Abstract
To achieve a multidimensional evaluation of chronic obstructive pulmonary disease (COPD) patients, the spirometry measures are supplemented by assessment of symptoms, risk of exacerbations, and CT imaging. However, the measurement of diffusing capacity of the lung for carbon monoxide (DLCO) is not included in most common used models of COPD assessment. Here, we conducted a meta-analysis to evaluate the role of DLCO in COPD assessment.
The studies were identified by searching the terms “diffusing capacity” OR “diffusing capacity for carbon monoxide” or “DLCO” AND “COPD” AND “assessment” in Pubmed, Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, Embase, Scopus, and Web of Science databases. The mean difference of DLCO % predict was assessed in COPD patient with different severity (according to GOLD stage and GOLD group), between COPD patients with or without with frequent exacerbation, between survivors and non-survivors, between emphysema dominant and non-emphysema dominant COPD patients, and between COPD patients with or without pulmonary hypertension.
43 studies were included in the meta-analysis. DLCO % predicted was significantly lower in COPD patients with more severe airflow limitation (stage II/IV), more symptoms (group B/D), and high exacerbation risk (group C/D). Lower DLCO % predicted was also found in exacerbation patients and non-survivors. Low DLCO % predicted was related to emphysema dominant phenotype, and COPD patients with PH.
The current meta-analysis suggested that DLCO % predicted might be an important measurement for COPD patients in terms of severity, exacerbation risk, mortality, emphysema domination, and presence of pulmonary hypertension. As diffusion capacity reflects pulmonary ventilation and perfusion at the same time, the predictive value of DLCO or DLCO combined with other criteria worth further exploration.
Chronic obstructive pulmonary disease (COPD) affects more than 328 million people 1 and is the third leading cause of death worldwide. 2 To evaluate the severity of COPD, a number of measurements are required. Spirometry has been the cornerstone of COPD assessment, including forced expiratory volume during the first second (FEV1), forced vital capacity (FVC), and the ratio between these two measurements (FEV1/FVC). Recently, to achieve a multidimensional evaluation, the spirometry measures are supplemented by assessment of symptoms, risk of exacerbations, as well as quantitative assessment of emphysema by CT imaging.3,4 However, one noninvasive and widely available tool is not included in commonly used prognostic models of COPD assessment, the measurement of diffusing capacity of the lung for carbon monoxide (DLCO).
Diffusing capacity of the lung for carbon monoxide is a measure of gas exchange reflective of the complex interactions occurring at the alveolar–capillary interface. 5 It reflects changes in functional lung volume and gas transport across the alveolar–capillary membrane at the same time. Pathological changes characteristic of COPD are found in the airways, lung parenchyma, and pulmonary vasculature. 6 Airflow limitation is the basic characteristic of COPD, and correlates with the reduction in the FEV1 and FEV1/FVC ratio. The peripheral airway limitation progressively traps gas during expiration, resulting in hyperinflation. Pulmonary hypertension may develop late in the course of COPD because of vasculature loss due to hyperinflation and hypoxic vasoconstriction of the small pulmonary arteries. Hyperinflation and pulmonary hypertension both influence the lung diffusing capacity, resulting in decrease of DLCO and DLCO/VA (DLCO divided by the alveolar volume). Thus, DLCO provides more information regarding respiratory physiology than spirometry alone.
However, the importance of DLCO in COPD assessment has not been paid enough attention. Some large cohort studies, such as ECLIPSE, did not include DLCO measurement mainly due to the cost. A few studies suggested an association between decreased DLCO and frequent exacerbation, but the sample size was relatively small.7,8 Studies evaluating DLCO as an independent predictor of mortality are conflicting.9-11 Thus, a meta-analysis integrating data from different center and increasing sample size might give more information and attract attention about this topic. Here, we conducted a meta-analysis of observational studies to explore the relation of DLCO and COPD severity, exacerbation, and mortality, the most important three factors of COPD assessment. As diffusing capacity has been showed to be related to emphysema and pulmonary hypertension, we also explored diffusing capacity in emphysema dominant phenotype and COPD—pulmonary hypertension phenotype.
