Abstract
This state-of-the-art review provides an overview of the history of home mechanical ventilation (HMV), including early descriptions of mechanical ventilation from ancient and Renaissance perspectives and the mass development of ventilators designed for long-term use during the poliomyelitis epidemic. Seminal data from key clinical trials supports the application of HMV in certain patients with chronic obstructive pulmonary disease, neuromuscular disease and obesity-related respiratory failure. Innovative engineering coupled with refined physiological understanding now permits widespread delivery of home mechanical ventilation to a global population, using portable devices with advanced ventilatory modes and telemonitoring capabilities. Exponential growth in digital technology continues, and ongoing research is needed to understand how to harness clinical and physiological data to benefit patients and healthcare services in a clinically- and cost-effective manner.
Keywords
Early history of ventilation – where did the concept of home ventilation come from?
Before embarking on a synopsis of modern-day home ventilation, it is important to understand the early physiological concepts, clinical experiments, and engineering techniques that have provided the foundations to evolve to the current devices in use today (Figure 1). Recognition of the central role of ventilation in maintaining life can be traced to references in ancient sacred Hindu text (Rig Veda, around 1500–1000 BC) and Egyptian writing (Ebers Papyrus, around 1550 BC) referencing incisions to access the windpipe.
1
More detailed descriptions are found in ancient Greek literature, including that of Empedocles (around 450 BC), Plato (Timaeus, 360 BC) and the Hippocratic Corpus (De Corde, Hellenistic period).
2
These authors demonstrate recognition of movement of breath and blood within body vessels and the relationship between cardio-pulmonary connections to transport blood to the lungs for nourishment. Galen (129–200 AD) further developed these concepts, which underpin modern respiratory physiology. He described the roles of the heart and lungs in exchange of air and blood and excretion of “fuliginous wastes”.
3
Following evaluation of injured gladiators, Galen also recognised the role of the diaphragm as the principle muscle of respiration, whereby complete C3-5 spinal cord injuries led to loss of phrenic nerve control with consequent diaphragm paralysis and rapid death.
4
Examples of negative pressure ventilation ‘iron lungs’, which were produced in large numbers and used to support patients with acute and chronic respiratory failure caused by the poliomyelitis pandemic in the mid-20th century. Patients lay in their ‘tank’, with their head protruding and sealed with a rubber collar. Lids were lifted to provide nursing and personal care. Image from the Lane Fox Respiratory Unit Collection, St Thomas’ Hospital, London UK.
Interest in respiratory physiology and mechanical ventilation re-emerged during the Renaissance, with Andreas Vesalius’ descriptions of positive pressure tracheostomy ventilation to sustain life in 1543: “But that life may, in a manner of speaking, be restored to the animal, an opening must be attempted in the trunk of the trachea, into which a tube or reed or cane should be put; you will then blow into this so that the lung may rise again and the animal take in air… and as I do this, and take care that the lung is inflated at intervals, the motion of the heart and arteries does not stop.”. 5 Michael Servetus and William Harvey recognised a closed loop double circulatory system, in which blood from the body is recirculated to the lungs via the right ventricle, where it mixes with inspired air before returning to the left ventricle.4,6 In the first description of sustained mechanical ventilation, in 1667 Robert Hooke delivered tracheal ventilation using bellows to a dog to prolong its life for over 1 hour. 7
The 19th century saw the development of pump systems to extract and quantify arterial oxygen and carbon dioxide content
8
and bellows ventilation, with Paul Bert identifying that increased ventilation increases blood oxygen content and lowers carbon dioxide content.
9
It was not until the polio epidemic of the 1950s, following the recent development of electrode measurement of blood pH,
10
that the high mortality rates observed in acute poliomyelitis were considered attributable to hypoventilation, with quantifiable hypercapnia and acidosis.
11
Prolonged mechanical ventilation during this period was largely delivered using negative pressure, which had been in development since the first box model built in Scotland in 1832.
12
Widespread clinical use of Philip Drinker and Louis Shaw’s 1928 “iron lung”
13
and John Haven Emerson’s iron lung and cuirass ventilators were rapidly implemented during the polio epidemic
14
(Figure 1). Mortality nevertheless remained high, particularly in those with bulbar disease. US physician Albert Bower and biomedical engineer Ray Bennet devised a valve that converted tank respirators to deliver intermittent positive pressure during inspiration, and its clinical application from 1949 improved survival from 21% to 84%.
