Abstract
Context:
The growing worldwide interest in early-stage type 1 diabetes (T1D) screening highlights the need for efficient and affordable approaches. Advanced analytical technologies such as Antibody Detection by Agglutination-PCR (ADAP) have improved large-scale sample handling, but optimal sample selection for simplified collection and processing remains a major challenge in screening designs.
Objective:
To assess ADAP for T1D-related autoantibody detection in different blood-derived samples to select the optimal sample type for large-scale screening.
Methods:
Autoantibodies against insulin (IAA), glutamate decarboxylase (GADA), protein tyrosine phosphatase IA-2 (IA2A), and zinc transporter 8 (ZnT8A) were analyzed by ADAP in 171 patients with stage 3 T1D (13.7 years; interquartile range [IQR]: 6.5–15.0) and 95 healthy controls (41.9 years; IQR: 29.5–56.7) previously studied by radiobinding assay (RBA). Comparison of sensitivity/specificity of ADAP and RBA in serum was assessed, as well as ADAP concordance across serum, plasma, EDTA-anticoagulated whole blood, and dried blood spot (DBS). Whole-blood stability was also investigated.
Results:
ADAP-based detection of two or more autoantibodies in serum demonstrated high sensitivity (87.1%) and specificity (100%), with strong concordance with RBA (Cohen’s kappa 0.90). Autoantibody analysis in whole blood by ADAP showed high agreement with serum, making it a consistent alternative. In contrast, DBS sampling had reduced concordance, which led to fewer positive detections and suggests a potential bias for T1D screening. A remarkable advantage was that autoantibodies in unrefrigerated whole blood remained stable for up to 7 days (IAA) and 20 days (GADA, IA2A, and ZnT8A), and also after four freeze–thaw cycles, considerably simplifying logistics.
Conclusion:
Pancreatic autoantibody detection in whole blood with ADAP provides a stable and more efficient diagnostic solution for T1D population screening, with simplified preparation, low-volume requirements, and high diagnostic consistency comparable with serum-based assays.
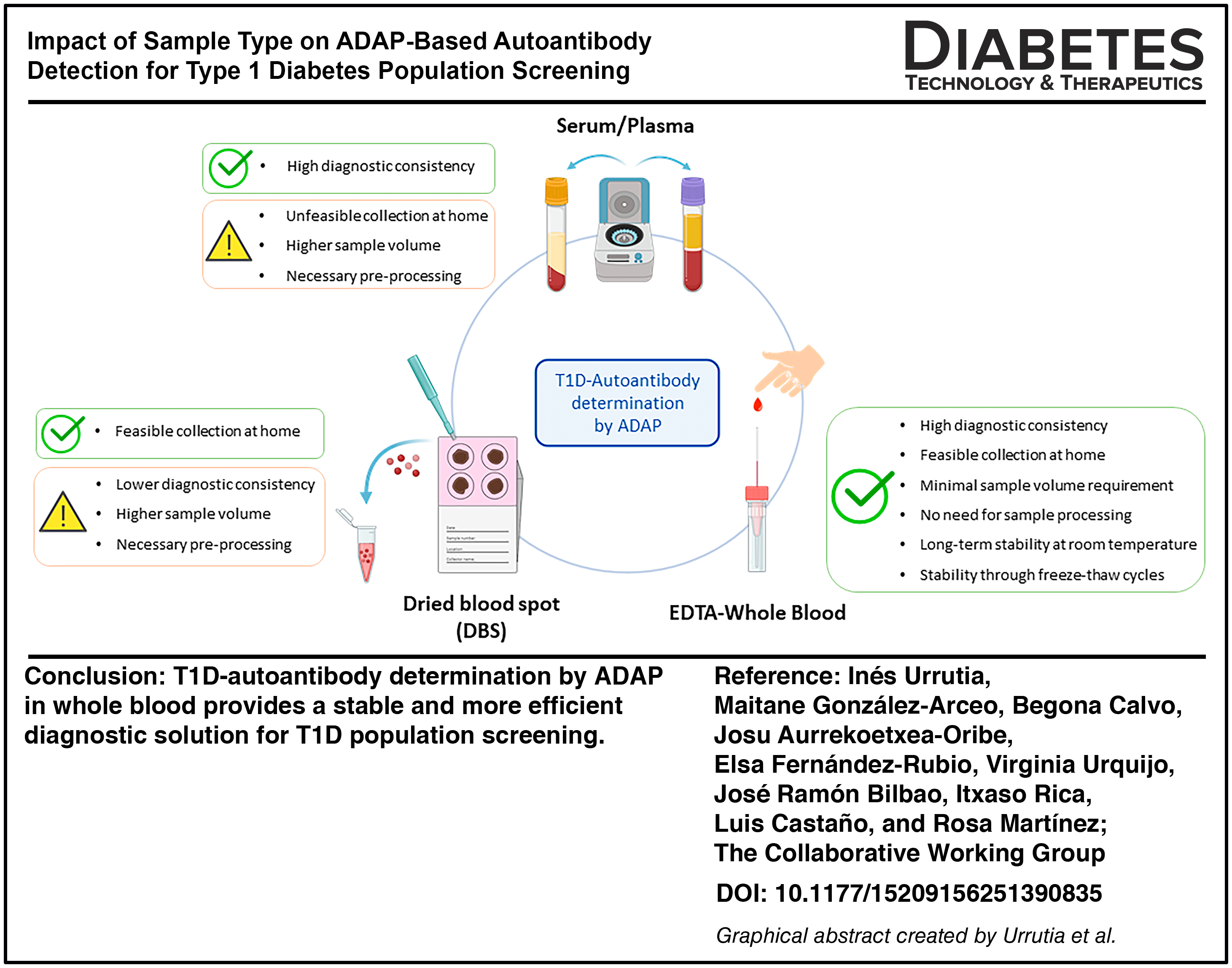
Introduction
Type 1 diabetes (T1D) is a chronic autoimmune disease characterized by progressive destruction of pancreatic beta cells, resulting in lifelong exogenous insulin dependence and risk of both acute and chronic complications. 1 The global incidence of T1D has risen in the last decades and by 2040, the number of people living with T1D is projected to reach 13.5–17.4 million. 2
The natural history of T1D starts with the onset of autoimmunity in individuals with normoglycemia (stage 1) and progresses at a variable rate to dysglycemia (stage 2) before becoming clinically symptomatic (stage 3). Autoantibodies against insulin (IAA), glutamate decarboxylase (GADA), protein tyrosine phosphatase IA-2 (IA2A), and zinc transporter 8 (ZnT8A) are well-established diagnostic biomarkers detectable in presymptomatic stages 1 and 2 of the disease.3–5
Early-stage diagnosis of T1D allows for timely patient education and training in disease management before overt symptoms appear. 6 This proactive approach minimizes the psychological impact and reduces the risk of serious complications at disease onset, such as diabetic ketoacidosis.7,8 Furthermore, emerging immunotherapy approaches targeting autoimmune processes underscore presymptomatic T1D identification, which may offer a therapeutic window to potentially preserve pancreatic cell mass.6,9 Consequently, early-stage T1D screening programs are gaining significant international attention10–14 and, therefore, optimizing the technique and sample type for measuring autoantibodies is crucial to facilitate large-scale population screening.
