Abstract
Introduction:
Achieving optimal glycemic control in patients with type 1 diabetes mellitus (PwT1DM) is essential to prevent complications. Continuous subcutaneous insulin infusion (CSII) systems combined with continuous glucose monitoring (CGM) have improved outcomes, but the effectiveness of additional technologies, such as mobile apps and hybrid closed-loop systems (HCLSs), remains unclear. This study evaluates glycemic control and quality of life (QoL) in adult PwT1DM transitioning from multiple daily injections (MDI) to a CSII, first with the Mylife™ Dose app and subsequently switching to an HCLS.
Materials and Methods:
This was a 10-month, multicenter, open-label sequential study involving 135 adults with type 1 diabetes (T1D), all of whom were using isCGM and MDI before transitioning to CSII, first with the Mylife Dose app and later to an HCLS. Glycemic control (glycated hemoglobin [HbA1c], time in range [TIR], time below range, time above range), insulin requirements, and QoL/treatment satisfaction/hypoglycemia perception (Diabetes Quality of Life questionnaire, Diabetes Treatment Satisfaction Questionnaire, Clarke’s test) were measured at each of the four study visits.
Results:
Transitioning from MDI to CSII showed modest improvements in HbA1c (7.57%–7.42%; P = 0.02) and TIR (56.3%–60.4%; P < 0.01). The introduction of the Mylife Dose app did not provide significant additional improvements in glycemic control or QoL, although it provided an additional tool for diabetes management. However, switching to an HCLS resulted in substantial improvements in HbA1c (6.7%), TIR (73.1%), and QoL, with over 70% of patients achieving an HbA1c <7%. Insulin requirements increased slightly with the HCLS, primarily due to basal insulin. Adherence was high, with 88.1% completing the study.
Conclusions:
The Mylife Dose app does not improve glycemic control or QoL significantly but offers convenience for patients with T1D. In contrast, HCLSs provide significant metabolic and QoL benefits, supporting their integration into T1D management with appropriate reimbursement policies.
Introduction
Type 1 diabetes (T1D) is characterized by a complete deficiency of insulin, requiring the patients to receive lifelong daily exogenous insulin administration. 1 Achieving optimal glycemic control is critical to minimizing diabetes-related complications. 1 However, despite significant progress in insulin therapies, many patients still struggle to meet the recommended goals for glycemic control. 1
The SED1 study conducted by the SED (Spanish Diabetes Society) underscores a concerning fact as follows: only 33% of individuals with T1D in our country achieve the recommended goal for good glycemic control (glycated hemoglobin [HbA1c] <7%). 1 This reality highlights the need to develop new strategies to improve glycemic control in our patients with T1D.
Technological innovations in diabetes management have grown remarkably in recent years, generating debates about their actual benefits. The “Exchange T1 study” revealed that an increase in the use of technology in T1D failed to produce a corresponding rise in the number of patients achieving optimal control. 2 In addition, the “Sweet study,” which followed a cohort from 19 countries over a decade, demonstrated a significant reduction in HbA1c levels with the use of these new technologies. 3
When examining advancements in diabetes technology, two key areas stand out as follows: continuous glucose monitoring (CGM) devices, which help track glucose levels in real-time and have demonstrated effectiveness in improving glycemic control in patients with T1D, 4 –6 and continuous subcutaneous insulin infusion (CSII) systems, which provide continuous rapid-acting insulin delivery based on a preset schedule. 7 When combined with CGM and a mathematical control algorithm, they form automated insulin delivery or hybrid closed-loop systems (HCLSs). 8
Research shows that while advanced systems improve glycemic control by increasing the time spent within target glucose levels, 9 their complexity and high costs often limit accessibility. Combining CSII with mobile dose calculation apps could offer a simpler, cost-effective alternative for achieving good glycemic control in select patients.