Method
Search Strategy
This meta-analysis has been performed in agreement with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Protocols.
12
Figure 1 shows the flow diagram. This quantitative synthesis satisfied all the recommended items reported by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Protocols checklist.
12
Flow chart of study inclusion.
Two reviewers performed a comprehensive literature search for observational studies evaluating DLCO in COPD assessment. The terms “diffusing capacity” OR “diffusing capacity for carbon monoxide” or “DLCO”AND “COPD” AND “assessment” were searched in Pubmed, Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, Embase, Scopus, and Web of Science databases to provide for relevant studies available up to 31 December 2019. Citations of recent published meta-analyses and relevant reviews were examined to identify further pertinent studies.11,13,14 Studies were limited to those published in English.
Study selection
High-quality observational studies investigating DLCO in patients with COPD were included in this meta-analysis. Two reviewers independently checked the relevant studies identified from literature searches obtained from the previously mentioned databases. The studies were selected in agreement with the previously mentioned criteria, and any difference in opinion about eligibility was resolved by consensus.
Data extraction
Two reviewers extracted information from all articles selected for inclusion in the meta-analysis. The extracted information included study design, sample size, DLCO % predict, COPD severity, acute exacerbation of COPD, smoking habit, clinical phenotype, cardiovascular morbidity, and COPD mortality. Two reviewers independently extracted the data, and then checked for accuracy; any inconsistency was resolved by consensus. Data have been extracted in agreement with Data Extraction for Complex Meta-Analysis recommendations. 15
Quality score, and risk of bias
The Newcastle–Ottawa Scale (NOS) 16 was used to assess the quality of cohort studies and case-control studies concerning the selection of cohorts/cases and controls, comparability of cohorts/cases and controls, and assessment of outcomes and exposures. 16 Quality of cross-section studies were assessed according to Agency for Healthcare Research and Quality (AHRQ) suggestion. 17 Two reviewers independently assessed the quality of individual studies; any difference in opinion about the quality score was resolved by consensus. The risk of publication bias was assessed by applying the funnel plot. The risk of small-study effect was assessed by Egger’s test.
Data synthesis and meta-analysis
The mean difference of DLCO % predict was assessed in COPD patient with different severity (according to GOLD stage and GOLD group 18 ), between COPD patients with or without frequent exacerbation, and between survivors and non-survivors. As diffusing capacity has been showed to be related to emphysema and pulmonary hypertension, we also assessed the mean difference of DLCO % predict between emphysema dominant and non-emphysema dominant COPD patients, and between COPD patients with or without pulmonary hypertension. Proper mean value and standard deviation (SD) were synthesized from original data as previous published, 15 if needed. More details were described in the results part. For those manuscripts without SD in certain groups, SD was synthesized from original SD in separated groups. For those manuscripts without SD, an average SD was estimated using the methods described by Hozo et al. 19 The hazard ratio of DLCO % predict for mortality is also investigated. Heterogeneity was assessed from visual inspection of the forest plots, χ2 tests, and the I2 statistic. 20 Statistically significant heterogeneity was considered present at P values < 0.10 and I2 > 50%. The analyses were performed with RevMan V 5.3 and Stata V 15.0.
Results
We retrieved a total of 459 references. 43 studiesfinally fulfilled the inclusion criteria and were further analyzed.9,11,14,21–59 A flow chart for the studies evaluated and the reasons for exclusion is shown in Figure 1.