15
This approach was replicated by anaesthetist Bjørn Ibsen in Denmark in 1952, who successfully applied intermittent positive pressure tracheostomy ventilation manually delivered via bag using pharmacological paralysis, with associated substantial reductions in mortality from acute polio.
16
Over the subsequent two decades, numerous case reports and observational studies of mechanical ventilation applied with tank respirators or invasive intermittent positive pressure to effectively correct gas exchange in patients with neuromuscular disease and COPD with acute respiratory acidosis emerged17–19 (Figure 2). History of mechanical ventilation, illustrating the developing understanding of cardiopulmonary anatomy and principles of ventilation, which paved foundations for biomedical engineering advances in home mechanical ventilation. Images reproduced from Wikimedia Commons: “Papyros Ebers” (1875), Georg Ebers in the Wellcome Collection (Creative Commons Attribution 4.0 License), Bust of Hippocrates, 1881 Young Persons’ Cyclopedia of Persons and Places (Public Domain), Galen Line Engraving (1973), R. Burgess, Portraits of doctors & scientists in the Wellcome Institute (Creative Commons Attribution 4.0 License), Illustration from Vesalius ‘Book I “De Humani Corporis Fabrica Libri Septum” (1543), Anatomical drawing from “De motu cordis et aneurysmatibus" (1728), William Harvey in the Wellcome Collection (Public Domain), London County Council Cuirass Respirator (1939), “Report of the Respirators (Poliomyelitis) Committee in the Wellcome Collection, Iron Lung (1955), US Food and Drug Administration (Public Domain).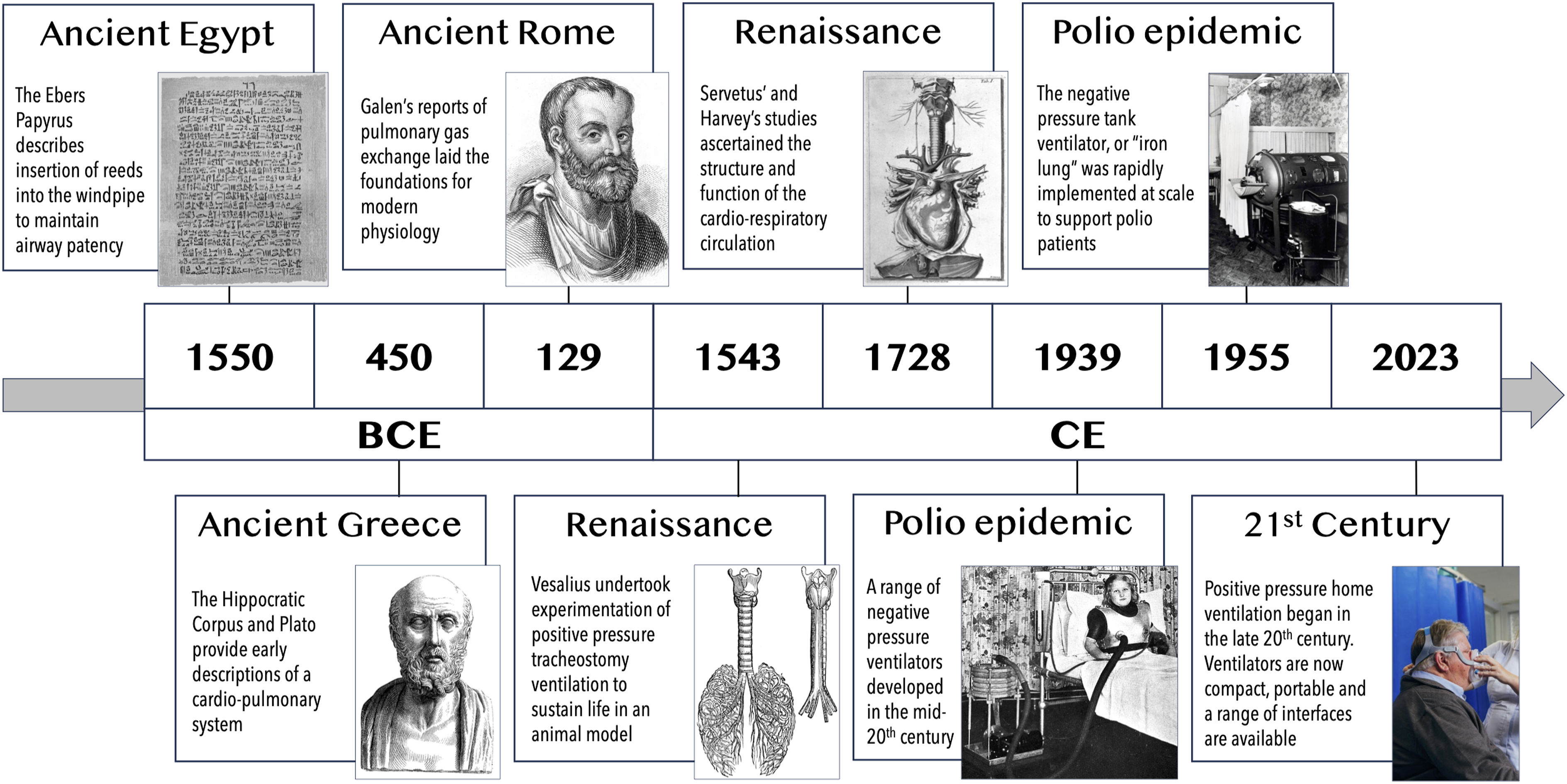
Modern home mechanical ventilation
The emergence of domiciliary ventilators
Technological advances led to commercially available automated blood gas analysers in 1973, with a corresponding increased recognition of patients with chronic respiratory failure. The concept of nocturnal home mechanical ventilation (HMV) to treat chronic respiratory failure emerged in the 1990s, in patients with inherited (Duchenne muscular dystrophy, spinal muscular atrophy) and acquired (post-polio, tuberculosis sequalae) restrictive lung disease and COPD.20,21 Observational data in patients with restrictive disease (neuromuscular or chest wall deformity) indicated that nocturnal non-invasive ventilation (NIV) conferred