Abstract
Introduction
Coronavirus disease (COVID-19) can reduce respiratory muscle function and functional capacity. 1 This can be through direct action of the virus (severe acute respiratory syndrome coronavirus-2) in the respiratory muscles causing muscle cell damage, inflammation and impaired muscle function2,3 and the resultant respiratory failure and increased work of breathing may necessitate mechanical ventilation. 4 Mechanical ventilation, while a life-saving intervention in patients with severe respiratory failure, 5 may also lead to disuse atrophy, weakness and damage to the respiratory muscles.6,7 This can reduce respiratory function and functional capacity after the patient is weaned from mechanical ventilation.6,7 COVID-19 causes a distinct pattern of lung damage, characterized by diffuse alveolar damage and impaired gas exchange.2,3 This unique pathology leads to persistent respiratory impairments, including reduced lung compliance, decreased inspiratory muscle strength, and impaired gas exchange efficiency, even after patients have been weaned from mechanical ventilation.2,3,8
As the respiratory muscles play an essential role in ventilation, damage to these muscles may have serious consequences and can lead to respiratory muscle dysfunction.9,10 This dysfunction can manifest as dyspnea and exercise intolerance through to ventilatory failure. 10 Previous studies have shown that the amount of respiratory muscle damage can be assessed by measuring concentrations of specific skeletal muscle damage biomarkers including creatine kinase muscle type (CKM), and slow and fast skeletal troponin I (sTnI) isoforms.11,12
Inspiratory muscle training (IMT) may help to restore muscle coordination lost during mechanical ventilation, by improving motor unit synchronization, resulting in more effective and efficient respiratory muscle function.13,14 This improved efficiency following training may result in lower concentrations of biomarkers of muscle damage. There is some evidence that 2 weeks of IMT may improve pulmonary function, dyspnea, functional capacity and Quality of life (QoL) in recovered COVID-19 patients after weaning from mechanical ventilation. 15 However, a placebo or sham IMT group was not used in this study which weakens internal validity and risks participant bias. Whether IMT compared to a sham IMT control group could reduce biomarkers of muscle damage and improve respiratory function and functional capacity in recovered COVID-19 patients after weaning from mechanical ventilation is unknown.
Accordingly, we investigated the effects of IMT on biomarkers of muscle damage, respiratory function and functional capacity in recovered COVID-19 patients after weaning from mechanical ventilation. We hypothesized that IMT would reduce muscle damage and improve respiratory function and functional capacity compared to a control group using sham IMT.
Methods
Participants
Anthropometrics and respiratory function for the inspiratory muscle training (IMT) and control (CON) groups at baseline. Values are mean ± SD.

FVC, forced vital capacity; FEV1, forced expiratory volume in 1 s; PMmax, maximal inspiratory mouth pressure.
Experimental design
The study adopted a single-blind, randomized controlled design. Participants attended the physiotherapy department of University of Lahore Teaching Hospital on four separate visits. All the participants were outpatients at the time of recruitment. The first was a screening visit where participants completed the Exercise and Sports Science Australia Adult Pre-Exercise Screening System. 16 Subsequently, participants provided informed consent and height, body mass, pulmonary function, and maximal inspiratory mouth pressure (PMmax) were assessed. Participants were then randomly, and equally divided into either an IMT or control (CON) group using Altman’s minimization method (prioritizing BMI and sex) to ensure that the groups were balanced. 17 During the second visit, participants had a blood sample taken and undertook questionnaires for dyspnea using the MRC Dyspnea Scale 18 and QoL, and undertook a six-minute walk test (6MWT), grip strength test and sit-to-stand test and were familiarized with the IMT or CON interventions. During visits three and four, participants repeated same assessments as in visit two. Visits one and two were separated by 24 h and visits two, three and four by 2 weeks.
Anthropometrics
Participants’ height was measured to the nearest 1 cm using a wall-mounted telescopic stadiometer (Seca220; Vogel & Halke, Hamburg, Germany) and body mass to the nearest 100 g using an electronic scale (Tanita Ultimate Scale 2000; Tanita, Tokyo, Japan).
