Abstract
Background
Despite proposed aetiological mechanisms involving stress-induced inflammation and behaviour, population-level evidence linking social isolation and loneliness to asthma is lacking. Therefore, we aimed to examine the associations of social isolation, loneliness, and genetic predisposition with asthma risk in middle-aged and older adults, and to quantify the contribution of various risk factors.
Methods
A total of 400,184 asthma-free participants were enrolled from the UK Biobank. Loneliness was evaluated using the short form of the Revised UCLA Loneliness Scale, and social isolation was assessed through a composite score based on key social network components. Asthma was evaluated using hospital inpatient records in UK Biobank. Genetic predisposition for asthma was characterized using weighted polygenic risk score (PRS). Cox proportional hazard models were utilized to assess the associations of social isolation and loneliness with the risk of asthma. The one-sample Mendelian randomization (MR) analysis was based on genome-wide association studies of UKB.
Results
Over an 11.6-years median follow-up, a total of 9453 new asthma cases were recorded. In the fully adjusted model, individuals with feelings of loneliness had a 1.27-fold higher risk of asthma (95% CI: 1.15–1.39) than those without. However, social isolation showed no significant association with asthma risk after several factor adjustments. No significant interactions of loneliness and asthma-PRS were detected. In the one-sample MR analyses, we observed suggestive genetic evidence supporting associations of feeling lonely, fewer leisure social activities, being willing to confide in others, and participating in more other group activities with asthma.
Conclusions
Our findings suggest that loneliness, but not social isolation is a risk factor for asthma that is independent of various factors and genetic predisposition. Mendelian randomization provides suggestive evidence that loneliness and reduced social activity may be causal risk factors for asthma.
Highlights
(1) Loneliness increases adult-onset asthma risk by 27%, independent of social isolation and genetics. (2) Mendelian randomization confirms loneliness and reduced social activity as causal risk factors for asthma. (3) Participation in sports clubs or gyms lowers asthma risk, highlighting social activity benefits. (4) Over half of loneliness-asthma risk is explained by shared factors like psychological and socioeconomic status. (5) With 400,184 participants and an 11.6-years follow-up, this study features methodological advantages that enhance the credibility of its findings.
Introduction
Asthma is a common chronic non-communicable disease, affects around 300 million individuals globally and gives rise to approximately 455,000 annual fatalities.1,2 The prevalence of asthma symptoms is around 10% in children and adolescents and 6–7% in adults.3–5 Although childhood wheezing is very common, the majority of asthma has its onset in adulthood, there is up to a 60% chance of remission of asthma in children, but only 5–15% for adult-onset asthma (AOA).6,7 Moreover, AOA tends to be more complex and associated with a wider range of risk factors than childhood asthma,8,9 making the management and treatment of AOA more challenging.
Growing efforts identify modifiable factors for preventing or delaying AOA, with established links to, 10 environmental factors, 11 and mental health. 12 Social isolation, characterized by disengagement from social ties, institutional connections, or community participation, is recognized as a significant risk factor for mental health problems. 13 Loneliness is a common mental health issue, which is defined as a subjective emotional state or a painful feeling of social isolation. 14 Both social isolation and loneliness can lead to poor dietary habits, failure of smoking cessation, weakening immune function, and depressive symptoms, which in turn contribute to systemic inflammation, metabolic dysregulation, and impaired immune function.15–17 Furthermore, the psychological distress associated with loneliness often manifests as depressive symptoms and chronic stress, both known to exacerbate airway inflammation and bronchoconstriction.18,19 Supporting evidence revealed that social isolation and loneliness were associated with risks of dementia, diabetes, cardiovascular disease (CVD), a variety of cancers, and mortality.20–23 However, no study has explored the associations of social isolation and loneliness with the risk of AOA, especially among middle-aged and older adults, who have the highest rates of asthma incidence and mortality.
Given asthma is influenced by the synergistic effects of genetic and environmental factors, 24 it is essential to account for the moderating effects of these additional factors when considering the association of social isolation and loneliness with the risk of AOA. Mendelian randomization (MR), an analytical method using the results of genome-wide association studies (GWAS), has the potential to inform our understanding of causal effects by considering or avoiding the possibility of a variety of confounding processes and reverse causality. 25 The application of MR can help establish a causal relationship between social isolation, loneliness, and AOA risk.