physiological benefits with respect to control of nocturnal hypoventilation and daytime gas exchange, as well as patient-reported improvements in daytime breathlessness, headaches and somnolence.22,23 Early randomised trials implementing NIV in COPD were less convincing, with the low-pressure approach (typical expiratory positive airway pressure (EPAP) 2 cm H2O, inspiratory positive airway pressure (IPAP) 10–15 cm H2O) failing to optimise gas exchange or breathlessness, and was poorly tolerated by patients.24,25 By the turn of the millennium, HMV was in widespread clinical use for patients with chronic respiratory failure, with a HMV prevalence of 6.6 per 100,000 people albeit with marked international variation, 26 and heterogeneity in the proportion of HMV users in each disease category, with more recent growth in HMV application amongst those with COPD and obesity-related respiratory failure.27,28
Where is home mechanical ventilation now?
Now in the 21st century, we have a robust evidence base demonstrating that HMV can improve mortality and morbidity in patients with COPD, neuromuscular disease (NMD) and obesity-related respiratory failure (ORRF). This has been through perseverance of clinical scientists and patients, who recognised the potential benefits of HMV and the need for personalised titration to treat nocturnal hypoventilation and optimise long-term adherence, collaboration with biomedical engineers and the healthtech industry to refine device technology and interfaces, and healthcare infrastructure to support setup and delivery of domiciliary ventilation. Seminal data from key clinical trials have informed current international guidelines and evidence-based clinical practice across COPD, NMD and ORRF
29
: Figure 3 outlines an example of local protocols for HMV initiation based on current evidence, Figure 4 demonstrates survival of HMV users by disease category in the 21st century.
28
In-patient setup protocols for home mechanical ventilation in patients with (A) COPD, (B) neuromuscular and chest wall disease and (C) obesity-related respiratory failure used at the Lane Fox Respiratory Unit, Guy’s and St Thomas’ NHS Foundation Trust. Survival after initiation on home non-invasive ventilation in different disease categories. Figure used with permission from Patout et al. Thorax 2020 Nov;75 (11):965-973. Doi: 10.1136/thoraxjnl-2019-214204.