To address this gap, our study evaluates the impact of transitioning patients from multiple daily injections (MDI) and isCGM (FreeStyle® Libre 2) to a CSII system paired with a mobile app. We aim to assess improvements in glycemic control and quality of life (QoL) while comparing outcomes to those achieved with HCLSs. By exploring these alternatives, we hope to contribute to a broader more inclusive strategy for optimizing T1D management.
Materials and Methods
Study design
We conducted a 10-month prospective, multicenter, national, descriptive, open-label, sequential observational study with four study visits.
The study included adult patients with T1D being treated with MDI and using the FreeStyle Libre 2 CGM system for the past 3 months at least. These patients were prescribed a CSII system by their endocrinologist as part of their treatment plan.
The protocol received approval from the Ethics and Research Committee of Asturias (No. 2021.593), and all participants provided written informed consent before enrollment. Personal data protection measures were implemented following applicable legislation.
Recruitment was conducted through the endocrinology departments of 25 hospitals across diverse regions in Spain ensuring a representative patient cohort.
Study visits
YpsoPump-Mylife is a free mobile app that has the potential to visualize insulin therapy data and propose bolus calculations for administration in ISCI users. It is configured with the patient’s personal parameters, such as the sensitivity factor or correction factor, to help calculate the correct amount of insulin to give before meals or as a corrective bolus. The Dose add-on has been designed to allow you to apply the recommended amount of insulin from your smartphone.
At each visit, clinicians collected a fasting venous blood sample to measure glucose and HbA1c levels. They downloaded the 14-day Ambulatory Glucose Profile report from the CGM system, with at least 70% of valid data. They measured weight and height, calculated body mass index (BMI), and documented total, basal, and prandial insulin doses, along with any concomitant illnesses or hospitalizations since the previous visit. Patients completed the Spanish versions of the Diabetes Quality of Life (DQoL) Questionnaire, 10 Diabetes Treatment Satisfaction Questionnaire (DTSQ), 11 and Clarke questionnaires. 12
Adverse events (AEs) were recorded during scheduled visits and unscheduled contacts, noting their onset, duration, severity, and relationship to the study devices. Events were classified as mild, moderate, or severe, with serious AEs promptly reported to the ethics committee and regulatory authorities within 24 h. All data were logged in an electronic system to ensure consistency and adherence to Good Clinical Practice guidelines.
All the information was recorded in an electronic data collection notebook. During the study, investigators conducted clinical review visits, which included uploading device data and reviewing CSII settings, as needed. To ensure consistency, investigators from each center received standardized training on data collection and device usage, with periodic audits performed during the study.
Participants
Individuals were eligible if they had T1D treated with MDI and were using the FreeStyle Libre 2 CGM system. A complete list of eligibility, inclusion, and exclusion criteria can be found in Supplementary Table S1.
Objectives
Primary objective
To evaluate whether the Mylife Dose application improves glycemic control, QoL, satisfaction, and risk of hypoglycemia in patients with T1D using CSII, as measured by the following: HbA1c Time in range (TIR), 70–180 mg/dL Time below range (TBR), <70 mg/dL Time above range (TAR), >180 mg/dL QoL, as measured by the DQoL questionnaire Diabetes Satisfaction, as measured by DTSQ Risk of hypoglycemia measured with Clarke’s test
Secondary objectives
To evaluate, first, the effects of initiating treatment with CSII and the Mylife Dose dosing application and, second, the outcomes after transitioning to the HCLS. These included the following: Changes in glucometry: Mean blood glucose, TIR, TBR, TAR, glucose management indicator (GMI), and coefficient of variation (CV). Changes in weight and BMI Changes in insulin requirements: Total daily insulin per kg of body weight, daily basal insulin dose, and prandial daily insulin dose. Changes in QoL as measured by the DQoL questionnaire Changes in satisfaction with diabetes care as measured by DTSQ. Changes in hypoglycemia perception as measured by Clarke’s test.
