Abstract
To synthesize evidence for prefrontal cortex (PFC), quadriceps, and respiratory muscle oxygenation using near-infrared spectroscopy (NIRS) during cycling in individuals with chronic obstructive pulmonary disease (COPD). A scoping review was performed searching databases (inception-August 2020): Ovid MEDLINE, EMBASE, Cochrane Systematic Reviews, Cochrane Central Register of Controlled Clinical Trials, CINAHL, SPORTDiscus and Pedro. The search focused on COPD, cycling, and NIRS outcomes. 29 studies (541 COPD participants) were included. Compared to healthy individuals (8 studies), COPD patients at lower cycling workloads had more rapid increases in vastus lateralis (VL) deoxygenated hemoglobin (HHb); lower increases in VL total hemoglobin (tHb) and blood flow; and lower muscle tissue saturation (StO2). Heliox and bronchodilators were associated with smaller and slower increases in VL HHb. Heliox increased VL and intercostal blood flow compared to room air and supplemental oxygen in COPD patients (1 study). PFC oxygenated hemoglobin (O2Hb) increased in COPD individuals during cycling in 5 of 8 studies. Individuals with COPD and heart failure demonstrated worse VL and PFC NIRS outcomes compared to patients with only COPD—higher or more rapid increase in VL HHb and no change or decrease in PFC O2Hb. Individuals with COPD present with a mismatch between muscle oxygen delivery and utilization, characterized by more rapid increase in VL HHb, lower muscle O2Hb and lower muscle StO2. PFC O2Hb increases or tends to increase in individuals with COPD during exercise, but this relationship warrants further investigation. NIRS can be used to identify key deoxygenation thresholds during exercise to inform PFC and muscle oxygenation.
Introduction
Chronic Obstructive Pulmonary Disease (COPD) is associated with significant morbidity and represents the third leading cause of mortality worldwide. 1 COPD patients experience decreased exercise tolerance as a function of their disease. 2,3 Such exercise intolerance is multifactorial and comprised of disease driven limitations involving the respiratory, cardiovascular, and musculoskeletal systems. 4,5 More recently, the contributions of pre-frontal cortical activity have been investigated to understand the effects of hypoxemia on neural tissue. 6
Skeletal muscle dysfunction is common in COPD patients. 7,8 Fiber atrophy, a shift in fiber type distribution, and mitochondrial abnormalities can contribute to skeletal muscle dysfunction. As such, individuals with COPD often experience decreased muscle strength, endurance and oxidative capacity; and increased muscle fatigability. 3 –5 Muscle oxidative capacity is of particular importance during periods of increased exertion, such as with exercise or performance of activities of daily living. These bioenergetic muscle abnormalities can have negative effects on both respiratory and locomotor muscles.
Other contributing factors associated with exercise intolerance are potentially mediated centrally. 9 –12 Specifically, the prefrontal cortex (PFC) activity has been associated with dyspnea and exercise intolerance in COPD patients. 13 It has been reported that PFC oxygenation and blood flow increase in response to submaximal exercise in healthy adults and patients with COPD. 14,15 This increase is needed to match the heightened level of neuronal metabolism during times of increased exertion 15 and with executive function (i.e. planning and strategizing), which are required during motor performance. 15,16
Near infrared spectroscopy (NIRS) is a non-invasive tool used to measure the changes in oxygenation of the chromophore hemoglobin. 17 The principles and calculations of NIRS-derived parameters have been previously described. 18 Briefly, NIRS emits two different wavelengths of infrared light through the target tissue. 19 Some infrared light is absorbed by oxygenated hemoglobin (O2Hb) and some by deoxygenated hemoglobin (HHb). 19 The absorbance, adjusted by the modified Beer-Lambert law, is used to calculate the concentration of the chromophores (i.e. O2Hb and HHb). The sum of these values is the total hemoglobin (tHb) that can provide an estimate of blood volume beneath the NIRS optode. 20 In skeletal muscle, ▵HHb is used as an index of oxygen extraction in the muscle, whereas the oxygen saturation of the tissue (StO2) calculated as the ratio of O2Hb to tHb in percentage, reflects the balance between oxygen delivery and utilization. 21 In the brain, StO2 is used as surrogate of tissue oxygenation, while an increase in ▵O2Hb is indicative of increased neural activation. 22
To our knowledge, there are no reviews that summarize the effect of cycling on the aforementioned NIRS parameters in individuals with COPD. Given the variety of NIRS outcomes in response to exercise, a scoping review was performed to characterize and describe its application to people with COPD. Specifically, the objective of this review was to evaluate changes in NIRS outcomes of respiratory and limb muscles and cerebral oxygenation in response to cycling in COPD patients. Through an explorative scoping review methodology, this review aims to synthesize evidence on the relationship among NIRS outcomes and intolerance during cycling in these patients with COPD. Further characterization of this relationship may help to inform practice geared toward management and rehabilitation practices in chronic lung disease patients.