28


COPD
The goals of ventilation in this patient population are to optimise gas exchange by overcoming the inspiratory threshold load imposed by intrinsic positive end expiratory pressure (PEEPi), airway resistance caused by inflammation, bronchospasm and sputum inspissation, and elastic load caused by emphysema to maintain alveolar ventilation. Early trials evaluating HMV in stable COPD adopted a low pressure approach (IPAP 10–18 cmH2O 30 ) and failed to demonstrated differences in gas exchange attributable to NIV compared to long-term oxygen therapy alone.31–33 Observational and short-term clinical trials indicated that a high-intensity approach, involving higher pressure support and backup respiratory rate to target PaCO2 reduction, was associated with improved daytime gas exchange and health-related quality of life.34–36 It was subsequently shown that high pressure NIV is as effective at targeting overnight transcutaneous carbon dioxide (TcCO2) and daytime PaCO2 as high pressure and high back-up rate ventilation, 37 which reflects the approach adopted in two landmark clinical trials in stable and post-acute COPD patients.38,39
Köhnlein et al. randomised stable COPD patients with daytime hypercapnia (PaCO2 ≥7 kPa) to receive usual care (including long-term oxygen therapy (LTOT), if indicated) or additional home NIV, using in-hospital titration, targeting PaCO2 <6.5 kPa or 20% reduction. 38 HMV (mean IPAP 22 cmH2O, EPAP 5 cmH2O, BUR 16 breaths/min) was found to reduce PaCO2 14 days post-setup, which was sustained at 12-months follow-up. HMV also conferred a survival advantage, with an absolute risk reduction of 21% at 12-month and a number needed to treat of 5 to prevent 1 death. The HOT-HMV study, delivered by Murphy et al., evaluated COPD patients with persistent hypercapnia 2–4 weeks after resolution of respiratory acidaemia due to COPD exacerbation. 39 Patients were randomised to receive home oxygen therapy (HOT) alone or with additional HMV (HOT-HMV), titrated to reduce overnight TcCO2 and increase oxygen saturation (SpO2). In this trial, HMV (median IPAP 24 cmH2O, EPAP 4 cmH2O, BUR 14 breaths/min) was associated with an absolute risk reduction of hospital readmission or death of 17% at 12 months and a number needed to treat of six to prevent one event. Health economic analyses demonstrate HOT-HMV to be a cost-effective intervention, with an incremental cost-effectiveness ratio of £10,259/quality-adjusted life year, which is comparable to pharmacological COPD interventions. 40 The HOT-HMV data contrast with earlier data from the RESCUE trial, which found no difference in 12-months admission-free survival amongst HMV users compared to usual care. 41 This is likely due to early recruitment of hypercapnic (>6.0 kPa) patients soon after liberation from acute ventilation, a proportion of whom may have spontaneously achieved normocapnia during exacerbation recovery. Notably, 50% of patients screened in the HOT-HMV study were eucapnic 2–4 weeks post-discharge, 39 and this cohort have comparable clinical mortality and readmission outcomes as patients with a eucapnic COPD exacerbation. 42
Key message
Data from clinical trials of HMV in COPD highlight the importance of patient selection and treatment target. Specifically, patients with persistent hypercapnia require a targeted approach to reduce carbon dioxide employing high pressure ventilation.
Neuromuscular and restrictive thoracic disease
NMD can be classed as rapidly or slowly progressive, and the onset of respiratory muscle weakness in the disease course depends on the underlying aetiology. Clinical trials in this patient cohort are challenging to conduct for several reasons, including disease rarity and progressive disability, which can pose barriers to recruitment, follow-up and protocols that may involve serial effort-dependent tests of respiratory muscle strength and lung volumes. There is wide geographical diversity on prevalence of non-invasive compared to invasive HMV in NMD, with a low prevalence of long-term tracheostomy ventilation (LTTV) in the UK and USA and relatively high numbers observed in Japan and Canada. 43 This may be attributable to healthcare systems, infrastructure and financial reimbursement, medical recommendations and patient preferences, and availability of specialist care delivered in either nursing homes or home-based care package and the time required to organise this. 44 Decisions to electively proceed with long-term tracheostomy ventilation are thus complex and should be approached on an individual basis.