Study characteristics
Characteristics of the included studies were summarized in Supplement Table S1, including publication year, authors, country, study design, inclusion and exclusion criteria, and classification of each study. Sixteen studies were included in meta-analysis of DLCO for COPD severity classification according to Global Initiative for Chronic Obstructive Lung Disease (GOLD) stage I, II, III, and IV. 60 All these studies evaluated stable COPD patients. Six studies were included in meta-analysis of DLCO for COPD severity classification according to GOLD group A, B, C, and D, of which 4 classified the patients according to GOLD 2011 group classification and two classified the patients according to GOLD 2017. All these studies evaluated stable COPD patients. In one study, 18 of 52 patients did not accomplished DLCO measurement. 33 No missing data of DLCO in other 21 studies included for COPD severity. Nine studies were included in the meta-analysis of DLCO for acute exacerbation of COPD (AECOPD), of which 2 compared between frequent exacerbators (more than 2 times for 1 year) and infrequent exacerbation ( < 1 time for 1 year), and seven compared between non-exacerbators and exacerbators. In two of these studies, DLCO data were missing in one of 189 patients 48 and in 76 of 311 patients, 46 and no missing data of DLCO in other seven studies. Five studies were included in the meta-analysis of DLCO for COPD mortality, of which 4 were prospective and one was retrospective. 36 of 93 patients in one study did not accomplish DLCO measurement, 14 and no missing data in other four studies.
Four studies evaluated DLCO % predicted between emphysema dominant COPD and non-emphysema dominant COPD. All these four studies used computed tomography (CT) to define emphysema, 1 with nonparametric scale, 2 with quantitative evaluation, and 1 with qualitative evaluation. In one study, 27% of patient did not accomplish DLCO measurement, 52 and no missing data of DLCO in other three studies. Four studies evaluated DLCO between COPD patient with pulmonary hypertension (PH) and without PH, of which 2 studies diagnosed PH according to the ratio of the main pulmonary artery diameter and the ascending aorta diameter (PA/A) and two studies measured mPAP directly with right heart catheterization. One study only enrolled severe COPD patients, and 154 of 362 patients did not accomplished DLCO measurement in this study. 58
Five studies enrolled in our analysis clearly mentioned that patients with cardiovascular diseases or primary cardiac diseases were excluded, while other studies did not state in their exclusion criteria. No enrolled study excluded obesity patients. And no enrolled study indicated if any patients had anemia.
We used funnel plots (Supplement Figure S1) and Egger’s test (Supplement Figure S2, Supplement Table S2) to check the publication bias and small-study effect, and the results showed that there was no obvious publication bias and no small-study effect.
Diffusing capacity and COPD severity, exacerbation, and mortality
To evaluate diffusing capacity in COPD airway limitation severity, we collected DLCO% predicted data from mild-moderate COPD (GOLD stage I–II) and severe-very severe COPD (GOLD stage III–IV). Most studies provided DLCO% predicted of each stage, so we combined the original data to obtain proper data for meta-analysis. DLCO % predicted was significantly lower in stage III–IV COPD patients (MD 22.02%, 95% CI: 18.10%–25.93%, p < 0.00,001) compared with stage I–II patients (Figure 2(a)). Furthermore, we evaluated diffusing capacity in symptomatic assessment of COPD and risk of exacerbation of COPD. We collected DLCO% predicted data from COPD group A/C (few symptomatic), group B/D (more symptomatic), group A/B (low exacerbation risk), and group C/D (high exacerbation risk), according to GOLD 2011 or 2017. All six studies included in this part of analysis provided DLCO% predicted data of each separate group, so we combined the original data to obtain proper data for meta-analysis. DLCO% predicted was significantly lower in more symptomatic group (group B/D) (MD 16.54%, 95% CI: 9.53%–23.54%, p < 0.00,001) compared to few symptomatic group (group A/C) (Figure 2(b)). Subgroup analysis showed this difference in both GOLD 2011 subgroup and GOLD 2017 subgroup. DLCO % predicted was also significantly lower in high exacerbation risk group (group C/D) (MD 9.92%, 95% CI: 5.55%–14.30%, p<0.00,001), compared to low exacerbation risk group (group A/B) (Figure 2(c)). Subgroup analysis showed this difference in both GOLD 2011 subgroup and GOLD 2017 subgroup. Forest plot of the mean difference of DLCO % predicted. 2(a). Between COPD patients of stage I/II and III/IV. 2(b). Between COPD patients of group A/C and group B/D. 2(c). Between COPD patiens of group A/B and C/D.