Statistical analysis
The required sample size to detect a mean difference of 0.5% in HbA1c with a standard deviation (SD) of 1.2, a significance level of 0.05, and a power of 80% was 54 participants. Up to 136 participants were recruited.
Summary data are reported as mean ± SD and interquartile range for continuous variables and as frequency and percentage for categorical variables. Between-group differences were compared using chi-square tests for categorical variables, whereas continuous outcomes were analyzed using repeated-measures linear mixed-effects models. An analysis of variance for repeated measures with a Greenhouse–Geisser correction was used to compare continuous data across the four visits.
A linear model was used to assess differences in HbA1c across visits. The adjusted models included age, sex, BMI, and disease evolution as covariates. All analyses were conducted using SPSS 27.0 statistical software, with statistical significance set at P < 0.05.
All data generated or analyzed during this study are included in the published article (and its online Supplementary Data).
Results
A total of 135 patients were recruited from 25 Spanish university hospitals (Supplementary Table S2), with 1 patient excluded due to a recent critical illness. The cohort consisted of 89 females and 46 males, aged 14–66 years, with a median diabetes duration of 17.5 years. Patients’ baseline characteristics are presented in Table 1.
Baseline Characteristics
Mean ± standard deviation (range).
BMI, body mass index; DPP4i, dipeptidyl peptidase-4 inhibitors; HbA1c, glycated hemoglobin; HDL, high-density lipoprotein; LDL, low-density lipoprotein; SGLT2i, sodium-glucose cotransporter-2 inhibitors.
After Visit 1, 6 patients (4.5%) withdrew from the study, 7 patients (5.2%) left after Visit 2, and 3 patients (2.2%) left after Visit 3. A total of 118 patients (88.1%) completed the study, resulting in a dropout rate of 11.9%. There were 128 active patients at Visit 2, 121 at Visit 3, and 118 at the final visit.