Motor neurone disease (MND) has a global incidence of 1.68 per 100,000 person-years, typically presents in the 7th to 8th decade of life, and respiratory muscle weakness is inevitable in the most common phenotypes.45,46 In the UK, home ventilation is usually delivered using NIV in this disease category. A small proportion of patients may receive long-term tracheostomy ventilation, with tracheostomy insertion arising more commonly in the context of acute illness and emergency mechanical ventilation, rather than on an elective basis. Patient selection for HMV in MND is guided by data from a key trial by Bourke et al., who randomised patients with orthopnoea, maximal inspiratory pressure <60% predicted and/or daytime hypercapnia to receive usual care or additional NIV. 47 NIV was found to confer a survival advantage and maintain HRQoL scores in those with more preserved bulbar function. Those with severe bulbar impairment were less tolerant of NIV, and no difference in survival or HRQoL was observed. This echoes observations of Gruis et al. of lower adherence amongst patients with bulbar-onset compared to limb-onset disease. 48 UK guidelines advocate for early discussions regarding HMV soon after diagnosis confirmation, and referral to a ventilation centre for consideration of NIV initiation when there is evidence of respiratory muscle weakness. This includes symptoms of breathlessness, orthopnoea, disturbed and unrefreshing sleep, daytime somnolence and frequent chest infections, which indicates ineffective secretion clearance, and physiological tests including peripheral oxygen saturation, PaCO2, forced vital capacity and sniff inspiratory nasal pressure or maximal inspiratory pressure. 49
Patients with Duchenne muscular dystrophy were amongst the earliest to receive HMV, which has translated into marked reductions in mortality and morbidity from respiratory failure and recurrent infections. Observational data from Simonds et al. indicate a survival advantage of those patients with daytime and nocturnal hypercapnia who were initiated on NIV, with 73% alive at 5 years and no discernible detrimental impact of NIV on HRQoL. 23 Furthermore, in adult slowly progressive NMD and nocturnal hypoventilation, chronic respiratory failure with daytime hypercapnia and progressive symptoms develops within 2 years. 50
Restrictive thoracic disease (RTD) may arise secondary to kyphoscoliosis or congenital or acquired chest wall deformity, and respiratory failure is the leading cause of death in this cohort. 51 Current clinical practice is based largely on observational, uncontrolled clinical trials undertaken around the turn of the millennium. These report physiological improvements in nocturnal hypoventilation and daytime PaCO2 with no improvements in lung volumes or inspiratory muscle strength, patient-reported reductions in daytime symptoms of breathlessness, somnolence and fatigue, and reduced incidence of acute hospitalisation following nocturnal NIV application.22,52–54 Recent international survey data highlight that home ventilation is initiated in NMD and RTD at a ratio of 5:1, which could be a consequence of surgical advances in early intervention in kyphoscoliosis and laparoscopic and targeted thoracic intervention to minimise consequent chest wall deformity, and that diurnal hypercapnia and frequent hospitalisations are clinicians’ leading reasons for home NIV initiation. 55
Complex ventilation units also support transitional care for children with neuromuscular weakness requiring HMV into adult services. Guidelines recommend initiation of NIV with the development of symptomatic nocturnal hypoventilation or daytime hypercapnia. 56 Indeed, pre-emptive initiation does not appear to influence the development of hypercapnia, vital capacity reduction or improve survival. 57 Multidisciplinary advances in managing extra-pulmonary disease manifestations, such as early surgical correction of scoliosis, enteral nutrition delivered through gastrostomy, and optimisation of cardiomyopathy with pharmacological and implantable device interventions have undoubtedly also contributed to the improved survival in this patient group.58,59
Neuromuscular and restrictive thoracic disease may be associated with impaired ability to effectively clear respiratory secretions, which predisposes to recurrent infection. This may arise due to bulbar weakness, which impairs laryngeal and pharyngeal muscle strength and coordination, and can lead to upper airway obstruction from impaired vocal cord coordination and/or adduction of supraglottic laryngeal structures during inspiration.60,61 Mechanical insufflation-exsufflation (MIE) or “cough assist” devices deliver positive pressure followed by a rapid switch to negative pressure to generate inspiratory and expiratory flows to simulate a cough. MIE can be delivered to the upper airways or via tracheostomy and facilitates mobilisation of secretions to the upper airway, where they can be suctioned. 62 Cough effectiveness can be quantified using peak cough flow (PCF), with values of 360-840 L/min expected in healthy adults, 63 and expert consensus advocating MIE initiation for those in whom manual clearance techniques are ineffective and/or those with PCF <160 L/min.64,65 However, recent data demonstrate that high pressure MIE generates more upper airway closure than low pressure MIE, which is missed if PCF is used as a sole titration target for clinical effectiveness. 66
Key message
Patient with neuromuscular disease should undergo respiratory muscle surveillance, using tests of inspiratory and expiratory muscle strength, daytime and overnight gas exchange and cough peak flow, to identify those at risk of developing respiratory failure and requiring home mechanical ventilation, and those at risk of secretion retention and chest sepsis requiring mechanical insufflation-exsufflation.