Respiratory function
Participants performed standardized forced vital capacity (FVC) maneuvers using a spirometer (Spirolab; Medical International Research, New Berlin, WI, USA) according to published guidelines. 19 PMmax was assessed using KH2 model IMT device (POWERbreathe International Ltd®, Warwickshire, UK) whilst participants were seated and wearing a nose clip and was initiated from residual volume. Repeat efforts were separated by 30 s and were performed until three serial measures differed by no more than 10% or 10 cmH2O, whichever was smallest. 20 Participants performed a minimum of five and a maximum of 10 efforts. The average of three highest values recorded were used for subsequent analysis. Predicted values for pulmonary volumes and capacities are from Quanjer et al. 21 and for PMmax are from Wilson et al. 22
Quality of life, handgrip strength, sit to stand and six-minute walk test
QoL was assessed utilizing the Euro Quality 5-Dimensions-3Levels (EQ-5D-3 L) questionnaire.23,24 Handgrip strength was determined using hand dynamometry (Jamar Digital Plus; Lafayette Instruments, Lafayette, IN, USA) as previously described. 25 One-minute sit to stand testing was performed with a chair of standard height and without armrests. The participant was seated upright on the chair positioned against a wall. The participants were required to get up from this chair with the legs straight and sit back down whilst continuing the repetitions as fast as possible within 1 min. 26 Exercise performance was assessed by using a 6MWT according to published guidelines.27,28 Predicted values for the 6MWT are from Rao et al. 29
Blood sampling and enzyme-linked immunosorbent assays
Twenty mL of venous blood was collected at each time point from an antecubital vein using a suitable method (either evacuated tube system or winged infusion) into serum separator tubes (BD Vacutainer® SST™ Tubes; Franklin Lakes, NJ, USA). The serum was separated, aliquoted and stored at −80°C until biochemical assays were performed. Enzyme-linked immunosorbent assays were performed for serum biomarkers using commercially available kits: Human CKM (Catalog No. RK01117 Abclonal, Woburn MA, USA); Fast sTnI (Catalog No. RK02421 Abclonal, Woburn MA, USA); and Slow sTnI (Catalog No. RK02420 Abclonal, Woburn MA, USA). ELISAs were performed by following manufacturers’ instructions for each specific kit. To minimize the effect of inter-assay variation, markers from both the IMT and CON groups were measured using the same assay plate.
Inspiratory muscle training and control interventions
The intervention lasted 4 weeks. The IMT and CON group were told they were a part of ‘respiratory muscle strength training’ OR ‘respiratory muscle endurance training’ intervention respectively. Both IMT and CON groups performed their respective training with an inspiratory pressure-threshold device (POWERbreathe Classic series 1st generation; Gaiam Ltd, Southam, UK). The IMT group performed 30 consecutive dynamic inspiratory efforts twice daily with initial training load set at 50% PMmax. Thereafter, participants were instructed to periodically increase the load so that 30 maneuvers could only just be completed. Each inspiratory effort was to be initiated from residual volume, and participants were asked to strive to maximize tidal volume. This regimen is known to be effective in eliciting an adaptive response.30–32 The CON group trained at a fixed intensity of 10% of PMmax once per day for a total of 60 repetitions, 5 days/week. Both groups performed one supervised session at the start of intervention and during week two of intervention to monitor the appropriateness of their training technique and intensity. Compliance was assessed using a register of completed sessions.
Statistical analysis
Statistical analyses were performed using SPSS 25 for Windows (IBM, Chicago, IL, USA). An initial power calculation was performed on the basis of our previous work, 11 showing that twelve participants would be required to demonstrate a 10% reduction in sTnI with an alpha of 0.05. Normality of the data was assessed by visual inspection of histograms. The data from both groups were analyzed using a two-way analysis of variance (ANOVA) procedure to determine the effects of ‘time’ (baseline, two and 4 weeks) and ‘group’ (IMT vs CON). Following significant interaction effects, pairwise comparisons were made using the Bonferroni method (adjustments for multiple comparisons). Statistical significance was set at p < 0.05. Results are presented as means ± SD.