Therefore, in this analysis using the UK Biobank study, a very large prospective population-based cohort study, we aimed to (1) examine the associations of social isolation and loneliness with first AOA; (2) calculate the percentage of excess risk mediated (PERM) to quantify the individual contributions of various confounding factors to the associations between social isolation and loneliness with AOA; (3) further explore whether social isolation and loneliness can increase the risk of AOA related to genetic factors; and (4) conduct one-sample MR analysis to address causality of the associations.
Materials and methods
Participants
This study utilized data from the UK Biobank, a prospective cohort of >500,000 participants aged 40–69 recruited from 22 UK assessment centers since 2006. Baseline data collection included comprehensive demographic, health, and lifestyle information obtained through interviews, touch-screen questionnaires, and physical examinations. 26 Ethical approval was granted by the NHS Northwest Research Ethics Committee (REC reference: 21/NW/0157), with full study protocols available at: https://biobank.ndph.ox.ac.uk/ukb/index.cgi. The final analytic sample included 400,184 participants, as depicted in Supplemental Method and Figure S1.
Cohort study
Defining social isolation and loneliness
Social isolation was assessed using a validated three-item scale (UK Biobank protocol; similar to prior UK studies 27 ), evaluating: (1) living situation, (2) family/friend contact frequency, and (3) leisure/social activity participation. Loneliness was measured via a two-item scale adapted from the UCLA Loneliness Scale, 28 inquiring about loneliness frequency and confiding in friends/family. For joint effect and sensitivity analyses, both measures were categorized into three-level variables (definitions in the Supplemental Method and Table S1).
Definition of AOA and polygenic risk score for asthma
The diagnosis of AOA was determined based on data from hospital admission records, and death records. We used the International Classification of Diseases (version 10; code ICD-10) coding system to record diagnoses. ICD-10 codes used for AOA included J45, J46, J450, J451, J458, and J459. In this study, data for hospital admissions and death registrations were updated to September 20, 2021 for England, February 28, 2018 for Wales, and July 31, 2021 for Scotland.
This study explored the genetic associations between social isolation, loneliness, and AOA risk using the standardized asthma Polygenic Risk Score (PRS) from the UK Biobank. The PRS is generated using Bayesian methods based on meta-analysis and summary statistics from the GWAS database. A higher PRS indicates a higher genetic predisposition to developing asthma. To facilitate subsequent analysis, asthma-PRS scores were categorized by tertiles: low, moderate, or high genetic risk.
Assessment of covariates
For this study, we included covariates including sociodemographic characteristics, lifestyle, body mass index (BMI), personal disease histories, and living environment. These covariates were obtained from standardized questionnaires and physical examinations at the baseline visit by trained staff. Detailed definitions and field IDs can be found in the Supplemental Method, Table S2 and Table S3, respectively.
Statistical analysis
Descriptive statistics appropriate to variable distributions were used to summarize baseline characteristics of the social isolation and loneliness states, respectively. Continuous variables were assessed for normality via Kolmogorov-Smirnov tests: normally distributed variables were reported as mean and standard deviation (SD), non-normal variables as median (IQR). Categorical variables were described using frequencies and percentages (n [%]). The follow-up period was calculated from the date of recruitment until the occurrence of an AOA diagnosis, death, or end of follow-up (December 21, 2022), whichever event occurred first.
Cox proportional hazards regression models estimated hazard ratios (HRs) with 95% confidence intervals (CIs) for social isolation/loneliness associations with AOA risk. Models were sequentially adjusted: Model 1 was a crude model; Model 2 adjusted for sex, age, and ethnicity; Model 3 further adjusted for education attainment, TDI, smoking status, alcohol consumption, regular physical activity, healthy diet score, healthy sleep patterns, BMI, disease history (hypertension, diabetes, and hyperlipidemia), secondhand smoke, PM2.5 and depressive symptoms; Model 4 adjusted for social isolation and loneliness mutually. All time-to-event analyses were performed with the use of Kaplan-Meier (K-M) methods and presented by K-M curves. The proportional hazards assumption was tested by the Schoenfeld residuals test, no significant deviations were observed. We assessed the linearity of all continuous covariates by examining Martingale residual plots and using likelihood ratio tests to compare models with linear terms to those with restricted cubic splines. No significant nonlinearity was detected (all p > 0.05); Thus, continuous variables were retained as linear terms in the final models. The multicollinearity among the covariates included in the model was evaluated by calculating the variance inflation factor (VIF). All VIF values were less than 3, indicating that there was no multicollinearity problem. Furthermore, the HRs for the primary exposures (loneliness and social isolation) remained stable across nested models with progressively added covariate sets, indicating that correlations among adjustments did not materially affect the core estimates. Missing covariate data were handled using multiple imputation by chained equations with the mice package in R. The imputation model included all analysis variables. We generated 10 imputed datasets (m = 10), a number sufficient to achieve stable estimates given the low overall fraction of missing information in our data. Cox models were fitted to each imputed dataset, and results were pooled using Rubin’s rules. The percentage of missing data for each variable is provided in Supplemental Method Table S4.
