome is not sex-specific and seems to improve over time, indicating that an endovascular-first approach in case of all AIOD lesions is supported by data from the current IPD analysis for KS treatment.
Supplemental Material
18-0175_Supplemental_File_1 – Supplemental material for Meta-analysis of Individual Patient Data After Kissing Stent Treatment for Aortoiliac Occlusive Disease
Supplemental material, 18-0175_Supplemental_File_1 for Meta-analysis of Individual Patient Data After Kissing Stent Treatment for Aortoiliac Occlusive Disease by Erik Groot Jebbink, Suzanne Holewijn, Michel Versluis, Frederike Grimme, Jan Willem Hinnen, Sebastian Sixt, John F. Angle, Walter Dorigo and Michel M. P. J. Reijnen in Journal of Endovascular Therapy
Footnotes
Authors’ Note
This study was presented at the Veith Symposium, (November 2017; New York, NY, USA) and published in a doctoral dissertation titled “Aortoiliac Stenting, How Blood Flow and Stents Interact.”
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