Methods
This review utilizes the established guidelines for scoping studies, including identifying the research question, searching and selecting relevant studies, charting the data, and summarizing and reporting the results. 23
Protocol and registration
This protocol was drafted using reported items for scoping reviews and was revised by members of the research team. The protocol was registered prospectively with Open Science Framework on May 22nd, 2020 (https://osf.io/w4b59/).
Identifying the research question
How do quadriceps, inspiratory muscles, and cerebral oxygenation levels respond to cycling in COPD patients?
Eligibility criteria
Papers that were eligible for this review were those that included COPD participants who performed a cycling intervention, and underwent evaluation using NIRS on quadriceps, inspiratory muscles, or the cerebral cortex. Included NIRS measures were: ▵HHb, ▵O2Hb, ▵tHb, StO2 (also termed tissue saturation index [TSI]), or blood flow (BF) measured by indocyanine green. To capture the possibility of much older yet fundamental NIRS literature, there were no date limitations assigned for this review. Only full text articles in English were included. Randomized control trials, systematic reviews and meta-analyses, controlled studies, cohort studies, case control studies, and case series and reports were eligible for this review to capture the range of study designs used to address this topic. Research in progress, conference proceedings and abstracts, dissertations and theses, and book chapters were excluded.
Information sources
A research librarian (AOC) from the University Health Network (UHN) was consulted to perform a comprehensive search strategy to identify English-language studies on near-infrared spectroscopy and cycling. The initial search strategy was developed for Ovid MEDLINE using a combination of database-specific subject headings and text words. The search strategy was then customized for each database. Searches were executed in the following databases on April 28, 2020 and updated August 14, 2020: Ovid MEDLINE, Ovid Embase, Cochrane Database of Systematic Reviews, Cochrane Central Register of Controlled Clinical Trials, CINAHL with Full Text, SPORTDiscus and Pedro. No date limits were imposed. Searches were limited to humans and adults. See Supplementary Appendix (Table A1) for a detailed search strategy.
Selection of sources of evidence
Two reviewers, MM and ST, independently screened the titles and abstracts to determine the eligibility of the studies. If this information could not be attained from reading the title and abstract, full text screening was performed. Disagreements were resolved by discussion with senior authors, WDR and DR, as needed.
Data charting process
An abstraction form developed by the research team was utilized. Data abstraction was performed independently by three reviewers (MM, ST, and LX) on included full text articles. MM performed data abstraction for all included articles with the other two reviewers serving as secondary abstractors. Any disagreements were resolved by discussion with WDR and DR. The data charting form was continuously revised through an iterative process until a consensus was reached.
Data items
The reviewers abstracted data on study design of the individual articles, inclusion and exclusion criteria, participant demographics, disease severity for COPD, cardiopulmonary parameters (heart rate, oxygen uptake), cycling protocol used, exercise progression, performance during cycling test, NIRS optode placement, and NIRS outcomes (O2Hb, HHb, StO2, tHb, BFI) as described above.
Critical appraisal of individual sources of evidence
A quality appraisal was performed using the modified Downs and Black assessment tool by three independent reviewers until a consensus was reached. 24 The modified Down’s and Black checklist consists of 15 items that assess reporting and internal and external validity that has been previously applied in several reviews. 25 –27 Quality assessments were expressed as the total of positive scores and were also expressed as a percentage of positive scores for each item and each included article. For the purposes of this scoping review, studies meeting 60–74% of the criteria was considered as moderate, ≥75% as high, and <60% as low. 28
Data synthesis
Studies were grouped by the target tissue, (skeletal muscle versus prefrontal cortex) and by type of cycling protocol (symptom-limited versus constant workload to exhaustion). These findings were used to generate three tables. The tables describe baseline patient demographics and characteristics of the cycling protocol, NIRS outcomes related to muscle, and NIRS outcomes related to cerebral oxygenation.