Serum-based radiobinding assay (RBA) remains the gold standard method for T1D antibody detection. 15 However, their reliance on radioactive isotopes, lengthy incubation times, and limited multiplexing capacity hinder scalability and automation. This significantly restricts their use in large population studies. In the last decade, advancements in T1D-related autoantibody assays have sought to provide improvements over traditional RBA. 15 Antibody Detection by Agglutination-PCR (ADAP) is a promising novel technique leveraging standard PCR protocols to simultaneously detect all four key autoantibodies associated with T1D in a single assay that also minimizes sample volume requirements, positioning it as a strong candidate for large-scale screening purposes. 16
In addition to the technical aspects of the assay, the selection of an optimal sample type significantly impacts the efficiency of T1D screening. Although capillary sampling is generally preferred over venipuncture in population studies,17–19 limited research has rigorously compared autoantibody measurements obtained across different blood-derived samples using modern, high-throughput methods. This study presents the first comprehensive evaluation of the ADAP technique for quantifying IAA, GADA, IA2A, and ZnT8A across various sample types: serum, plasma, whole blood, and dried blood spot (DBS). The diagnostic accuracy and processing feasibility for each sample type were compared to determine their suitability for early-stage T1D screening programs.
Methods
Study design and participants
T1D-related autoantibodies (IAA, GADA, IA2A, and ZnT8A) were analyzed by ADAP in a cohort of 266 participants: 171 patients with new-onset T1D and at least one positive autoantibody (median age 13.7 years, interquartile range [IQR]: 6.5–15.0; 55% male) and 95 healthy controls (median age 41.9 years, IQR: 29.5–56.7; 37% male). T1D-related autoantibodies had previously been analyzed using RBA and all patients were diagnosed according to the American Diabetes Association criteria. 20 The study was conducted in accordance with the principles of the Declaration of Helsinki (2008) of the World Medical Association. Ethical approval was obtained from the local ethics committee and informed consent was obtained from all participants and/or their legal guardians.
In the initial phase of this study, diagnostic sensitivity and specificity of the ADAP assay for each autoantibody in serum were evaluated, along with concordance with the gold standard RBA in the entire cohort of 266 participants.
Subsequently, the concordance of ADAP results across serum, plasma, whole blood, and DBS was assessed in a subgroup of 66 participants (52 patients with T1D and 14 healthy controls) selected from the original cohort, based on the availability of all sample types. All participants concurrently provided serum (BD Vacutainer SSTII Advance tube) and whole blood (BD Vacutainer K2 EDTA tube). From the EDTA tube, aliquots were taken for autoantibody measurement in whole blood and for the preparation of DBS cards. The remaining blood was then centrifuged to obtain plasma.
DBS cards were prepared by applying 65 μL of EDTA-anticoagulated whole blood onto preprinted 13-mm-diameter circles on Whatman 903 protein saver cards. Cards were air-dried for 2 h at room temperature and then stored individually in zipped plastic bags containing desiccant at 4°C until autoantibody analysis. For the elution of DBS samples, six 3 mm discs were incubated in 1 mL of PBS on a shaking platform, at 37°C for 90 min. The supernatant was then concentrated using the Amicon Ultra-0.5 centrifugal filters 100 kDa MWCO (Millipore). Initially, 400 µL of the eluant was added to the filter and centrifuged at 14,000× g for 2.5 min. Then, an additional 400 µL was added to the same filter and centrifuged at 14,000× g for 10 min. The resulting concentrated eluant (30–50 µL), as well as the serum, plasma, and whole-blood samples, was stored at −80°C until analysis.
Finally, the stability of autoantibodies in whole blood was assessed in a group of 18 patients with T1D from the original cohort, characterized by the following positivity rates: IAA (88.9%), GADA (100%), IA2A (66.7%), and ZnT8A (44.4%). For analysis, only positive results for each autoantibody were used. Stability was evaluated under two conditions: (1) prolonged storage at room temperature (20°C–25°C) for up to 20 days, and (2) exposure to four consecutive freeze–thaw cycles. To assess the impact of unrefrigerated storage time, each EDTA-collected whole-blood sample was divided into 100 μL aliquots. A subset of these aliquots was frozen at −80°C on days 1, 2, 3, 4, 5, 7, 10, 14, and 20 postcollection. In addition, a separate set of aliquots from each sample on day 0 was subjected to four consecutive freeze–thaw cycles.
Autoantibody analyses
T1D-related autoantibodies were measured in serum by RBA and in serum, plasma, whole blood, and DBS by ADAP.
Radiobinding assay
Pancreatic-autoantibodies (IAA, GADA, IA2A, and ZnT8A) were determined in serum using standardized RBA protocols, as previously described. 21 In brief, IAA was determined using a competitive fluid-phase radioassay that uses [125I]-labeled recombinant human insulin (Revvity, Waltham, MA, USA) as antigen. Specificity was 98%–100%, with a sensitivity of 60%–65% (internal validation, last 5 years). GADA, IA2A, and ZnT8A followed the same standard RBA protocol with in vitro transcribed and translated, [35S]-labeled, recombinant human full-length glutamate decarboxylase, IA2ic (amino acids 605–979), and ZnT8 antigens. The ZnT8A assay simultaneously measures autoantibodies against both variants of the COOH-terminal domain (Arg325/Trp325). Our laboratory participates regularly in the International Islet Autoantibody Standardization Program (IASP), obtaining a specificity of 99%–100% for all three autoantibody assays and sensitivity of 68%–88% for GADA, 62%–78% for IA2A, and 62%–74% for ZnT8A in the last 5 years.