Results
Participant characteristics
Figure 1 shows the CONSORT participant flow diagram. Compliance with the interventions was good with 78 ± 4% and 91 ± 5% of the IMT and CON sessions completed, respectively. There were no differences in baseline participant characteristics between the groups (Table 1). The CONSORT flow diagram of the study.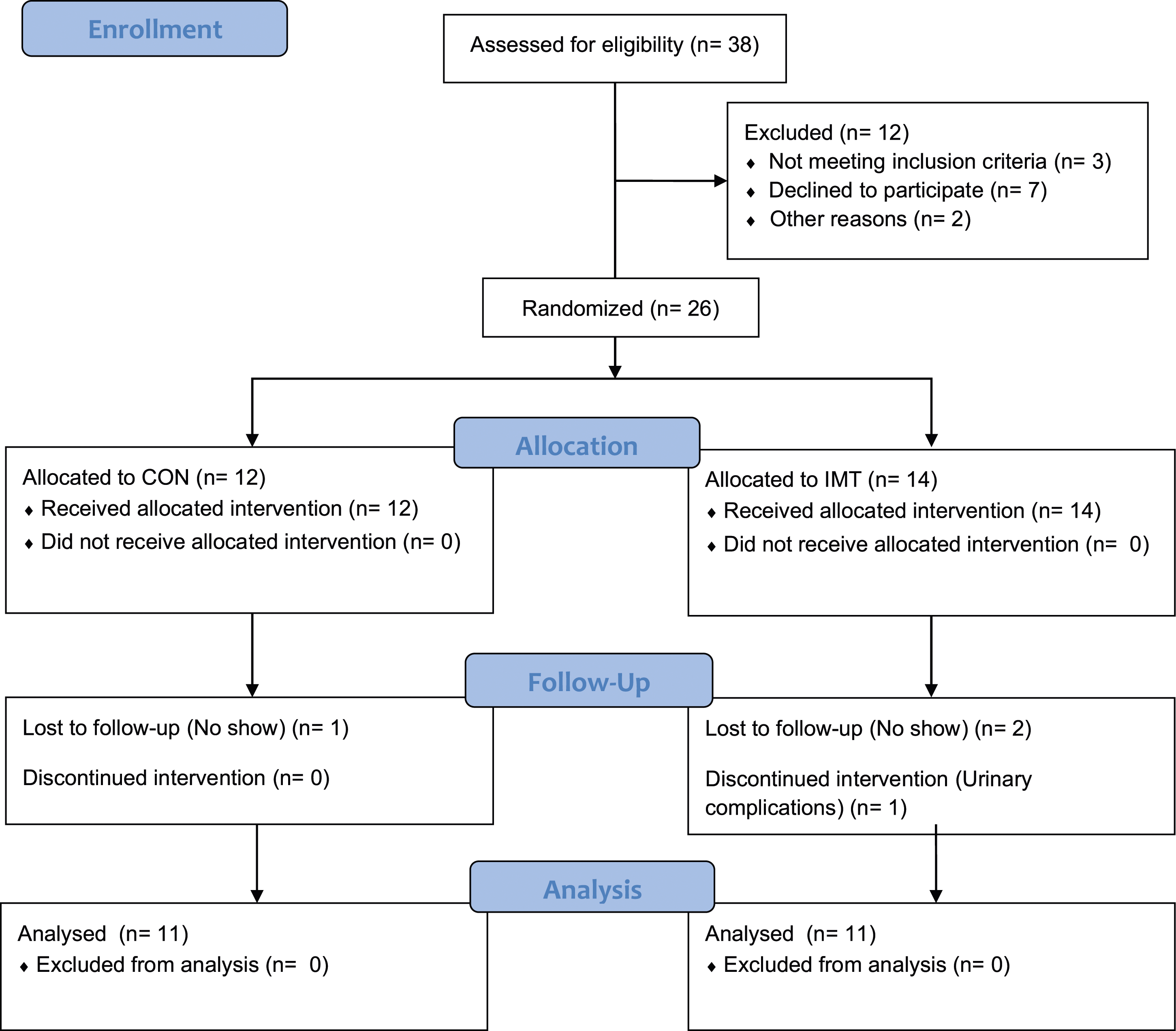
Respiratory function and functional capacity
Respiratory function and functional capacity for the inspiratory muscle training (IMT) and control (CON) groups at baseline, week two and week four. Values are mean ± SD.

FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; PMmax, maximal inspiratory mouth pressure; 6MWTD, six-minute walk test distance. *Significantly different between IMT and CON (p < 0.05).
A two-way ANOVA revealed time × group interaction effects for the distance covered during 6MWT (p = 0.001), sit to stands repetitions (p < 0.001) and right-hand (p < 0.001) and left-hand (p = 0.001) grip strength. There were no pairwise differences between the IMT and CON groups for the 6MWT distance and sit to stand repetitions. Right and left-hand grip strength was higher for the IMT compared to the CON group at week two (p = 0.05 p = 0.031) and week four (p = 0.013, p = 0.012) during the intervention.
Quality of life and dyspnea
Quality of life (visual analog scale and Euro Quality 5-Dimensions-3Levels (EQ-5D-3L)) and dyspnea (MRC Dyspnea Scale) for the inspiratory muscle training (IMT) and control (CON) groups at baseline, week two and week four. Values are mean ± SD.

*Significantly different between IMT and CON (p < 0.05).
Muscle damage biomarkers
A two-way ANOVA revealed time × group interaction effects for CKM (p < 0.001) and slow sTnI (p = 0.030). Both were lower for the IMT compared to the CON group at week two (p = 0.001, p = 0.002) and week four (p = 0.020, p = 0.001) during the intervention. There were no significant time × group interaction effects for fast sTnI (p = 0.130) (Figure 2). Serum slow skeletal troponin I (sTnI) (A), fast sTnI (B) and creatine kinase muscle type (CKM; C) for the inspiratory muscle training (IMT) and control (CON) groups at baseline, week two and week four. Values are means ± SD. *Significantly different between IMT and CON (p < 0.05).
Discussion
Main findings
The main findings were that compared to the CON group, 4 weeks of IMT resulted in a reduction in muscle damage biomarkers sTnI and CKM. IMT improved pulmonary function and inspiratory muscle strength, measured through PMmax. Four weeks of IMT also resulted in an increase in some components of physical functional capacity, as indicated by increased hand grip strength.
Muscle damage biomarkers
To our knowledge, we are the first to report the effects of IMT on muscle damage biomarkers including CKM, fast sTnI, and slow sTnI in COVID-19 recovered patients, who had been weaned from mechanical ventilation. Our results showed that serum concentrations of CKM, fast sTNI, and slow sTNI were higher at baseline in recovered COVID-19 patients who had been weaned from mechanical ventilation in the last 4 weeks than concentrations observed in healthy individuals.11,12 The results for the serum concentrations of CKM, fast sTNI, and slow sTNI were also similar to those reported in patients being weaned from mechanical ventilation 33 and the CKM results from patients with COVID-19. 34
Previous studies11,12 have reported that these markers are more sensitive and specific biomarkers to evaluate respiratory muscle damage following conditions that elevate inspiratory muscle work or activity and causes respiratory muscle damage (i.e., inspiratory pressure threshold loading; ITL). Respiratory muscle damage may occur during and following excessive loading, which exceeds the usual requirements of the muscle. Excessive loading can be categorized in two ways - overload or overactivity. 9 In respiratory disease, the respiratory muscles can experience both overload and overactivity because each breath may require a higher inspiratory muscle force and a higher breathing frequency (i.e., number of respiratory muscle contractions). ITL has been used previously to load respiratory muscles beyond their normal capacity and this loading was intended to produce respiratory muscle damage.11,12 ITL-induced respiratory muscle damage could be particular to just a few macromolecules of muscle tissue, or could result in small tears in the sarcolemma, z-disk, basal lamina, or supportive connective tissues, and damage to the cytoskeleton and contractile elements.9,12
Our results showed that the serum concentrations of CKM, fast and slow sTnI were higher at baseline in recovered COVID-19 patients who had been weaned from mechanical ventilation in the last 4 weeks than concentrations reported in healthy individuals.11,12,35 COVID-19 patients may experience respiratory muscle damage either through direct viral invasion of the respiratory muscles causing muscle cell damage, inflammation and impaired muscle function.1–3 The resultant respiratory failure and increased work of breathing may necessitate mechanical ventilation. 4 COVID-19 patients, weaned from mechanical ventilation, may have been experiencing respiratory muscle weakness and damage due to COVID-19 and the effects of mechanical ventilation.