We calculated PERM to assess how much the associations of social isolation and loneliness with AOA weakened when the seven baseline explanatory variables were successively added to the model. PERM is calculated as follows:
Using Cox proportional hazards regression, we examined the association between an asthma-PRS and AOA risk, then evaluated genetic predisposition’s modifying effect on social isolation/loneliness-AOA associations by: (1) categorizing asthma-PRS into tertiles, (2) assessing isolation/loneliness-AOA relationships across PRS strata, and (3) incorporating asthma-PRS × isolation/loneliness interaction terms in Model 4; joint effects were assessed through nine groups stratified by PRS tertiles and isolation/loneliness categories, with hazard ratios (95% CIs) estimated relative to the lowest-risk group.
We conducted several secondary analyses to enhance the comprehensiveness of our study: (1) Stratification by sex, age, and ethnicity, with formal interaction testing by including exposure-by-covariate product terms in the Cox models. The statistical significance of effect modification was assessed using the p value for each interaction term (all p > 0.05). Stratified HRs are presented for descriptive illustration, and the primary statistical inference is based on these interaction tests rather than on multiple comparisons across subgroups; (2) Sensitivity analyses using 3-category isolation/loneliness variables; (3) Testing linear trends via continuous scores; (4) Excluding AOA cases occurring within 2 years of follow-up to address reverse causality.
All statistical analyses were conducted using the Statistical Analysis System 9.4 edition for Windows, provided by SAS Institute Inc. In Cary, NC, USA. Each statistical test was two-sided, and p < 0.05 was considered statistically significant.
Mendelian randomization study
In this study, we conducted one-sample Mendelian Randomization (MR) analyses to examine the causal effects of social isolation and loneliness, on the risk of AOA. The exposures in the MR analysis encompassed all aspects of social isolation and loneliness, utilizing instrumental variables derived from the UK Biobank genome-wide association studies (GWAS). Ethical approval, which included obtaining informed consent, was secured for all of the original studies.
Genetic instruments between social isolation and loneliness
A range of 4 to 23 single nucleotide polymorphisms (SNPs) were identified for each exposure trait. These SNPs were all independent of each other (i.e. linkage equilibrium) and were used to construct the genetic risk score (GRS). The GRS for each participant was calculated by summing the number of risk alleles and weighted by the corresponding effect size (β-coefficients) derived from the GWAS study. Detailed information about the SNPs included as genetic instruments can be found in the Supplemental Method and Table S5.
One-sample mendelian randomization
In the one-sample MR analysis, we utilized the two-stage least squares method to assess the relationships between genetically predicted social isolation, loneliness, and the risk of AOA. Both regression stages were adjusted for age, sex, the top 20 genetic principal components, assessment center, and genotyping chip. We conducted sensitivity analyses to assess instrument strength (using the F-statistic) and the robustness of estimates to measured confounding. Detailed calculation methods and variable adjustments are provided in the Supplemental Method.