Results
The search generated 2552 titles and abstracts (Figure 1). 29 Removal of duplicates using Covidence online software resulted in 1325 records. 30 1284 studies were excluded for the following reasons: non-COPD sampling, non-cycling exercise intervention, and research presented as abstract or conference proceedings. One study evaluated both COPD and interstitial lung disease (ILD) patients, 3 but given only five ILD patients were described in this study, no further inclusion or data analysis was performed. One additional article was brought to the attention of the research team that was not captured in the search. Full-text screening was performed on 41 articles and 12 were excluded because the NIRS parameters were not measured during a cycling protocol. Twenty-nine articles were included in this scoping review. 2 –5,9,13,17,20,21,31 –50

Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram of the literature search.
29
Quality assessment score of articles ranged between 63% and 88% (10–13 maximum points) with a mean of 72%. 17 studies scored between 60% and 74%, (Table A2). The remaining studies scored 75% or greater. No studies scored less than 60%. All studies received points for: clearly stating the hypothesis and methods; describing the main outcomes in the introduction and methods section; describing the characteristics of included patients; describing the main findings; addressing if results were based on data dredging; appropriate statistical testing; and valid and reliable measurement of results. For the one question scored out of two points that addressed the distribution of principle cofounders, 86% of studies scored 1 point. Fewer than half of the articles described: the inclusion of participants that were representative of the populations they were drawn from; blinding of assessors measuring the main outcomes; recruiting participants over the same time period; and adjustment for confounding variables. The items involving reporting of actual probability values, and the inclusion of subjects that were prepared to participate were reported 52% and 90% of the time, respectively.