Antibody detection by agglutination-PCR assay
Serum and plasma samples were processed using ADAP assay (Enable Biosciences, CA, USA) by incubating 4 μL of the sample with 8 μL of conjugated mixture at 37°C for 30 min. For whole-blood and DBS extracts, the conjugation reaction consisted of 6 μL of sample and 6 μL of conjugate mixture. After conjugation, 4 μL of the reaction mixture was combined with 116 μL of ligation mixture and incubated at 37°C for 15 min. Following ligation, 25 μL of the resulting mixture was added to 25 μL of a PCR amplification mixture containing specific primers for the four target antibodies (IAA, GADA, IA2A, and ZnT8A). Amplification was performed under the following conditions: 95°C for 5 min followed by 13 cycles of 95°C for 30 s and 56°C for 90 s. Conjugation, ligation, and preamplification steps were carried out on a Verity 96-well Thermal Cycler (Applied Biosystems, MA, USA). The resulting preamplified products were then diluted 20-fold with molecular biology-grade water and quantified with different primer pairs using SYBR green-based real-time qPCR on a QuantStudio 6 Pro Real-Time PCR System (Thermo Fisher Scientific, MA, USA). Conditions for qPCR were 95°C for 10 min, followed by 40 cycles of 95°C for 15 s and 56°C for 60 s. Results were expressed as cycle threshold (Ct) relative to buffer control (ΔCt).
Positivity thresholds were determined as the mean value plus three standard deviations of the measurements from 100 healthy controls.
Statistical analyses
Qualitative variables are presented as percentages, while quantitative variables are expressed as median and IQR: P25–P75.
Sensitivity was defined as the percentage of positive autoantibody results among patients with T1D and specificity as the percentage of negative results among healthy controls. Concordance for positive/negative results between methods (ADAP vs. RBA) and sample types (serum, plasma, whole blood, and DBS) was assessed using Cohen’s kappa test, and the following thresholds were used for interpretation: kappa values ≥0.81 indicated strong concordance, values from 0.61 to 0.80 indicated substantial concordance, values between 0.41 and 0.60 indicated moderate concordance. Autoantibody levels in serum by ADAP were compared with those in plasma, whole blood, and DBS using Pearson correlation analysis.
For the stability study, median values for each autoantibody were calculated for positive cases at each time point. Differences among these medians were assessed using nonparametric Friedman analysis of variance. Post hoc Wilcoxon signed-rank tests were applied where appropriate. The P-values were corrected for multiple comparisons using the false discovery rate (FDR). All statistical analyses were performed using SPSS software (v.29; SPSS Inc., Chicago, IL).
Results
To establish the autoimmune status of each participant, serum levels of the four autoantibodies were quantified using RBA, which served as the gold standard for this study.
Diagnostic sensitivity/specificity and concordance of ADAP compared with RBA
Serum samples from the entire cohort (171 patients with stage 3 T1D and 95 healthy controls) were analyzed to assess the clinical sensitivity, specificity and concordance between ADAP and RBA methods for quantifying T1D-associated autoantibodies (Table 1). ADAP demonstrated slightly higher sensitivity than RBA across all autoantibodies tested, with strong concordance between methods (Cohen’s kappa >0.81 for each autoantibody) validating ADAP as a reliable alternative to the current gold standard.
Comparison of Autoantibody Detection Performance Achieved with RBA and ADAP in Serum
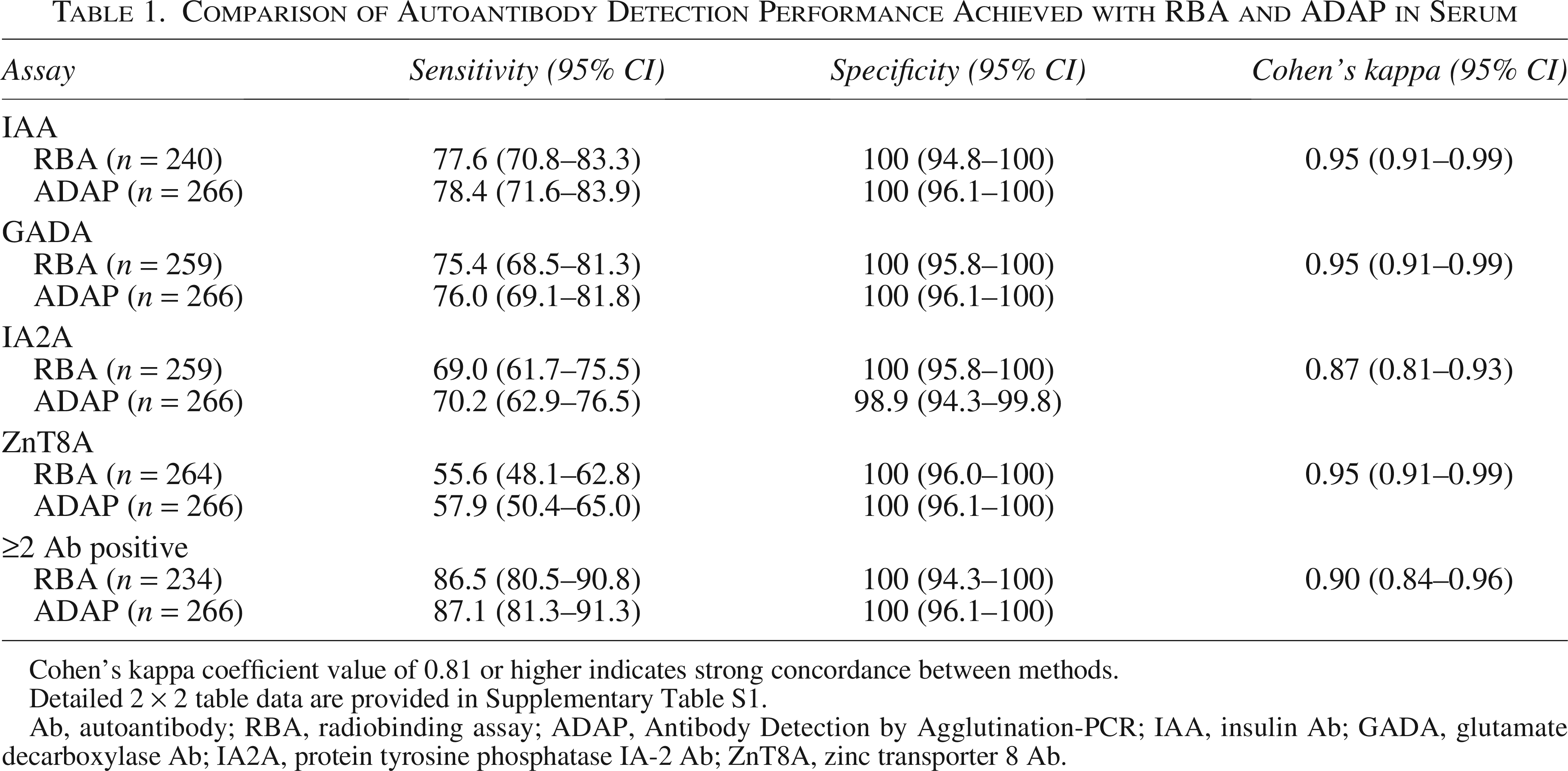
Cohen’s kappa coefficient value of 0.81 or higher indicates strong concordance between methods.
