Abstract
Diabetic retinopathy (DR) is a common and potentially sight-threatening complication of diabetes. Early detection of DR through screening can prevent visual loss. Handheld fundus cameras combined with artificial intelligence (AI) technology may improve DR screening. We evaluated the Aireen AI algorithm’s performance in grading DR in fundus images captured by the handheld Optomed Aurora. Two retina specialists and Aireen graded 624 fundus images for DR. Sensitivity, specificity, and predictive values were measured against the ophthalmologists’ grading. Overall, 97% of images were sufficient for DR classification. Aireen demonstrated 94.8% sensitivity, 91.4% specificity, and 92.7% diagnostic accuracy for DR. Aireen showed high diagnostic accuracy in detecting DR in Optomed Aurora images, suggesting its potential for effective screening. The validated use of AI with a handheld fundus camera may streamline the screening process, reduce the burden on health care professionals, and improve access to screening and patient outcomes through enhanced diagnostic accuracy.
Keywords
Introduction
The global diabetes epidemic affects an increasing number of patients worldwide. 1 Diabetic retinopathy (DR) is the most common complication of diabetes. It can lead to irreversible visual loss, which can be prevented by early detection and timely intervention. 2 Traditional DR screening methods rely on specialized equipment and trained personnel, limiting accessibility. Telemedicine technology, portable fundus cameras, and artificial intelligence (AI)-based grading for DR offer promising solutions to alleviate the burden and improve cost-effectiveness of screening. 3 Handheld fundus cameras like Optomed Aurora are promising for screening due to their low cost, portability, ease of use, and integration with AI algorithms for automated DR grading. 4 They also enable screening for populations lacking access to conventional procedures.
AI algorithms have recently shown high accuracy in detecting DR, often matching or surpassing human graders.5,6 However, results vary based on the camera, image modality, and AI algorithm used. External validation in real-world settings is essential to ensure compliance with DR screening recommendations before large-scale clinical application. 6 This study aimed to compare the Aireen AI solution for DR detection against ophthalmologist experts using fundus images from the handheld Optomed Aurora in a real-world clinical setting.
Materials and Methods
We evaluated the performance of the Aireen AI algorithm in grading DR using images captured by the handheld Optomed Aurora fundus camera in a real-world clinical setting. This study was conducted at Oulu University Hospital with the approval of the Oulu University Hospital Ethical Research Committee, adhering to the tenets of the Declaration of Helsinki.
In the first phase, 106 consecutive patients with either type 1 or type 2 diabetes attending DR screening were evaluated. Since most patients attending DR screening have no DR or only mild nonproliferative DR (NPDR), an additional 50 patients with more severe DR attending follow-up visits at the hospital’s outpatient eye clinic were included. Two retina specialists independently graded all images and were blinded to each other’s and AI’s grading results. Images were captured using standardized methods: fundus photography was performed after mydriasis, first with a traditional table-top fundus camera as part of a routine screening protocol, followed by 50° photography with Optomed Aurora by the same photographer. Papilla- and macula-centered fundus images were obtained from both eyes of a total of 156 participants. A total of 624 fundus images were graded for DR by two retina specialists and Aireen. Five out of 156 patients had insufficient image quality for AI analysis and were graded only by human graders.
Aireen is an autonomous, high-speed, noninvasive solution that uses AI to analyze retinal images captured by a fundus camera. Aireen classifies images into two categories: DR and non-DR. Before classification, AI algorithms review the image quality within seconds, allowing immediate upload of a new image if the quality is poor. Quality control is performed through neural network models that verify the presence of the macula and optic disk, assess image sharpness, and confirm the use of an optical camera.
EfficientNet V2 is a convolutional neural network architecture designed for high performance and efficiency, combining model scaling with advanced training techniques. To ensure clinical transparency and support decision-making, Aireen incorporates explainability features alongside its classification output. Although the underlying EfficientNet V2 model is inherently a black-box architecture, the system is augmented with heatmap-based visualizations that highlight regions of the retinal image contributing most significantly to the DR classification. These heatmaps are made available to physicians, enabling them to visually verify the presence of key pathological features such as microaneurysms, hemorrhages, or exudates. This approach fosters trust in the AI output and facilitates the integration of Aireen into routine clinical workflows by providing clinicians with interpretable cues linked to model predictions. The Aireen AI model has been trained, validated, and tested on more than 800,000 fundus images sourced from a large and ethnically diverse population.
Sensitivity, specificity, and predictive values were measured against the ophthalmologists’ grading. DR classification followed the five-scale grading system from the Finnish Current Care Guideline, which is equal to the International Classification of Diabetic Retinopathy (ICDR) scale.7,8 The AI-based results were categorized as non-DR, any-DR, or ungradable.
Results
Among the participants, five patients (3.2%) had poor-quality images in both eyes due to cataract formation, which were deemed nongradable by the Aireen solution. The remaining 151 patients (97%) had images of sufficient quality for DR classification. Based on human grading assessments, all DR stages and various lesion types were represented in the dataset: Non-DR was identified in 183 eyes, mild NPDR in 21 eyes, moderate NPDR in 66 eyes, severe NPDR in 1 eye, and proliferative DR in 41 eyes. Although the assessments were conducted at the eye level, the AI-based grading was performed at the patient level, whereby the presence of DR in either eye was considered indicative of DR for the patient. The Aireen AI algorithm demonstrated 94.8% sensitivity, 91.4% specificity, and 92.7% diagnostic accuracy for DR. Of the 151 patients assessed, the Aireen system evaluated 55 true positives, 8 false positives, 85 true negatives, and 3 false negatives. In six of the eight false-positive cases, other ocular abnormalities or diseases were detected. The performance of the Aireen algorithm in detecting DR in fundus images is summarized in Table 1.