Detailed information regarding participant demographics are shown in Table 1. The forced expiratory volume (FEV1) for COPD patients ranged from 19–66% predicted. The mean age range of patient participants for the included studies was 60 to 73 years old. Overall, there were 541 COPD patients (83.4% male; 14.4% female; and 2.2% not reported in one study 39 ) and 120 healthy adults (88.3% male) evaluated in the 29 papers. Fifteen studies included only male populations and 11 studies reported a higher male to female ratio. Two studies reported a higher female to male ratio 5,45 ; no studies reported solely a female population.
Characteristics of participants and cycle ergometer test.
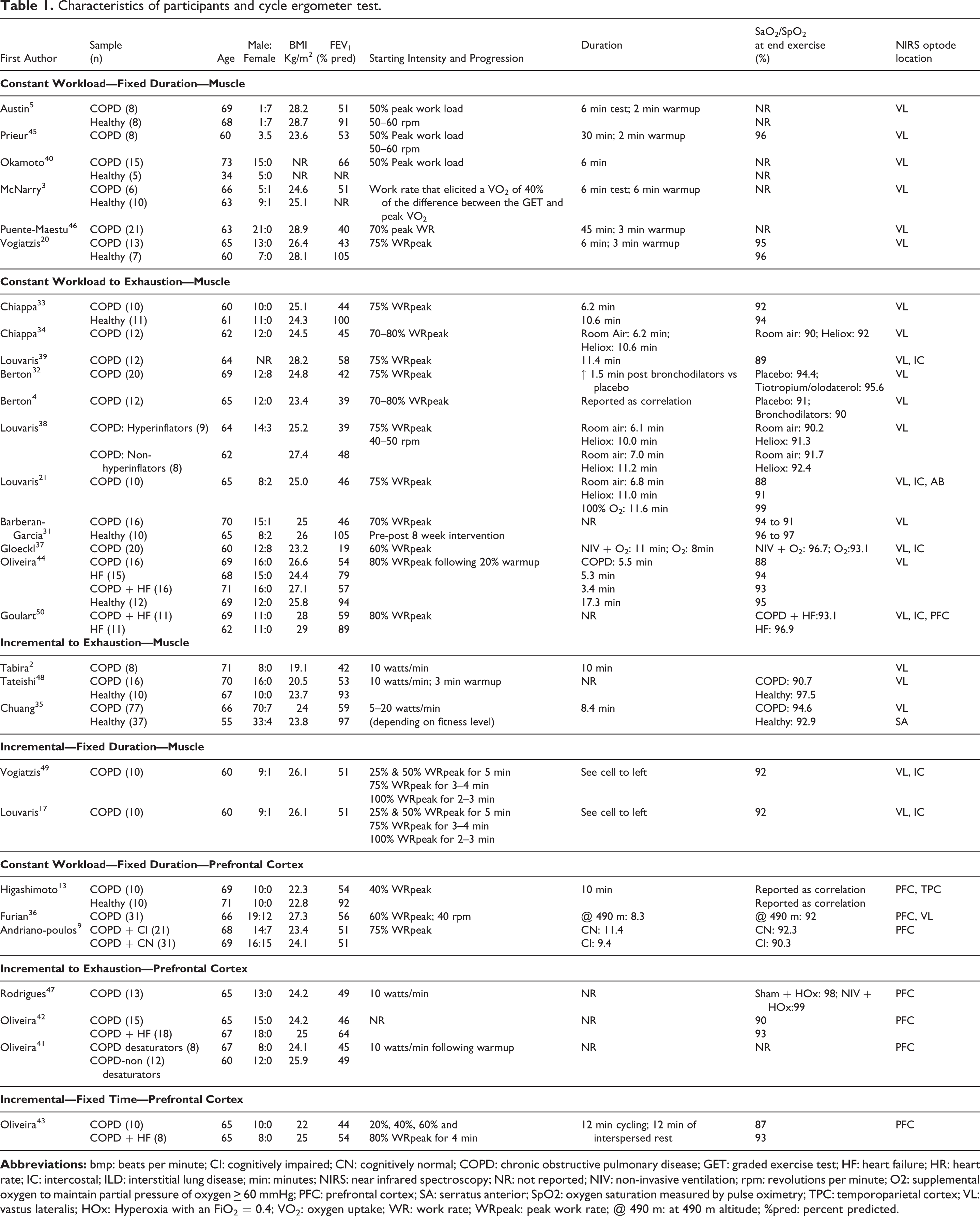
With respect to the exercise intervention, the majority of studies (n = 20) utilized constant work rate cycling. The remaining studies (n = 9) reported use of a symptom-limited incremental cycling protocol. Of the 12 studies that utilized constant work rate cycling, nine studies reported a pre-determined exercise time, ranging from 6 to 45 minutes. All remaining studies utilizing constant work rate and incremental protocols required participants to cycle to exhaustion, ranging from 6 to 17 minutes. Cycling workloads for COPD patients were consistently lower than those performed by healthy participants (see further details below).
In terms of placement of the NIRS optodes, 23 studies monitored NIRS parameters of the vastus lateralis (VL) and one study monitored the vastus medialis (VM). 31 Six studies evaluated the 7th intercostal muscle (IC), 17,21,37,39,49,50 one study monitored the serratus anterior (SA), 35 and one study examined the abdominal muscles, over the upper rectus abdominis. 21 Eight studies evaluated oxygenation of the cerebral cortex 9,13,36,41 –43,47,50 and two studies examined the VL in conjunction with the cerebral cortex. 36,50
Vastus lateralis oxygenation and deoxygenation responses to exercise
Of the studies that reported changes in oxygen extraction (HHb) of VL (n = 8), all reported a statistically significant increase in this value in response to cycling in COPD (Table 2). Of the studies that monitored VL or VM StO2 (n = 11), most studies 2,20,31,35,36,38,39,45,48 reported a decrease in this value, whereas two studies reported unchanging levels of VL StO2 (Table 2). 5,37
Muscle oxygenation/deoxygenation and blood volume/flow outcomes—changes from baseline to peak exercise, between treatments or between groups.