Detailed 2 × 2 table data are provided in Supplementary Table S1.
Ab, autoantibody; RBA, radiobinding assay; ADAP, Antibody Detection by Agglutination-PCR; IAA, insulin Ab; GADA, glutamate decarboxylase Ab; IA2A, protein tyrosine phosphatase IA-2 Ab; ZnT8A, zinc transporter 8 Ab.
Detection of two or more autoantibodies, the established consensus for the diagnosis of stages 1 and 2 T1D, achieved 87.1% sensitivity and 100% specificity in ADAP, with 95.3% overall agreement with RBA and Cohen’s kappa coefficient of 0.90 (Table 1).
ADAP assay performance across different blood-derived samples compared with serum
To evaluate the performance of the ADAP assay across different sample types, 66 participants (52 patients with T1D and 14 healthy controls) from the original cohort were selected. Each participant provided concurrent serum, plasma, whole blood, and DBS samples. Pancreatic autoantibody levels measured by ADAP in different sample types were compared with the results obtained in serum (Figs. 1–4).
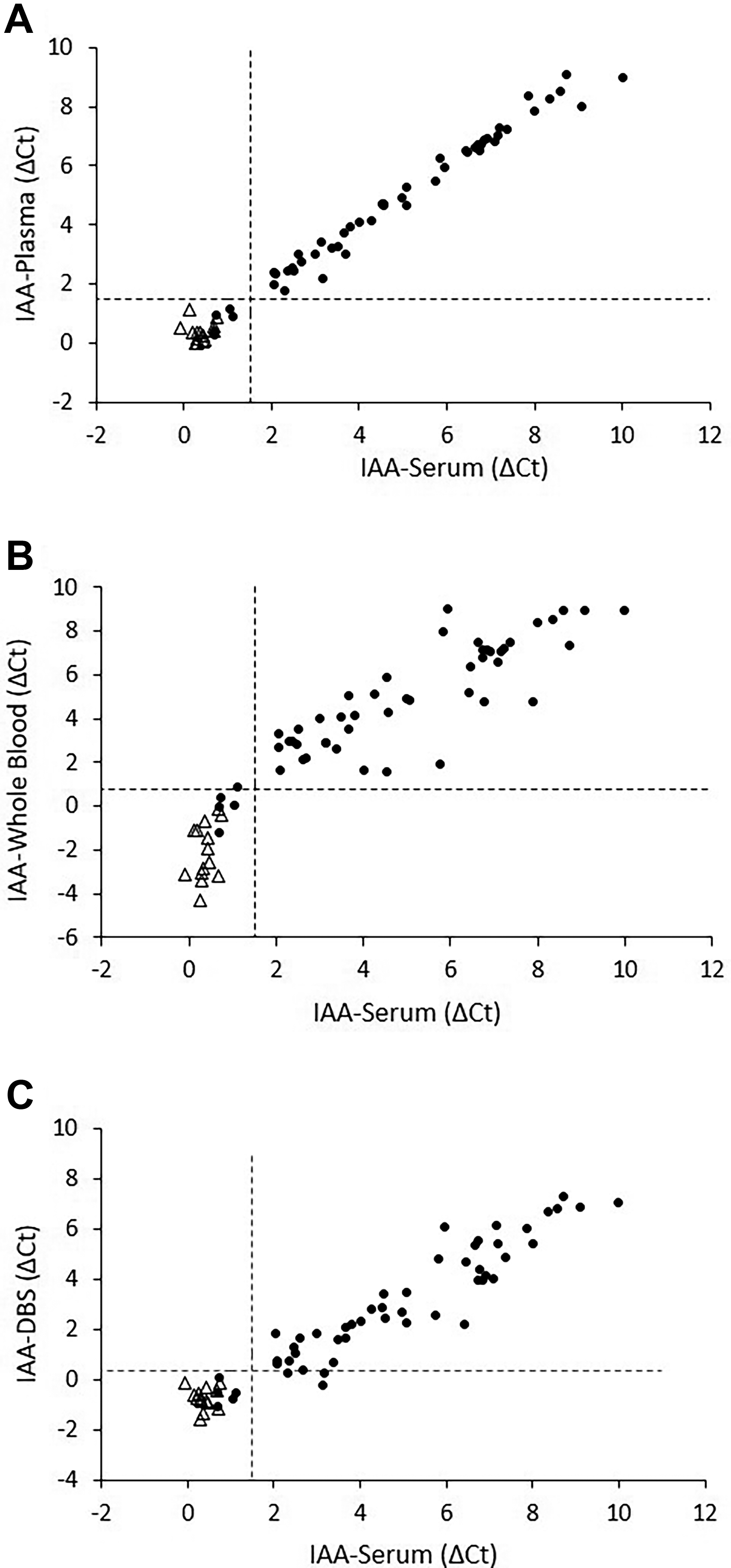
Correlation of IAA levels by antibody detection by ADAP in serum versus plasma
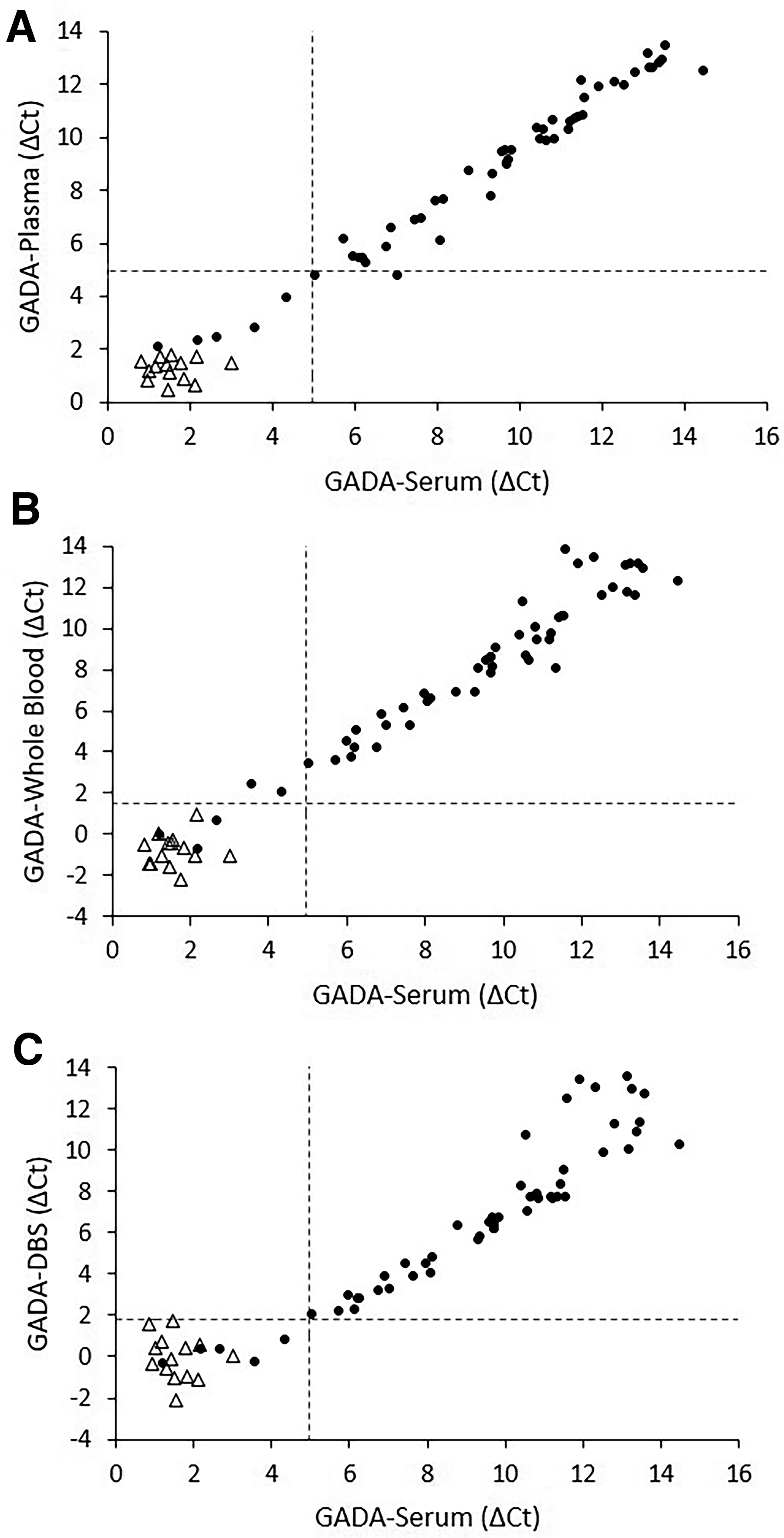
Correlation of GADA levels by ADAP in serum versus plasma
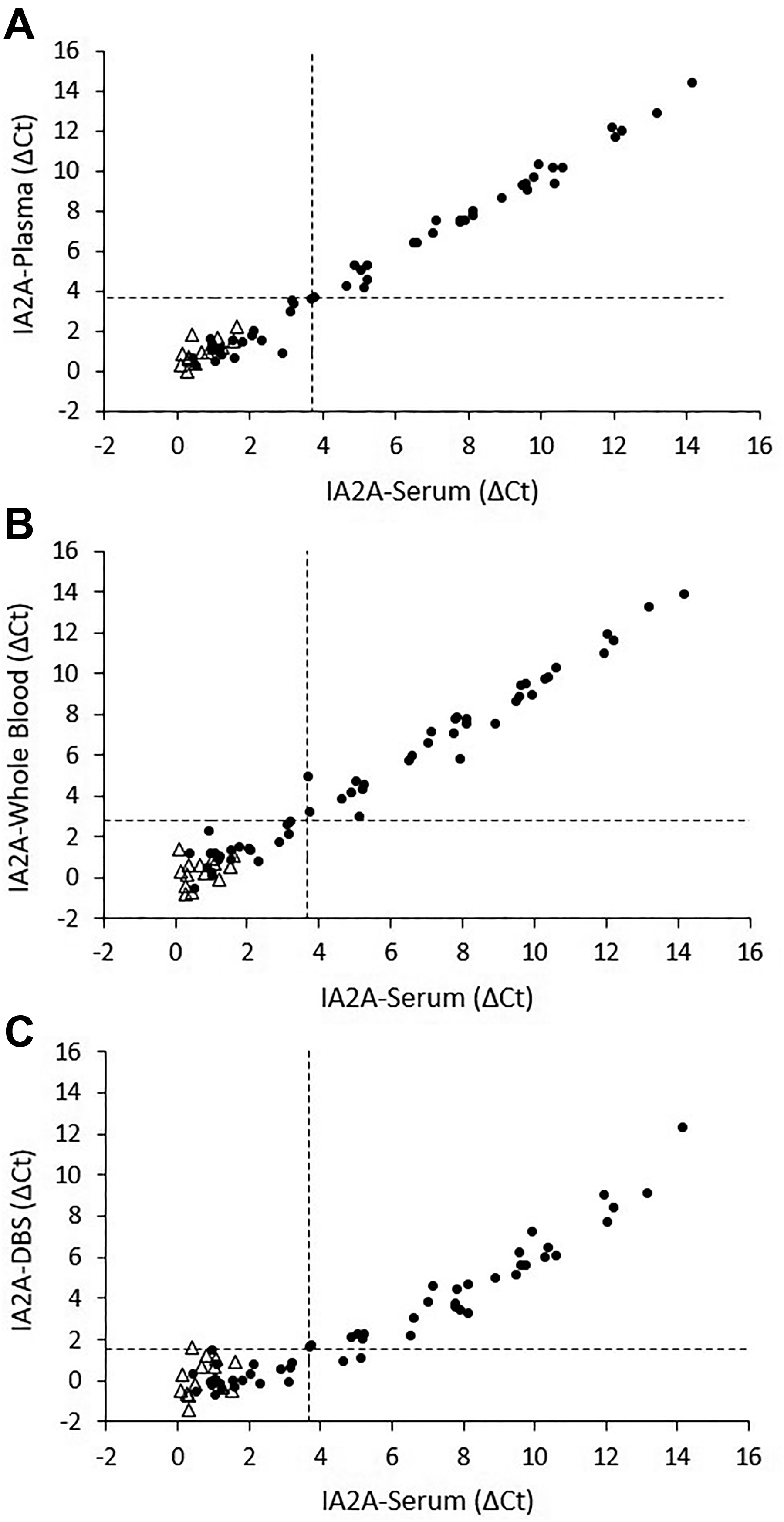
Correlation of IA2A levels by ADAP in serum versus plasma
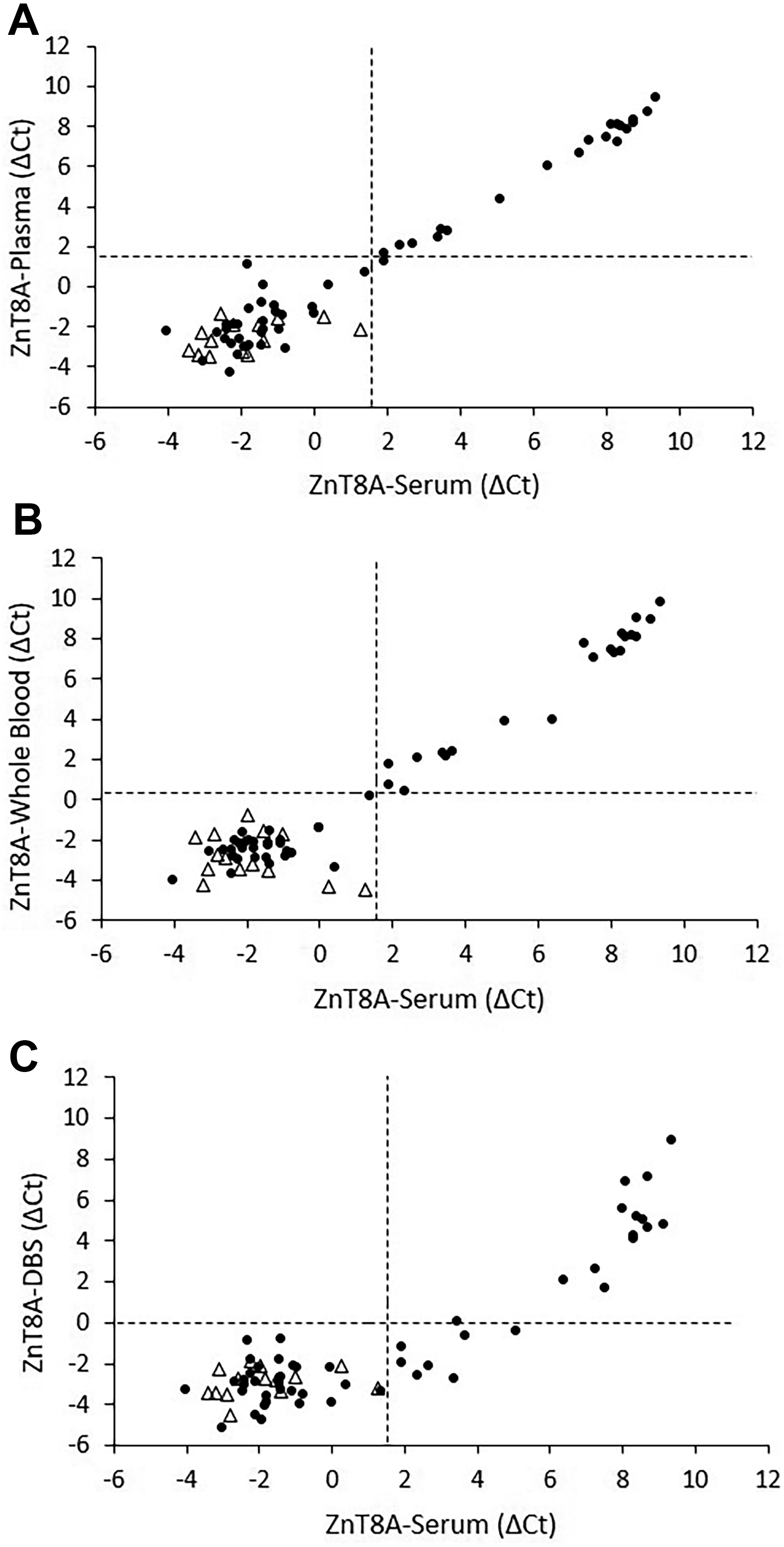
Correlation of ZnT8A by ADAP in serum versus plasma
Serum and plasma measurements achieved strong correlation for all tested autoantibodies (Figs. 1–4). Strong concordance was also maintained, although a slight reduction was observed for GADA and ZnT8A (Table 2). Specifically, two