Results
Cohort analyses
Baseline characteristics of participants
Participant characteristics by social isolation and loneliness at baseline in UK Biobank (n = 400,184). a
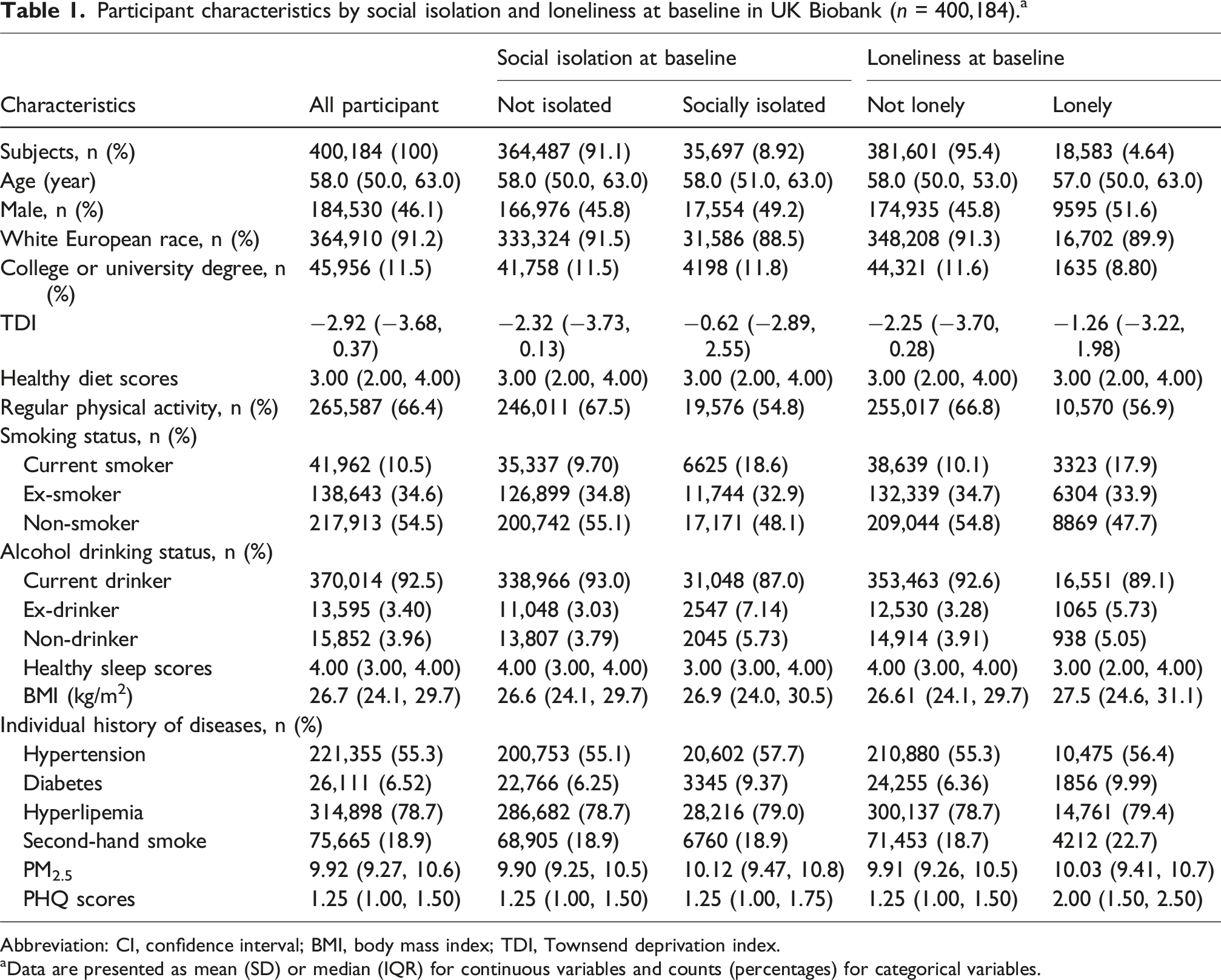
Abbreviation: CI, confidence interval; BMI, body mass index; TDI, Townsend deprivation index.
aData are presented as mean (SD) or median (IQR) for continuous variables and counts (percentages) for categorical variables.
Associations between isolation and loneliness with the risk of AOA
Hazard ratios for associations of social isolation and loneliness with incident adult-onset asthma in the UK Biobank. a

aAnalysis by Cox proportional hazards regression model.
bModel 1 was the crude model.
cHazard ratios (95% confidence interval) (all such values).
dModel 2 was adjusted for age, sex, and ethnicity.
eModel 3 was additionally adjusted for education attainment, Townsend deprivation index, smoking status, alcohol drinking status, regular physical activity, health diet scores, health sleep scores, body mass index, individual history of diseases (hypertension, diabetes, and hyperlipemia), inhalation of secondhand smoke, PM2.5 and depressive symptoms.
fModel 4 was further adjusted for loneliness (for social isolation as the exposure) or social isolation (for loneliness as the exposure).