*BOM-L1TRW, Omegawave, Tokyo, Japan; CWS 2000 Runman, Philadelphia, USA; Inspectra Tissue Spectrometer Model 325 System, Hutchinson Technology Inc., Hutchinson, MN; OxiplexTSTM system Frequency Domain ISS, Champaign, IL, USA; NIRO-200NX, Hamamatsu, Photonics Japan; Oxymon, Artinis Medical Systems, Elst, The Netherlands; PortaLite, Artinis Medical Systems, Elst, The Netherlands; Portamon, Artinis Medical Systems, Elst, The Netherlands
** units for O2Hb, HHb and tHb can be expressed as arbitrary units (AU) or in micromolar (μM), which in essence have a similar meaning because the volume through which NIRS transmits light is not determined by many devices.
Vastus lateralis tHb and blood flow responses to exercise
Of the studies that examined VL tHb or BFI measured by indocyanine green (ICG), all reported an increase in this value throughout cycling (Table 2). 2,17,21,32,38,39,43,45,49
Muscle oxygenation and blood flow in COPD compared to healthy controls
Of the 23 studies examining muscle oxygenation, eight articles compared COPD to healthy participants (Table 1). Patients with COPD demonstrated a shorter VL HHb time constant in one report 33 and the response magnitude of VL HHb relative to workload did not differ from healthy controls in one study. 35 Moreover, of the studies that measured StO2 (n = 5), one study reported that VL StO2 levels were lower in healthy controls compared to COPD patients, 20 and two studies observed a larger or more rapid decrease in response to exercise in healthy adults. 20,48 BFI was lower in limb muscles of COPD patients compared to healthy individuals. 44 With respect to the cycling protocol, two studies utilized incremental cycling, with peak workloads that were lower in COPD patients compared to healthy controls (COPD range: 73 to 87 watts versus Controls: 113 to 144 watts; relative work rate difference of 35 to 40% lower in COPD). 35,48 For constant workload cycling, COPD patients and healthy controls cycled at a similar percentage of maximum effort generally ranging from 50 to 75% as shown in Table 1, but this occurred at a lower absolute workload (watts) for COPD patients (peak work rate difference of 30 to 76% lower). 3,5,20,31,33,40
Muscle deoxygenation and blood flow in COPD compared to congestive heart failure
COPD patients with congestive heart failure (HF) experienced a more marked increase in VL and IC HHb compared to those with COPD (Table 2). 44,50 BFI was lower in individuals with COPD and HF overlap compared to those with only COPD. 44
The effect of respiratory aids on muscle oxygenation
Two studies examined the effects of heliox on exercise performance. One study demonstrated that the use of heliox was effective in slowing the onset of the VL HHb response 34 and the other showed greater VL, IC and rectus abdominis blood flow compared to room air. 21 Other interventions utilized were bronchodilators 4,32 and non-invasive ventilation (NIV). 37 Only one of these three studies reported that the use of respiratory aids, namely bronchodilators, 4 elicited a VL HHb response that was slower than the placebo treatment.
Pre-frontal cortical oxygenation and deoxygenation response to exercise
Of the studies that examined cerebral oxygenation (n = 8), five studies 13,41 –43,47 indicated that oxygen delivery (O2Hb) levels increased during exercise and one study 9 reported no significant changes to O2Hb in COPD patients (Table 3). Two studies reported a decline in cerebral O2Hb in response to cycling in the patient group. 42,50 Oliveira et al reported that COPD patients with HF demonstrated a decline in PFC O2Hb, which is consistent with the findings of Goulart et al who reported a decrease in the PFC O2Hb in the HF group. 42,50
Prefrontal cortex oxygenation/deoxygenation and blood volume/flow outcomes—changes from baseline to the endpoint of exercise.

*ETG-7100, Hitachi Medical Corporation, Tokyo, Japan; NIRO 200, Hamamatsu Photonics KK, Hamamatsu, Japan; Oxymon, Artinis Medical Systems, Elst, The Netherlands; PortaLite, Artinis Medical Systems, Elst, The Netherlands.
** units for O2Hb, HHb and tHb can be expressed as arbitrary units (AU) or in micromolar (μM), which in essence have a similar meaning because the volume through which NIRS transmits light is unknown.