serum-positive GADA cases were not detected in plasma (2/47), likely due to their autoantibody levels being close to the positivity threshold. A similar issue was observed for ZnT8A in one case (1/21).
Performance of Autoantibody Detection by ADAP in Different Blood-Derived Sample Types Compared with Serum
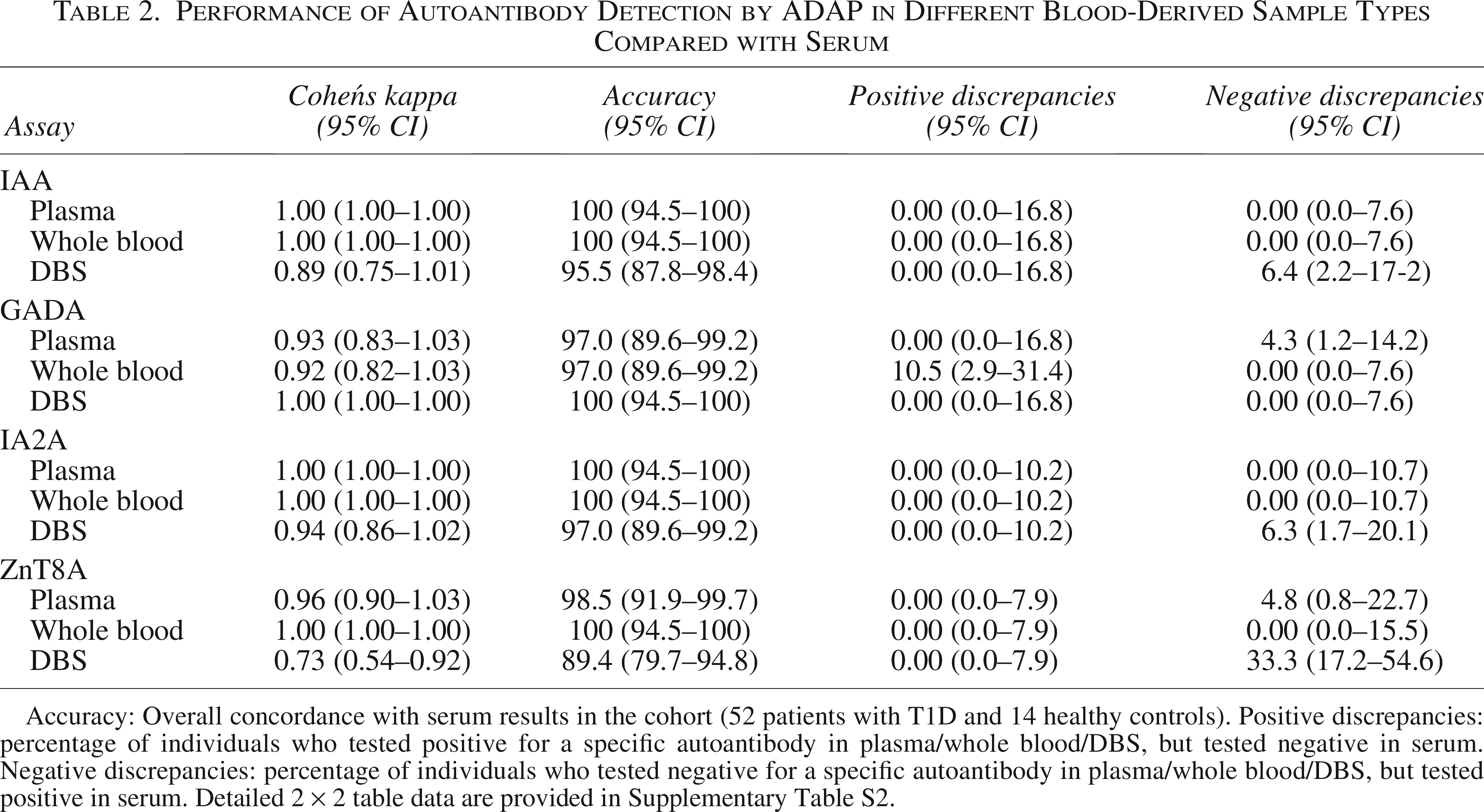
Accuracy: Overall concordance with serum results in the cohort (52 patients with T1D and 14 healthy controls). Positive discrepancies: percentage of individuals who tested positive for a specific autoantibody in plasma/whole blood/DBS, but tested negative in serum. Negative discrepancies: percentage of individuals who tested negative for a specific autoantibody in plasma/whole blood/DBS, but tested positive in serum. Detailed 2 × 2 table data are provided in Supplementary Table S2.