The PERM for seven groups of risk factors
Figure 1 illustrates the proportionate contribution of different risk factors to the associations of social isolation (Panel A) and loneliness (Panel B) with AOA risk. Sequential adjustment for seven sets of covariates attenuated both associations: for social isolation, attenuation ranged from 9.60% to 57.2% (PERM), with socioeconomic factors contributing most significantly (57.2% attenuation; adjusted HR reduced to 1.07, 95% CI: 1.00-1.15); full adjustment abolished the association (p > 0.05). For loneliness, attenuation ranged from 5.40% to 26.1% (PERM), with psychological factors accounting for the largest proportion (26.1% attenuation; HR reduced to 1.49, 95% CI: 1.37-1.61); after full adjustment, the association remained significant but was attenuated by 60.0% (adjusted HR: 1.27, 95% CI: 1.15-1.37). Associations of social isolation (A)/loneliness (B) with incident all-cause risk of AOA and proportions attributable to different risk factors.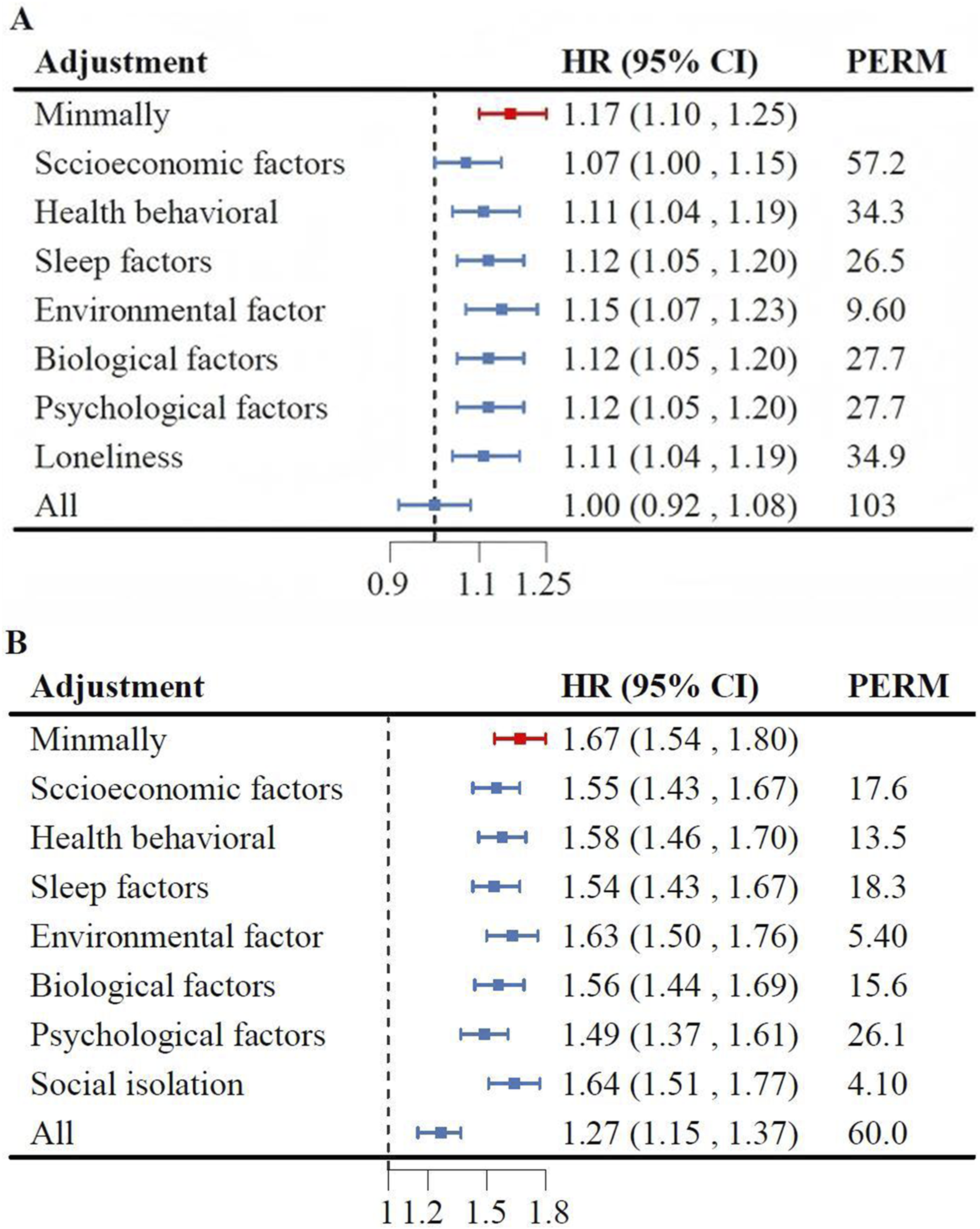
Association of social isolation/loneliness and asthma-PRS
The asthma-PRS demonstrated a significant positive dose-response relationship with AOA risk (p-trend <0.001; Table S6). Compared to the low-PRS group, intermediate and high PRS groups showed progressively higher AOA risk (HR 1.24, 95% CI 1.16–1.31 and HR 1.59, 95% CI 1.50–1.69, respectively). In the fully adjusted model, for every one standard deviation increase in the polygenic risk score, the risk of adult asthma onset increased by 25% (HR 1.25, 95% CI 1.22 - 1.28; Table S6). The fully adjusted model (Model 4) explained approximately 0.7% of the variance in AOA risk (Nagelkerke R2 = 0.007). Loneliness consistently increased AOA risk across all genetic susceptibility strata, while social isolation showed no association (Table S7), with no significant additive interactions (all p > 0.10). Figure 2 illustrates joint exposure effects: participants with high genetic predisposition and maximum isolation had HR 1.69 (95% CI 1.55–1.84) versus the low-risk reference group (low PRS + least isolation), whereas those with high genetic predisposition and maximum loneliness showed HR 2.06 (95% CI 1.77–2.41) versus the low-risk reference group (low PRS + least