Three studies examined the tHb response to cycling. 9,13,43 Two studies 9,43 reported an increase in PFC tHb compared to baseline in COPD patients, while one study reported that COPD patients with HF demonstrated a decrease in PFC blood flow in response to exercise compared to COPD controls. 43 Rodrigues et al. demonstrated that the change in PFC oxygenation in COPD patients was greater in those performing cycling using a combination of non-invasive ventilatory support and heliox compared to heliox alone, suggesting a possible central hemodynamic benefit with non-invasive ventilatory support. 47
Pre-frontal cortical oxygenation changes in COPD patients compared to control
Only one study compared PFC O2Hb levels in COPD patients to that of healthy controls. 13 Although both groups demonstrated increases in this variable, between group differences were not significant.
Discussion
Summary of results
The main findings of this scoping review of 541 COPD patients (83% males) provides evidence that oxygen extraction (HHb) increases more quickly in the vastus lateralis at lower absolute workloads (range of 30 to 76% lower) during cycling compared to healthy persons. Although this response is faster in COPD patients compared to healthy adults, the increase in oxygen extraction experienced by patients can be mitigated by the use of respiratory aids such as heliox and bronchodilators. Of the studies that reported VL StO2, most demonstrated a decrease during both constant workload and incremental cycling. As expected during exercise, VL blood flow increased during cycling as indicated by an increase in VL tHb or ICG blood flow measures, but these increases were lower in COPD compared to healthy individuals. Even though there were fewer reports evaluating PFC compared to peripheral muscle oxygenation, cortical oxygen levels were observed to increase during cycling in COPD patients, but a diminished response to exercise was shown in COPD patients with HF.
Mechanisms of oxygen extraction
Our scoping review revealed that VL HHb increased more quickly during cycling in patients with COPD compared to healthy people with submaximal exercise, typically using a constant workload protocol ranging from 50 to 75% peak work rate. Even though relative work intensity was commonly used to compare between COPD and controls, several studies accounted for the lower workload in COPD participants by evaluating the rate of change in HHb. The quicker rate of oxygen extraction (HHb) may be attributed to increased local acidosis, 51 which reduces hemoglobin’s affinity for oxygen by virtue of the “Bohr effect.” 52 In addition, it has been proposed that the heightened levels of peripheral muscle deoxygenation at lower absolute workloads observed in COPD patients may be a result of impaired oxygen delivery due to low gas exchange and impaired cardiovascular function. 44 The altered mechanics of breathing in these patients may cause disturbances in cardiac output leading to an imbalance between oxygen delivery and extraction, reflected as lower StO2 levels. 53 Furthermore, the increased rate of oxygen extraction can’t be attributed to metabolic oxidative capacity, given the decreased oxidative enzymatic activity reported in skeletal muscle of COPD patients. 51,54
The effect of respiratory aids on muscle oxygenation
Several studies indicate that blood flow to the quadriceps increases during cycling in patients with COPD . Notably, this muscle perfusion can be enhanced by treatments such as NIV and Heliox. 4,34,38 These interventions improve oxygen delivery by countering the characteristic exercise onset of dynamic hyperinflation in COPD, and reducing the work of breathing. 38 Furthermore, lung hyperinflation may increase ventricular afterload and decrease pre-load, resulting in a decrease in stroke volume and cardiac output. 32 This effect on cardiac output may have downstream effects on peripheral muscle oxygen saturation, especially during periods of heightened cardiac and ventilatory requirements with exercise. Additional benefits of heliox include a reduction of diaphragmatic activity, and increase in exercise tolerance, and less leg discomfort. 21,34,38 In addition, treatment with inhaled bronchodilators can reduce airflow obstruction leading to slower oxygen extraction kinetics following treatment. 4 Thus, as demonstrated in several studies, treatment aimed at reducing lung hyperinflation and airway obstruction may improve limb muscle oxygen saturation and slow the kinetics of oxygen extraction by alleviating the mechanical burden of breathing in patients with COPD. 4,32,34
Blood flow distribution