Glycemic control
Glycemic control improved after the implantation of the CSII, as evidenced by the following outcomes at Visit 2: HbA1c decreased from 7.57% to 7.42% (−0.15 ± 0.76%; P < 0.05), the percentage of patients with HbA1c < 7% increased from 29.1% to 38.5% (P < 0.01), and there were no changes in the percentage of patients with HbA1c <6.5% (25.4% vs. 23.4%; P = NS) (Fig. 1). This improvement was consistent across subgroups stratified by sex, age, BMI, and disease duration (Supplementary Table S3).
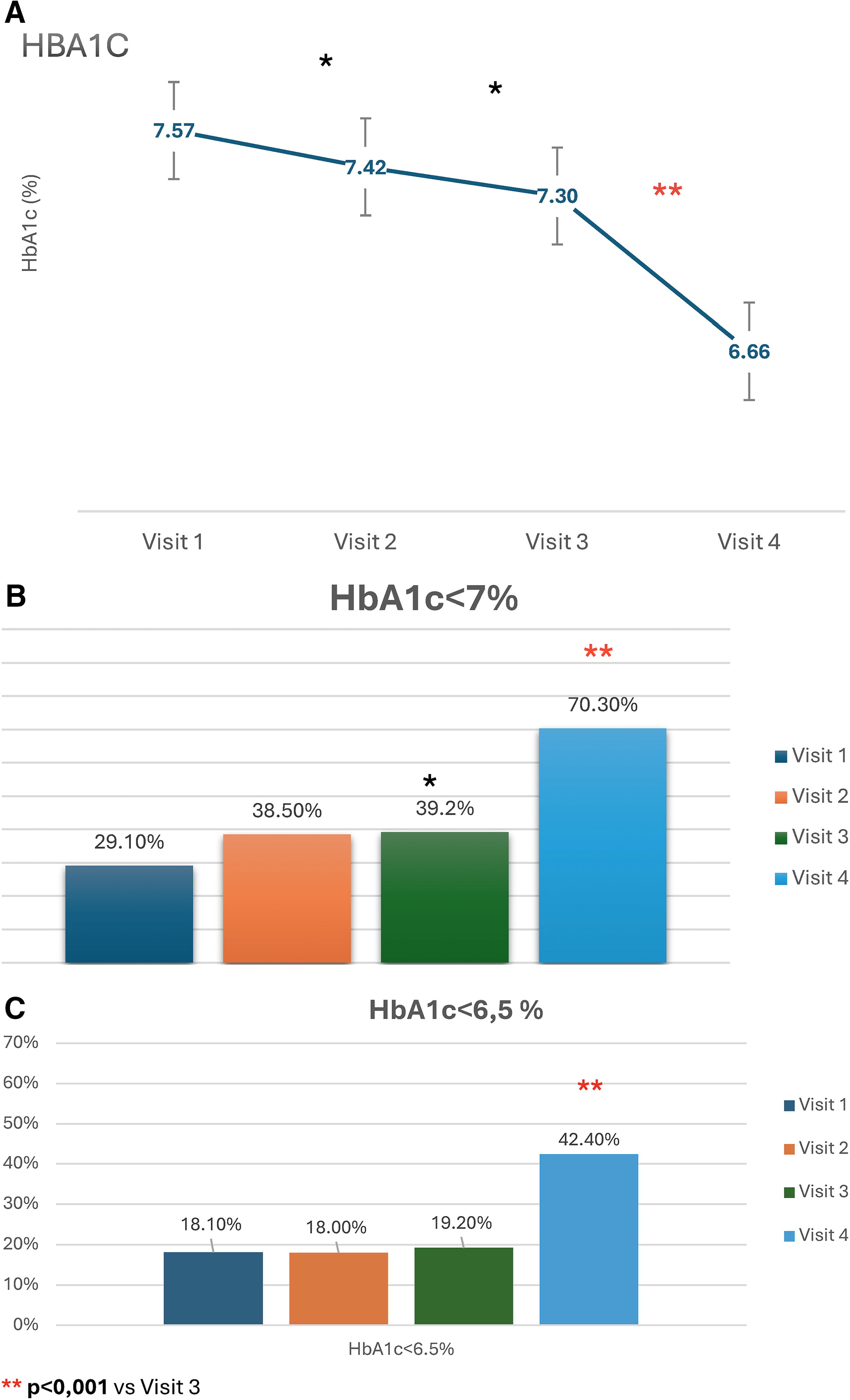
HbA1c evolution. Visit 1 baseline. Visit 2 90 days post Ypsomed insulin pump (YpsoPump). Visit 3: 90 days post Mylife Dose application. Visit 4 90 days post Mylife Loop hybrid system.
The mean glucose levels decreased from 171.8 mg/dL to 166.2 mg/dL (−0.56 ± 25.23 mg/dL; P < 0.05), and TIR increased from 56.3% to 60.4% (+4.1 ± 12.8%; P < 0.01) (Table 2). These improvements were sustained with the use of the MyLife Dose application; however, no further improvements were observed in any parameters at Visit 3.
Clinical, Laboratory, and Glucometric Evolution Data
P < 0.05 vs. V1.
P < 0.01 vs. V3.
CV, coefficient of variation; GMI, glucose management indicator; SD, standard deviation; TAR, time above range; TBR, time below range; TIR, time in range.
After transitioning to the HCLSs, glycemic control improved further by Visit 4, with a significant reduction in mean HbA1c to 6.66% (HbA1c Visit 3 7.30 ± 0.92 vs. HbA1c Visit 4 6.66 ± 0.76%; P < 0.01), an increase in the patients with HbA1c < 7% of 70.3% (percentage of patients with HbA1c < 7% Visit 3: 39.2% vs. Visit 4 70.3; P < 0.01), an increase in TIR to 73.1% (TIR Visit 3 60.92 ± 16.51 vs. Visit 4 73.11 ± 12.88; P < 0.01), and notable reductions in TAR, TBR, GMI, and CV (Table 2 and Fig. 2). By the end of the study 42.4% achieved an HbA1c < 6.5% (Table 2 and Fig. 1). These changes occurred regardless of sex, age, basal BMI, or years of disease evolution (Supplementary Table S5).