In parallel, serum and whole-blood measurements exhibited strong correlation and high Cohen′s kappa coefficients (ranging 0.92–1.0), comparable with plasma results. In this case, two serum-negative GADA cases were detected as positive in whole blood, a finding potentially attributable again to their proximity to the analytical cutoff. Importantly, using whole blood did not lead to any loss of detection for autoantibodies found positive in serum (Table 2).
For DBS sampling, results were comparable with plasma and whole blood only for GADA. For the other autoantibodies, the proportion of undetected serum-positive cases was higher with DBS than with whole blood, particularly for ZnT8A, where seven serum-positive cases (7/21) were not detected (Table 2). These findings suggest that DBS sampling may introduce significant bias in autoantibody quantification.
Whole-blood stability study
The stability of autoantibodies in whole blood was assessed in 18 patients with T1D, specifically focusing on the effects of unrefrigerated storage time and repeated freeze–thaw cycles.
As shown in Table 3, at least three autoantibodies remained stable in whole blood for up to 20 days without refrigeration. The only exception was IAA, which showed a significant 23% decrease in detection between days 10 and 20. These results could potentially simplify transport and storage protocols. Moreover, after four freeze–thaw cycles, whole blood remained stable for autoantibody detection, with no significant decrease in performance compared with unfrozen samples (Table 4), supporting that whole blood is a reliable sample type for autoantibody detection under different storage conditions.
Stability of Autoantibody Detection in Whole Blood Stored at Room Temperature
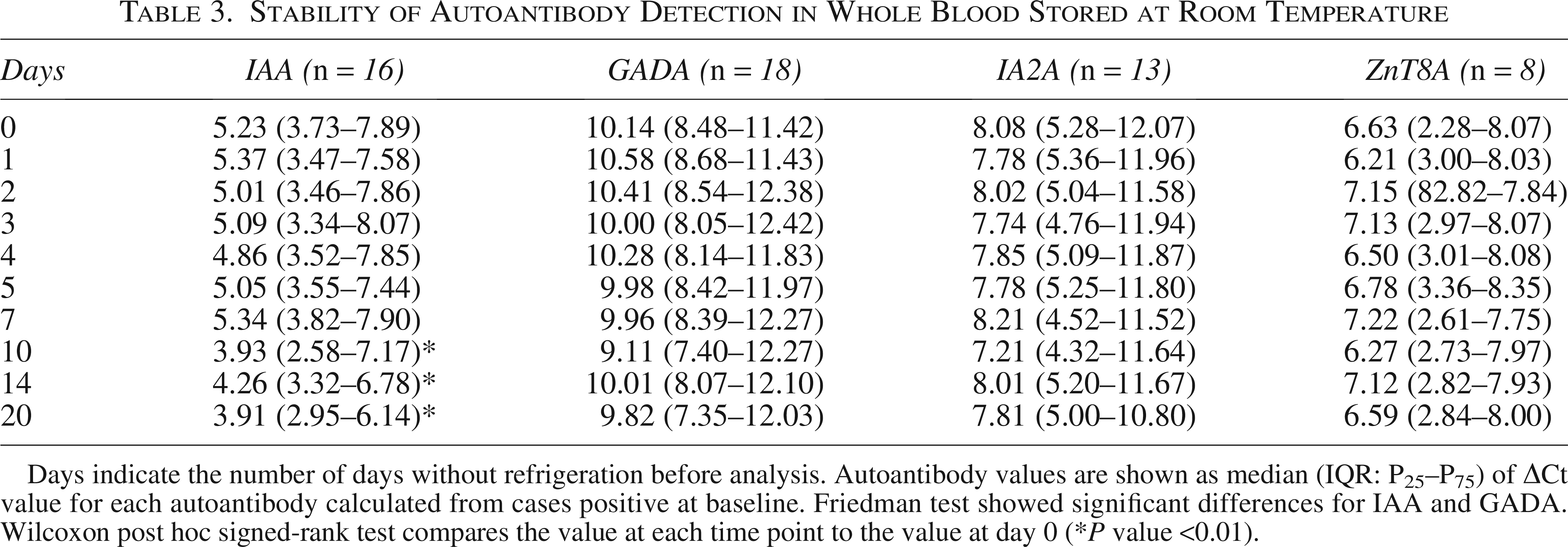
Days indicate the number of days without refrigeration before analysis. Autoantibody values are shown as median (IQR: P25–P75) of ΔCt value for each autoantibody calculated from cases positive at baseline. Friedman test showed significant differences for IAA and GADA. Wilcoxon post hoc signed-rank test compares the value at each time point to the value at day 0 (*P value <0.01).
Impact of Freeze–Thaw Cycles on Autoantibody Detection in Whole Blood

Cycles indicate the number of freezing–thawing events the sample underwent before analysis. Autoantibody values are shown as median (IQR: P25–P75) of ΔCt value for each autoantibody calculated from cases positive at baseline. The Friedman test showed no statistically significant difference in any autoantibody.