The rationale for the aforementioned benefits of respiratory aids coincide with findings of blood flow distribution relative to respiratory muscle loading. 5 It has been postulated that blood flow is redistributed from limb to respiratory muscles when the respiratory load is increased in COPD and healthy controls, or from the respiratory to the limb muscle when the respiratory load is decreased. 55 This postulate is supported by a study of healthy adults exposed to expiratory flow limitation (EFL) during exercise; they demonstrated an increased intercostal muscle blood flow index (BFI), but a decrease in quadriceps BFI suggesting a redistribution of blood flow from the locomotor to the respiratory muscles. 56 This occurred at an exercise level that demanded submaximal cardiac output when leg muscle blood flow could have been sustained, if cardiac output was the rate limiting factor. Alternatively, other factors such as hypoxemia and hypercapnia associated with EFL may have resulted in increased sympathetic discharge and consequent vasoconstriction, leading to locomotor blood flow reduction, as opposed to redistribution. 55
More recent studies have investigated blood flow distribution during cycling in COPD patients. When individuals with COPD were given 100% oxygen or heliox (helium 79% and oxygen 21%), in an attempt to reduce the work of breathing during constant load cycling at 75% of peak capacity, no redistribution of blood flow from the intercostals relative to the vastus lateralis was observed. 21 Moreover, 100% oxygen and heliox compared to room air, resulted in longer cycling endurance times compared to the room air condition. At the end of cycling, intercostal and quadriceps muscle blood flow were both greater with heliox compared with room air or 100% oxygen. 21 Although the work of breathing and dyspnea was reduced with the use of heliox, redistribution of blood flow from intercostal muscles to the locomotor muscles was not observed. Further work investigating other mechanisms of how heliox decreases dyspnea and improves exercise endurance is needed. The changes in blood flow redistribution of other inspiratory muscles (i.e. diaphragm, sternocleidomastoid) could be one potential mechanism.
Blood flow distribution among respiratory and locomotor muscles may vary during different types of exercise stimuli. A recent study conducted on 18 COPD patients by Louvaris et al. 57 demonstrated that during high intensity constant-load cycling (80% peak work rate), intercostals, scalene, and abdominal muscle blood flow did not change from rest with cycling despite ongoing cardiac output reserve, and the muscle oxygenation saturation in these muscles was lower compared to the hyperpneic state. This contrasts with the isocapnic hypernea protocol performed without locomotor muscle movement, where increases in muscle perfusion of the aforementioned muscles was observed for a given minute ventilation. 57 Thus, these results highlight that high-intensity exercise impairs the blood flow to the extra-diaphragmatic respiratory muscles in COPD patients and may in part account for the dyspnea experienced by COPD patients with exercise. 55,57 However, an important limitation of the blood flow distribution theory is that blood flow to the diaphragm and accessory muscles of inspiration have not been measured simultaneously. Furthermore, to our knowledge there have been no reports describing potential differences in blood flow redistribution in COPD compared to healthy controls utilizing a similar ventilatory and exercise protocol. This is an area in need of future investigation to further understand the blood flow distribution theory between respiratory and locomotor muscles. 55
Cerebral oxygenation
Compared to the breadth of studies examining peripheral muscle oxygenation and blood flow in respiratory patients, investigations of prefrontal cortical oxygenation response to exercise is less conclusive. However, several reports demonstrate that cortical oxygen levels (O2Hb) of COPD patients increased in response to exercise, 13,41,43,44,47 albeit to a lesser extent or decreased in COPD patients with HF. 44,50 When neural activity and associated metabolism increases, cerebral vessels dilate to enhance regional oxygen availability. 9 This results in higher tHb values in addition to increased O2Hb. 13 In contrast, prefrontal cortical oxygenation increases during incremental cycling in healthy persons but decreases preceding task failure due to exhaustion. 58 Although this has been considered a mechanism of “central fatigue,” it may be attributed to hyperventilation induced hypocapnia and consequent cerebral vasoconstriction. 58 In contrast, hyper- rather than hypocapnia may occur at task failure in COPD patients in those with marked ventilatory limitations, postulated to be related to gas exchange abnormalities and significant hyperinflation. 59,60 The specific mechanisms contributing to cerebral oxygenation during cycling in COPD patients with and without HF require further delineation.