Performance of the Aireen System in Terms of Sensitivity, Specificity, Positive Predictive Value, Negative Predictive Value, Agreement, and Accuracy
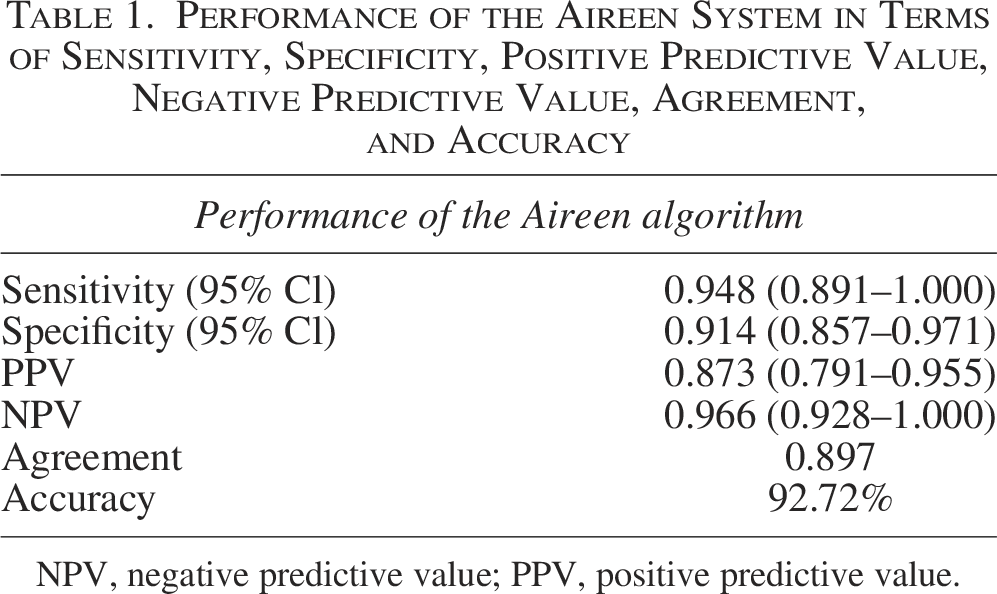
NPV, negative predictive value; PPV, positive predictive value.
Discussion
The combined use of the Optomed Aurora camera and Aireen AI algorithm showed high accuracy in detecting DR, surpassing the widely recommended benchmarks for DR screening, which aim for 80% sensitivity and 90%–95% specificity. 9 Recent evidence supports the feasibility of the Optomed Aurora for DR screening due to its sufficient image quality and high diagnostic accuracy. 4 The integration of portable fundus cameras, teleophthalmology, and AI technology, as demonstrated by Aireen’s performance, offers a promising and reliable solution for DR screening in real-world clinical practice. The system’s high accuracy, rapid results, and ease of use can enhance screening rates and early detection of DR, potentially improving visual outcomes for patients with diabetes and reducing the burden of diabetic eye disease. In parallel, recent studies have demonstrated the utility of machine learning for predicting incident DR in adults with T1D, reinforcing the growing clinical value of AI-driven approaches in DR risk stratification and screening. 10 The Aireen AI algorithm is among the AI-based systems developed for the classification of DR into two categories: non-DR and any-DR. Depending on the specific AI screening methodology employed, DR can also be categorized as nonreferable DR (including no DR and mild NPDR) and referable DR (defined as more than mild NPDR).
Despite the proven benefits of DR screening in reducing visual impairment, the implementation of nationwide programs remains inconsistent across developed countries, primarily due to resource constraints. Large-scale DR screening requires investments in infrastructure, trained health care professionals, and resources for follow-up, referral, and treatment. The increasing global prevalence of diabetes underscores the urgent need for scalable screening solutions. 1 The integration of portable fundus cameras, teleophthalmology, and AI technology, as evidenced by Aireen’s performance, offers a promising avenue to address these challenges.
To prevent unnecessary referrals to ophthalmologists, AI solutions for DR screening must maintain a balance between high sensitivity and specificity to ensure the cost-effectiveness of screening programs. Training and careful validation of algorithms by AI providers are crucial for the clinical usability and reliability of AI in DR screening. Further investigation and broader implementation are needed to address issues such as data sharing and privacy, transparency of AI algorithms, data standardization and interoperability across platforms, and concerns about patient safety for the implementation and acceptance of AI in everyday clinical practice.
Conclusions
The handheld Optomed Aurora fundus camera, combined with the Aireen AI algorithm, offers a promising and reliable solution for DR screening in real-world clinical practice. Its potential to streamline the screening process and improve patient outcomes warrants further investigation and broader implementation. The system’s high accuracy, rapid results, and ease of use can enhance screening rates and early detection of DR, ultimately improving visual outcomes for patients with diabetes and reducing the burden of diabetic eye disease.
Authors’ Contributions
All authors have substantially contributed to the conception of the present study and the acquisition, analysis, and interpretation of the data. They have reviewed the article critically for important intellectual content and approved the final version to be published. All authors have agreed to be accountable for all aspects of the study in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Footnotes
Acknowledgment
Imaging engineer Antti Keskitalo is greatly acknowledged for his expert help in fundus photography.
Author Disclosure Statement
The authors declare no conflicts of interest.
Funding Information
This work was supported by PPSHP VTR under grant number K71769 and the Finnish Eye Foundation under grant number 00082022.