loneliness). Multi-adjusted hazards ratios (HRs) and 95% confidence interval (CI) of adult-onset asthma by joint effect of social isolation (A)/loneliness (B) and genetic predisposition. The analysis was performed in Model 4 (adjusted with age, sex, ethnicity, education attainment, Townsend deprivation index, smoking status, alcohol drinking status, regular physical activity, health diet scores, health sleep scores, body mass index, individual history of diseases [hypertension, diabetes, and hyperlipemia], secondhand smoke, PM2.5, depressive symptoms and social isolation/loneliness). Abbreviations: AOA, adult-onset asthma; HR, hazard ratio.
Secondary analyses
Subgroup analyses (Table S8) revealed no significant interactions between loneliness and sex, age, or ethnicity on AOA risk (all P-interaction >0.1). Sensitivity analyses categorizing participants by social isolation and loneliness scores revealed significant dose-response relationships with AOA risk (Figure S4). The loneliest group exhibited a significantly higher AOA risk than the least lonely group (fully adjusted HR: 1.31, 95% CI: 1.19–1.45), with each 1-point increase in loneliness score associated with a 13% elevated risk (Table S9). Excluding participants who developed AOA within the first 2 years of follow-up (Table S10) did not substantially alter the primary findings.
Mendelian randomization results
Genetic instruments for social isolation, loneliness and AOA
Genetic instruments for feeling lonely, being willing to confide in others, not living alone, contact with friends or family, fewer leisure social activities, engaging in more sports clubs or gyms, pubs or social clubs, religious activities, adult education classes, and other group activities explained 0.13%, 0.11%, 0.02%, 0.18%, 0.08%, 0.05%, 0.17%, 0.18%, 0.03% and 0.03% of the variance, respectively. The F-statistic was used to eliminate bias in the results caused by weak instrumental variables. The F-statistic is calculated as F = (N-K-1)/K *R2/(1-R2), K = n SNP merged, N = sample size in the discovery GWAS, and R2 reflects the degree to which the instrumental variable explains the exposure. In this study, genetic instrumentation showed strong associations with social isolation and loneliness, with F statistics greater than 10.