The influence of heart failure on cerebral oxygenation
COPD patients with HF demonstrated either a decrease in cerebral oxygenation or had no change in oxygenation compared to COPD controls, 43,44,50 which differed from responses of COPD patients without this comorbidity. Furthermore Oliveira et al. reported that in addition to the diminished cerebral oxygenation increase in COPD patients with HF, prefrontal cortical blood flow decreased, while the COPD-only group increased blood flow to this region. 43 Impairments in cerebral blood flow and oxygenation in COPD patients with HF may be attributed to the decrease in cardiac output and lower amounts of cerebral vasodilation associated with the HF state. 43 In addition, it has been suggested that cerebral hypoperfusion can result in the abnormalities in cerebral metabolism that are commonly observed in patients with advanced lung disease. 61,62
Limitations
This scoping review has some limitations. For one, NIRS parameters were expressed in a variety of ways across the included studies. Thus, a meta-analysis was not possible due to the diverse NIRS measurement techniques, lack of reporting of absolute changes, and several studies not reporting participant adipose tissue thickness beneath NIRS optode sites. Adipose tissue thickness (>10 mm) is an important exclusion criteria because thicker adipose can confound NIRS measurements by increasing resting oxygenation values, reducing optical density, and decreasing sensitivity to detect oxygenation changes in the tissue of interest, in this case muscle or prefrontal cortex. 63 Moreover, this review is limited to the effects of cycle ergometry on NIRS outcomes, and did not investigate other methods of exercise. In addition, male COPD patients were overwhelmingly represented in the aforementioned studies (83% men), possibly masking the muscle and PFC oxygenation response to cycling in women, and concealing potential sex differences, if any. Lastly, there were limited reports of cerebral oxygenation response to cycling in COPD patients, and thus prefrontal cortical oxygenation studies may underrepresent a full spectrum of outcomes in the current review.
Clinical relevance
There are several benefits associated with NIRS technology when applied to exercise. For one, the utility of NIRS for examining prefrontal cortical changes becomes apparent when compared to traditional imaging techniques such as functional magnetic resonance imaging (fMRI). Unlike fMRI, NIRS can measure oxygenation changes during whole body movement, making it more suitable for exercise. 64 Since exercise is often a component of many programs aimed at rehabilitation, the ability to detect physiological changes during activity highlights the advantages of using NIRS in a clinical setting. NIRS may help identify oxygen extraction thresholds with various therapeutic strategies (i.e. exercise training, oxygen supplementation), but important to account in the analysis the lower absolute workloads observed in COPD patients compared to controls. Furthermore, NIRS oxygenation outcomes are relevant beyond the scope of exercise. For example, impaired matching between oxygen delivery and consumption and resultant fatigue could be evaluated to determine its influence on COPD patient’s ability to perform activities of daily living such as dressing or bathing. 65 Moreover, the ability of NIRS to detect changes in cerebral oxygenation in COPD patients becomes relevant when assessing cognitive health. As lower cerebral O2Hb may be associated with reduction in cognitive performance, there is a need to evaluate ways that may enhance or at least maintain cerebral oxygenation. 66
Conclusion
COPD patients experience faster quadriceps oxygen desaturation at lower workloads during cycling exercise compared to healthy controls. Faster dynamics of oxygen extraction together with diminished oxygen delivery may play a role in exercise intolerance. Although studies show that cerebral oxygenation increases during exercise in patients with COPD, the coexistence of heart failure may diminish this response. NIRS appears to be a promising modality for assessment of prefrontal cortical neural activity and peripheral oxygen extraction during cycling and potentially for other physical activities in COPD patients.
Supplemental material
Supplemental Material, sj-docx-1-crd-10.1177_1479973121993494 - Muscle and cerebral oxygenation during cycling in chronic obstructive pulmonary disease: A scoping review
Supplemental Material, sj-docx-1-crd-10.1177_1479973121993494 for Muscle and cerebral oxygenation during cycling in chronic obstructive pulmonary disease: A scoping review by Melissa Miles, Antenor Rodrigues, Shirin Tajali, Yijun Xiong, Ani Orchanian-Cheff, W Darlene Reid and Dmitry Rozenberg in Chronic Respiratory Disease
Footnotes
Authors’ note
WD Reid and D Rozenberg contributed equally as co-senior authors. Work performed at: Physical Therapy, University of Toronto, Toronto, Canada; Toronto General Hospital Research Institute, Toronto, Canada.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MM and DR were funded in part by Sandra Faire and Ivan Fecan Professorship in Rehabilitation Medicine. ST was funded in part by a grant from the Canadian Pulmonary Fibrosis Foundation and the Ontario Respiratory Care Society. AR was funded from a Fellowship from the Faculty of Medicine, University of Toronto.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