Both the IMT and CON groups observed reduction in the concentrations of muscle damage biomarkers, but IMT resulted in a greater reduction in CKM and slow sTnI compared to the CON group. The decrease in all biomarkers in both groups could be due to the natural recovery of muscles post-ventilation, but the significantly greater decrease in the IMT group compared to the CON group could be attributed to the IMT intervention. IMT has been found to be beneficial in the recovery from mechanical ventilation-induced respiratory muscle dysfunction by increasing respiratory muscle strength and endurance.36,37 IMT might have helped to restore the muscle coordination lost during mechanical ventilation by potentially training the muscles to work together in a synchronized manner, possibly resulting in more effective and efficient respiratory muscle function.13,14 This improved efficiency following training may have resulted in lower concentrations of biomarkers of muscle damage (CKM and slow sTnI). IMT can also improve overall metabolic function and could enhance the body’s ability to clear metabolites.38,39 This could contribute to a more rapid clearance of muscle damage biomarkers from the bloodstream over time and IMT could have potentially accelerated their removal. This mechanism should remove all biomarkers, but just a significant removal was observed in slow sTnI and CKM and not fast sTnI.
Our findings may also indicate that our IMT protocol preferentially targeted slow fibers, whereas this protocol had less effect on fast fibers (i.e., fast sTnI). This could be explained on results presented by another study analyzing the structural adaptations and physiological outcomes of IMT in patients with chronic obstructive pulmonary disease (COPD). 40 The proportion of type I fibers (∼38%) and the size of type II fibers (21%) of the external intercostal muscles increase after IMT in COPD patients. 40 These findings establish that the external intercostal muscles of patients with COPD have the capacity to express differential structural remodeling after IMT. It must be noted though that the IMT protocols used by Ramirez-Sarmiento et al. 40 and the present study differ, and this potentially impacts the structural changes that may be observed. The participants in the Ramirez-Sarmiento et al. 40 study underwent 30 min of IMT that included intermittent IMT for 3 min with 2 min rest periods, starting at 60% PMmax, for 5 days per week over 5 weeks. In contrast, our study used a 4 week intervention where the IMT group performed 30 consecutive dynamic inspiratory efforts twice daily, starting at 50% of PMmax and adjusting the load periodically.
Respiratory function
The PMmax at baseline and throughout the intervention indicates inspiratory muscle weakness in both the IMT and CON groups. 41 We observed an increase in FEV1, FVC and PMmax, but not FEV1/FVC following IMT. FVC was higher for the IMT compared to the CON group at weeks two and four during the intervention. FEV1 and PMmax were higher for the IMT compared to the CON group at week four during the intervention. IMT may have improved lung function by increasing the strength and endurance of the inspiratory muscles resulting in an increase in total lung capacity. 42 In healthy individuals, the principal limitation on total lung capacity is inspiratory muscle strength. Thus, if the inspiratory muscles are stronger, they can oppose the combined elastic recoil of the chest wall and lungs to a greater extent, and thereby a higher end-inspiratory lung volume can be achieved at full inflation (i.e., total lung capacity). Also, with a larger initial lung volume, there is slightly greater tethering of the airway, which would also facilitate a higher FEV1.43,44 Our findings support a recent study that found an increase in PMmax, FVC and FEV1 following 2 weeks of IMT in COVID-19 patients following their weaning from mechanical ventilation. 15 We recognize that patients recovering from COVID-19 would also regain their respiratory muscle function without IMT. 45 Nevertheless, based upon our findings, IMT may have accelerated this recovery.