Causal effect of individual social isolation, loneliness on the risk of AOA
One-sample Mendelian randomization analyses revealed that feeling lonely (OR 1.18, 95% CI 1.07–1.30; p < 0.001) and fewer leisure social activities (OR 1.29, 95% CI 1.14–1.47; p < 0.001) increased AOA risk, whereas willingness to confide in others (OR 0.84, 95% CI 0.73–0.96; p = 0.01) and participation in group activities (OR 0.56, 95% CI 0.35–0.91; p = 0.02) reduced risk (Figure 3). Sensitivity analyses adjusting for confounders attenuated these associations: only fewer leisure social activities remained significantly associated with elevated AOA risk (OR 1.21, 95% CI 1.02–1.43; p = 0.03), with no causal relationships detected for other exposures. The limited explanatory power of genetic variants (0.03%–0.18% for loneliness/social isolation) suggests potential weak instrument bias affecting AOA risk estimates (Table S11). Mendelian randomization analyses for the causal effects of loneliness and social isolation on the risk of AOA by using two-stage least squares method based on GWAS of UKB. Forest plots show the odds ratio (OR) and 95% confidence interval (95% CI) for the associations of loneliness and social isolation with AOA. OR, odds ratio; 95% CI, 95% confidence interval; AOA, adult-onset asthma; GWAS, genome-wide association studies; UKB, the UK biobank.
Discussion
In this prospective cohort study, we explored the associations between social isolation, loneliness and AOA risk. Our results demonstrate that loneliness significantly increases AOA risk independently of social isolation. After full adjustment for seven groups of risk factors, the loneliness-AOA association remained statistically significant despite substantial attenuation (overall attenuation: 57.1%). Participants with social isolation/loneliness and high genetic risk had the highest risk of AOA, even though there was no statistically significant interaction between social isolation/loneliness and genetic susceptibility to AOA. The robustness of our results is further supported by a variety of MR methods and extensive sensitivity analyses, providing a comprehensive understanding of the impact of loneliness on AOA risk.
Previous studies have verified the detrimental impact of social isolation and loneliness on various chronic conditions, such as type 2 diabetes mellitus, myocardial infarction, stroke, later dementia, cardiovascular disease, and even mortality.22,29,30 Consistent with previous studies, we found that loneliness was associated with a 1.27 fold increased risk of developing AOA, which was independent of social isolation and other risk factors. Social isolation was not related to the incidence of AOA after removing all covariates including loneliness, and 57.2% of the relationship was attributable to socioeconomic factors. To our knowledge, this is the first prospective study to assess the associations of social isolation and loneliness with the risks of AOA.
To elucidate the potential causal pathways and guide our analytic approach, we propose an integrative framework. First, the chronic psychological stress of loneliness may exert a direct psycho-neuro-immune effect. Evidence suggests that such stress can dysregulate the hypothalamic-pituitary-adrenal axis, promoting systemic and airway inflammation, which may represent a key biological mechanism through which loneliness impacts health.31,32 Second, social isolation often leads to or coexists with intermediate psychological and behavioral factors. Our PERM results indicate that depressive symptoms play a role of approximately 26.1% in the association between loneliness and AOA, underscoring a significant psychological pathway. 33 Although correlated unhealthy behaviors (e.g., poor diet, inactivity) showed minimal confounding effect in our data, they remain confounding factors. Furthermore, our MR analysis suggested that structured social activities (e.g., sports clubs) are protective.34,35 Therefore, we treated variables such as depressive symptoms, health behaviors (smoking, alcohol, diet, physical activity, sleep), and BMI as key adjustment variables. Third, shared socio-environmental determinants could confound the observed relationship. Low socioeconomic status may simultaneously increase the risk of social isolation, limit healthcare access, and elevate exposure to environmental pollutants (e.g., PM2.5, secondhand smoke).16,17 Consequently, we adjusted for factors including education, TDI, PM2.5, and secondhand smoke exposure as potential confounders. Finally, demographic factors (age, sex, ethnicity) and comorbid conditions (hypertension, diabetes, hyperlipidemia) were adjusted for as standard epidemiological confounders related to both exposure and outcome. To disentangle their independent effects, loneliness and social isolation were also mutually adjusted in the model.
AOA is a complex heterogeneous disease caused by gene–environment interactions. Previous studies have identified a number of psychological factors that may be intertwined with genetic factors, such as anxiety, depression and post-traumatic stress disorder.36,37 However, in our study, we did not find significant interaction between social isolation/loneliness and asthma-PRS on the risk of AOA. This suggests that social isolation/loneliness and genetic factors may act independently on asthma risk.