ORRF
Obesity hypoventilation has been defined as the combination of obesity (body mass index (BMI) ≥30 kg/m2) with sleep-disordered breathing and daytime hypercapnia (PaCO2 > 6 kPa) in the absence of an alternative neuromuscular, mechanical or metabolic explanation. 67 The prevalence of chronic respiratory failure in the obese population increases with BMI, affecting 33% of morbidly obese patients.68,69 ORRF patients have poorer clinical outcomes in-hospital outcomes compared to eucapnic obese patients, greater comorbidity and worse outcomes compared with eucapnic patients with or without sleep-disordered breathing.70–74
Recent studies have cast focus on ORRF phenotyping based on the predominant underlying cause of chronic respiratory failure and response to positive airway pressure (PAP) therapy. The goal of domiciliary PAP therapy in this context is to treat the phenotype of sleep-disordered breathing to improve symptoms. This can be achieved by overcoming the loads on the respiratory muscle pump (imposed by upper airway resistance, reduced lung compliance and PEEPi), impaired capacity (reduced expiratory reserve volume and functional residual capacity) and blunted ventilatory response.
75
Three phenotypes are described: (A) Obstructive sleep apnoea (OSA) without hypoventilation (apnoea-hypopnoea index >30 events per hour without hypoventilation during overnight continuous positive airway pressure (CPAP) trial) (B) Combination of OSA and OHS (obesity hypoventilation syndrome) (evidence of hypoventilation; apnoea-hypopnoea index >30 events per hour) requiring NIV (C) OHS without severe OSA (evidence of hypoventilation; apnoea-hypopnoea index <30 events per hour) requiring NIV
Piper et al. demonstrated comparable change in PaCO2 and PaO2 in hypercapnic OSA patients without hypoventilation, after 3 months of CPAP (mean pressure of 14 cmH2O) and NIV (mean IPAP 16 cmH2O EPAP 10 cmH2O). 76 Based on these clinical effectiveness data, CPAP should be considered first-line therapy for OSA patients with chronic respiratory failure without evidence of hypoventilation (Phenotype A). Although Masa et al. demonstrated a lack of difference in terms of hospitalisation days per patient year, with similar gas exchange and symptom burden at 12 months in patients with combination OSA and OHS (Phenotype B), it must be acknowledged that this clinical trial was underpowered as the event rate was lower than expected.77,78 Furthermore, the study design may have impacted on the treatment effect size, since the home CPAP arm received inpatient overnight polysomnographic titration whilst the home NIV arm received outpatient daytime setup without targeted CO2 reduction. Current guidance is therefore to start PAP therapy in this group with NIV and then consider converting to CPAP after 3–6 months following attainment of normocapnia. 79 Long-term follow-up is key, as chronic respiratory failure may recur in a proportion of patients, necessitating conversion back to NIV. 80 Finally, in patients with OHS without severe OSA (Phenotype C), NIV is first line therapy with previous data demonstrating improvements in PaCO2 and Epworth sleepiness score. 81
Key message
Phenotyping patients with obesity related respiratory failure is key to determine suitable first-line positive airway pressure therapy. 82 CPAP should be considered first-line therapy in those with OSA without hypoventilation and moderate hypercapnia (daytime PaCO2 <6.7 kPa). NIV should be considered first-line therapy in those with OSA and OHS and those with OHS without severe OSA. Long-term monitoring and adherence to therapy is essential and switching therapy can be considered.
The future – where is home mechanical ventilation going?