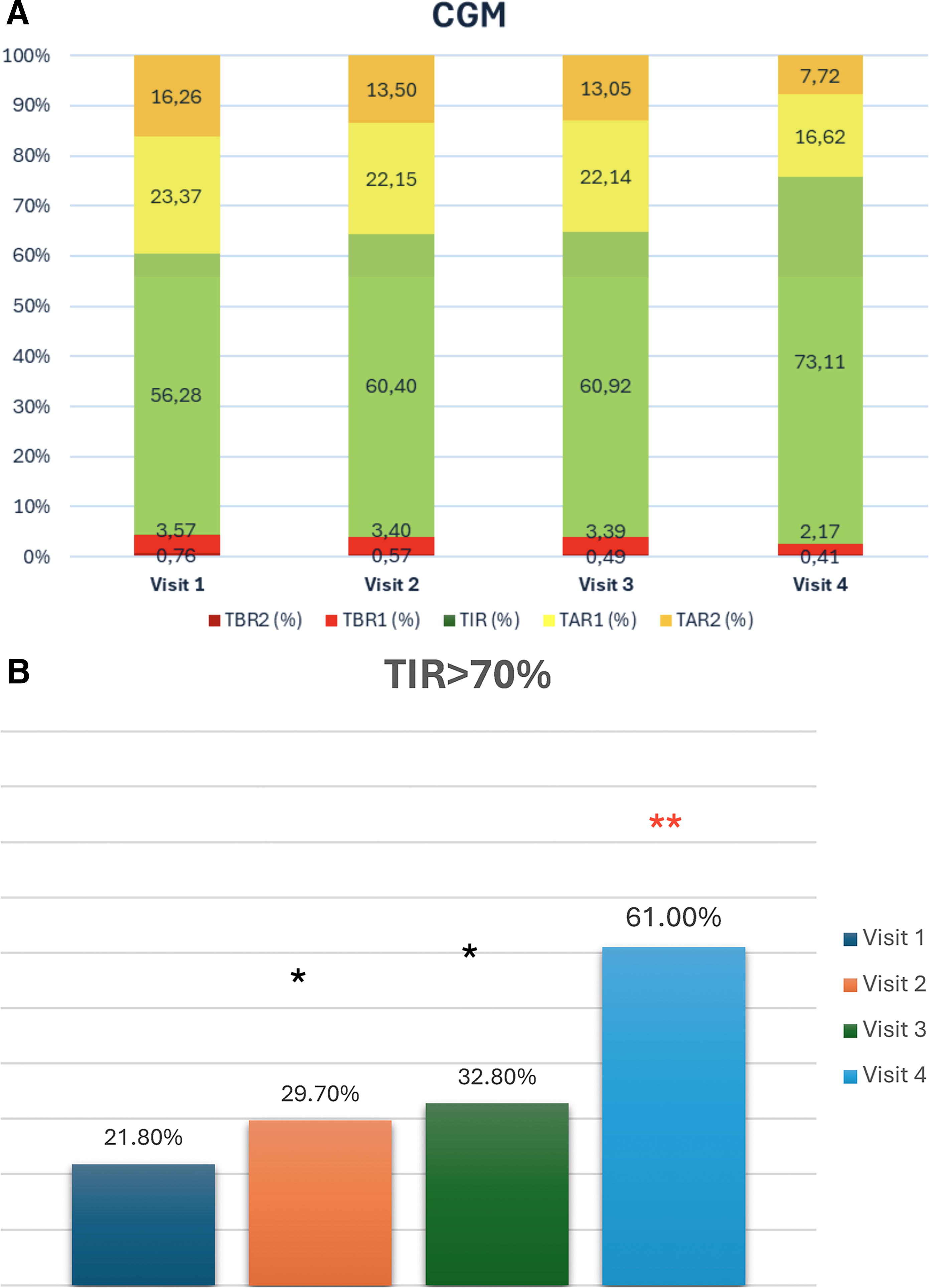
CGM results
Body weight/BMI
In our sample, over 20% of participants were classified as obese and more than 35% were overweight (Table 1). After starting treatment with CSII, no changes in weight were observed. However, switching to the automatic system was associated with a significant weight gain. This weight gain was similar in men and women.