Functional capacity
We observed time × group interaction effects for the distance covered during 6MWT, sit to stands repetitions and right- and left-hand grip strength. However, there were no pairwise differences between the IMT and CON groups for the 6MWT distance and sit to stand repetitions. Our results suggest that as little as 4 weeks of IMT can improve grip strength, but this may not have been a long enough duration to improve 6MWT distance and sit to stand repetitions. The 6MWT and sit to stand tests are more complex physical function tests compared to hand grip strength. It is unclear why grip strength was improved following IMT. However, we speculate that improved motor unit synchronization post IMT may result in improved neuromuscular recruitment patterns and postural stability.46,47
Quality of life and dyspnea
We observed a higher QoL score for the VAS for the IMT compared to the CON group at week two and week four during the intervention. However, none of the five dimensions of QoL measured using the EQ-5D-3 L changed significantly during intervention. Abodonya et. al 15 reported that total QoL VAS score in EQ-5D-3 L questionnaire was increased in COVID-19 patients after 2 weeks of IMT. However, this study did not report on the five dimensions of QoL nor did they use a sham IMT (control group). IMT may not directly address or significantly influence dimensions such as mobility, self-care, usual activities, pain/discomfort, and anxiety/depression, specifically in recovered COVID-19 patients after weaning from mechanical ventilation, who were critically ill previously. IMT may have a narrower scope of impact compared to interventions targeting these specific dimensions.
There were no time × group interaction effects for dyspnea and this was improved in both IMT and CON groups. Our dyspnea results are in accordance with a meta-analysis conducted by Figueiredo et al 48 in which they analyzed 12 studies on the effects of IMT in COPD patients and reported that IMT did not change dyspnea. Reductions in dyspnea were though greater in studies that conducted IMT for 8 weeks while studies with IMT durations of four to 6 weeks reported no significant improvement in dyspnea. 48 Thus, our IMT duration may not be long enough to elicit reductions in dyspnea.
Limitations
Due to the restricted availability of suitable patients, a smaller number of female patients were recruited, preventing the comparison of potential sex-related differences in outcome measures. Furthermore, the study had a limited duration of supervision and follow-up observation. Furthermore, the study had a limited duration of supervision and follow-up observation which is a limitation. As our study involved outpatient participants who visited the clinic at weekly intervals rather than daily, closely supervising their IMT or CON sessions in-person was not possible. We only monitored the appropriateness of the participants’ training technique and intensity during each of the participants first two visits to the hospital and intensity adjustments were not documented in training diaries. The duration of the study was limited to 4 weeks. This was because our study was a proof of concept of the effects of IMT in recovered COVID-19 patients after weaning from mechanical ventilation and there is evidence of adaptations to IMT after just 4 weeks.30–32 In an ideal situation, the duration of the intervention would have lasted 8-12 weeks, and we would have more regular supervised training sessions which documented intensity adaptations during the intervention. Thus, it is essential to conduct randomized controlled trials with a larger patient cohort and an extended supervision and observation period for comprehensive evaluation. Additionally, early initiation of IMT during mechanical ventilation is recommended to prevent complications associated with post COVID sequalae and mechanical ventilation.
Conclusion
The present study found that 4 weeks of IMT results in a reduction of the muscle damage biomarkers slow sTnI and CKM. IMT also improved pulmonary function, inspiratory muscle strength and hand grip strength. The inclusion of IMT into the management of COVID-19 patients, particularly for intensive care patients, could assist with their recovery. Randomized controlled trials with a larger patient cohort and an extended supervision and observation period are required to support our findings.
Footnotes
Author contributions
E.W., B.H., D.E.M., conceived and designed the experiments; M.I., K.H, performed the experiments; M.I., D.E.M., analyzed the data; M.I., D.E.M. wrote the paper. E.W., B.H., D.E.M., reviewed the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the UniSQ International Research Scholarship, University of Southern Queensland.