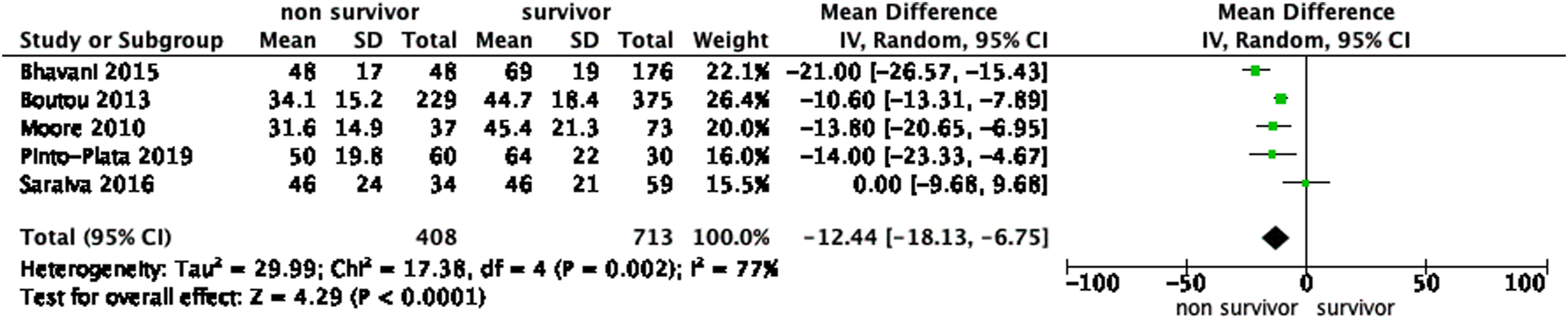
Nine studies were included in meta-analysis to evaluate diffusing capacity in AECOPD (Figure 3). Seven studies compared DLCO % predicted between non-exacerbators and exacerbators, showing a decreased DLCO % predicted in exacerbators (MD 4.56%, 95% CI: 2.54%–6.59%, p < 0.00,001). Two studies compared DLCO % predicted between non-frequent exacerbators and frequent exacerbators, indicating that DLCO % predicted was lower in frequent exacerbators (MD 7.20%, 95% CI: 3.14%–11.27%, p = 0.0005). The meta-analysis of DLCO % predicted in COPD mortality showed a significantly decreased DLCO % predicted in non-survivors (MD 12.44%, 95% CI: 6.75%–18.13%, p < 0.0001) (Figure 4). Forest plot of the mean difference of DLCO % predicted between COPD patients with or without acute exacerbation. Forest plot of the mean difference of DLCO % predicted between survivors and non-survivors.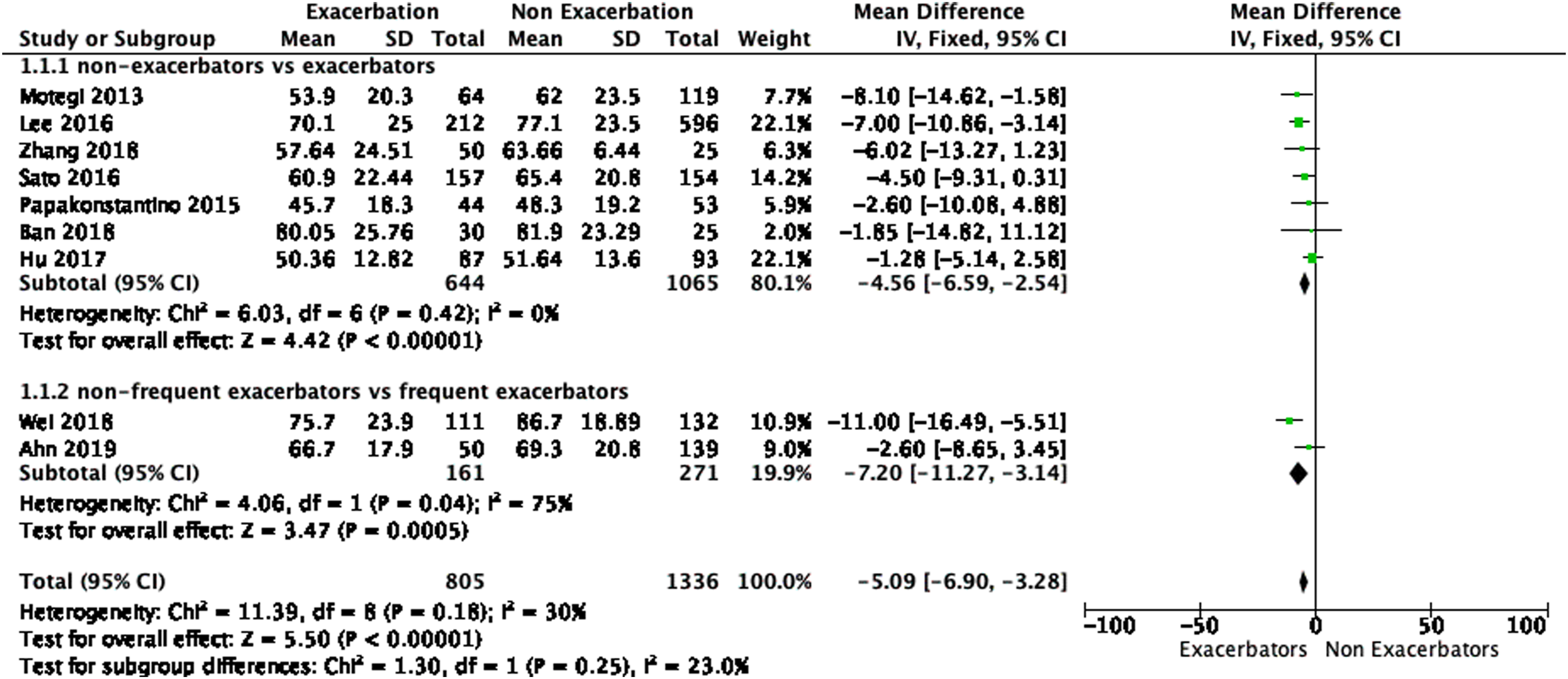
Diffusing capacity, emphysema and pulmonary hypertension
Figure 5(a) showed a decreased DLCO % predicted in emphysema dominant COPD patients (MD 23.86%, 95% CI: 17.51%–30.21%, p < 0.00,001), compared to non-emphysema dominant COPD patients. Figure 5(b) showed that DLCO % predicted was significantly decreased in COPD patients with pulmonary hypertension (PH) (MD 12.52%, 95% CI: 23.76%–1.29%, p < 0.00,001), compared with COPD without PH. One study
57
included in analysis only enrolled severe COPD patients and did not show a great difference of DLCO % predicted between groups. This might related to the great percentage of patients who did not accomplished DLCO measurement. (5a)Forest plot of the mean difference of DLCO % predicted between emphysema dominant and non-emphysema dominant COPD patients (5b) between COPD patients with or without PH.