Discussion
The ability to identify individuals at high risk for T1D before the onset of clinical symptoms offers a significant opportunity to improve patient outcomes. The development and validation of reliable and scalable methods for pancreatic autoantibody detection, with careful consideration of optimal sample types, are crucial for optimizing early T1D diagnosis in large population studies. In response to this critical need, this study successfully applied the novel ADAP method to different blood-derived samples, demonstrating its efficacy for direct autoantibody detection in whole blood. This approach holds great promise for early-stage T1D population screening programs.
A high-throughput method that requires a minimal sample volume and ensures diagnostic reliability and scalability is essential for effective screening strategies. ADAP addresses this challenge because it requires only a small sample volume for the simultaneous measurement of four pancreatic autoantibodies. Moreover, as shown in the present study and consistent with previous reports,16,22 ADAP demonstrated results in serum that are comparable with the established gold standard RBA. However, despite good overall agreement, the concordance for positive/negative results between assays is compromised at low autoantibody levels. This highlights the importance of establishing robust positivity thresholds and testing all four autoantibodies for more reliable diagnoses, since the established consensus for stages 1 and 2 T1D diagnosis requires the presence of at least two positive autoantibodies. 6 In addition, while the slightly increased sensitivity of ADAP is an advantage for detecting low autoantibody levels, it could also compromise its diagnostic specificity. This is a critical challenge in population screening for T1D because it could lead to false positives. Therefore, to minimize the possibility of false positives, any ADAP result that is positive or close to the positivity threshold should be confirmed with a second blood draw from the patient, using a different method. This process follows “the rule of twos,” a widely accepted clinical guideline that requires two different autoantibodies confirmed in two tests from two separate samples. If the result remains positive, close patient monitoring would be advisable in accordance with the consensus guidelines for individuals who are positive for islet autoantibodies. 6
In addition to serum, this study further demonstrated the versatility of ADAP in detecting autoantibodies in plasma, whole-blood, and DBS samples. However, its suitability for large-scale sample processing and the reliability of diagnostic results varied across sample types.
Previous studies have evidenced the feasibility of measuring T1D-associated autoantibodies in easily collected samples by finger prick, such as DBS17,18 and whole blood.23,24 However, to the best of our knowledge, no prior research has directly compared the performance of these two sample types using ADAP for T1D autoantibody detection. While DBS appears to be an attractive alternative to venipuncture due to its ease of collection and transport, our analysis revealed significant limitations. We found lower concordance between serum and DBS than between serum and whole blood, indicating a risk of missing positive cases when using DBS for autoantibody screening. Furthermore, it should be noted that DBS sampling requires a more laborious preprocessing, which adds to costs, time, and complexity for automation. Another challenge with DBS is ensuring full saturation of the collection card, a condition frequently unmet in practice. Notably, our findings demonstrate the robust stability of whole blood in nonrefrigerated storage for up to 20 days in most autoantibodies, and for 7 days in the case of IAA. This stability considerably simplifies sample transport and storage, making it ideal for multicenter programs. Overall, whole blood testing by ADAP offers significant advantages over DBS, as it is a more rapid, reliable, and cost-effective diagnostic solution for large-scale studies.
Serum and plasma showed high correlation for autoantibody measurements, howewer, these approaches are limited by two practical challenges: centrifugation is not always possible at the collection site, and obtaining sufficient sample volume for target blood component separation can often be difficult, especially when venipuncture is not feasible. These limitations make serum and plasma impractical for large-scale population screening. Thus, measuring autoantibodies in small, centrifugation-free samples obtained through minimally invasive methods, such as finger or earlobe pricks, can significantly reduce the drawbacks associated with traditional blood collection techniques and improve population accessibility for large-scale screening. The use of specialized EDTA tubes for capillary whole-blood sampling aligns well with the goal of minimizing these barriers, since ADAP-based technology only requires 6 µL of whole-blood sample. A recent study has shown that home-based capillary collection of whole blood is feasible, achieving both acceptable participation rates and adequate sample volumes for analysis. 25
Our study accentuates the risk of missing positive cases when using DBS in screening programs, a key strength of our findings, as it highlights a potential caveat of current screening protocols. In addition, a low rate of false positives could be detected by ADAP in the analysis of pancreatic autoantibodies in whole blood; therefore, confirmation of positive results using another method and two separate blood draws is recommended.
Although participant preference often favors capillary blood collection for screening, this study used venous blood to ensure sufficient sample volume for robust and comprehensive comparisons. However, this potential limitation is mitigated by the demonstrated equivalence in autoantibody measurements between venous and capillary blood samples. 23 Furthermore, the fixed blood volume used for DBS collection in our experimental design minimized the variability arising from differing sample volumes collected on the card.
In conclusion, the ADAP assay demonstrates reliable detection of IAA, GADA, IA2A, and ZnT8A in whole blood, achieving results comparable with the standard RBA in serum. This study highlights the benefits of using whole blood as a stable and practical sample for T1D screening with the ADAP assay. The low sample volume requirement, simplified preparation, and high diagnostic consistency are particularly valuable properties for early-stage T1D screening programs.
Authors’ Contributions
I.U.: Conceptualization (equal), writing—original draft (lead), methodology (equal), formal analysis (lead), and writing—review and editing (equal). M.G.-A.: Conceptualization (equal), writing—original draft (supporting), investigation (equal), formal analysis (supporting), and writing—review and editing (equal). B.C.: Conceptualization (equal), writing—original draft (supporting), and writing—review and editing (equal). J.A.-O.: Formal analysis (supporting) and writing—review and editing (equal). E.F.-R.: Resources (equal) and writing—review and editing (equal). V.U.: Resources (equal) and writing—review and editing (equal). J.R.B.: Formal analysis (supporting) and writing—review and editing (equal). I.R.: Resources (equal), funding acquisition (equal), and writing—review and editing (equal). L.C.: Conceptualization (equal), funding acquisition (equal), supervision, and writing—review and editing (equal). R.M.: Conceptualization (equal), funding acquisition (equal), writing—original draft (supporting), methodology (equal), investigation (equal), and writing—review and editing (equal).
Footnotes
Acknowledgments
The authors thank Cristina Llinares, Lorena Ansual, and María Teresa Pérez de la Puente for their technical assistance. They also thank all the patients who voluntarily agreed to take part in this study, as well as the participating doctors and nurses.
Author Disclosure Statement
The authors have nothing to disclose.
Funding Information
This work was partially supported by grants from the University of the Basque Country, EHU (IT1739-22), and the Department of Health of the Basque Government (GV2024111044).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