Insulin requirements
Regarding insulin requirements, there were no changes in the total daily insulin dose administered from T1 to T3, which remained at 0.58 IU/kg of body weight. However, a decrease was observed in the basal-to-prandial insulin ratio, with a reduction in the proportion of basal insulin. By Visit 4, an increase in the total daily insulin dose was noted, primarily driven by an increase in basal insulin (9.78%), whereas prandial insulin decreased (Table 2).
Quality of life
Patients’ QoL, as measured by the DQoL, improved during the first 3 months of insulin pump treatment, with the total score increasing from 65.2 to 68.9 points, primarily driven by improvements in the satisfaction section. No further changes were observed following the use of the MyLife Dose application. However, after transitioning to the HCLS, a new improvement was observed, with the total score reaching 73.2 points. This enhancement reflected gains not only in the satisfaction section but also in the impact and diabetes-specific sections of the questionnaire (Table 3).
Diabetes Quality of Life
P < 0.05 vs. V1.
P < 0.05 vs. V3.
P < 0.01 vs. V3.
DQoL, Diabetes Quality of Life.
Satisfaction with the treatment and perception of hypoglycemia
The total DTSQ score increased following the initiation of CSII therapy, improving from Visit 1 to Visit 2. No significant changes were observed after introducing the smartphone application at Visit 3. However, a significant improvement was recorded 3 months after transitioning to the hybrid system at Visit 4 (Table 4). Results from the Clarke’s test showed an increase in the number of patients who regained hypoglycemia perception after Visit 3, with no further changes following HCLS (Table 5).
Diabetes Treatment Satisfaction Questionnaire
P < 0.05 vs. V1.
P < 0.05 vs. V3.
P < 0.01 vs. V3.
DTSQ, Diabetes Treatment Satisfaction Questionnaire.
Clarke’s Test
P < 0.05 vs. V1.
Adverse events
In our study, 85.8% of patients did not experience any AEs. Among the reported AEs, the most common were related to metabolic or nutritional disorders and product-related issues; however, all were mild in nature. There was a total of six cases of severe AEs, which included diabetic ketoacidosis (3 cases), hyperglycemia (2 cases), and hypoglycemia (1 case) (Supplementary Table S4a and S4b).
Discussion
Over the past few years, technological developments have transformed insulin administration and blood glucose management in the daily lives of individuals with T1D. 13 On one hand, CSII has transformed insulin administration, 14 reducing hypoglycemia and improving both glycemic control 15 and QoL. 16,17 However, the number of patients using CSII remains relatively low in our country. 1 On the other hand, continuous glucose measurement with modern interstitial glucose sensors has demonstrated improvements in glycemic control, as measured by HbA1c, while also reducing hypoglycemic episodes and enhancing the QoL for patients with T1D. 17,18 Due to their lower cost, these devices are widely financed for all T1D individuals in many developed countries, including Spain.