In the last decade, there have been exponential advances in electrical and mechanical engineering that have resulted in smaller, more portable home ventilators with extended battery capabilities. In combination with our understanding of pathophysiological phenotyping of chronic respiratory failure across restrictive and obstructive respiratory disease, modes of ventilation have been designed to meet the demands of a personalised approach to clinical care, with the goal of enhancing ventilator adherence and improving clinical outcomes.
Where to start HMV?
HMV has conventionally been initiated in-hospital, which permits overnight physiological observation of the patient, ventilation titration and treatment response as well as optimisation of mask fit and comfort. This approach requires inpatient capacity and expert clinician capability. Recent evident supports both outpatient NIV setup and home setup as safe and effective options in certain contexts. In patients with ORRF, Murphy et al. demonstrated that ambulatory NIV set up with an automated NIV mode was similar to inpatient setup employing nurse-led titration in terms of cost-effectiveness and daytime gas exchange.83,84 Furthermore, in patients with COPD and neuromuscular disease, home-based NIV setup has been shown as comparable to inpatient setup in terms of PaCO2 reduction, HRQoL and cost-effectiveness.84,85 However, the limited monitoring available in outpatient and home environments can lead to more frequent unscheduled visits to optimise ventilator settings and interfaces and for acute care.83,86 With rising numbers of patients suitable for HMV and limited in-hospital resources, a bespoke patient-centred approach to HMV setup with established outpatient pathways should be adopted to provide safe and clinically- and cost-effective care.
Modes of ventilation
HMV is typically delivered using a pressure-targeted approach, whereby clinicians pre-set inspiratory and expiratory pressures, inspiratory time and backup rate based on the underlying clinical condition, and settings are titrated to optimise gas exchange, targeting CO2 reduction. Pulmonary mechanics and alveolar ventilation vary throughout different sleep stages (rapid eye movement (REM) and non-REM), which fixed pressure-targeted devices cannot adjust to. Algorithms have therefore been engineered to deliver volume-targeted pressure support ventilation, whereby the target tidal volume is pre-set and IPAP auto-titrated based on estimated expiratory tidal volume. Volume-targeted pressure support ventilation may be implemented in patients with obesity hypoventilation, in whom changes in pulmonary mechanics occur in the supine position and during different stages of sleep.87,88 This auto-titrating mode has been demonstrated as comparable to in-hospital nurse-led titration, producing similar changes in daytime PaCO2 at 3 months. This auto-titrating mode is well tolerated, with similar improvements in subjective and objective sleep quality observed. 89 Ongoing advancements have led to modes whereby IPAP, EPAP and back up rate are auto-titrated, such that combination of these settings can target tidal volume, abolish upper airway obstruction, and provide an adequate level of mandatory breaths. This auto-NIV mode has been shown to achieve similar reduction in PaCO2 reduction without impacting objective sleep quality in patients with COPD-OSA overlap syndrome and OHS.90,91 Depending on the frequency of oscillation used in forced oscillation, EPAP can be auto-titrated to abolish upper airways obstruction or expiratory flow limitation.75,77 The use of the forced oscillation technique has been shown in a physiological study to titrate auto-titrate EPAP and abolish expiratory flow limitation in patients with severe COPD with an accompanying maximal unloading of the respiratory muscles as evidenced by a reduction intrinsic positive end-expiratory pressure (PEEPi), neural respiratory drive and transdiaphragmatic pressure. 92 As these novel ventilator modes, which target underlying pathophysiology, pass through safety, efficacy and clinical effectiveness testing, clinicians will need to adopt an approach of physiologically-optimised pressure support ventilation.
Physiological monitoring
The goal of HMV is targeted reduction in daytime and nocturnal carbon dioxide, which can be monitored using transcutaneous or blood gas monitoring. It is noteworthy that a recent survey identified clinicians’ preference for SpO2 monitoring alone, over combined SpO2 and TcCO2 monitoring, to diagnose nocturnal hypoventilation, and SpO2 monitoring and downloaded data employed more frequently than CO2 measurements prior to discharge after HMV setup.