Discussion
With the results of our analysis, DLCO % predicted could be a predictor of several aspects of COPD. We analyzed DLCO % predicted in COPD patients with different severity classification, according to GOLD stage, GOLD 2011 groups, or GOLD 2017 groups. Consistent to previous study, 60 lower DLCO % predicted was found in COPD patients with lower FEV1 % predicted (stage III/IV). Lower DLCO % predicted was also found in patients with high exacerbation risk (Group C/D) no matter that the patients were classified according to GOLD 2011 or to GOLD 2017. The predictive value of DLCO% predicted in exacerbation might be just because of the correlation between DLCO% predicted and FEV1% predicted. However, compared to GOLD 2011, the evaluation of lung function impairment is separated from the “ABCD groups” in GOLD 2017 version. Study indicated that this modification led to a shift of patients from high risk (C and D) to low-risk categories (A and B). 61 But in our analysis, lower DLCO % predicted was also found in group C/D according to GOLD 2017. This suggested that DLCO % predicted might have a role in predicting exacerbation risk, independently to FEV1 level. Previous studies3,40,42 indicated DLCO % predicted or DLCO/VA as a predictor of AECOPD. Regrettably, since previous studies used different grouping of DLCO to evaluate predictive value, it is not possible to conduct a meta-analysis to get a synthesis HR.
Diffusing capacity of the lung for carbon monoxide or DLCO % predicted might also predict COPD mortality. Ward et al.’s study 18 showed that DLCO was the best lung function predictor of all-cause mortality, but this study was not only limited in COPD patients. Other studies62–64 with relative smaller sample sizes found a predictive value of DLCO or DLCO % predicted for COPD mortality. However, neither DLCO nor DLCO % predicted is currently integrated in commonly used predict models. A recent systematic review 65 of prognostic models for COPD outcomes included 408 models. Among the 12 most validated prognostic models (ADO index, APACHE II, BOD index, BODE index, BODEx index, CODEX index, COTE index, CURB-65, DOSE index, LACE index, updated ADO index, and updated BODE index), none of them included DLCO or DLCO % predicted. Due to different grouping of DLCO in previous studies, we were not able to conduct a meta-analysis to get a synthesis HR, and only showed a significantly lower DLCO % predicted in non-survivors when compared to survivors. However, in our opinion, we should still keep in mind that DLCO% predicted could be a predictive factor of COPD mortality.
It is well known that DLCO is related to emphysema. Our analysis also showed a great degree of decrease in DLCO % predicted in emphysema dominant COPD patients, when compared to non-emphysema dominant COPD patients. Beside emphysema, DLCO is also related to pulmonary perfusion. A recent study showed that decrease of DLCO % predicted was related with poorer quality of life, more severe exacerbation, and increased COPD morbidity, independent of FEV1 and CT-based emphysema percentage. 3 The authors believed that DLCO % predicted provided more clinical information than spirometry and CT imaging and provide a window into the interactions between vascular and pulmonary physiology. Pulmonary hypertension is one of the most common vascular comorbidities in COPD patients. We found that DLCO % predicted was significantly lower in COPD patients with PH compared to COPD patients without PH. A subset of COPD-PH patients deemed the “vascular phenotype” is defined as those patients who have minimal airflow obstruction but severe hemodynamic derangement. 66 This subset of patients was characterized with lower DLCO and worse mortality compared to COPD patients without PH. And study showed that DLCO % predicted was an independent predictor for survival in COPD patients with PH. 67
Current assessment of COPD is mainly based on GOLD 2017 group classification. GOLD 2017 group classification includes spirometry, symptoms, and exacerbation history; however, the evaluation of symptoms with mMRC or CAT is subjective, and, to some extent, the evaluation of exacerbation is also subjective. Our analysis showed that diffusion capacity may be a useful objective assessment criterion for COPD. As diffusion capacity reflects pulmonary ventilation and perfusion at the same time, it may be able to explain why some patients have more symptoms while their FEV1 is not quite low. But the predictive value of DLCO or DLCO combined with other criteria still needs to be further explored. Another obstacle is that it is challenging for some patients to perform the DLCO maneuver, especially patients with severe airflow limitation.