As the first large-scale study to quantify risk factor contributions to associations between social isolation/loneliness and AOA, this research demonstrates that controlling conventional risk factors substantially attenuates loneliness-related AOA risk. Mendelian randomization analysis further provided suggestive evidence supporting causal links between these psychosocial factors and AOA. Strengths include the large sample size, longitudinal design, comprehensive sensitivity analyses, and robust statistical power—highlighting the imperative to integrate loneliness mitigation strategies into healthcare education. Nevertheless, certain constraints should be considered when interpreting our results. Firstly, the assessment of social isolation and loneliness relied on three-item and two-item questionnaires, respectively, rather than validated scales, which may introduce potential bias. Future research should utilize well-designed scales to validate our findings. Furthermore, participants in UK Biobank are often healthier, have higher socioeconomic status, and exhibit healthier lifestyle behaviors compared to the general population. 38 This selection bias may lead to an underrepresentation of individuals with severe health conditions or those most affected by social isolation and loneliness. Secondly, our study population was predominantly of White European ancestry (90%), which limits the generalizability of our findings to other racial and ethnic groups. Caution is therefore warranted when extrapolating to younger adults, non-European ancestries, or settings with different health-care and social-security systems. Thus, it is unclear if our results are generalizable to racial/ethnic minorities outside of the UK. Furthermore, the limited diversity implies that the specific risk estimates we report, and the performance of the genetic instruments used in our MR analysis, may not directly translate to other populations. These factors underscore that our findings primarily elucidate relationships within a specific demographic context and highlight the necessity for validation in more heterogeneous cohorts. Thirdly, risk factors such as socioeconomic status, health behaviors and biological factors were assessed through baseline self-report questionnaires without accounting for changes over time, potentially introducing recall bias and misclassification. Fourth, our MR analysis used a one-sample design within the UK Biobank. This violates the assumption of sample independence, limits standard pleiotropy checks, and risks bias from overfitting and the upward bias in genetic effect estimates inherent to using variants discovered in the same sample. Therefore, while the MR results provide suggestive evidence, causal interpretation requires caution, and generalizability to other ethnic groups may be limited. Fifth, although this study controlled for multiple potential confounders, residual confounding factors may still be present. Sixth, it should be noted that incident AOA cases in this study were ascertained through hospital inpatient and death registry data. While this ensures diagnostic rigor, it may not capture milder cases managed in primary or outpatient care, potentially leading to an underestimation of the total incidence. Consequently, our findings primarily reflect associations with more severe asthma outcomes that required hospitalization. Finally, the UK Biobank’s age restriction of 37-74 years limits the generalizability of our findings to other age groups. Future research across diverse populations is necessary to validate these findings.
Conclusion
The findings of this cohort study indicate that loneliness, rather than social isolation, is a risk factor for AOA after accounting for multiple confounders and genetic predisposition. One-sample MR Analysis confirmed the association and further revealed that increased social activity (particularly sports club/gym participation) reduced AOA risk. Notably, >50% of the loneliness-AOA risk association appeared attributable to shared modifiable factors, suggesting their management could mitigate loneliness-related AOA incidence.
Supplemental material
Supplemental material - Associations of social isolation and loneliness with the risk of adult-onset asthma: A prospective cohort and mendelian randomization study
Supplemental material for Associations of social isolation and loneliness with the risk of adult-onset asthma: A prospective cohort and mendelian randomization study by Juanjuan Zhang, Yuqian Li, Xiangdong Sun, Ruiqi An, Hongjie Zhang, Jianping Gong, Yuanda Zhang, Yijue Li, Yanfeng Gu, Yang Xia in Chronic Respiratory Disease
Footnotes
Acknowledgments
The authors gratefully acknowledge all the people that have made this study.
Ethical considerations
This study was approved by the National Information Governance Board for Health and Social Care and the National Health Service North West Multicenter Research Ethics Committee (REC reference: 21/NW/0157).
Consent to participate
The privacy rights of human subjects have been observed and that informed consent was obtained for experimentation with human subjects.
Author contributions
Study concept and design: J.Z., Y.X. Acquisition, analysis, or interpretation of data: J.Z., Y.L, J.G. Drafting of the manuscript: X.S., R.A., J.Z. Critical revision of the manuscript for important intellectual content: J.Z., Y.L. Statistical analysis: J.Z., Y.L, R.A, H.Z. Visualization: Y.X. Obtained funding: Z.J., Y.G. Administrative, technical, or material support: J.G., Y.Z. Study supervision: Y.G., Y.X. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Science Research Project of Hebei Education Department (grant number QN2025221 to Juanjuan Zhang).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