93
Clinicians should be cognisant of the substantial evidence base and international guidelines supporting targeted CO2 reduction to improve patient outcomes.29,39,40,94–97 Patients established on HMV should thus undergo regular clinical review to evaluate patient symptom burden, anthropometrics (weight and BMI), spirometry, gas exchange (including CO2 measurement) and to troubleshoot interface and technical issues (Figure 5). Initial follow-up after HMV setup is undertaken within 3 months,
93
with subsequent reviews planned every 3–12 months depending on the underlying aetiology of their respiratory failure.49,94 Conventional methods of monitoring overnight gas exchange include (A) overnight pulse oximetry (peripheral oxygen saturation and heart rate), which can be performed in the home setting, and (B) transcutaneous monitoring to obtain measurements of carbon dioxide (green), oxygen (red) and heart rate (blue), more conventionally used in-hospital.
Modern home ventilators include remote monitoring capabilities, whereby a cloud-based system is used to collate continuously collected data using internet connectivity. 98 Commonly reviewed data include usage time and patterns and estimates of unintentional leak, tidal volume, minute ventilation and residual oxygen desaturation index or apnoea hypopnea index. 93 These data are accessed at the time of clinical encounters and can be used to supplement patients’ reported adherence and issues relating to mask fit and ventilator settings. 99 Retrospective evaluation of ventilator data highlights that changes in respiratory rate, proportion of triggered breaths, and usage time can predict COPD exacerbation.100,101 However, the practical value of these findings and their influence of clinical outcomes warrants further evaluation, given limited clinical resources to continuously monitor such data.
Home-based monitoring of physiological parameters in conjunction with patient-reported outcomes may have a role in early detection of clinical deterioration and hospital admission prevention amongst HMV users. Wearable technology that captures physical activity and sleep quality indices have been applied in COPD. Data demonstrate reductions in daytime activity and impairments in sleep quality occur during exacerbations,102,103 and are associated with symptom burden, expiratory flow limitation and neural respiratory drive during COPD exacerbation recovery. 104 Such data may be integrated into web-based platforms, such as the myCOPD application or in the RECEIVER trial,105,106 and the impact of digital technology and machine learning to optimise disease control and early COPD exacerbation detection requires further prospective evaluation. 107 Use of domiciliary physiological monitoring is feasible to deliver and is adopted by patients irrespective of digital literacy and socioeconomic demographics. 106 However, patient-led data collection may increase unscheduled healthcare contacts, which could relate to reduced self-efficacy and increased dependence on clinicians to interpret day-to-day variability in measured parameters. 108 Home-based measures of forced oscillation technique and parasternal electromyography, which are techniques that presently reside solely within the research domain, may have a role in identifying and abolishing expiratory flow limitation and monitoring clinical change in HMV users in the future, and research is ongoing.92,109
Key message
Digital technology, remote monitoring and machine learning are rapidly advancing. Clinicians should adopt a patient-centred approach to HMV initiation, which may include outpatient or home-based setup, where resources safely permit. Alternative modes of ventilation may be considered to optimise gas exchange where conventional pressure-targeted ventilation has been ineffective. Further research is needed to understand the clinical- and cost-effectiveness of domiciliary physiological to optimise gas exchange, predict clinical deterioration and prevent hospital admission.
Conclusions
In this state-of-the-art review, we have provided an overview of ancient and Renaissance perspectives that paved the way for our ability to diagnose and manage chronic respiratory failure. The polio epidemic placed extraordinary burdens onto healthcare services but was a stimulus for innovative approaches to sustained mechanical ventilation and mass delivery, echoes of which we saw during the COVID-19 pandemic. Innovative engineering coupled with refined physiological understanding now permits widespread delivery of home mechanical ventilation to tens of thousands of patients globally, using portable devices with advanced ventilatory modes and telemonitoring capabilities (Figure 6). Research to understand how to harness rapid advances in digital technology to benefit patients and healthcare services in a clinically- and cost-effective manner is underway. We look forward to ongoing collaboration between innovative clinician scientists, biomedical engineers and patients to enhance patient-centred delivery of home mechanical ventilation to improve clinical outcomes and the patient experience of living with chronic respiratory failure. Home mechanical ventilation technology continues to advance, with compact, portable and battery powered devices enabling and empowering users of home ventilation. Image taken on a family holiday and provided with gratitude from the Home Mechanical Ventilation in Partnership Group (HMViP), which provides resources for HMV users and clinicians (HMViP.co.uk).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