All studies enrolled in our meta-analysis calculated DLCO % predicted by using Global Lung Initiative reference equations, 68 with DLCO values adjusted for hemoglobin and altitude. However, another factor which may also influence DLCO measurement is smoking. Acute intense smoking results in a decrease in DLCO. 69 None of the enrolled studies indicated the time interval between last smoking and DLCO measurement. Although some studies provided smoking status of patients, but it is not sufficient to explain how much smoking influenced DLCO data. Besides COPD and pulmonary vascular involvement due to COPD, the cardiovascular diseases can also influence diffusing capacity of lung. Five studies enrolled in our analysis clearly mentioned that patients with cardiovascular diseases or primary cardiac diseases were excluded, while other studies did not state in their exclusion criteria. The cardiovascular disease might be a confounder of our meta-analysis. Exclusion of heart diseases, especially coronary heart disease and left heart failure, should be paid attention in future studies about DLCO in COPD patients. No enrolled studies specifically excluded obesity patients. Most of the studies provided BMI data and most were in a normal range. No enrolled studies indicated if patients had anemia or if anemia patients were excluded. But as all studies calculated DLCO % predicted with DLCO adjusted for hemoglobin, we suppose that the anemie should not influence DLCO measurement too much. DLCO % predicted, DLCO and carbon monoxide transfer coefficient (KCO) can all reflect lung diffusing capacity but in different ways and they can be influenced by many physiologic or pathologic reasons. How to use these parameters correctly in COPD assessment need to be further investigated.
Although our analysis provided interesting findings about DLCO % predicted in COPD assessment, there are still some limitations. Our analysis is a meta-analysis of observational studies. Due to the nature of meta-analysis, the quality can be influenced by the primary studies. Besides, although there were no significant publication bias or small-study effects found in funnel plot and Egger’s test, the numbers of studies were still small, especially these included for emphysema and pulmonary hypertension, thus potential publication bias may exist. And our meta-analysis included both cross-sectional studies and cohort studies. Different design of studies may be a confounder, although no significant difference was found in subgroup analysis of study design (data not shown). Finally, since the predictive role of DLCO% predicted in exacerbation risk and mortality was given in different modality, we were not able to synthesize an overall HR of DLCO% predicted.
Conclusion
The current meta-analysis showed that DLCO % predicted was an important measurement for COPD patients in terms of severity, exacerbation risk, mortality, emphysema domination, and presence of pulmonary hypertension. Although current prognostic models assessing mortality and morbidity do not include DLCO % predicted, findings from this study suggest that inclusion of DLCO % predicted in such models should be considered. DLCO % predicted is a readily available and frequently obtained test. Future studies investigating the clinical utility of DLCO % predicted in prognostic models, the relationship between vascular disease in COPD and DLCO % predicted, and the implications of longitudinal changes in diffusing capacity are of great necessity.
Supplemental Material
sj-pdf-1-crd-10.1177_14799731211056340 – Supplemental Material for Diffusing capacity in chronic obstructive pulmonary disease assessment: A meta-analysis
Supplemental Material, sj-pdf-1-crd-10.1177_14799731211056340 for Diffusing capacity in chronic obstructive pulmonary disease assessment: A meta-analysis by Yingmeng Ni, Youchao Yu, Ranran Dai and Guochao Shi in Chronic Respiratory Disease
Footnotes
Authors contributions
Yingmeng Ni: Study searching, data extraction and manuscript writingYouchao Yu: Study searchingRanran Dai: Data extractionGuochao Shi: Study design and manuscript writing
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by NSFC 81800019, 81970020, 81770025 and program of National Innovative Research Team of High-level Local Universities in Shanghai, Shanghai Municipal Key Clinical Speciality (shslczdzk02202) and Shanghai Top-Priority Clinical Kew Disciplines Construction Project (2017ZZ02014).
Ethical approval
The paper is exempt from ethical committee approval. This paper is a meta-analysis and only used published data.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