Automatic insulin infusion systems, also known as HCLSs, which combine CGM with a CSII and a control algorithm that automates insulin delivery, have been available since the early 2010s. These systems have proven effective at improving blood glucose control and QoL, albeit at a higher cost. 19 More recently, mobile phone applications have been developed to connect with insulin pumps and CGM systems. 20 These apps enable data sharing, insulin dose calculations, and the transmission of commands to the insulin pump, eliminating the need for the individual to interact with the device directly. 20
To date, few studies have evaluated the outcomes of patients already using CGM after transitioning to a CSII system. A systematic review and meta-analysis 21 found that real-time continuous glucose monitoring (RT-CGM) combined with MDI had a similar impact on HbA1c, weight, insulin usage, and TIR compared with RT-CGM combined with an open-loop CSII system. Furthermore, RT-CGM with CSII was associated with higher costs and a decrease in QoL. However, in our study, transitioning from MDI to CSII in patients already using continuous glucose sensors resulted in a modest but significant improvement in HbA1c and TIR, despite the participants having already acceptable baseline HbA1c levels (7.57%). In addition, the transition led to improved QoL, as measured by the DQoL questionnaire, and greater satisfaction with treatment, as assessed by the DTSQ. We found no differences according to gender, BMI, or years of disease evolution when we looked at subgroups.
In our study, the main objective was to evaluate the evolution of glucose control and QoL in the same group of T1D adults in a sequential manner after using a specific smartphone application (MyLife Dose app). Although there was a slight improvement of 0.12% in HbA1c, this difference was not statistically significant. Similarly, no improvements were observed in any CGM glucometric parameters, QoL as measured by the DQoL questionnaire, satisfaction with treatment, or perception of hypoglycemia. Therefore, we conclude that the use of the MyLife Dose app does not provide a statistically significant impact on glucose control or QoL.
As observed in previous studies 22,23 and meta-analyses, 24,25 this multicenter real-life study with patients with T1D shows that transitioning from an open-loop CSII + CGM system to an HCLS results in significant improvement both in glycemic control compared with previous treatment with a 0.5% improvement in HbA1c and in QoL. However, the transition does not lead to improvements in treatment satisfaction or perception of hypoglycemia.
With the closed-loop system, we also observed an increase in body weight and total daily insulin dose, consistent with findings reported by other researchers. 22
It is important to note that this weight gain can present a new challenge for patients with T1D. In our sample, at the start of the study, more than half of the patients were overweight and obese. Considering this, other strategies associated with these closed-loop systems may be explored to address this major problem.
Insulin pumps reduce the subcutaneous insulin depot, potentially increasing the risk of ketoacidosis and hyperglycemic complications. In our study 5 patients were affected with 2 cases of severe complications due to ketoacidosis requiring hospitalization—a slightly higher percentage compared with other studies. 26 It is evident that the occurrence of ketoacidosis has increased among the patient cohort. A potential explanation for this phenomenon could be the utilization of insulin pump technology by all patients, a novel approach for which they had not previously been exposed. However, further research is required to elucidate these findings.
A potential study limitation could be the relatively short follow-up period (3 months between each visit). However, previous research has shown that improvements in metabolic control achieved within this time frame are generally sustained over time. 27,28 In addition, the dropout rate was low, with only a few cases due to treatment-related complications.
In conclusion, introducing a CSII in patients with T1D already using isCGM results in an improvement in metabolic control and QoL. The introduction of a mobile phone application for CSII management (Mylife Dose) did not add any benefit to metabolic control or QoL. In contrast, transitioning from isCGM + CSII to HCLS (CamAPS) leads to significant improvements in glycemic control, TIR, and glycemic variability while also enhancing QoL, treatment satisfaction, and perception of hypoglycemia.
Conclusions
This study suggests that this technology has the potential to be useful in the management of adult patients with T1D. However, the use of a mobile phone application for insulin delivery (Mylife Dose) did not provide a significant benefit in metabolic control and QoL for people with T1D. In the future, the development of innovative applications designed to facilitate patient decision-making may help these patients to improve their glycemic control and QoL.
Footnotes
Acknowledgments
The authors thank the patients who participated in the study and the research staff at the investigational centers.
Authors’ Contributions
P.P.A. and E.L.M.T. performed the protocol preparation, systematic search, data extraction, and statistical analyses. All authors were involved in data interpretation, article writing, and editing. All authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
Funding for the development of this article was provided by Ypsomed Spain.
Supplementary Material
Supplementary Data
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
